The Impact of Introducing Successive Biosimilars on Changes in Prices of Adalimumab, Infliximab, and Trastuzumab—Polish Experiences
Abstract
:1. Introduction
2. Materials and Methods
2.1. Phase 1: Medicine Selection
- The original drug should have been approved for marketing in the European Union.
- The original drug should have at least three biosimilars approved for marketing in the European Union.
- The original drug and its biosimilars should be covered by drug-program reimbursement.
2.2. Phase 2: Identification of Price-Level Changes
2.3. Phase 3: Analysis of Price Change Levels
- price change in reference to basic observation (single-base indices, 100 = July 2012);
- price change in reference to a prior price change (single-base indices, 100 = prior price change).
3. Results
3.1. Adalimumab
3.2. Infliximab
3.3. Trastuzumab
3.4. Comparative Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Medicines Agency. Guideline on Similar Biological Medicinal Products. CHMP/437/04 Rev 1. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-similar-biological-medicinal-products-rev1_en.pdf (accessed on 3 March 2021).
- Marklowska Dzierżak, M. Co trzeba wiedzieć o lekach biologicznych referencyjnych i biopodobnych. In Leki Biologiczne Biopodobne w Praktyce Klinicznej; Puls Medycyny: Warsaw, Poland, 2017; p. 8. [Google Scholar]
- Art. 15 Section 2 Point 15 of the Act of 27 August 2004 on Healthcare Services Financed from Public Funds (Journal of Laws 2004 no. 210, Item 2135). Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20042102135 (accessed on 9 May 2021).
- Art. 2 Point 18 of the Act of 12 May 2011 on Reimbursement of Medicines, Foodstuffs Intended for Particular Nutritional Uses and Medical Devices. Available online: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20111220696/T/D20110696L.pdf (accessed on 9 May 2021).
- Art. 48 Section 2 of the Act of 27 August 2004 on Healthcare Services Financed from Public Funds (Journal of Laws 2004 no. 210, Item 2135). Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20042102135 (accessed on 9 May 2021).
- Ministry of Health Gov.pl. Available online: https://www.gov.pl/web/zdrowie/programy-lekowe (accessed on 7 May 2021).
- Lurka, K. Oto Najdroższe Programy Lekowe, Termedia.pl. Available online: https://www.termedia.pl/mz/Oto-najdrozsze-programy-lekowe,36052.html (accessed on 7 May 2021).
- Programy Lekowe. Choroby Nieonkologiczne. Available online: https://www.gov.pl/web/zdrowie/choroby-nieonkologiczne (accessed on 7 May 2021).
- Announcements of the Minister of Health—List of Reimbursed Drugs. Ministry of Health Gov.pl. Available online: https://www.gov.pl/web/zdrowie/obwieszczenia-ministra-zdrowia-lista-lekow-refundowanych (accessed on 6 April 2021).
- Jakie i ile Leków Refunduje NFZ? Część Pierwsza—Programy Lekowe, Pacjentinfo.pl. Available online: https://pacjentinfo.pl/tag/programy-lekowe/ (accessed on 3 March 2021).
- Rogowski, W.; Zyśk, R.; Krzakowski, M. Programy Lekowe W Onkologii. Jak Optymalnie Wykorzystać ich Możliwości? Onkologia w Praktyce Klinicznej—Edukacja. 2018, 4, pp. 323–325. Available online: https://journals.viamedica.pl/onkologia_w_praktyce_klin_edu/article/view/61495 (accessed on 3 March 2021).
- Ile Wydał NFZ na Refundację w 2020 r.? politykazdrowotna.com. Available online: https://www.politykazdrowotna.com/67870,wydatki-nfz-na-refundacje-w-2020-r (accessed on 3 March 2021).
- Cykl refundacja—Ile NFZ Wydaje na leki?, Pacjentinfo.pl. Available online: https://pacjentinfo.pl/2019/12/06/cykl-refundacja-ile-nfz-wydaje-na-leki/ (accessed on 5 March 2021).
