Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review
Abstract
1. Introduction
- (1)
- action potential transmission along the sarcolemma;
- (2)
- excitation–contraction coupling;
- (3)
- actin-myosin interaction.
2. Methods
2.1. Literature Search Strategies
2.2. Inclusion and Exclusion Criteria
2.3. Study Selection
2.4. Outcome Measures
2.5. Quality Assessment of the Experiments
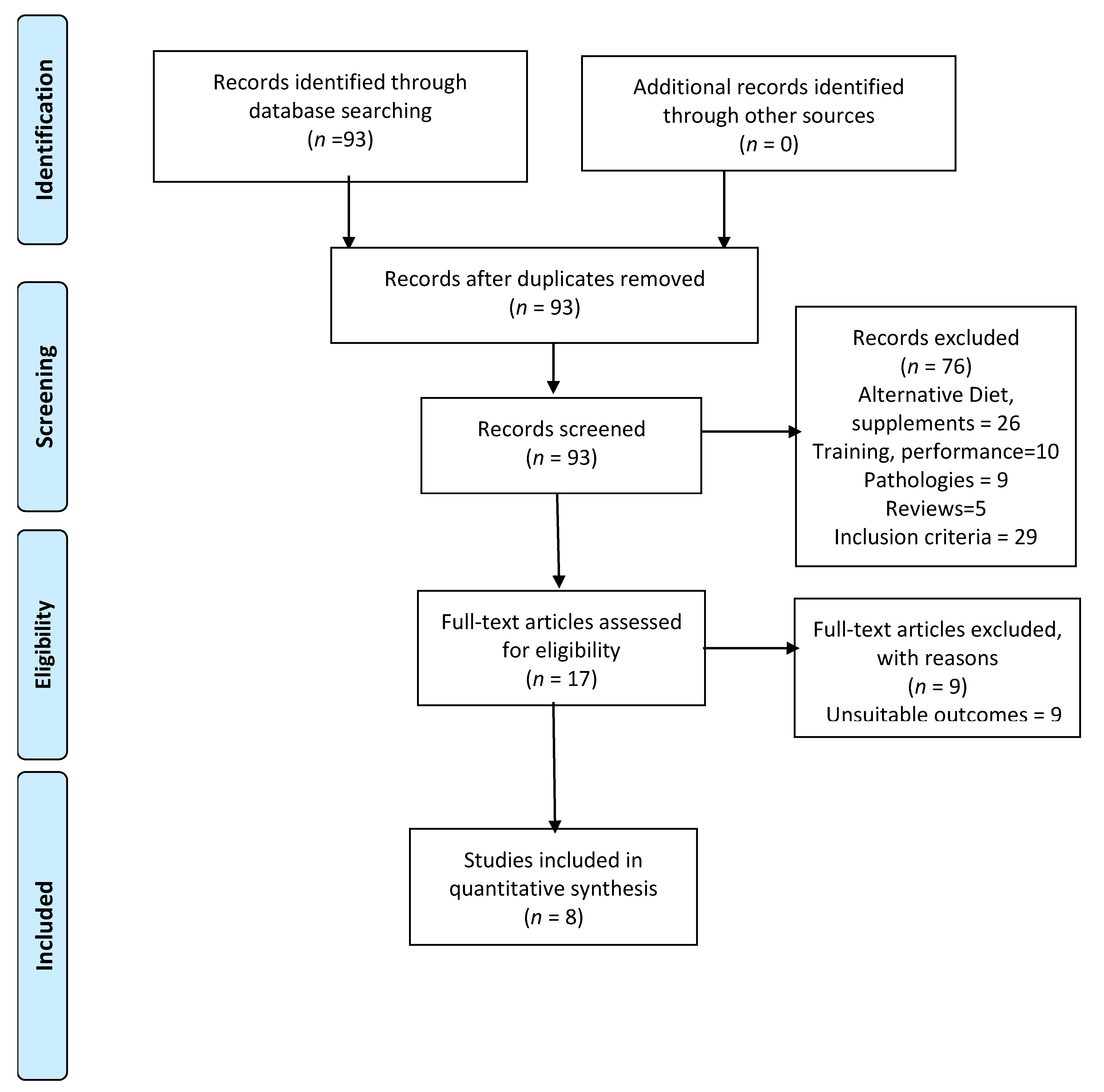
3. Results
3.1. Main Search
3.2. Characteristics of SOUT Events
3.3. Characteristics of SOUT Runners
3.4. Carbohydrate Intake
3.5. Fatigue Outcomes
3.6. GI Symptoms
4. Discussion
4.1. CHO Intake by Athletes during SOUT Events
4.2. Fatigue Outcomes
4.2.1. Internal Exercise Load
4.2.2. EIMD Markers
4.2.3. Post-Exercise Recovery
4.3. Effects of CHO Intake on GI Problems
5. Limitations and Strengths and Future Lines of Research
6. Practical Applications
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Knechtle, B.; Lepers, R. Participation and performance trends in ultra-triathlons from 1985 to 2009. Scand. J. Med. Sci. Sports 2010, 21, e82–e90. [Google Scholar] [CrossRef]
- Urdampilleta, A.; Arribalzaga, S.; Viribay, A.; Castañeda-Babarro, A.; Seco-Calvo, J.; Mielgo-Ayuso, J. Effects of 120 vs. 60 and 90 g/h Carbohydrate Intake during a Trail Marathon on Neuromuscular Function and High Intensity Run Capacity Recovery. Nutrients 2020, 12, 2094. [Google Scholar] [CrossRef]
- Lavoué, C.; Siracusa, J.; Chalchat, E.; Bourrilhon, C.; Charlot, K. Analysis of food and fluid intake in elite ultra-endurance runners during a 24-h world championship. J. Int. Soc. Sports Nutr. 2020, 17, 1–12. [Google Scholar] [CrossRef]
- Viribay, A.; Arribalzaga, S.; Mielgo-Ayuso, J.; Castañeda-Babarro, A.; Seco-Calvo, J.; Urdampilleta, A. Effects of 120 g/h of Carbohydrates Intake during a Mountain Marathon on Exercise-Induced Muscle Damage in Elite Runners. Nutrients 2020, 12, 1367. [Google Scholar] [CrossRef] [PubMed]
- Wardenaar, F.C.; Hoogervorst, D.; Versteegen, J.J.; Van Der Burg, N.; Lambrechtse, K.J.; Bongers, C.C.W.G. Real-Time Observations of Food and Fluid Timing During a 120 km Ultramarathon. Front. Nutr. 2018, 5, 32. [Google Scholar] [CrossRef]
- Wardenaar, F.C.; Dijkhuizen, R.; Ceelen, I.J.; Jonk, E.; De Vries, J.H.; Witkamp, R.F.; Mensink, M. Nutrient Intake by Ultramarathon Runners: Can They Meet Recommendations? Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Stellingwerff, T. Competition Nutrition Practices of Elite Ultramarathon Runners. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.S.; Gill, S.K.; Hankey, J.; Wright, A.; Marczak, S. Perturbed energy balance and hydration status in ultra-endurance runners during a 24 h ultra-marathon. Br. J. Nutr. 2014, 112, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Moran, S.T.; Dziedzic, C.E.; Cox, G. Feeding Strategies of a Female Athlete During an Ultraendurance Running Event. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Fallon, K.E.; Broad, E.; Thompson, M.W.; Reull, P.A. Nutritional and Fluid Intake in a 100-km Ultramarathon. Int. J. Sport Nutr. 1998, 8, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, P.T.; Veniamakis, E.; Rosemann, T.; Knechtle, B. Nutrition in Ultra-Endurance: State of the Art. Nutrients 2018, 10, 1995. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E. Nutritional implications for ultra-endurance walking and running events. Extreme Physiol. Med. 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E. Nutrition for endurance sports: Marathon, triathlon, and road cycling. J. Sports Sci. 2011, 29, 37–41. [Google Scholar] [CrossRef]
- Millet, G.Y.; Tomazin, K.; Verges, S.; Vincent, C.; Bonnefoy, R.; Boisson, R.-C.; Gergelé, L.; Féasson, L.; Martin, V. Neuromuscular Consequences of an Extreme Mountain Ultra-Marathon. PLoS ONE 2011, 6, e17059. [Google Scholar] [CrossRef] [PubMed]
