Pilot Study to Assess the Feasibility of a Mobile Unit for Remote Cognitive Screening of Isolated Elderly in Rural Areas
Abstract
1. Introduction
2. Materials and Methods
2.1. Equipment
- -
- PPG Sensor: Measures Blood Volume Pulse (BVP), from which heart rate variability can be derived.
- -
- Three-axis Accelerometer: Captures motion-based activity.
- -
- EDA Sensor (GSR Sensor): Measures the constantly fluctuating changes in certain electrical properties of the skin.
- -
- Infrared Thermopile: Reads peripheral skin temperature.
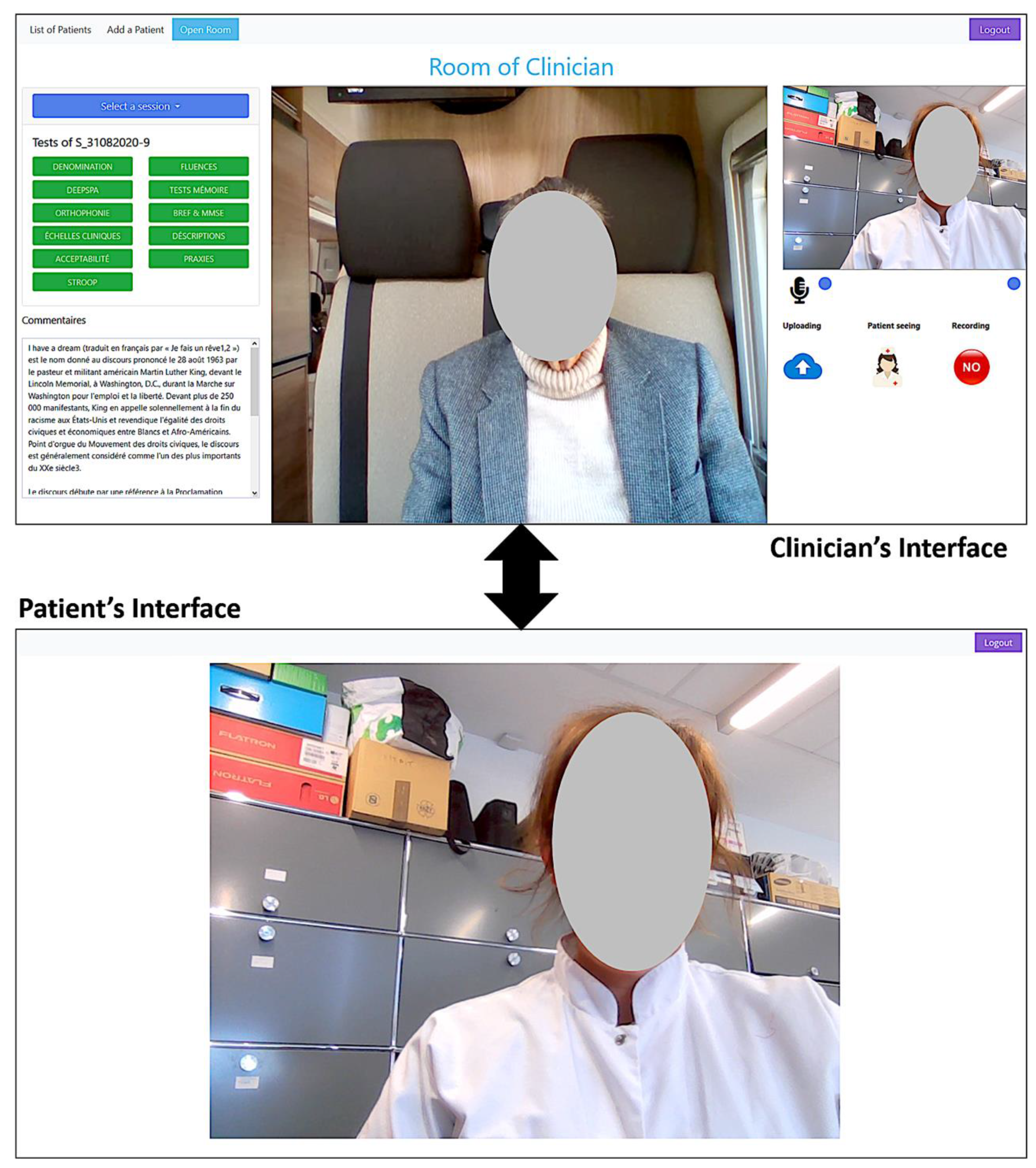
2.2. The Video Conference (VC) System
- -
- Clinical interviewing with the patient
- ○
- POption to video and/or audio record the conversation
- -
- Performing the clinical tests:
- ○
- More than 20 known and widely used neuropsychological tests and clinical scales are already implemented and validated (in French) such as: Mini Mental State Examination (MMSE) ([22]), Free Cued Selective Recall Task (FCSRT), such as the RL/RI 16 items [23] and the five-word screening test” test [24], Semantic and Phonemic verbal fluencies, denomination tasks, description tasks, and neuropsychiatric scales, such as the Geriatric Depression Scale [25] and Apathy Inventory [26].
