Enhance Access to Pulmonary Rehabilitation with a Structured and Personalized Home-Based Program—reabilitAR: Protocol for Real-World Setting
Abstract
1. Introduction
2. Materials and Methods
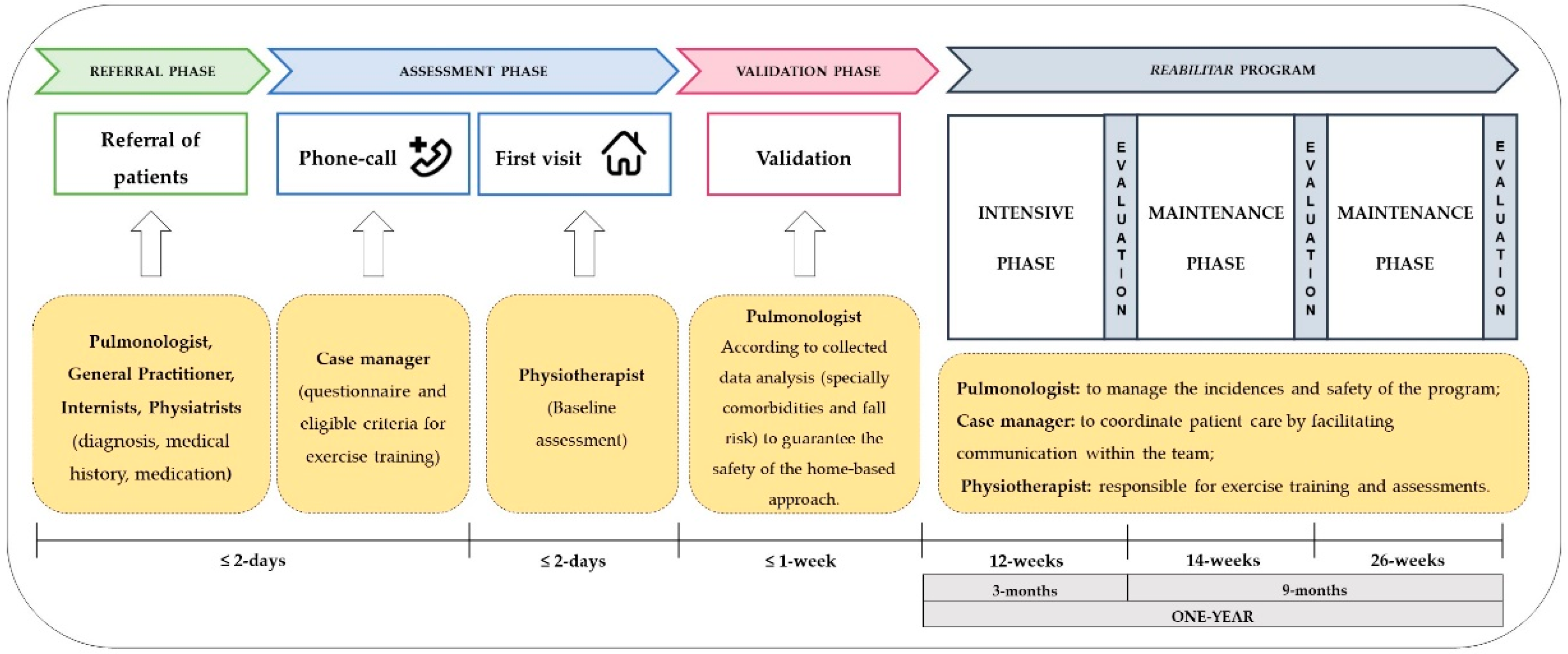
2.1. Design, Setting and Medical Referrals
2.2. Eligibility Criteria
2.3. Data Collection
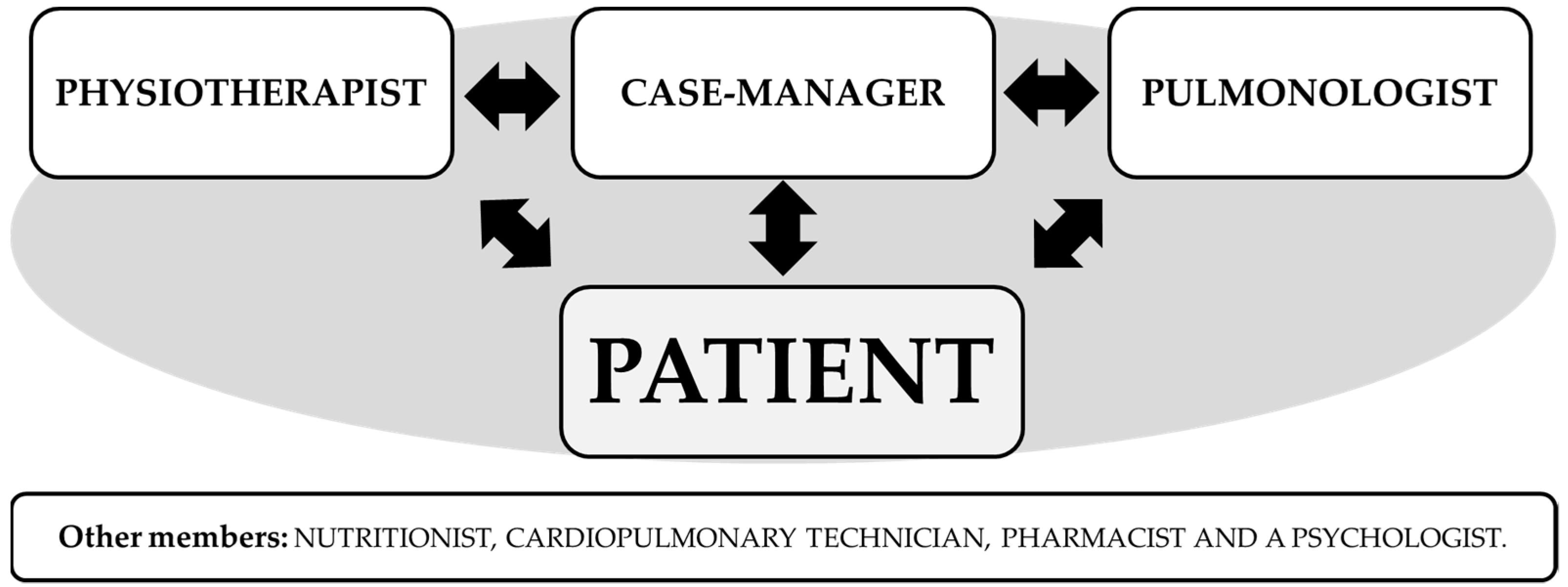
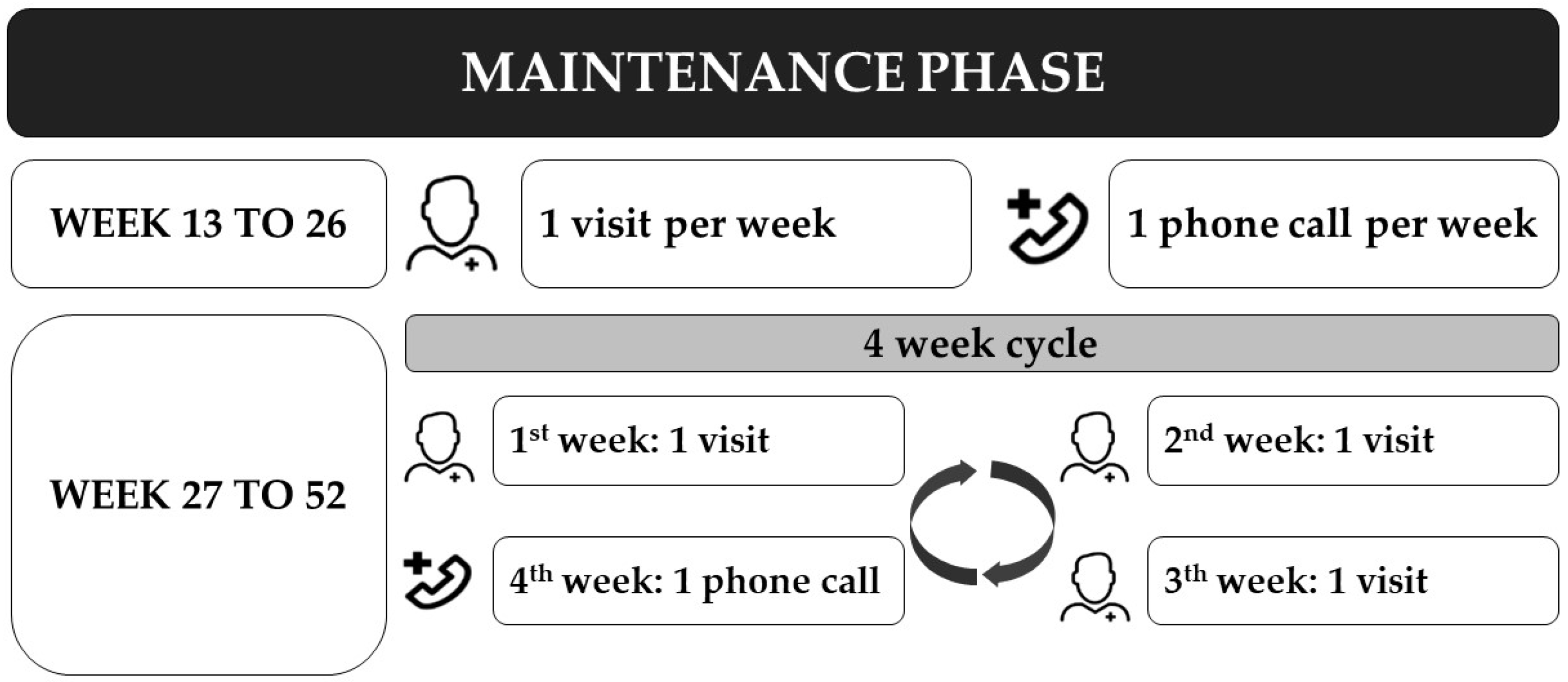
2.4. Intervention
2.5. Statistical Analysis
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Chronic Respiratory Diseases. Available online: https://www.who.int/health-topics/chronic-respiratory-diseases (accessed on 21 September 2020).
- Machado, A.; Marques, A.; Burtin, C. Extra-pulmonary manifestations of COPD and the role of pulmonary rehabilitation: A symptom-centered approach. Expert Rev. Respir. Med. 2021, 15, 131–142. [Google Scholar] [CrossRef]
- Maltais, F.; Decramer, M.; Casaburi, R.; Barreiro, E.; Burelle, Y.; Debigaré, R.; Dekhuijzen, P.N.R.; Franssen, F.; Gayan-Ramirez, G.; Gea, J.; et al. An Official American Thoracic Society/European Respiratory Society Statement: Update on Limb Muscle Dysfunction in Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2014, 189, e15–e62. [Google Scholar] [CrossRef]