- Giandolini, M.; Vernillo, G.; Samozino, P.; Horvais, N.; Edwards, W.B.; Morin, J.B.; Millet, G.Y. Fatigue associated with pro-longed graded running. Eur. J. Appl. Physiol. 2016, 116, 1859–1873. [Google Scholar] [CrossRef]
- Stuempfle, K.J.; Hoffman, M.D.; Hew-Butler, T. Association of gastrointestinal distress in ultramarathoners with race diet. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 103–109. [Google Scholar] [CrossRef]
- Jeukendrup, A.E. Periodized Nutrition for Athletes. Sports Med. 2017, 47, 51–63. [Google Scholar] [CrossRef]
- Coyle, E.F.; Hagberg, J.M.; Hurley, B.F.; Martin, W.H.; Ehsani, A.A.; Holloszy, J.O. Carbohydrate feeding during prolonged strenuous exercise can delay fatigue. J. Appl. Physiol. 1983, 55, 230–235. [Google Scholar] [CrossRef]
- Coggan, A.R.; Coyle, E.F. Carbohydrate ingestion during prolonged exercise: Effects on metabolism and performance. Exerc. Sport Sci. Rev. 1991, 19, 1–40. [Google Scholar] [CrossRef]
- Pfeiffer, B.; Stellingwerff, T.; Hodgson, A.B.; Randell, R.; Pottgen, K.; Res, P.; Jeukendrup, A.E. Nutritional intake and gastro-intestinal problems during competitive endurance events. Med. Sci. Sports Exerc. 2012, 44, 344–351. [Google Scholar] [CrossRef]
- King, A.J.; O’Hara, J.P.; Morrison, D.J.; Preston, T.; King, R.F.G.J. Carbohydrate dose influences liver and muscle glycogen oxidation and performance during prolonged exercise. Physiol. Rep. 2018, 6, e13555. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.F.; Coggan, A.R.; Hemmert, M.K.; Ivy, J.L. Muscle glycogen utilization during prolonged strenous exercise when fed carbohydrate. J. Appl. Physiol. 1986, 61, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Stellingwerff, T.; Boon, H.; Gijsen, A.P.; Stegen, J.H.C.H.; Kuipers, H.; Van Loon, L.J.C. Carbohydrate supplementation during prolonged cycling exercise spares muscle glycogen but does not affect intramyocellular lipid use. Pflugers Arch. 2007, 454, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.T.; Fuchs, C.J.; Smith, F.E.; Thelwall, P.E.; Taylor, R.; Stevenson, E.J.; Trenell, M.I.; Cermak, N.M.; Van Loon, L.J.C. Ingestion of glucose or sucrose prevents liver but not muscle glycogen depletion during prolonged endurance-type exercise in trained cyclists. Am. J. Physiol. Metab. 2015, 309, E1032–E1039. [Google Scholar] [CrossRef]
- Burke, L.M.; Jeukendrup, A.E.; Jones, A.M.; Mooses, M. Contemporary Nutrition Strategies to Optimize Performance in Dis-tance Runners and Race Walkers. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 117–129. [Google Scholar] [CrossRef]
- Jeukendrup, A. A Step Towards Personalized Sports Nutrition: Carbohydrate Intake During Exercise. Sports Med. 2014, 44, 25–33. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.J.; Viguie, C.A.; Mazzeo, R.S.; Brooks, G.A. Carbohydrate Dependence During Marathon Running. Med. Sci. Sports Exerc. 1993, 25, 1009–1017. [Google Scholar]
- Thomas, D.T.; Erdman, K.A.; Burke, L. American College of Sports Medicine Joint Position Statement. Nutrition and Athletic Performance. Med. Sci. Sport Exerc. 2016, 48, 543–568. [Google Scholar]
- Martinez, S.; Aguilo, A.; Rodas, L.; Lozano, L.; Moreno, C.; Tauler, P. Energy, macronutrient and water intake during a mountain ultramarathon event: The influence of distance. J. Sports Sci. 2017, 36, 333–339. [Google Scholar] [CrossRef]
- Hansen, E.A.; Emanuelsen, A.; Gertsen, R.M.; Sørensen, S.S.R. Improved Marathon Performance by In-Race Nutritional Strategy Intervention. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 645–655. [Google Scholar] [CrossRef]
- Stellingwerf, T. Case study: Nutrition and training periodization in three elite marathon runners. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 392–400. [Google Scholar] [CrossRef]