- ○
- Content sharing with the study participant: image, video, and text.
- ○
- Reporting and saving test scores.
- ○
- Video and/or audio recording of the test session (patient side).
- -
- During a clinical test:
- ○
- The patient can remain passive and does not have to interact with the system (everything is done with verbal communication).
- ○
- The patient can interact with the system if they are comfortable with the technology: click on buttons, check boxes, or select an image.
2.3. Participants
2.4. Procedure/Protocol
2.5. Data Collection and Availability
2.6. Acceptability Evaluation
2.7. Data Analysis
3. Results
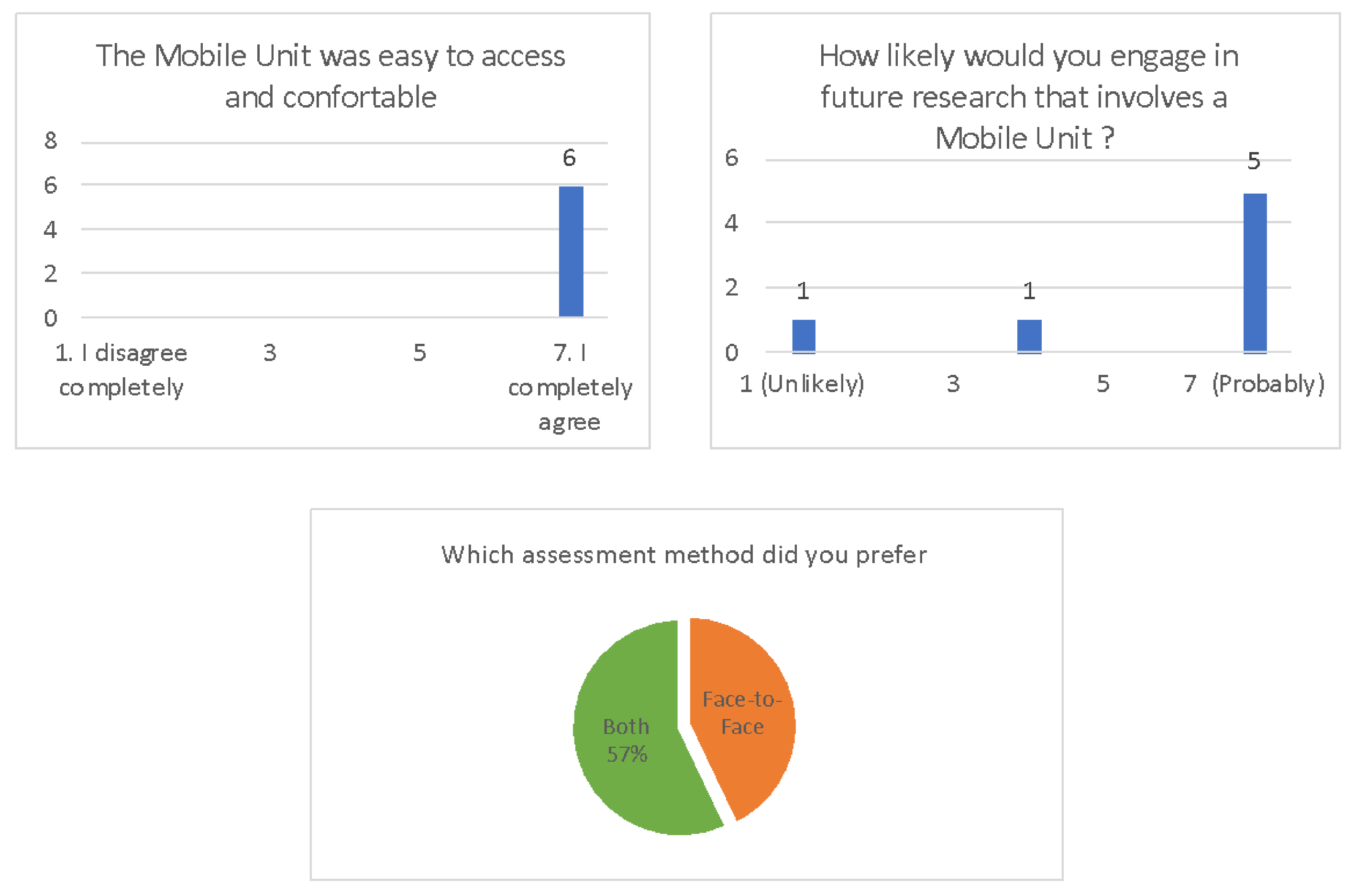
Acceptability Surveys
- -
- At the beginning of the study, next to the assistant, an engineer was present to make sure that the technical equipment was used properly. However, since it was so easy to use, for the rest of study, no further assistance was required.
- -
- In the MU, the laptop’s built-in camera had a narrow angle, which limited the view of the participants’ upper body.
- -
- As these areas can have limited internet access, the VC system was not able to video record one subject during some tests. The system was designed so that video streaming and connection to the VC system are prioritized over video recording.
- -
- One of the participants’ house location did not allow internet access to 4G. The MU was then moved, with the participant in it, to the nearest village hall, allowing a 3G connection.
4. Discussion
4.1. Principal Findings
4.2. Advantages and Challenges of a VC System within an MU
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hantke, N.C.; Gould, C. Examining Older Adult Cognitive Status in the Time of COVID-19. J. Am. Geriatr. Soc. 2020, 68, 1387–1389. [Google Scholar] [CrossRef] [PubMed]
- Chevillard, G.; Lucas-Gabrielli, V.; Mousquès, J. “Medical deserts” in France: Current state of research and future directions. Espac. Geogr. 2018, 47, 362–380. [Google Scholar] [CrossRef]
- Legendre, B. En 2018, Les Territoires Sous-Dotés En Médecins Généralistes Concernent Près De 6% De La Population. Published 2020. Available online: https://drees.solidarites-sante.gouv.fr/publications/etudes-et-resultats/en-2018-les-territoires-sous-dotes-en-medecins-generalistes (accessed on 23 March 2021).