- O’Donnell, D.E.; Milne, K.M.; James, M.D.; de Torres, J.P.; Neder, J.A. Dyspnea in COPD: New Mechanistic Insights and Management Implications. Adv. Ther. 2020, 37, 41–60. [Google Scholar] [CrossRef] [PubMed]
- Spruit, M.A.; Vercoulen, J.H.; Sprangers, M.A.G.; Wouters, E.F.M. Fatigue in COPD: An important yet ignored symptom. Lancet Respir. Med. 2017, 5, 542–544. [Google Scholar] [CrossRef]
- Paddison, J.S.; Effing, T.W.; Quinn, S.; Frith, P.A. Fatigue in COPD: Association with functional status and hospitalisations. Eur. Respir. J. 2013, 41, 565–570. [Google Scholar] [CrossRef]
- Agnieszka, L.; Penelope, B.; Andy, J.; Rachel, G. A Comprehensive Literature Review of COPD-Related Fatigue. Curr. Respir. Med. Rev. 2012, 8, 370–382. [Google Scholar] [CrossRef]
- Scioscia, G.; Blanco, I.; Arismendi, E.; Burgos, F.; Gistau, C.; Foschino Barbaro, M.P.; Celli, B.; O’Donnell, D.E.; Agustí, A. Different dyspnoea perception in COPD patients with frequent and infrequent exacerbations. Thorax 2017, 72, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Miravitlles, M.; Ribera, A. Understanding the impact of symptoms on the burden of COPD. Respir. Res. 2017, 18, 67. [Google Scholar] [CrossRef] [PubMed]