- Pugh, J.N.; Kirk, B.; Fearn, R.; Morton, J.P.; Close, G.L. Prevalence, Severity and Potential Nutritional Causes of Gastrointestinal Symptoms during a Marathon in Recreational Runners. Nutrients 2018, 10, 811. [Google Scholar] [CrossRef]
- Kruseman, M.; Bucher, S.; Bovard, M.; Kayser, B.; Bovier, P.A. Nutrient intake and performance during a mountain marathon: An observational study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2005, 94, 151–157. [Google Scholar] [CrossRef]
- De Oliveira, E.P.; Burini, R.C.; Jeukendrup, A. Gastrointestinal Complaints During Exercise: Prevalence, Etiology, and Nutritional Recommendations. Sports Med. 2014, 44, 79–85. [Google Scholar] [CrossRef]
- Glace, B.; Murphy, C.; McHugh, M. Food and Fluid Intake and Disturbances in Gastrointestinal and Mental Function during an Ultramarathon. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 414–427. [Google Scholar] [CrossRef]
- Ross, B.M. The Emerging Role of Eicosapentaenoic Acid as an Important Psychoactive Natural Product: Some Answers but a Lot more Questions. Lipid Insights 2008, 2, LPI.S1013. [Google Scholar] [CrossRef]
- Costa, R.J.; Knechtle, B.; Tarnopolsky, M.; Hoffman, M.D. Nutrition for Ultramarathon Running: Trail, Track, and Road. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 130–140. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Snipe, R.; Camões-Costa, V.; Scheer, V.; Murray, A.J.; Snipe, R. The Impact of Gastrointestinal Symptoms and Dermatological Injuries on Nutritional Intake and Hydration Status During Ultramarathon Events. Sports Med. Open 2016, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rehrer, N.J.; Van Kemenade, M.; Meester, W.; Brouns, F.; Saris, W.H. Gastrointestinal Complaints in Relation to Dietary Intake in Triathletes. Int. J. Sport Nutr. 1992, 2, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Glace, B.W.; Murphy, C.A.; McHugh, M.P. Food intake and electrolyte status of ultramarathoners competing in extreme heat. J. Am. Coll. Nutr. 2002, 21, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Stuempfle, K.J.; Hoffman, M.D.; Weschler, L.B.; Rogers, I.R.; Hew-Butler, T. Race diet of finishers and non-finishers in a 100 mile (161 km) mountain footrace. J. Am. Coll. Nutr. 2011, 30, 529–535. [Google Scholar] [CrossRef]
- Jeukendrup, A.E. Training the Gut for Athletes. Sports Med. 2017, 47, 101–110. [Google Scholar] [CrossRef]
- Costa, R.J.; Miall, A.; Khoo, A.; Rauch, C.; Snipe, R.; Camões-Costa, V.; Gibson, P. Gut-training: The impact of two weeks repetitive gut-challenge during exercise on gastrointestinal status, glucose availability, fuel kinetics, and running performance. Appl. Physiol. Nutr. Metab. 2017, 42, 547–557. [Google Scholar] [CrossRef]
- Miall, A.; Khoo, A.; Rauch, C.; Snipe, R.M.J.; Camões-Costa, V.L.; Gibson, P.R.; Costa, R.J. Two weeks of repetitive gut-challenge reduce exercise-associated gastrointestinal symptoms and malabsorption. Scand. J. Med. Sci. Sports 2017, 28, 630–640. [Google Scholar] [CrossRef]
- Tiller, N.B.; Roberts, J.D.; Beasley, L.; Chapman, S.; Pinto, J.M.; Smith, L.; Wiffin, M.; Russell, M.; Sparks, A.; Duckworth, L.; et al. International Society of Sports Nutrition Position Stand: Nutritional considerations for single-stage ultra-marathon training and racing. J. Int. Soc. Sports Nutr. 2019, 16, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Hillman, K. Health systems research & intensive care. Intensiv. Care Med. 1999, 25, 1353–1354. [Google Scholar]
- Saaiq, M.; Ashraf, B. Modifying “Pico” Question into “Picos” Model for More Robust and Reproducible Presentation of the Methodology Employed in A Scientific Study. World J. Plast. Surg. 2017, 6, 390–392. [Google Scholar]