- Steven, S. Biosimilar Medicines and cost-effectiveness. Clin. Econ. Outcomes Res. 2011, 3, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Roger, S.D. Biosimilars: Current status and future directions. Expert Opin. Biol. Ther. 2010, 10, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Vakil, N.; Fanikos, J. Regulatory and Clinical Perspective on Biosimilars: A Comparison of the US and European Experiences. Curr. Emerg. Hosp. Med. Rep. 2019, 7, 111–117. [Google Scholar] [CrossRef]
- European Medicines Agency. Medicines. Available online: https://www.ema.europa.eu/en/medicines/field_ema_web_categories%253Aname_field/Human/ema_group_types/ema_medicine/field_ema_med_status/authorised-36/ema_medicine_types/field_ema_med_biosimilar/ search_api_aggregation_ema_medicine_types/ field_ema_med_biosimilar (accessed on 3 March 2021).
- Obarska, I. Biologiczne leki równoważne i ich znaczenie dla systemu ochrony zdrowia w Polsce. In Dostęp do leczenia biologicznego w Polsce; Modern Healthcare Institute: Warsaw, Poland, 2019. [Google Scholar]
- European Medicines Agency and the European Commission. Biosimilars in the EU. Information Guide for Healthcare Professionals; European Medicines Agency: Amsterdam, The Netherlands, 2019; p. 29. [Google Scholar]
- The IQVIA Institute. A Multi-Stakeholder Assessment. Advancing Biosimilar Sustainability in Europe. Warsaw. 2018. Available online: https://www.iqvia.com/insights/the-iqvia-institute/reports/advancing-biosimilar-sustainability-in-europe (accessed on 14 June 2021).
- Art. 2 Point 7 of the Act of 12 May 2011 on Reimbursement of Medicines, Foodstuffs Intended for Particular Nutritional Uses and Medical Devices. Available online: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20111220696/T/D20110696L.pdf (accessed on 9 May 2021).
- Domański Zakrzewski Palinka Limited Partnership. Raport Regulacyjny Dotyczący Leków Biologicznych i Biopodobnych. Available online: https://www.dzp.pl/files/shares/Publikacje/Raport_Leki_Biologiczne.pdf (accessed on 6 April 2021).
- European Medicines Agency (EMA). Humira (Adalimumab) An Overview of Humira and Why It Is Authorised in the EU. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/humira (accessed on 9 November 2020).
- European Medicines Agency. An Overview of Remicade and Why It Is Authorised in the EU. Available online: https://www.ema.europa.eu/en/documents/overview/remicade-epar-summary-public_en.pdf (accessed on 7 April 2021).
- National Bank of Poland. Table No. 109/A/NBP/2021 of 2021-06-09. Available online: https://www.nbp.pl/homen.aspx?f=/kursy/ratesa.html (accessed on 14 June 2021).
- Łącka-Gaździk, B.; Śnit, M.; Grzeszczak, W. Insuliny biopodobne jako alternatywa dla produktów oryginalnych. Diabetol. Prakt. 2016, 2, 143. [Google Scholar] [CrossRef]
- Chen, Y.; Lan, Y.; Huang, Z. Government Subsidy Strategies for Biosimilars RD Based on Dynamic Game Theory. IEEE Access 2020, 8, 5817–5823. [Google Scholar] [CrossRef]
- Zielińska, J.; Bialik, W. Recent changes on the biopharmaceutical market after the introduction of biosimilar G-CSF products. Oncol. Clin. Pract. 2016, 12, 144–152. [Google Scholar] [CrossRef]
- Od Czego Zależy Cena Leku Refundowanego, Ministerstwo Zdrowia. Ministry of Health Gov.pl. Available online: https://www.gov.pl/web/zdrowie/od-czego-zalezy-cena-leku-refundowanego (accessed on 5 April 2021).
- Ministry of Health. National Drug Policy 2018–2022; Ministry of Health: Warsaw, Poland, 2018; p. 58. [Google Scholar]
- Religioni, U. Zasady Ustalania i Wysokość Budżetu Refundacyjnego w Polsce—Część 2 Farmakoekonomika Szpitalna” no. 46/2019. Available online: http://www.farmakoekonomika.com.pl/zasady-ustalania-i-wysokosc-budzetu-refundacyjnego-w-polsce-czesc-2/ (accessed on 6 April 2021).