- Spruit, M.A.; Singh, S.J.; Garvey, C.; ZuWallack, R.; Nici, L.; Rochester, C.; Hill, K.; Holland, A.E.; Lareau, S.C.; Man, W.D.; et al. An official American Thoracic Society/European Respiratory Society statement: Key concepts and advances in pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2013, 188, e13–e64. [Google Scholar] [CrossRef]
- McCarthy, B.; Casey, D.; Devane, D.; Murphy, K.; Murphy, E.; Lacasse, Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Puhan, M.A.; Gimeno-Santos, E.; Cates, C.J.; Troosters, T. Pulmonary rehabilitation following exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Spruit, M.A.; Clini, E.M. Towards health benefits in chronic respiratory diseases: Pulmonary rehabilitation. Eur. Respir. Rev. 2013, 22, 202–204. [Google Scholar] [CrossRef]
- Desveaux, L.; Janaudis-Ferreira, T.; Goldstein, R.; Brooks, D. An International Comparison of Pulmonary Rehabilitation: A Systematic Review. COPD J. Chronic Obstr. Pulm. Dis. 2015, 12, 144–153. [Google Scholar] [CrossRef]
- Direção-Geral da Saúde. Relatório de Análise da Capacidade Instalada de Reabilitação Respiratória nos Hospitais do Serviço Nacional de Saúde. 2015. Available online: https://www.dgs.pt/paginas-de-sistema/saude-de-a-a-z/programa-nacional-para-as-doencas-respiratorias/relatorios-e-publicacoes (accessed on 21 October 2020).