- Letts, L.; Wilkins, S.; Law, M.; Stewart, D.; Bosch, J.; Westmorland, M. Guidelines for Critical Review form: Qualitative Studies (Version 2.0); McMaster University Occupational Therapy Evidence-Based Practice Research Group: Hamilton, ON, Canada, 2007; Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.600.223&rep=rep1&type=pdf (accessed on 19 December 2019).
- Burke, L.M.; Hawley, J.A.; Wong, S.H.S.; Jeukendrup, A.E. Carbohydrates for training and competition. J. Sports Sci. 2011, 29, S17–S27. [Google Scholar] [CrossRef]
- Stellingwerff, T.; Cox, G. Systematic review: Carbohydrate supplementation on exercise performance or capacity of varying durations. Appl. Physiol. Nutr. Metab. 2014, 39, 998–1011. [Google Scholar] [CrossRef]
- King, A.J.; O’Hara, J.P.; Arjomandkhah, N.C.; Rowe, J.; Morrison, D.J.; Preston, T.; King, R.F.G.J. Liver and muscle glycogen oxidation and performance with dose variation of glucose–fructose ingestion during prolonged (3 h) exercise. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 119, 1157–1169. [Google Scholar] [CrossRef]
- Jentjens, R.L.P.G.; Moseley, L.; Waring, R.H.; Harding, L.K.; Jeukendrup, A.E. Oxidation of combined ingestion of glucose and fructose during exercise. J. Appl. Physiol. 2004, 96, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E. Carbohydrate and exercise performance: The role of multiple transportable carbohydrates. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Moseley, L. Multiple transportable carbohydrates enhance gastric emptying and fluid delivery. Scand. J. Med. Sci. Sports 2010, 20, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Birkenhead, K.L.; Slater, G. A Review of Factors Influencing Athletes’ Food Choices. Sports Med. 2015, 45, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Zhu, X.; Deng, S. Sweet Taste Receptor Expression and Its Activation by Sucralose to Regulate Glucose Absorption in Mouse Duodenum. J. Food Sci. 2021, 86, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, B.; Stellingwerff, T.; Zaltas, E.; Jeukendrup, A.E. Oxidation of Solid versus Liquid CHO Sources during Exercise. Med. Sci. Sports Exerc. 2010, 42, 2030–2037. [Google Scholar] [CrossRef]
- Dashty, M. A quick look at biochemistry: Carbohydrate metabolism. Clin. Biochem. 2013, 46, 1339–1352. [Google Scholar] [CrossRef]
- Pfeiffer, B.; Stellingwerff, T.; Zaltas, E.; Jeukendrup, A.E. CHO Oxidation from a CHO Gel Compared with a Drink during Exercise. Med. Sci. Sports Exerc. 2010, 42, 2038–2045. [Google Scholar] [CrossRef] [PubMed]
- Eden, B.D.; Abernethy, P.J. Nutritional Intake during an Ultraendurance Running Race. Int. J. Sport Nutr. 1994, 4, 166–174. [Google Scholar] [CrossRef]
- Fuchs, C.J.; Gonzalez, J.T.; Van Loon, L.J. Fructose co-ingestion to increase carbohydrate availability in athletes. J. Physiol. 2019, 597, 3549–3560. [Google Scholar] [CrossRef]
- Fornasiero, A.; Savoldelli, A.; Fruet, D.; Boccia, G.; Pellegrini, B.; Schena, F. Physiological intensity profile, exercise load and performance predictors of a 65-km mountain ultra-marathon. J. Sports Sci. 2017, 36, 1287–1295. [Google Scholar] [CrossRef]
- Vernillo, G.; Savoldelli, A.; Zignoli, A.; Skafidas, S.; Fornasiero, A.; La Torre, A.; Bortolan, L.; Pellegrini, B.; Schena, F. Energy cost and kinematics of level, uphill and downhill running: Fatigue-induced changes after a mountain ultramarathon. J. Sports Sci. 2015, 33, 1998–2005. [Google Scholar] [CrossRef]
- Halson, S.L. Monitoring Training Load to Understand Fatigue in Athletes. Sports Med. 2014, 44, 139–147. [Google Scholar] [CrossRef]
- Ørtenblad, N.; Nielsen, J. Muscle glycogen and cell function—Location, location, location. Scand. J. Med. Sci. Sports 2015, 25, 34–40. [Google Scholar] [CrossRef]
