Neurophysiological Stress Response and Mood Changes Induced by High-Intensity Interval Training: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Approach
2.2.1. Session 1: Graded Exercise Test
2.2.2. Session 2: HIIT Intervention
2.3. Statistical Analysis
3. Results
3.1. Sample Selection
3.2. Graded Exercise Test
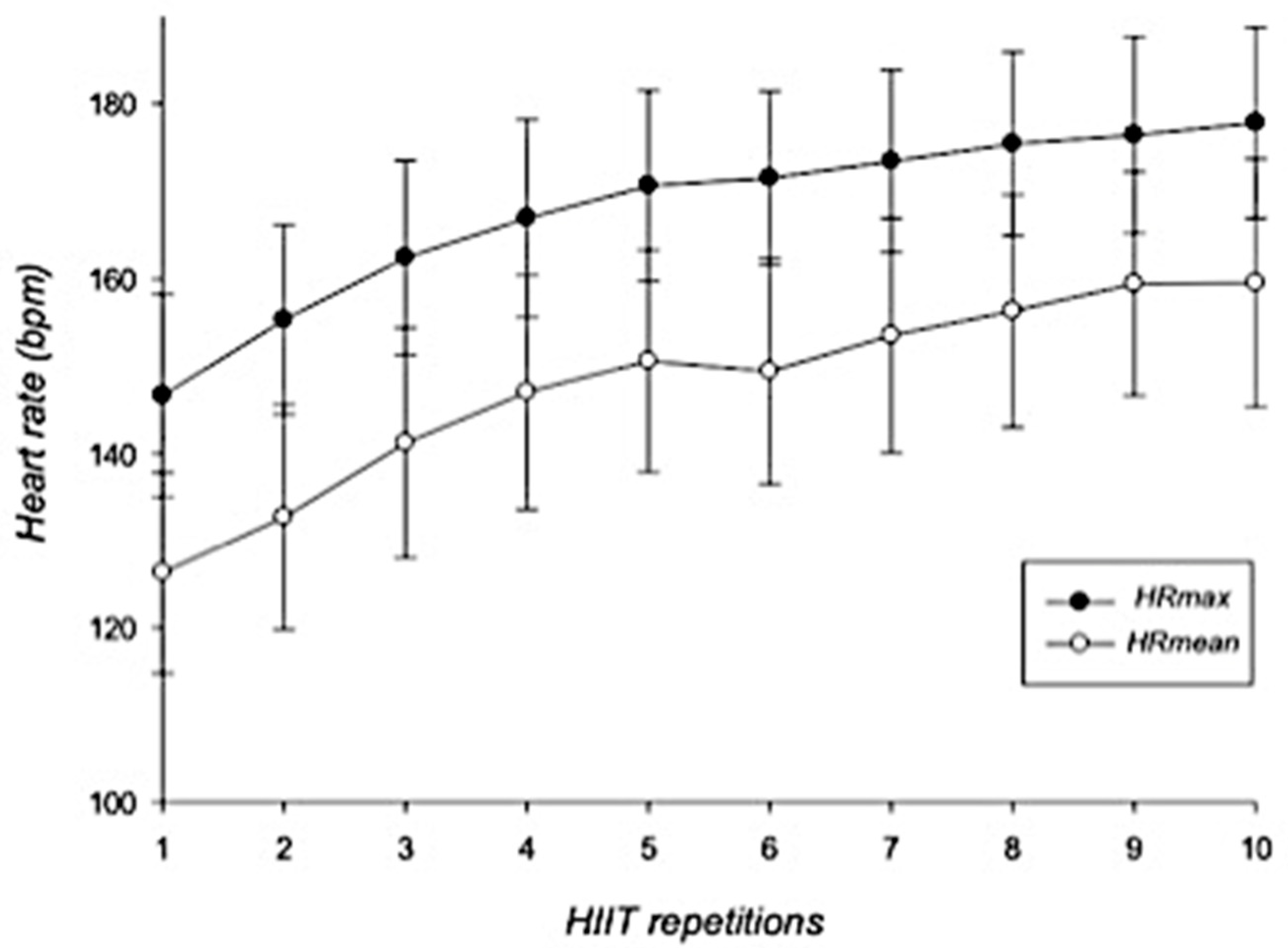
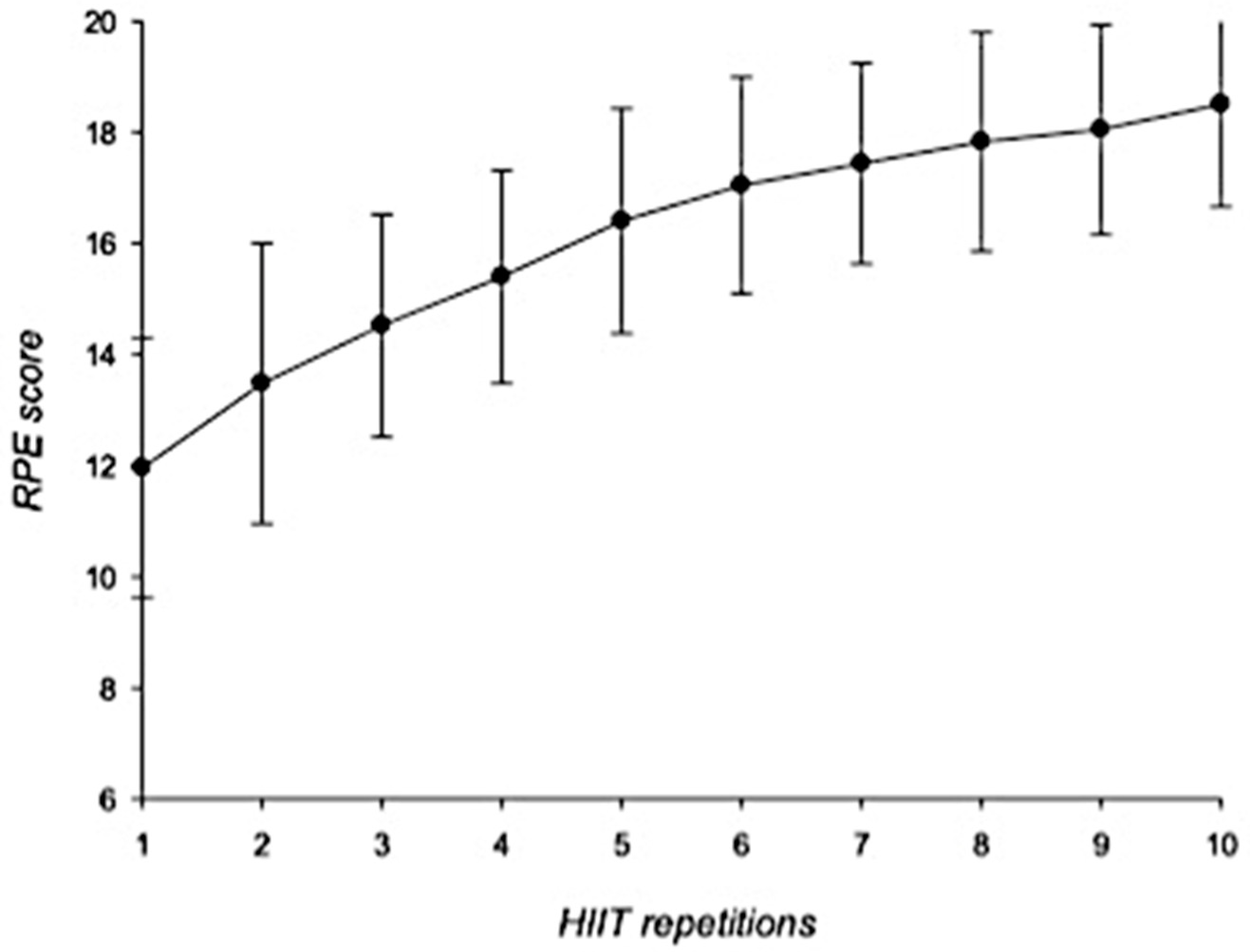
3.3. HIIT Session: Cardiovascular and RPE Responses
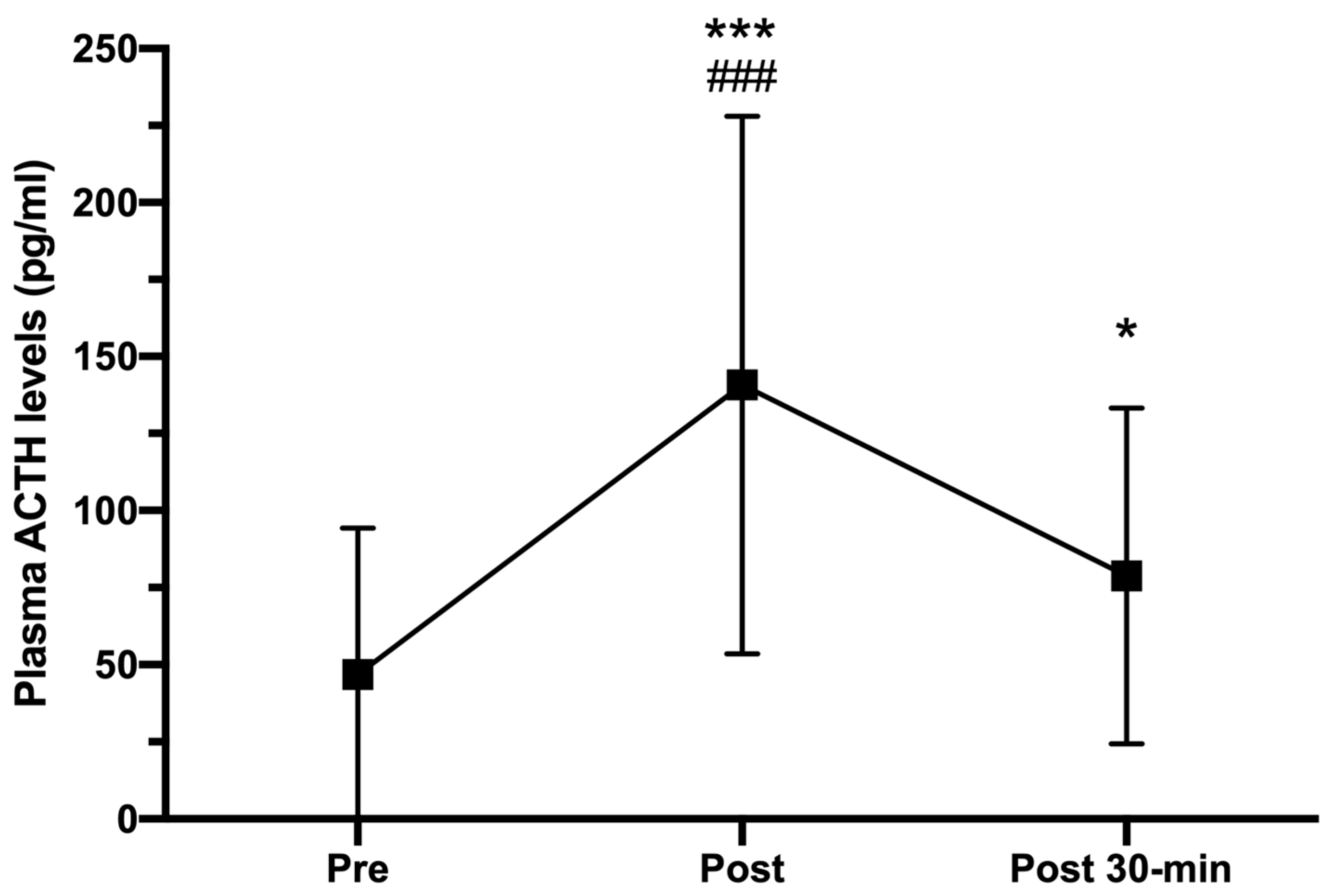
3.4. Plasma ACTH and Cortisol Levels
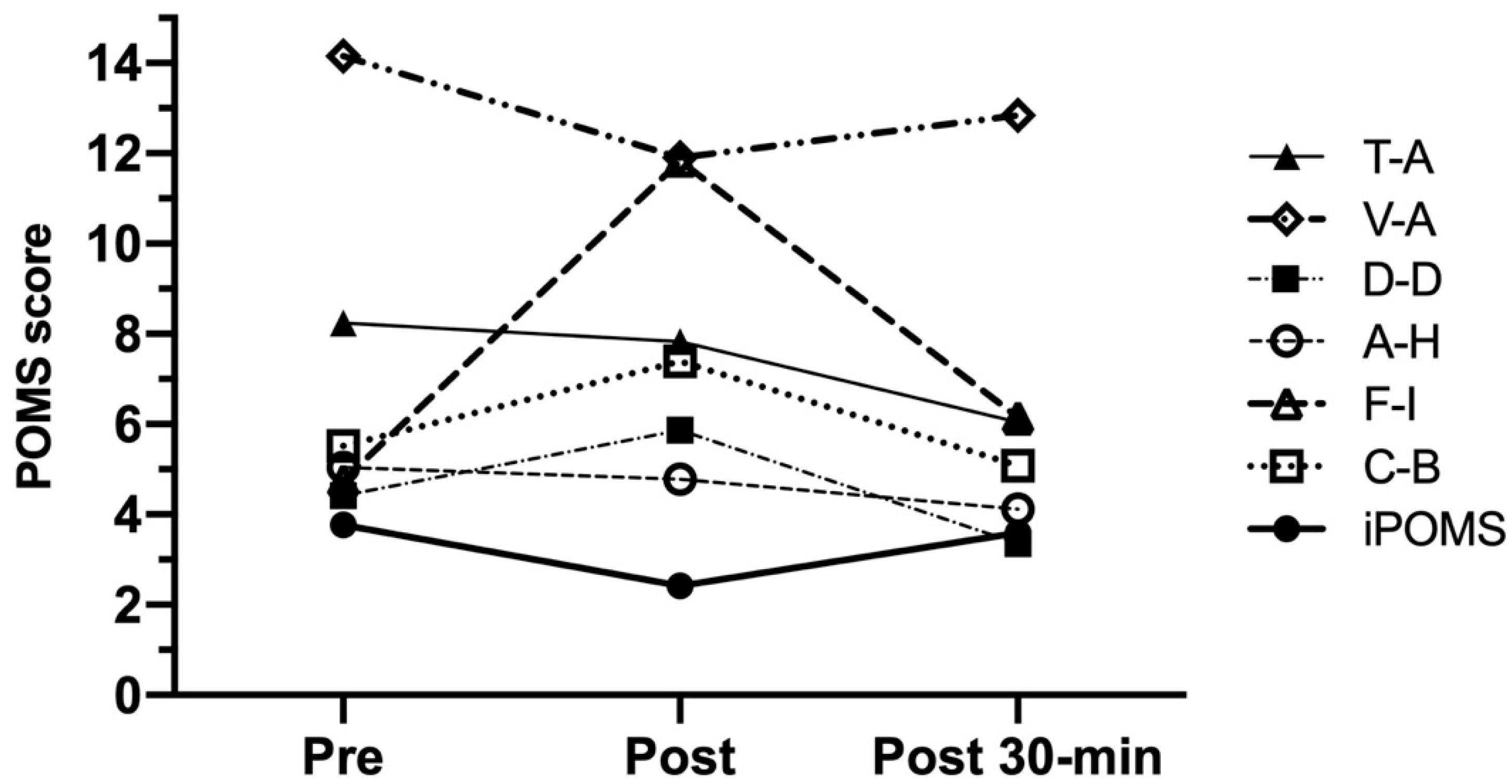
3.5. Exercise-Induced Mood Changes
3.6. Relationships between ACTH, Cortisol and Mood States
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Helgerud, J.; Høydal, K.; Wang, E.; Karlsen, T.; Berg, P.; Bjerkaas, M.; Simonsen, T.; Helgesen, C.; Hjorth, N.; Bach, R.; et al. Aerobic high-intensity intervals improve VO2max more than moderate training. Med. Sci. Sports Exerc. 2007, 39, 665–671. [Google Scholar] [CrossRef]
- Buchheit, M.; Laursen, P. High-intensity interval training, solutions to the programming puzzle. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef]
- Henckens, M.J.; Wingen, G.A.; Joels, M.; Fernandez, G. Time-dependent effects of cortisol on selective attention and emotional interference: A functional MRI study. Front. Integr. Neurosci. 2012, 6, 66. [Google Scholar] [CrossRef]
- Spencer, R.L.; Deak, T. A users guide to HPA axis research. Physiol. Behav. 2017, 178, 43–65. [Google Scholar] [CrossRef] [PubMed]
- Meeusen, R.; Piacentini, M.F.; Van Den Eynde, S.; Magnus, L.; De Meirleir, K. Exercise performance is not influenced by a 5-HT reuptake inhibitor. Int. J. Sports Med. 2001, 22, 329–336. [Google Scholar] [CrossRef]