- Keating, A.; Lee, A.; Holland, A.E. What prevents people with chronic obstructive pulmonary disease from attending pulmonary rehabilitation? A systematic review. Chronic Respir. Dis. 2011, 8, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Rochester, C.L.; Vogiatzis, I.; Holland, A.E.; Lareau, S.C.; Marciniuk, D.D.; Puhan, M.A.; Spruit, M.A.; Masefield, S.; Casaburi, R.; Clini, E.M.; et al. An Official American Thoracic Society/European Respiratory Society Policy Statement: Enhancing Implementation, Use, and Delivery of Pulmonary Rehabilitation. Am. J. Respir. Crit. Care Med. 2015, 192, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Maltais, F.; Bourbeau, J.; Shapiro, S.; Lacasse, Y.; Perrault, H.; Baltzan, M.; Hernandez, P.; Rouleau, M.; Julien, M.; Parenteau, S.; et al. Effects of home-based pulmonary rehabilitation in patients with chronic obstructive pulmonary disease: A randomized trial. Ann. Intern. Med. 2008, 149, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.E.; Mahal, A.; Hill, C.J.; Lee, A.L.; Burge, A.T.; Cox, N.S.; Moore, R.; Nicolson, C.; O’Halloran, P.; Lahham, A.; et al. Home-based rehabilitation for COPD using minimal resources: A randomised, controlled equivalence trial. Thorax 2017, 72, 57–65. [Google Scholar] [CrossRef]
- Vestbo, J.; Hurd, S.S.; Agustí, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Barnes, P.J.; Fabbri, L.M.; Martinez, F.J.; Nishimura, M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef]
- Vogiatzis, I.; Rochester, C.L.; Spruit, M.A.; Troosters, T.; Clini, E.M. Increasing implementation and delivery of pulmonary rehabilitation: Key messages from the new ATS/ERS policy statement. Eur. Respir. J. 2016, 47, 1336–1341. [Google Scholar] [CrossRef]
- ACSM. American College of Sports Medicine’s Guidelines for Exercise Testing and Prescription; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Fonseca, F.R.; Karloh, M.; Araujo, C.L.P.d.; Reis, C.M.d.; Mayer, A.F. Validation of a bioelectrical impedance analysis system for body composition assessment in patients with COPD. J. Bras. Pneumol. 2018, 44, 315–320. [Google Scholar] [CrossRef]
- World Health Organization. Waist Circumference and Waist–Hip Ratio: Report of a WHO Expert Consultation. Available online: https://www.who.int/publications/i/item/9789241501491 (accessed on 21 October 2020).