- Ørtenblad, N.; Nielsen, J.; Saltin, B.; Holmberg, H.-C. Role of glycogen availability in sarcoplasmic reticulum Ca2+ kinetics in human skeletal muscle. J. Physiol. 2011, 589, 711–725. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.A.; Sherman, W.M.; Strauss, R.L. Efects of eccentric and concentric exercise on muscle glycogen replenishment. J. Appl. Physiol. 1993, 74, 1848–1855. [Google Scholar] [CrossRef] [PubMed]
- Asp, S.; Daugaard, J.R.; Richter, E. Eccentric exercise decreases glucose transporter GLUT4 protein in human skeletal muscle. J. Physiol. 1995, 482, 705–712. [Google Scholar] [CrossRef]
- Khong, T.K.; Selvanayagam, V.S.; Sidhu, S.K.; Yusof, A. Role of carbohydrate in central fatigue: A systematic review. Scand. J. Med. Sci. Sports 2016, 27, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.D.; Duhamel, T.A.; Foley, K.P.; Ouyang, J.; Smith, I.C.; Green, H.J. Protection of muscle membrane excitability during prolonged cycle exercise with glucose supplementation. J. Appl. Physiol. 2007, 103, 331–339. [Google Scholar] [CrossRef]
- Owens, D.J.; Twist, C.; Cobley, J.N.; Howatson, G.; Close, G.L. Exercise-induced muscle damage: What is it, what causes it and what are the nutritional solutions? Eur. J. Sport Sci. 2019, 19, 71–85. [Google Scholar] [CrossRef]
- Fatouros, I.G.; Jamurtas, A.Z. Insights into the molecular etiology of exercise-induced inflammation: Opportunities for opti-mizing performance. J. Inflamm. Res. 2016, 9, 175–186. [Google Scholar] [CrossRef]
- Clarkson, P.M.; Hubal, M. Exercise-Induced Muscle Damage in Humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef]
- Schwane, J.; Johnson, S.R.; Vandenakker, C.B.; Armstrong, R.B. Delayed-onset muscular soreness and plasma CPK and LDH activities after downhill running. Med. Sci. Sports Exerc. 1983, 15, 51–56. [Google Scholar] [CrossRef]
- Alghannam, A.F.; Jedrzejewski, D.; Bilzon, J.; Thompson, D.; Tsintzas, K.; Betts, J.A. Influence of post-exercise carbohydrate-protein ingestion on muscle glycogen metabolism in recovery and subsequent running exercise. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 572–580. [Google Scholar] [CrossRef]
- Nielsen, L.L.K.; Lambert, M.N.T.; Jeppesen, P.B. The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2020, 12, 1483. [Google Scholar] [CrossRef]
- Betts, J.A.; Williams, C.C. Short-term recovery from prolonged exercise exploring the potential for protein ingestion to accentuate the benefits of carbohydrate supplements. Sport Med. 2012, 40, 941–959. [Google Scholar] [CrossRef]
- Garbisu-Hualde, A.; Santos-Concejero, J. What are the Limiting Factors During an Ultra-Marathon? A Systematic Review of the Scientific Literature. J. Hum. Kinet. 2020, 72, 129–139. [Google Scholar] [CrossRef]
- Burke, L.M.; van Loon, L.J.; Hawley, J.A. Postexercise muscle glycogen resynthesis in humans. J. Appl. Physiol. 2017, 122, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Beelen, M.; Burke, L.M.; Gibala, M.J.; Van Loon, L.J. Nutritional Strategies to Promote Postexercise Recovery. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B. Nutrition behaviors, perceptions, and beliefs of recent marathon finishers. Physician Sportsmed. 2016, 44, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.; Hoffman, M.D.; Stellingwerff, T. Considerations for ultra-endurance activities: Part 1 nutrition. Res. Sports Med. 2019, 27, 166–181. [Google Scholar] [CrossRef] [PubMed]
- Keeffe, E.B.; Lowe, D.K.; Goss, J.R.; Wayne, R. Gastrointestinal Symptoms of Marathon Runners. West. J. Med. 1984, 141, 481–484. [Google Scholar] [PubMed]
- Jeukendrup, A.E.; Vet-Joop, K.; Sturk, A.; Stegen, J.H.J.C.; Senden, J.; Saris, W.H.M.; Wagenmakers, A.J.M. Relationship be-tween gastro-intestinal complaints and endotoxaemia, cytokine release and the acute-phase reaction during and after a long-distance triathlon in highly trained men. Clin. Sci. 2000, 98, 47–55. [Google Scholar] [CrossRef]