- Helmer, C.; Berr, C.; Dartigues, J.F. Epidémiologie des démences et facteurs de risque modifiables. In Démences; Doin Editions: France, 2015; Volume 33. [Google Scholar]
- Defontaines, B.; Denolle, S.; Helene, L.; Bonnet, N. Diagnostic de la maladie d’Alzheimer: Géographie de l’offre de soins actuelle et modélisation/projection des flux en 2030. Rev. Neurol. 2016, 17, A10. [Google Scholar] [CrossRef]
- Martin, S.; Kelly, S.; Khan, A.; Cullum, S.; Dening, T.; Rait, G.; Fox, C.; Katona, C.; Cosco, T.; Brayne, C.; et al. Attitudes and preferences towards screening for dementia: A systematic review of the literature. BMC Geriatr. 2015, 15, 66. [Google Scholar] [CrossRef]
- De Roeck, E.E.; De Deyn, P.P.; Dierckx, E.; Engelborghs, S. Brief cognitive screening instruments for early detection of Alzheimer’s disease: A systematic review. Alzheimer’s Res. Ther. 2019, 11, 21. [Google Scholar] [CrossRef]
- Prince, M.; Acosta, D.; Albanese, E.; Arizaga, R.; Ferri, C.P.; Guerra, M.; Huang, Y.; Jacob, K.S.; Jimenez-Velazquez, I.Z.; Rodriguez, J.L.; et al. Ageing and dementia in low and middle income countries—Using research to engage with public and policy makers. Int. Rev. Psychiatry 2008, 20, 332–343. [Google Scholar] [CrossRef][Green Version]
- Robert, P.; Koening, A.; Andrieu, S.; Bremond, F.; Chemin, I.; Chung, P.C.; Dartigues, J.F.; Dubois, B.; Feutren, G.; Guillemaud, R.; et al. Recommendations for ICT use in Alzheimer’s Disease assessment: Monaco CTAD expert meeting. J. Nutr. Health Aging 2013, 17, 653–660. [Google Scholar] [CrossRef]
- Kim, H.; Jhoo, J.H.; Jang, J.W. The effect of telemedicine on cognitive decline in patients with dementia. J. Telemed. Telecare 2017, 23, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Timpano, F.; Pirrotta, F.; Bonanno, L.; Marino, S.; Marra, A.; Bramanti, P.; Lanzafame, P. Videoconference-based mini mental state examination: A validation study. Telemed. e-Health 2013, 19, 931–937. [Google Scholar] [CrossRef]
- Carotenuto, A.; Rea, R.; Traini, E.; Ricci, G.; Fasanaro, A.M.; Amenta, F. Cognitive assessment of patients with Alzheimer’s disease by telemedicine: Pilot study. J. Med. Internet Res. 2018, 20, e8097. [Google Scholar] [CrossRef] [PubMed]
- Martin-Khan, M.; Flicker, L.; Wootton, R.; Loh, P.K.; Edwards, H.; Varghese, P.; Byrne, G.J.; Klein, K.; Gray, L.C. The Diagnostic Accuracy of Telegeriatrics for the Diagnosis of Dementia via Video Conferencing. J. Am. Med. Dir. Assoc. 2012, 13, 487.e19–487.e24. [Google Scholar] [CrossRef]