- Pieniążek-Osińska, B. Mniej na Refundację Leków w Przyszłym Roku? Available online: https://www.politykazdrowotna.com/65509,mniej-na-refundacje-lekow-w-przyszlym-roku (accessed on 6 April 2021).
- Art. 3 Point 1 of the Act of 29 January 2001 on Public Procurement Law. Available online: https://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20040190177/U/D20040177Lj.pdf (accessed on 10 June 2021).
- Stajszczyk, M.; Obarska, I.; Brzosko, M.; Kucharz, E.J.; Samborski, W. Leki Biologiczne w Chorobach Reumatycznych w Polsce—Jak Zwiększyć Dostęp Pacjentów do Terapii. Forum Reumatologiczne 2018, 4, 118–125. Available online: https://journals.viamedica.pl/forum_reumatologiczne/article/view/58264 (accessed on 10 December 2020).
- Curtis, J.R.; Xie, F.; Kay, J.; Kallich, J.D. Will Savings from Biosimilars Offset Increased Costs Related to Dose Escalation? A Comparison of Infliximab and Golimumab for Rheumatoid Arthritis. Arthritis Res. Ther. 2019, 21, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inotai, A.; Csanadi, M.; Petrova, G.; Dimitrova, M.; Bochenek, T.; Tesar, T.; York, K.; Fuksa, L.; Kostyuk, A.; Lorenzovici, L.; et al. Patient Access, Unmet Medical Need, Expected Benefits, and Concerns Related to the Utilisation of Biosimilars in Eastern European Countries: A Survey of Experts. BioMed Res. Int. 2018, 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barszczewska, O.; Piechota, A.; Suchecka, J. Management of the National Drug Administration Through the Use of Biosimilar Medicines. Expenditures, Numbers of Reimbursed Packages and Shares of Biosimilar Products in the Infliximab Market a Year Prior to and a Year After the Introduction of the National Drug Policy 2018–2022. Probl. Zarządzania (Manag. Issues) 2020, 18, 121–138. [Google Scholar] [CrossRef]
- Kucharz, E.J.; Stajszczyk, M.; Batko, B.; Brzosko, M.; Jeka, S.; Kotulska, A.; Majdan, M.; Samborski, W.; Świerkot, J.; Wiland, P.; et al. Biopodobne Leki Biologiczne w Reumatologii Biosimilars in Rheumatology. Forum Reumatol. 2018, 3, 98–109. [Google Scholar]
- Konstantinidou, S.; Papaspiliou, A.; Kokkotou, E. Current and Future Roles of Biosimilars in Oncology Practice (Review). Oncol. Lett. 2020, 19, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Infarma, HTA Consulting, Koszty pośrednie w ocenie technologii medycznych. Metodyka, badanie pilotażowe i rekomendacje. Warszawa. 2014. Available online: https://www.infarma.pl/assets/files/raporty/Raport_Koszty_posrednie_w_ocenie_technologii_medycznych_01.pdf (accessed on 15 April 2021).