- Schulz, A.; Harbach, H.; Katz, N.; Geiger, L.; Teschemacher, H. β-endorphin immunoreactive material and authentic β-endorphin in the plasma of males undergoing anaerobic exercise on a rowing ergometer. Int. J. Sports Med. 2000, 21, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Harada, T.; Kurono, M.; Matsui, N. Effect of exercise-induced acidosis on aldosterone secretion in men. Eur. J. Appl. Physiol. Occup. Physiol. 1998, 77, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Fragala, M.; Kraemer, W.J.; Denegar, C.R.; Maresh, C.M.; Mastro, A.M.; Volek, J.S. Neuroendocrine-Immune Interactions and Responses to Exercise. Sports Med. 2011, 41, 621–639. [Google Scholar] [CrossRef]
- Rojas-Vega, S.; Struder, H.K.; Wahrmann, B.V.; Schmidt, A.; Bloch, W.; Hollmann, W. Acute BDNF and cortisol response to low intensity exercise and following ramp incremental exercise to exhaustion in humans. Brain Res. 2006, 1121, 59–65. [Google Scholar] [CrossRef]
- Tanner, A.V.; Nielsen, B.V.; Allgrove, J. Salivary and plasma cortisol and testosterone responses to interval and tempo runs and a bodyweight-only circuit sesión in endurance-trained men. J. Sports Sci. 2014, 32, 680–690. [Google Scholar] [CrossRef]
- Hough, J.P.; Papacosta, E.; Wraith, E.; Gleeson, M. Plasma and salivary steroid hormone response of men to high-intensity cycling and resistance exercise. J. Strength Cond. Res. 2011, 25, 23–31. [Google Scholar] [CrossRef]
- Vuorimaa, T.; Ahotupa, M.; Hakkinen, K.; Vasankari, T. Different hormonal response to continuous and intermittent exercise in middle-distance and maratón runners. Scand. J. Med. Sci. Sports 2008, 18, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Gerber, M.; Minghetti, A.; Beck, J.; Zahner, L.; Donath, L. Sprint Interval Training and Continuous Aerobic Exercise Training Have Similar Effects on Exercise Motivation and Affective Responses to Exercise in Patients With Major Depressive Disorders: A Randomized Controlled Trial. Front. Psychiatry 2018, 9, 694. [Google Scholar] [CrossRef] [PubMed]