- Wilson, P.B. Perceived life stress and anxiety correlate with chronic gastrointestinal symptoms in runners. J. Sports Sci. 2017, 36, 1713–1719. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Snipe, R.M.J.; Kitic, C.M.; Gibson, P.R. Systematic review: Exercise-induced gastrointestinal syndrome—Implications for health and intestinal disease. Aliment. Pharmacol. Ther. 2017, 46, 246–265. [Google Scholar] [CrossRef]
- Goyal, R.K.; Guo, Y.; Mashimo, H. Advances in the physiology of gastric emptying. Neurogastroenterol. Motil. 2019, 31, e13546. [Google Scholar] [CrossRef] [PubMed]
- Heikura, I.A.; Stellingwerff, T.; Burke, L.M. Self-Reported Periodization of Nutrition in Elite Female and Male Runners and Race Walkers. Front. Physiol. 2018, 9, 1732. [Google Scholar] [CrossRef] [PubMed]
- Dorling, J.; Broom, D.R.; Burns, S.F.; Clayton, D.J.; Deighton, K.; James, L.J.; King, J.A.; Miyashita, M.; Thackray, A.E.; Bat-terham, R.L.; et al. Acute and chronic effects of exercise on appetite, energy intake, and appetite-related hormones: The mod-ulating effect of adiposity, sex, and habitual physical activity. Nutrients 2018, 10, 1140. [Google Scholar] [CrossRef]
- Brandsborg, O.; Christensen, N.J.; Galbo, H.; Brandsborg, M.; Lovgreen, N.A. The effect of exercise, smoking and propranolol on serum gastrin in patients with duodenal ulcer and in vagotomized subjects. Scand. J. Clin. Lab. Investig. 1978, 38, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Stellingwerff, T. Contemporary Nutrition Approaches to Optimize Elite Marathon Performance. Int. J. Sports Physiol. Perform. 2013, 8, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Jentjens, R.L.P.G.; Moseley, L. Nutritional Considerations in Triathlon. Sports Med. 2005, 35, 163–181. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Leiper, J.B. Limitations to Fluid Replacement During Exercise. Can. J. Appl. Physiol. 1999, 24, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.D.; Ingwerson, J.L.; Rogers, I.R.; Hew-Butler, T.; Stuempfle, K.J. Increasing Creatine Kinase Concentrations at the 161-km Western States Endurance Run. Wilderness Environ. Med. 2012, 23, 56–60. [Google Scholar] [CrossRef] [PubMed]
| Evaluated Items | Urdampilleta et al., 2020 [2] | Lavoué et al., 2020 [3] | Viribay et al., 2020 [4] | Wardenaar et al., 2018 [5] | Martínez et al., 2018 [27] | Wardenaar et al., 2015 [6] | Costa et al., 2014 [8] | Kruseman et al., 2005 [33] |
|---|---|---|---|---|---|---|---|---|
| Purpose | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Literature Review | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Study Design | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 |
| Blinding | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Sample Description | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 |
| Sample Size | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 |
| Ethics and Consent | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Validity of Outcomes | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 |
| Reliability of Outcomes | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 |
| Intervention Description | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Statistical Significance | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| Statistical Analysis | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Clinical Importance | 1 | 0 | 1 | 1 | 0 | 0 | 1 | NR |
| Conclusions | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Clinical Implications | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 |
| Study Limitations | 1 | 0 | 1 | 1 | NR | NR | NR | 1 |
| TOTAL | 12 | 8 | 12 | 13 | 10 | 12 | 11 | 11 |
| % | 75% | 61.53% | 75% | 81.25 | 62.5 | 75.0 | 68.8 | 68.8 |
| Methodological Quality | Good | Poor | Good | Very Good | Acceptable | Good | Good | Good |