- Loh, P.K.; Donaldson, M.; Flicker, L.; Maher, S.; Goldswain, P. Development of a telemedicine protocol for the diagnosis of Alzheimer’s disease. J. Telemed. Telecare 2007, 13, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Hilty, D.M.; Ferrer, D.C.; Parish, M.B.; Johnston, B.; Callahan, E.J.; Yellowlees, P.M. The effectiveness of telemental health: A 2013 review. Telemed. e-Health 2013, 19, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Dario, C.; Luisotto, E.; Dal Pozzo, E.; Mancin, S.; Aletras, V.; Newman, S.; Gubian, L.; Saccavini, C. Assessment of patients’ perception of telemedicine services using the service user technology acceptability questionnaire. Int. J. Integr. Care 2016, 16, 13. [Google Scholar] [CrossRef]
- Brearly, T.W.; Shura, R.D.; Martindale, S.L.; Lazowski, R.A.; Luxton, D.D.; Shenal, B.V.; Rowland, J.A. Neuropsychological Test Administration by Videoconference: A Systematic Review and Meta-Analysis. Neuropsychol. Rev. 2017, 27, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Attipoe-Dorcoo, S.; Delgado, R.; Gupta, A.; Bennet, J.; Oriol, N.E.; Jain, S.H. Mobile health clinic model in the COVID-19 pandemic: Lessons learned and opportunities for policy changes and innovation. Int. J. Equity Health 2020, 19, 73. [Google Scholar] [CrossRef]
- Fils-Aimé, J.R.; Grelotti, D.J.; Thérosmé, T.; Kaiser, B.N.; Raviola, G.; Alcindor, Y.; Severe, J.; Affricot, E.; Boyd, K.; Legha, R.; et al. A mobile clinic approach to the delivery of community-based mental health services in rural Haiti. Kidd SA, ed. PLoS ONE 2018, 13, e0199313. [Google Scholar] [CrossRef]
- Greenwald, Z.R.; El-Zein, M.; Bouten, S.; Ensha, H.; Vazquez, F.L.; Franco, E.L. Mobile screening units for the early detection of cancer: A systematic review. Cancer Epidemiol. Biomark. Prev. 2017, 26, 1679–1694. [Google Scholar] [CrossRef]
- Nelson, R.; Bhattacharya, S.D.; Hart, S. Combined in-person and tele-delivered mobile school clinic: A novel approach for improving access to healthcare during school hours. J. Telemed. Telecare 2020, 1357633X2091749. [Google Scholar] [CrossRef]
- Folstein, M.F.; Robins, L.N.; Helzer, J.E. The Mini-Mental State Examination. Arch. Gen. Psychiatry 1983, 40, 812. [Google Scholar] [CrossRef]
- Van der Linden, M.; Coyette, F.; Poitrenaud, J.; Kalafat, M.; Calicis, F.; Wyns, C. L’épreuve de Rappel Libre/Rappel Indicé à 16 Items (RL/RI 16) BT-L’évaluation Des Troubles de La Mémoire: Présentation de Quatre Tests de Mémoire Épisodique (Avec Leur Étalonnage); Solal: Marseille, France, 2004; pp. 25–47. [Google Scholar]