- Kim, H.U.; Alten, R.; Avedano, L.; Dignass, A.; Gomollón, F.; Greveson, K.; Halfvarson, J.; Irving, P.M.; Jahnsen, J.; Lakatos, P.L.; et al. The Future of Biosimilars: Maximizing Benefits Across Immune-Mediated Inflammatory Diseases. Drugs 2020, 80, 99–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Substance | Original Drug | First Biosimilar | Second Biosimilar | Third Biosimilar | Fourth Biosimilar | Fifth Biosimilar |
|---|---|---|---|---|---|---|
| Adalimumab | Humira | Imraldi | Amgevita Hyrimoz | Idacio | x | x |
| Infliximab | Remicade | Remsima Inflectra | Flixabi | Zessly | x | x |
| Trastuzumab | Herceptin | Kanjinti | Herzuma Ontruzant | Ogivri | Trazimera | New dose Ogivri |
| Drug | Humira | Imraldi | Amgevita and Hyrimoz | Idacio | |
|---|---|---|---|---|---|
| Event | Original Drug | First Introduction of Biosimilars | Second Introduction of Biosimilars | Third Introduction of Biosimilars | |
| Date | July 2012 | January 2019 | March 2019 | March 2020 | |
| Price per package | 4446.75 | 2102.67 | 2041.2 | 800.66 | |
| Price per 1 mg | 55.58 | 26.28 | 25.52 | 20.02 | |
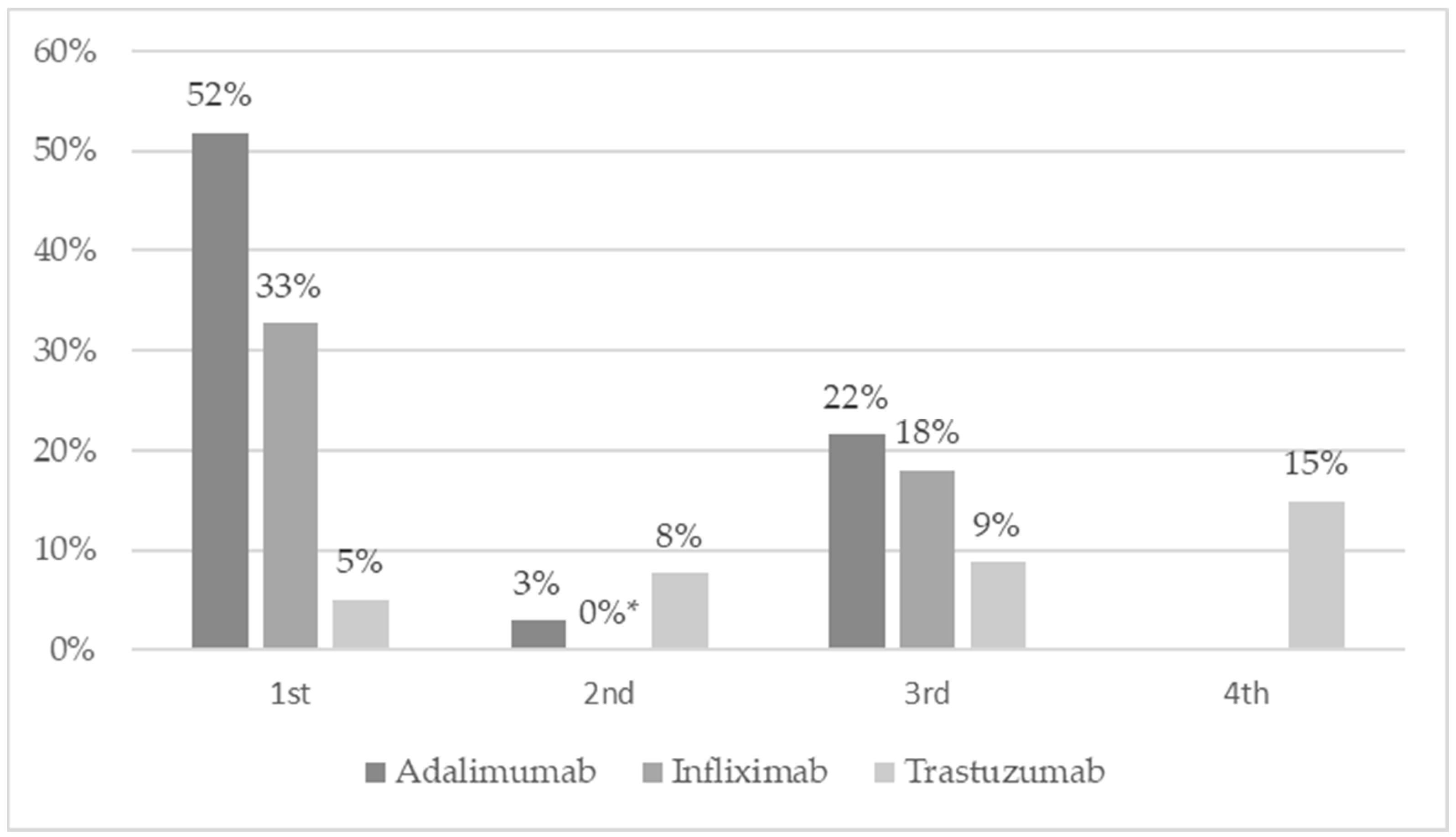
| Current price vs. first price from July 2012 | PLN | x | −29.30 | −30.07 | −35.57 |
| % | x | −53% | −54%s | −64% | |
| Current price vs price from previous reimbursement list | PLN | x | x | −0.77 | −5.50 |
| % | x | x | −3% | −22% | |
| Drug | Remicade | Remsima and Inflectra | Flixabi | Zessly | ||
|---|---|---|---|---|---|---|
| Event | Original Drug | First Introduction of Biosimilars | Second Introduction of Biosimilars | Becomes Limit Basis—Price Drop | Third Introduction of Biosimilars | |
| Date | July 2012 | January 2014 | January 2018 | July 2018 | March 2019 | |
| Price per package | 2261.77 | 1508.22 | 1508.22 | 1048.95 | 850.50 | |
| Price per 1 mg | 22.62 | 15.08 | 15.08 | 10.4895 | 8.51 | |
| Current price vs first price from July 2012 | PLN | x | −7.54 | −0.00 | −12.13 | −14.11 |
| % | x | −33% | −33% | −54% | −62% | |
| Current price vs price from the previous reimbursement list | PLN | x | x | 0.00 | −4.59 | −1.87 |
| % | x | x | −0% * | −30% | −18% | |