- Crisafulli, E.; Clini, E.M. Measures of dyspnea in pulmonary rehabilitation. Multidiscip. Respir. Med. 2010, 5, 202. [Google Scholar] [CrossRef]
- Jones, P.W.; Harding, G.; Berry, P.; Wiklund, I.; Chen, W.H.; Kline Leidy, N. Development and first validation of the COPD Assessment Test. Eur. Respir. J. 2009, 34, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.W.; Hogg, L.; Nolan, J.; Jefford, H.; Grant, A.; Lord, V.M.; Falzon, C.; Garrod, R.; Lee, C.; Polkey, M.I.; et al. The COPD assessment test (CAT): Response to pulmonary rehabilitation. A multicentre, prospective study. Thorax 2011, 66, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Pais-Ribeiro, J.; Silva, I.; Ferreira, T.; Martins, A.; Meneses, R.; Baltar, M. Validation study of a Portuguese version of the Hospital Anxiety and Depression Scale. Psychol. Health Med. 2007, 12, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Garrod, R.; Bestall, J.C.; Paul, E.A.; Wedzicha, J.A.; Jones, P.W. Development and validation of a standardized measure of activity of daily living in patients with severe COPD: The London Chest Activity of Daily Living scale (LCADL). Respir. Med. 2000, 94, 589–596. [Google Scholar] [CrossRef]
- Vaidya, T.; de Bisschop, C.; Beaumont, M.; Ouksel, H.; Jean, V.; Dessables, F.; Chambellan, A. Is the 1-min sit-to-stand test a good tool for the evaluation of the impact of pulmonary rehabilitation? Determination of the minimal important difference in COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 2609–2616. [Google Scholar] [CrossRef] [PubMed]
- Watz, H.; Pitta, F.; Rochester, C.L.; Garcia-Aymerich, J.; ZuWallack, R.; Troosters, T.; Vaes, A.W.; Puhan, M.A.; Jehn, M.; Polkey, M.I.; et al. An official European Respiratory Society statement on physical activity in COPD. Eur. Respir. J. 2014, 44, 1521–1537. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Guerreiro, M.; Silva, A.P.; Botelho, M.A.; Leitão, O.; Castro-Caldas, A.; Garcia, C. Adaptação à população portuguesa da tradução do Mini Mental State Examination (MMSE). Rev. Port. Neurol. 1994, 1, 9–10. [Google Scholar]
- Chou, C.Y.; Chien, C.W.; Hsueh, I.P.; Sheu, C.F.; Wang, C.H.; Hsieh, C.L. Developing a short form of the Berg Balance Scale for people with stroke. Phys. Ther. 2006, 86, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.O.; Wood-Dauphinee, S.L.; Williams, J.I.; Maki, B. Measuring balance in the elderly: Validation of an instrument. Can. J. Public Health 1992, 83 (Suppl. 2), S7–S11. [Google Scholar]
- Bui, K.L.; Nyberg, A.; Maltais, F.; Saey, D. Functional Tests in Chronic Obstructive Pulmonary Disease, Part 1: Clinical Relevance and Links to the International Classification of Functioning, Disability, and Health. Ann. Am. Thorac. Soc. 2017, 14, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, M.K.; Janaudis-Ferreira, T.; Parreira, V.; Romano, J.M.; Woon, L.; Goldstein, R.S.; Brooks, D. A randomized controlled trial of balance training during pulmonary rehabilitation for individuals with COPD. Chest 2013, 144, 1803–1810. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.L.; Holland, A.E. Time to adapt exercise training regimens in pulmonary rehabilitation—A review of the literature. Int. J. Chron. Obstruct. Pulm. Dis. 2014, 9, 1275–1288. [Google Scholar] [CrossRef]
- Fischer, M.J.; Scharloo, M.; Abbink, J.J.; van’t Hul, A.J.; van Ranst, D.; Rudolphus, A.; Weinman, J.; Rabe, K.F.; Kaptein, A.A. Drop-out and attendance in pulmonary rehabilitation: The role of clinical and psychosocial variables. Respir. Med. 2009, 103, 1564–1571. [Google Scholar] [CrossRef]
- Steurer-Stey, C.; Dalla Lana, K.; Braun, J.; ter Riet, G.; Puhan, M.A. Effects of the “Living well with COPD” intervention in primary care: A comparative study. Eur. Respir. J. 2018, 51, 1701375. [Google Scholar] [CrossRef] [PubMed]