- Saanijoki, T.; Nummenmaa, L.; Koivumäki, M.; Löyttyniemi, E.; Kalliokoski, K.K.; Hannukainen, J.C. Affective adaptation to repeated SIT and MICT protocols in insulin-resistant subjects. Med. Sci. Sports Exerc. 2018, 50, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Stork, M.J.; Gibala, M.J.; Martin-Ginis, K.A. Psychological and behavioral responses to interval and continuous exercise. Med. Sci. Sports Exerc. 2018, 50, 2110–2121. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, P.J.; Carraca, E.V.; Markland, D.; Silva, M.N.; Ryan, R.M. Exercise, physical activity, and self-determination theory: A systematic review. Int. J. Behav. Nutr. Phys. Activ. 2012, 9, 78. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, B.R.R.; Oliveira, B.R.R.; Santos, T.M.; Kilpatrick, M.; Pires, F.O.; Deslandes, A.C. Affective and enjoyment responses in high intensity interval training and continuous training: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0197124. [Google Scholar]
- Di Corrado, D.; Agostini, T.; Bonifazi, M.; Perciavalle, V. Changes in mood states and salivary cortisol levels following two months of training in elite female water polo players. Mol. Med. Rep. 2014, 9, 2441–2446. [Google Scholar] [CrossRef]
- Schelling, X.; Calleja-González, J.; Terrados, N. Variación de la testosterona y el cortisol en relación al estado de ánimo en jugadores de baloncesto de élite. Rev. Int. Cienc. Deporte 2013, 34, 342–359. [Google Scholar] [CrossRef]
- Kilian, Y.; Engel, F.; Wahl, P.; Achtzehn, S.; Sperlich, B.; Mester, J. Markers of biological stress in response to a single session of high-intensity interval training and high-volume training in young athletes. Eur. J. Appl. Physiol. 2016, 116, 2177–2186. [Google Scholar] [CrossRef]