| Drug | Herceptin | Kanjinti (2 Doses) | Herzuma and Ontruzant | Ogivri | Trazimera | Ogivri | |
|---|---|---|---|---|---|---|---|
| Event | Original Drug | First Introduction of Biosimilars | Second Instroduction of Biosimilars | Third Instroduction of Biosimilars | Fourth Instroduction of Biosimilars | New Dose | |
| Date | July 2012 | July 2018 | March 2019 | July 2019 | September 2019 | March 20 | |
| Price per package (PLN) | 2889 | 7271.78 | 1678.32 | 1530.9 | 1302.74 | 3319.22 | |
| Price per 1 mg (PLN) | 19.26 | 12.12 | 11.19 | 10.21 | 8.68 | 7.9 | |
| Current price vs first price from July 2012 | PLN | x | −7.14 | −8.07 | −9.05 | −10.58 | −11.36 |
| % | x | −37.07% | −41.91% | −47.01% | −54.91% | −59% | |
| Current price vs price from the previous reimbursement list | PLN | x | −0.64 | −0.93 | −0.98 | −1.52 | −0.78 |
| % | x | −5% | −8% | −9% | −15% | −9% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barszczewska, O.; Piechota, A. The Impact of Introducing Successive Biosimilars on Changes in Prices of Adalimumab, Infliximab, and Trastuzumab—Polish Experiences. Int. J. Environ. Res. Public Health 2021, 18, 6952. https://doi.org/10.3390/ijerph18136952
Barszczewska O, Piechota A. The Impact of Introducing Successive Biosimilars on Changes in Prices of Adalimumab, Infliximab, and Trastuzumab—Polish Experiences. International Journal of Environmental Research and Public Health. 2021; 18(13):6952. https://doi.org/10.3390/ijerph18136952
Chicago/Turabian StyleBarszczewska, Olga, and Anna Piechota. 2021. "The Impact of Introducing Successive Biosimilars on Changes in Prices of Adalimumab, Infliximab, and Trastuzumab—Polish Experiences" International Journal of Environmental Research and Public Health 18, no. 13: 6952. https://doi.org/10.3390/ijerph18136952
APA StyleBarszczewska, O., & Piechota, A. (2021). The Impact of Introducing Successive Biosimilars on Changes in Prices of Adalimumab, Infliximab, and Trastuzumab—Polish Experiences. International Journal of Environmental Research and Public Health, 18(13), 6952. https://doi.org/10.3390/ijerph18136952