| Reference | Event | Distance/Elevation Gain Temperature | Participants | |
|---|---|---|---|---|
| N (M + F) (Age) | Experience | |||
| Ultra-Trail | ||||
| Lavoué et al., 2020 [3] | Albi, France | 24 h/ (distance range 133–272 km) 11.9–35.1 °C | 11 (5 M + 6 F) (46 ± 7 years) | Elite |
| Wardenaar et al., 2018 [5] | Sixty of Texel, Netherlands | 120 km/4.3–9.6 °C | 5 M (47 ± 6 years) | Completed at least 10 (ultra) marathons |
| Martínez et al., 2018 [29] | Ultra Mallorca Serra de Tramuntana, Spain | 112 km/ 4.448 m of ascent 11–21 °C | 54 (53 M + 1 F) (35.7 ± 7.9 years) | Data not shown |
| 67 km/2521 m of ascent 11–21 °C | 109 (98 M + 11 F) (35.2 ± 8.4 years) | |||
| Wardenaar et al., 2015 [6] | Sixty of Texel, Netherlands | 120 km/2.2 °C | 8 (7 M + 1 F) (46.6 ± 6.3 years) | Data not shown |
| 60 km/2.2 °C | 60 (48 M + 12 F) (46.5 ± 7.2 years) | |||
| Costa et al., 2014 [8] | Glenmore 24 Trail Race, Scottish Highlands, Scotland, UK | 24 h/(distance range: 122–208 km) 0–20 °C | 25 (19 M + 6 F) (39 ± 7 years) | Data not shown |
| Kruseman et al., 2005 [33] | Tour des Dents du Midi race, Switzerland | 44 km/2890 m of ascent 18–30 °C | 42 (39 M + 3 F) (42 ± 9.7 years) | Completed at least 10 trail marathons |
| Trail Marathon | ||||
| Urdampilleta et al., 2020 [2] | Oiartzun, Spain | 10 °C/Cumulative elevation gain: 3980.80 m | 20 M LOW: 37.8 ± 9.4 years CON: 37.2 ± 5.4 years EXP: 38 ± 6.8 years | 5 years in SOUT (2 World champions) |
| Viribay et al., 2020 [4] | Oiartzun, Spain | 10 °C/Cumulative elevation gain: 3980.80 m | 20 M LOW: 37.8 ± 9.4 years CON: 37.2 ± 5.4 years EXP: 38 ± 6.8 years | 5 years in SOUT (2 World champions) |
| Martínez et al., 2018 [29] | Ultra Mallorca Serra de Tramuntana, Spain | 11.0–21.6 °C/1424 m of ascent | 63 (51 M + 12 F) (36.6 ± 8 years) | Unshown data |
| Reference | Fluid Intake | CHO Intake | Type of CHO | Vs. Recommendations (90 g/h) | Nutritional Strategies | Observations |
|---|---|---|---|---|---|---|
| Ultra-Trail | ||||||
| Lavoué et al., 2020 [3] | 274 ± 115 mL/h | 13.9–105.4 g/h (62.2 ± 29.6 g/h) | Sports drink, cake, fruit, and mashed potatoes. | ↓ | YES | Higher rates of energy intake for finishers relative to those of non-finishers and for fast runners compared to slow runners |
| 685 ± 290 mL/kg BM | ||||||
| Wardenaar et al., 2018 [5] | 354–765 mL/h | 46.5 ± 14.1 g/h (range: 22.1–62.6 g/h) | Fruit, gels, sports drink, chocolate milk | ↓ | NO | ↑ CHO intake in the section 75–90 km because ↓lower running speed |
| Martínez et al. 2018 [29] | 6.319 ± 4214 L | (a) 112 km: 534.9 ± 279.3 g/total (31.2 ± 17.8 g/h) | (a) & (b) Sandwiches, fruit (mainly bananas), gels, pasta, energy bars CHO-electrolyte drinks | (a) ↓ | NO | No difference among distances. The slow paces during race could mean that participants did not require as much CHO. |
| 351 ± 239 mL/h | (b) 67 km: 326.7 ± 157.2 g/total (32.1 ± 14.8 g/h) | (b) ↓ | ||||
| Wardenaar et al., 2015 [6] | 2.9 ± 0.9 L | (a) 60 km: 274 ± 133 g/total | No data shown | (a) ↓ | NO | (a) 22% of runners kept to CHO recommendations (21.2% males and 12.5% females) |
| (b) 120 km: 67.3 ± 31.7 g/h | (b) ↓ | (b) Only 1 runner kept to CHO recommendations | ||||
| Costa et al., 2014 [8] | 9.1 ± 4.0 L | (a) <160 km: 31 ± 9 g/h | Mono/di/oligosaccharide, polysaccharide sources | (a) ↓ | NO | CHO rates ranged from 16 to 53 g/h (only 1 runner of (≥160 km) consumed 126 g/h). |
| 118 ± 51 mL/kg BM | (b) ≥160 km: 44 ± 33 g/h | (b) ↓ | ||||
| Kruseman, et al., 2005 [33] | 3.777 ± 1.146 L | 31 ± 14 g/h | Sweet drinks and glucose. Slowest: soup, fruits and cereal bars. Fastest: Gels | ↓ | NO | >50% runners: <30 g/h 3 runners = 60 g/h insufficient palatability of fluid and food, the practical difficulty of drinking or eating while running/walking remains possible |
| 545 ± 158 mL/h | ||||||