- Lopes, A.; de Toledo, R.A.G.; Luksevicius, R.; Fernandes, A.; Miranda, J.M.Q.; Teixeira, C.V.L.S.; Lopes, C.R.; Bocalini, D.S. Effects of high-intensity calisthenic training on mood and affective responses. J. Exerc. Physiol. 2017, 20, 15–23. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- McNair, D.M.; Lorr, M.; Droppleman, L.F. Manual for the Profile of Mood States; Educational and Industrial Testing Services: San Diego, CA, USA, 1971. [Google Scholar]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sport Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Fontani, G.; Lodi, L.; Migliorini, S.; Corradeschi, F. Effect of Omega-3 and policosanol supplementation on attention and reactivity in athletes. J. Am. Coll. Nutr. 2009, 28, 473–481. [Google Scholar] [CrossRef]
- Spielberger, C.D. Manual for the State-Trait Anxiety Inventory: STAI (Form I); Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Earlbaum Associates: Hillsdale, MI, USA, 1988. [Google Scholar]
- Van der Pompe, G.; Bernards, N.; Kavelaars, A.; Heijnen, C. An exploratory study into the effect of exhausting bicycle exercise on endocrine and immune responses in post-menopausal women: Relationships between vigour and plasma cortisol concentrations and lymphocyte proliferation following exercise. Int. J. Sports Med. 2001, 22, 447–453. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Davranche, K.; Jones, G.; Hall, B.; Corbett, J.; Minter, C. Acute incremental exercise, performance of a central executive task, and sympathoadrenal system and hypothalamic-pituitary-adrenal axis activity. Int. J. Psychophysiol. 2009, 73, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Marquet, P.; Lac, G.; Chassain, A.P.; Habrioux, G.; Galen, F.X. Dexamethasone in resting and exercising men. I. Effects on bioenergetics, minerals and related hormones. J. Appl. Physiol. 1999, 87, 175–182. