Fecundability and Sterility by Age: Estimates Using Time to Pregnancy Data of Japanese Couples Trying to Conceive Their First Child with and without Fertility Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
2.2. Statistical Analyses
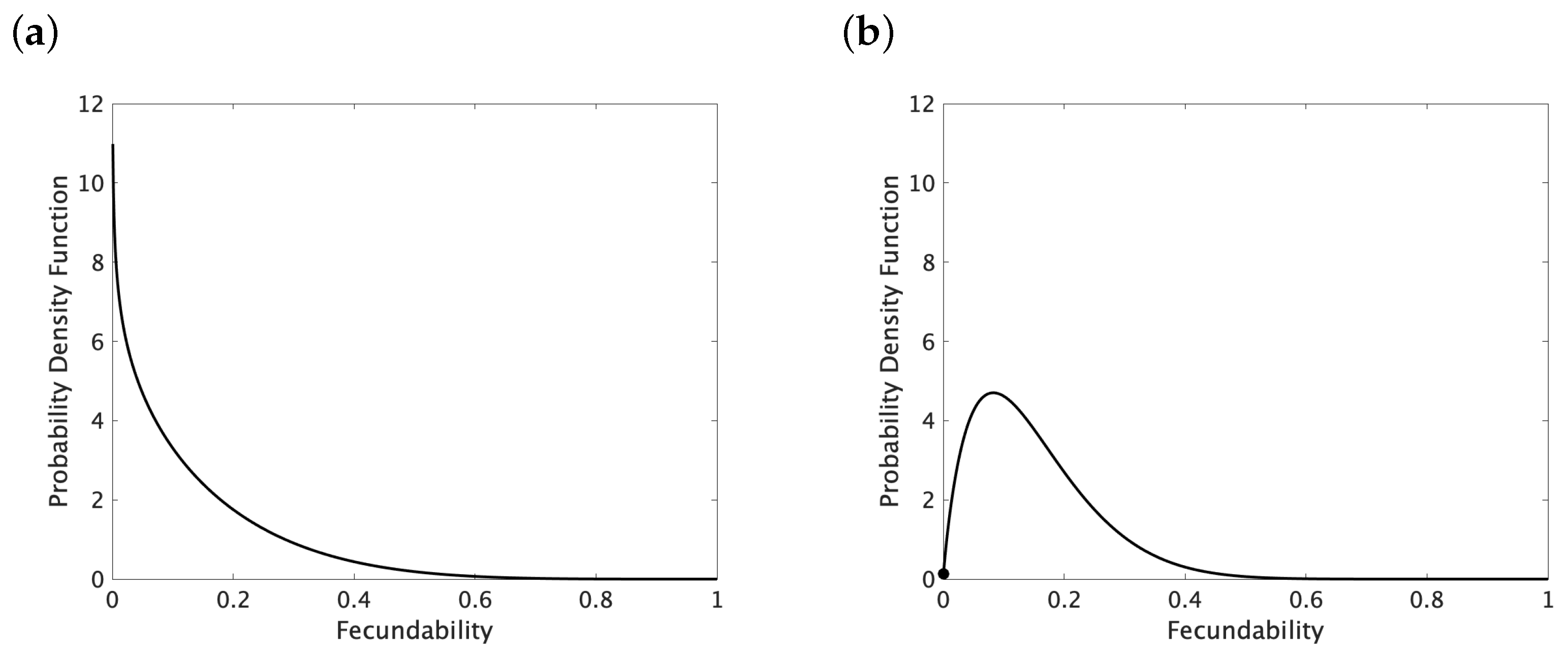
2.3. Maximum Likelihood Method
2.4. Sensitivity Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. Detailed Description About the Maximum Likelihood Method
Appendix A.1. One-Parameter Model
Appendix A.2. Two-Parameter Model
Appendix A.3. Three-Parameter Model
Appendix A.4. Maximum Likelihood Estimate
Appendix A.4.1. Two-Parameter Models
Appendix A.4.2. Three-Parameter Models
| TTP-All | |||||
|---|---|---|---|---|---|
| Fecundability, Mean (Variance) | |||||
| Age (y) Group | n | Two-Parameter Models | One-Parameter Models | ||
| All | 1264 | 0.13 (0.01) | 0.07 (0.00) | ||
| 24 or younger | 91 | 0.21 (0.03) | 0.11 (0.00) | ||
| 25–29 | 561 | 0.14 (0.01) | 0.09 (0.00) | ||
| 30–34 | 417 | 0.12 (0.01) | 0.06 (0.00) | ||
| 35–39 | 174 | 0.09 (0.01) | 0.04 (0.00) | ||
| 40+ | 21 | 0.02 (0.00) | 0.01 (0.00) | ||
| Maximum Log-Likelihood | |||||
| Age (y) Group | n | Two-Parameter Models (a) | One-Parameter Models (b) | 2{(a)–(b)} | p-Value |
| All | 1264 | −3350.67 | −3498.01 | 294.69 | 0.000 |
| 24 or younger | 91 | −239.40 | −252.39 | 25.98 | 0.000 |
| 25–29 | 561 | −1507.31 | −1555.25 | 95.88 | 0.000 |
| 30–34 | 417 | −1111.52 | −1162.00 | 100.96 | 0.000 |
| 35–39 | 174 | −427.49 | −444.62 | 34.26 | 0.000 |
| 40+ | 21 | −22.62 | −22.73 | 0.21 | 0.646 |
| TTP-Natural | |||||
| Fecundability, Mean (Variance) | |||||