- Cowppli-Bony, P.; Fabrigoule, C.; Letenneur, L.; Ritchie, K.; Alpérovitch, A.; Dartigues, J.F.; Dubois, B. Validity of the five-word screening test for Alzheimer’s disease in a population based study. Rev. Neurol. 2005, 16, 1205–1212. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982, 17, 37–49. [Google Scholar] [CrossRef]
- Robert, P.H.; Clairet, S.; Benoit, M.; Koutaich, J.; Bertogliati, C.; Tible, O.; Caci, H.; Borg, M.; Brocker, P.; Bedoucha, P. The Apathy Inventory: Assessment of apathy and awareness in Alzheimer’s disease, Parkinson’s disease and mild cognitive impairment. Int. J. Geriatr. Psychiatry 2002, 17, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Brooke, J. SUS: A “Quick and Dirty’s Usability”. Usability Eval. Ind. 1996. [Google Scholar] [CrossRef]
- Menachemi, N.; Burke, D.E.; Ayers, D.J. Factors affecting the adoption of telemedicine—A multiple adopter perspective. J. Med. Syst. 2004, 28, 617–632. [Google Scholar] [CrossRef] [PubMed]
- Geddes, M.R.; O’Connell, M.E.; Fisk, J.D.; Gauthier, S.; Camicioli, R.; Ismail, Z. Remote cognitive and behavioral assessment: Report of the Alzheimer Society of Canada Task Force on dementia care best practices for COVID-19. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2020, 12, e12111. [Google Scholar] [CrossRef] [PubMed]
- Brown-Connolly, N.E.; Concha, J.B.; English, J. Mobile health is worth it! Economic benefit and impact on health of a population-based mobile screening program in new Mexico. Telemed. e-Health 2014, 20, 18–23. [Google Scholar] [CrossRef]
- Malone, N.C.; Williams, M.M.; Smith Fawzi, M.C.; Bennet, J.; Hill, C.; Katz, J.N.; Oriol, N.E. Mobile health clinics in the United States. Int. J. Equity Health 2020, 19, 40. [Google Scholar] [CrossRef]


| n | % | ||||
|---|---|---|---|---|---|
| Sex | |||||
| Females | 4 | 50 | |||
| Males | 4 | 50 | |||
| Education | |||||
| Primary | 2 | 25 | |||
| Secondary | 3 | 38 | |||
| High | 3 | 38 | |||
| Age, years | mean | sd | min | max | |
| 76.7 | 6.12 | 69 | 86 |
| N = 8 | Mobile Unit | Face to Face | |||
|---|---|---|---|---|---|
| Global Functioning | MMSE | 27.38 | (3.20) | 26.88 | (3.31) |
| Executive functions | FAB | 12.50 | (0.93) | 12.63 | (1.92) |
| Memory (5 words) | Total recall | 9.88 | (0.35) | 9.88 | (0.35) |
| Free recall | 8.13 | (1.73) | 8.88 | (0.99) | |
| Delayed recall | 4.88 | (0.35) | 4.88 | (0.35) | |
| Verbal Fluency | SVF | 21.88 | (11.04) | 19.75 | (2.60) |
| PVF | 20.25 | (8.12) | 18.00 | (7.50) | |
| Working memory (Digit Span) | Onwards | 5.63 | (1.51) | 5.38 | (1.30) |
| Backwards | 4.63 | (1.19) | 4.00 | (0.76) | |
| Tests | Wilcoxon Paired Sample | ICC | ||
|---|---|---|---|---|