| Trail Marathon | ||||||
| Urdampilleta et al., 2020 [2] | No data shown | (a) LOW: 60 g/h | 30 g/h maltodextrin (glucose) and fructose gels (ratio 2:1) | (a) ↓ | YES | ↑ CHO intake decreasing internal exercise load and neuromuscular fatigue |
| (b) CON: 90 g/h | (b)  | |||||
| (c) EXP: 120g/h | (c) ↑ | |||||
| Viribay et al., 2020 [4] | No data shown | (a) LOW: 60 g/h | 30 g/h maltodextrin (glucose) and fructose gels (ratio 2:1) | (a) ↓ | YES | ↑ CHO intake decreasing internal exercise load and EIMD. |
| (b) CON: 90 g/h | (b)  | |||||
| (c) EXP: 120g/h | (c) ↑ | |||||
| Martínez et al. 2018 [29] | 4.727 ± 2694 L | 205.2 ± 81.2 g (33.4 ± 13.5 g/h) | Sandwiches, fruit (mainly bananas), gels, pasta, energy bar, CHO-electrolyte drinks | ↓ | NO | No difference between distances. The slow paces during the race could mean that participants did not require as much CHO |
| 459 ± 256 mL/h | ||||||
 : equal; ↑: Higher.
: equal; ↑: Higher.| Author | Fatigue Related Outcomes Studied | Conclusions |
| Internal exercise load | ||
| Urdampilleta et al., 2020 [2] |
|
|
| Viribay et al., 2020 [4] |
|
|
| EIMD markers | ||
| Viribay et al., 2020 [4] |
|
|
| Post-exercise recovery | ||
| Urdampilleta et al., 2020 [2] |
|
|
| Autor | Upper GI Symptoms | Lower GI Symptoms | Conclusions |
|---|---|---|---|
| Ultra-Trail | |||
| Lavoué et al., 2020 [3] | 8 participants experienced at least one GI (nausea = 4, difficulty swallowing = 3 vomiting = 1) | Diarrhea (N = 2) | The episodes of GI were transient and did not cause any major decreases in performance or dropping out |
| Wardenaar et al., 2018 [5] | Nausea (N = 1) | Urge to defecate (N = 3) Flatulence (N = 3) Side (N = 1) | Lower amount of GI complaints during the race than the post-race |
| Wardenaar et al., 2015 [6] | Reflux (16.3%) Heartburn (9.3%) Belching (41.9%) Bloating (16.3%) Stomach cramps (14.0%) Nausea (20.9%) | Intestinal cramp (9.3%) Flatulence (34.9%) Urge to defecate (16.3%) Side ache (11.6%) Abdominal pain (9.3%) Loose stool (4.6%) Diarrhea (2.3%) | Higher nutrient intake, except fiber intake, was in general associated with lower frequency of GI distress |
| Costa et al., 2014 [8] | 65% reporting at least one severe symptom (nausea; GI pain; vomiting; indigestion; bloating; abnormal bowel movements (e.g., urgency to defecate) | No association between GI and energy and CHO intake was evident. A 2·5-fold greater occurrence of Gl symptoms was observed in the fast group vs. slow group | |
| Trail Marathon | |||
| Urdampilleta et al., 2020 [2] | 3 with flatulence and/ or reflux | Athletes with gut training did not evidence any GI problem | |
| Viribay et al., 2020 [4] | 3 with flatulence and/ or reflux | Athletes with gut training did not evidence any GI problem | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arribalzaga, S.; Viribay, A.; Calleja-González, J.; Fernández-Lázaro, D.; Castañeda-Babarro, A.; Mielgo-Ayuso, J. Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 5737. https://doi.org/10.3390/ijerph18115737
Arribalzaga S, Viribay A, Calleja-González J, Fernández-Lázaro D, Castañeda-Babarro A, Mielgo-Ayuso J. Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(11):5737. https://doi.org/10.3390/ijerph18115737
Chicago/Turabian StyleArribalzaga, Soledad, Aitor Viribay, Julio Calleja-González, Diego Fernández-Lázaro, Arkaitz Castañeda-Babarro, and Juan Mielgo-Ayuso. 2021. "Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 11: 5737. https://doi.org/10.3390/ijerph18115737
APA StyleArribalzaga, S., Viribay, A., Calleja-González, J., Fernández-Lázaro, D., Castañeda-Babarro, A., & Mielgo-Ayuso, J. (2021). Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review. International Journal of Environmental Research and Public Health, 18(11), 5737. https://doi.org/10.3390/ijerph18115737