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Veldhuis, J.D.; Iranmanesh, A.; Naftolowitz, D.; Tatham, N.; Cassidy, F.; Carroll, B.R. Corticotropin secretory dynamics in humans under low glucocorticoid feedback. J. Clin. Endocrinol. Metab. 2001, 86, 5554–5563. [Google Scholar] [CrossRef] [PubMed]
- Peak, J.M.; Tan, S.J.; Markworth, J.F.; Broadbent, J.A.; Skinner, T.L.; Cameron-Smith, D. Metabolic and hormonal responses to isoenergetic high-intensity interval exercise and continuous moderate-intensity. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E539–E552. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Ratamess, N.A. Hormonal responses and adaptations to resistance exercise and training. Sports Med. 2005, 35, 339–361. [Google Scholar] [CrossRef]
- Schwarz, L.; Kindermann, W. Beta-endorphin, adrenocorticotropic hormone, cortisol and catecholamines during aerobic and anaerobic exercise. Eur. J. Appl. Physiol. Occup. Physiol. 1990, 61, 165–171. [Google Scholar] [CrossRef]
- Ratamess, N.A.; Kraemer, W.J.; Volek, J.S.; Maresh, C.M.; VanHeest, J.L.; Sharman, M.J.; Rubin, M.R.; French, D.N.; Vescovi, J.D.; Silvestre, R.; et al. Androgen receptor content following heavy resistance exercise in men. J. Steroid Biochem. Mol. Biol. 2005, 93, 35–42. [Google Scholar] [CrossRef]
- Kujach, S.; Olek, R.A.; Byun, K.; Suwabe, K.; Sitek, E.J.; Ziemann, E.; Laskowski, R.; Soya, H. Acute sprint interval exercise increases both cognitive functions and peripheral neurotrophic factors in humans: The possible involvement of lactate. Front. Neurosci. 2020, 13, 1455. [Google Scholar] [CrossRef]
- Basso, J.C.; Suzuki, W.A. The effects of acute exercise on mood, cognition, neurophysiology, and neurochemical pathways: A review. Brain Plast. 2017, 2, 127–152. [Google Scholar] [CrossRef] [PubMed]