| p-Value | Coefficient | Lower Bound | Upper Bound | |
| MMSE | 0.279 | 0.964 *** | 0.835 | 0.993 |
| FAB | 0.863 | 0.297 | −4.697 | 0.869 |
| 5 words—Total recall | 1 | −0.4 | −33.787 | 0.758 |
| SVF | 0.674 | 0.117 | −5.598 | 0.834 |
| PVF | 0.115 | 0.924 *** | 0.630 | 0.985 |
| DS Onwards | 0.414 | 0.894 ** | 0.505 | 0.979 |
| DS Backwards | 0.236 | 0 | −2.937 | 0.788 |
| Face-to-Face | Mobile Unit | |||
|---|---|---|---|---|
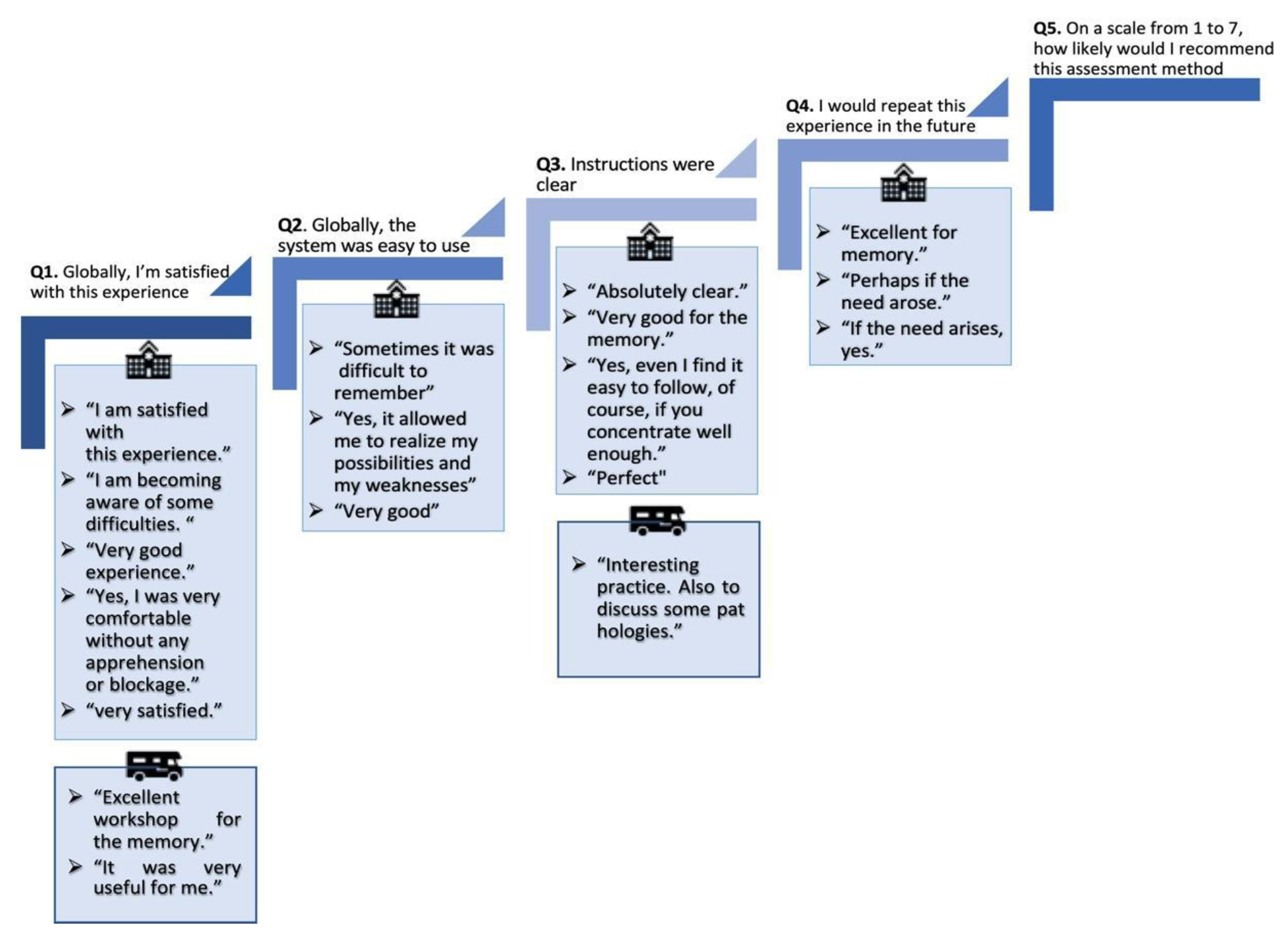
| Q1. Globally, I’m satisfied with this experience | 7.0 | (0.0) | 7.0 | (0.0) |
| Q2. Globally, the system was easy to use | 6.6 | (1.1) | 7.0 | (0.0) |
| Q3. Instructions were clear | 6.4 | (1.5) | 7.0 | (0.0) |
| Q4. I would repeat this experience in the future | 6.6 | (1.1) | 6.6 | (1.1) |
| Q5. On a scale from 1 to 7, how likely would I recommend this assessment method? | 6.7 | (0.6) | 7.2 | (0.4) |
| Q6. Globally, my stress level was | 4.8 | (1.0) | 5.6 | (1.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeghari, R.; Guerchouche, R.; Tran Duc, M.; Bremond, F.; Lemoine, M.P.; Bultingaire, V.; Langel, K.; De Groote, Z.; Kuhn, F.; Martin, E.; et al. Pilot Study to Assess the Feasibility of a Mobile Unit for Remote Cognitive Screening of Isolated Elderly in Rural Areas. Int. J. Environ. Res. Public Health 2021, 18, 6108. https://doi.org/10.3390/ijerph18116108
Zeghari R, Guerchouche R, Tran Duc M, Bremond F, Lemoine MP, Bultingaire V, Langel K, De Groote Z, Kuhn F, Martin E, et al. Pilot Study to Assess the Feasibility of a Mobile Unit for Remote Cognitive Screening of Isolated Elderly in Rural Areas. International Journal of Environmental Research and Public Health. 2021; 18(11):6108. https://doi.org/10.3390/ijerph18116108
Chicago/Turabian StyleZeghari, Radia, Rachid Guerchouche, Minh Tran Duc, François Bremond, Maria Pascale Lemoine, Vincent Bultingaire, Kai Langel, Zeger De Groote, Francis Kuhn, Emmanuelle Martin, and et al. 2021. "Pilot Study to Assess the Feasibility of a Mobile Unit for Remote Cognitive Screening of Isolated Elderly in Rural Areas" International Journal of Environmental Research and Public Health 18, no. 11: 6108. https://doi.org/10.3390/ijerph18116108
APA StyleZeghari, R., Guerchouche, R., Tran Duc, M., Bremond, F., Lemoine, M. P., Bultingaire, V., Langel, K., De Groote, Z., Kuhn, F., Martin, E., Robert, P., & König, A. (2021). Pilot Study to Assess the Feasibility of a Mobile Unit for Remote Cognitive Screening of Isolated Elderly in Rural Areas. International Journal of Environmental Research and Public Health, 18(11), 6108. https://doi.org/10.3390/ijerph18116108