- Yohannes, A.M.; Willgoss, T.G.; Baldwin, R.C.; Connolly, M.J. Depression and anxiety in chronic heart failure and chronic obstructive pulmonary disease: Prevalence, relevance, clinical implications and management principles. Int. J. Geriatr. Psychiatry 2010, 25, 1209–1221. [Google Scholar] [CrossRef] [PubMed]
- Vu, G.V.; Ha, G.H.; Nguyen, C.T.; Vu, G.T.; Pham, H.Q.; Latkin, C.A.; Tran, B.X.; Ho, R.C.M.; Ho, C.S.H. Interventions to Improve the Quality of Life of Patients with Chronic Obstructive Pulmonary Disease: A Global Mapping During 1990–2018. Int. J. Environ. Res. Public Health 2020, 17, 3089. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, T.; Chambellan, A.; de Bisschop, C. Sit-to-stand tests for COPD: A literature review. Respir. Med. 2017, 128, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.E.; Spruit, M.A.; Troosters, T.; Puhan, M.A.; Pepin, V.; Saey, D.; McCormack, M.C.; Carlin, B.W.; Sciurba, F.C.; Pitta, F.; et al. An official European Respiratory Society/American Thoracic Society technical standard: Field walking tests in chronic respiratory disease. Eur. Respir. J. 2014, 44, 1428–1446. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.L.; Neves, I.; Luís, G.; Camilo, Z.; Cabrita, B.; Dias, S.; Ferreira, J.; Simão, P. Is the 1-Minute Sit-To-Stand Test a Good Tool to Evaluate Exertional Oxygen Desaturation in Chronic Obstructive Pulmonary Disease? Diagnostics 2021, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Ozalevli, S.; Ozden, A.; Itil, O.; Akkoclu, A. Comparison of the Sit-to-Stand Test with 6min walk test in patients with chronic obstructive pulmonary disease. Respir. Med. 2007, 101, 286–293. [Google Scholar] [CrossRef]
- Crook, S.; Büsching, G.; Schultz, K.; Lehbert, N.; Jelusic, D.; Keusch, S.; Wittmann, M.; Schuler, M.; Radtke, T.; Frey, M.; et al. A multicentre validation of the 1-min sit-to-stand test in patients with COPD. Eur. Respir. J. 2017, 49, 1601871. [Google Scholar] [CrossRef] [PubMed]
- Gephine, S.; Bergeron, S.; Tremblay Labrecque, P.-F.; Mucci, P.; Saey, D.; Maltais, F. Cardiorespiratory Response during the 1-min Sit-to-Stand Test in Chronic Obstructive Pulmonary Disease. Med. Sci. Sports Exerc. 2020, 52, 1441–1448. [Google Scholar] [CrossRef]
- Bui, K.-L.; Nyberg, A.; Maltais, F.; Saey, D. Functional Tests in Chronic Obstructive Pulmonary Disease, Part 2: Measurement Properties. Ann. Am. Thorac. Soc. 2017, 14, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Franssen, F.M.E.; Rochester, C.L. Comorbidities in patients with COPD and pulmonary rehabilitation: Do they matter? Eur. Respir. Rev. 2014, 23, 131–141. [Google Scholar] [CrossRef]
- Santos, C.D.; das Neves, R.C.; Ribeiro, R.M.; Caneiras, C.; Rodrigues, F.; Spruit, M.A.; Bárbara, C. Novel Input for Designing Patient-Tailored Pulmonary Rehabilitation: Telemonitoring Physical Activity as a Vital Sign—SMARTREAB Study. J. Clin. Med. 2020, 9, 2450. [Google Scholar] [CrossRef]
- Sánchez Castillo, S.; Smith, L.; Díaz Suárez, A.; López Sánchez, G.F. Associations between Physical Activity and Comorbidities in People with COPD Residing in Spain: A Cross-Sectional Analysis. Int. J. Environ. Res. Public Health 2020, 17, 594. [Google Scholar] [CrossRef]
- Meys, R.; Stoffels, A.A.F.; Houben-Wilke, S.; Janssen, D.J.A.; Burtin, C.; van Hees, H.W.H.; Franssen, F.M.E.; van den Borst, B.; Wouters, E.F.M.; Spruit, M.A.; et al. Association between patient-reported outcomes and exercise test outcomes in patients with COPD before and after pulmonary rehabilitation. Health Qual. Life Outcomes 2020, 18, 300. [Google Scholar] [CrossRef]
- Diário da República. Saúde: Despacho nº 6300/2016. Available online: https://dre.pt/home/-/dre/74425578/details/ (accessed on 12 December 2020).
- Direção Geral da Saúde. Orientação Técnica—Programas de Reabilitação Respiratória nos Cuidados de Saúde Primários. Available online: https://www.dgs.pt/directrizes-da-dgs/orientacoes-e-circulares-informativas/orientacao-n-0142019-de-07082019.aspx (accessed on 12 December 2020).