- Berger, B.G.; Motl, R.W. Exercise and mood: A selective review and synthesis of research employing the profile of mood states. J. Appl. Sport Psychol. 2000, 12, 69–92. [Google Scholar] [CrossRef]
- Selmi, O.; Haddad, M.; Majed, L.; Ben Khalifa, W.; Hamza, M.; Chamari, K. Soccer Training: High-Intensity Interval Training Is Mood Disturbing While Small Sided Games Ensure Mood Balance. J. Sports Med. Phys. Fit. 2019, 58, 1163–1170. [Google Scholar]
- Parfitt, G.; Rose, E.A.; Burgess, W.M. The psychological and physiological responses of sedentary individuals to prescribed and preferred intensity exercise. Br. J. Health Psychol. 2006, 11, 39–53. [Google Scholar] [CrossRef]
- Oliveira, B.R.; Slama, F.A.; Deslandes, A.C.; Furtado, E.S.; Santos, T.M. Continuous and high-intensity interval training: Which promotes higher pleasure? PLoS ONE 2013, 8, e79965. [Google Scholar] [CrossRef] [PubMed]
- Martinez, N.; Kilpatrick, M.W.; Salomon, K.; Jung, M.E.; Little, J.P. Affective and enjoyment responses to high-intensity interval training in overweight-to-obese and insufficiently active adults. J. Sport Exerc. Psychol. 2015, 37, 138–149. [Google Scholar] [CrossRef]
- Hall, E.E.; Ekkekakis, P.; Petruzzello, S.J. The affective beneficence of vigorous exercise revisited. Br. J. Health Psychol. 2002, 7, 47–66. [Google Scholar] [CrossRef]
- Crush, E.A.; Frith, E.; Loprinzi, P.D. Experimental effects of acute exercise duration and exercise recovery on mood state. J. Affect. Disord. 2018, 229, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, R.R.; Dzewaltowski, D.A.; Rinehard, K.F.; Castracane, V.D. Mood alteration from treadmill running and its relationship to beta-endorphin, corticotropin, and growth hormone. J. Sports Med. Phys. Fit. 1990, 30, 241–246. [Google Scholar]
- Garcia, J. Association of Exercise Induced Salivary Cortisol Levels to Exertional Perception and Affect. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, 2008. [Google Scholar]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A.; PAFS Consensus Group. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. BMJ 2016, 355, i5239. [Google Scholar] [CrossRef] [PubMed]

| Mean | sd | |
|---|---|---|
| Subjects’ characteristics (n = 25) | ||
| Age (yr) | 21.7 | 2.1 |
| Height (cm) | 177 | 6.1 |
| Weight (Kg) | 72.6 | 8.4 |
| BMI (Kg/m2) | 23.1 | 1.4 |
| Body fat (%) | 13.4 | 3.6 |
| IPAQ-total score (MET/min/wk) | 5877.6 | 1668.2 |
| Graded Exercise Test | ||
| Time to exhaustion (min) | 13.6 | 1.8 |
| HRmax (bpm) | 181.5 | 8.4 |
| VO2peak (mL/kg/min) | 45.3 | 9.3 |
| pVO2peak (W) | 275.0 | 48.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Díaz, I.C.; Carrasco, L. Neurophysiological Stress Response and Mood Changes Induced by High-Intensity Interval Training: A Pilot Study. Int. J. Environ. Res. Public Health 2021, 18, 7320. https://doi.org/10.3390/ijerph18147320
Martínez-Díaz IC, Carrasco L. Neurophysiological Stress Response and Mood Changes Induced by High-Intensity Interval Training: A Pilot Study. International Journal of Environmental Research and Public Health. 2021; 18(14):7320. https://doi.org/10.3390/ijerph18147320
Chicago/Turabian StyleMartínez-Díaz, Inmaculada C., and Luis Carrasco. 2021. "Neurophysiological Stress Response and Mood Changes Induced by High-Intensity Interval Training: A Pilot Study" International Journal of Environmental Research and Public Health 18, no. 14: 7320. https://doi.org/10.3390/ijerph18147320
APA StyleMartínez-Díaz, I. C., & Carrasco, L. (2021). Neurophysiological Stress Response and Mood Changes Induced by High-Intensity Interval Training: A Pilot Study. International Journal of Environmental Research and Public Health, 18(14), 7320. https://doi.org/10.3390/ijerph18147320