| Timepoint | Baseline T0 | Post 12 Weeks T1 | Post 26 Weeks T2 | Post 52 Weeks T3 |
|---|---|---|---|---|
| Intervention | Home-Based Pulmonary Rehabilitation—reabilitAR | |||
| Outcomes | ||||
| Anthropometric measures | X | X | X | X |
| Impact of the disease | X | X | X | X |
| Emotional status | X | X | X | X |
| Balance and fall risk | X | X | X | X |
| Cognitive function | X | |||
| Functional capacity | X | X | X | X |
| Number of exacerbations and hospital admissions | X | X | X | X |
| Level of physical activity | X | X | X | X |
| Self-Management Educational Intervention | ||
|---|---|---|
| Visit | Module | Themes |
| 1 | Integrating an Exercise Program into Your Life | Exercise program, pursed-lip breathing technique and scale of perceived exertion |
| 2 | Being Healthy with COPD—Preventing your Symptoms and Taking Your Medication | Anatomy, physiology and cause of COPD and factors that can make your symptoms worse |
| 3 | Medications and inhalation techniques | |
| 4 | Managing your Breathing and Saving Your Energy | Understanding how breathing works, reducing shortness of breath (breathing techniques and body positions) |
| 5 | Clearing your airways (coughing tecniques, active cycle of breathing technique and devices) | |
| 6 | Clearing your airways (devices) | |
| 7 | Applying energy conservation principles | |
| 8 | Review of previous themes (doubts) and reinforce the importance of medications and breathing techniques/body positions for shortness of breath | |
| 9 | Managing Your Stress & Anxiety | Identifying the stressors in your life and understanding your reaction, breaking the anxiety-breathlessness cycle |
| 10 | Associate the topics of visit 4 and visit 9 | |
| 11 | Managing Your Stress & Anxiety | Integrating relaxation exercises and applying rules to live a healthier life |
| 12 | Integrating a Healthy Diet into Your Life | The link between COPD and what you eat, maintaining a healthy weight and healthy and balanced eating |
| 13 | Keeping a Healthy and Fulfilling Lifestyle | Being healthy, quitting smoking (optional), sleeping better, satisfying sex life, leisure activities and travelling |
| 14 | Review of previous themes (doubts) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernard, S.; Vilarinho, R.; Pinto, I.; Cantante, R.; Coxo, R.; Fonseca, R.; Mayoralas-Alises, S.; Diaz-Lobato, S.; Carvalho, J.; Esteves, C.; et al. Enhance Access to Pulmonary Rehabilitation with a Structured and Personalized Home-Based Program—reabilitAR: Protocol for Real-World Setting. Int. J. Environ. Res. Public Health 2021, 18, 6132. https://doi.org/10.3390/ijerph18116132
Bernard S, Vilarinho R, Pinto I, Cantante R, Coxo R, Fonseca R, Mayoralas-Alises S, Diaz-Lobato S, Carvalho J, Esteves C, et al. Enhance Access to Pulmonary Rehabilitation with a Structured and Personalized Home-Based Program—reabilitAR: Protocol for Real-World Setting. International Journal of Environmental Research and Public Health. 2021; 18(11):6132. https://doi.org/10.3390/ijerph18116132
Chicago/Turabian StyleBernard, Sarah, Rui Vilarinho, Inês Pinto, Rosa Cantante, Ricardo Coxo, Rosa Fonseca, Sagrario Mayoralas-Alises, Salvador Diaz-Lobato, João Carvalho, Cátia Esteves, and et al. 2021. "Enhance Access to Pulmonary Rehabilitation with a Structured and Personalized Home-Based Program—reabilitAR: Protocol for Real-World Setting" International Journal of Environmental Research and Public Health 18, no. 11: 6132. https://doi.org/10.3390/ijerph18116132
APA StyleBernard, S., Vilarinho, R., Pinto, I., Cantante, R., Coxo, R., Fonseca, R., Mayoralas-Alises, S., Diaz-Lobato, S., Carvalho, J., Esteves, C., & Caneiras, C. (2021). Enhance Access to Pulmonary Rehabilitation with a Structured and Personalized Home-Based Program—reabilitAR: Protocol for Real-World Setting. International Journal of Environmental Research and Public Health, 18(11), 6132. https://doi.org/10.3390/ijerph18116132