| Age (y) Group | n | Two-Parameter Models | One-Parameter Models | ||
| All | 1264 | 0.13 (0.02) | 0.07 (0.00) | ||
| 24 or younger | 91 | 0.21 (0.02) | 0.12 (0.00) | ||
| 25–29 | 561 | 0.14 (0.01) | 0.09 (0.00) | ||
| 30–34 | 417 | 0.13 (0.02) | 0.06 (0.00) | ||
| 35–39 | 174 | 0.10 (0.02) | 0.04 (0.00) | ||
| 40+ | 21 | 0.02 (0.00) | 0.01 (0.00) | ||
| Maximum Log-Likelihood | |||||
| Age (y) Group | n | Two-Parameter Models (c) | One-Parameter Models (d) | 2{(c)–(d)} | p-Value |
| All | 1264 | −2870.59 | −3000.66 | 260.14 | 0.000 |
| 24 or younger | 91 | −234.63 | −246.43 | 23.61 | 0.000 |
| 25–29 | 561 | −1346.07 | −1382.87 | 73.60 | 0.000 |
| 30–34 | 417 | −907.64 | −966.84 | 118.39 | 0.000 |
| 35–39 | 174 | −323.91 | −339.84 | 31.86 | 0.000 |
| 40+ | 21 | −15.67 | −15.70 | 0.06 | 0.813 |
| TTP-All | ||||
|---|---|---|---|---|
| Maximum Log-Likelihood | ||||
| Age (y) Group | Three-Parameter Models (a) | Two-Parameter Models (b) | 2{(a)–(b)} | p-Value |
| All | −3350.60 | −3350.67 | 0.14 | 0.710 |
| 24 or younger | −239.30 | −239.40 | 0.21 | 0.648 |
| 25–29 | −1507.04 | −1507.31 | 0.54 | 0.463 |
| 30–34 | −1111.52 | −1111.52 | −0.01 | 1.000 |
| 35–39 | −427.49 | −427.49 | 0.00 | 1.000 |
| 40+ | −22.63 | −22.62 | 0.00 | 1.000 |
| TTP-Natural | ||||
| Maximum Log-Likelihood | ||||
| Age (y) Group | Three-Parameter Models (c) | Two-Parameter Models (d) | 2{(c)–(d)} | p-Value |
| All | −2865.78 | −2870.59 | 9.64 | 0.002 |
| 24 or younger | −234.51 | −234.63 | 0.24 | 0.625 |
| 25–29 | −1344.58 | −1346.07 | 2.99 | 0.084 |
| 30–34 | −904.84 | −907.64 | 5.61 | 0.018 |
| 35–39 | −323.55 | −323.91 | 0.72 | 0.398 |
| 40+ | −15.67 | −15.67 | 0.00 | 1.000 |
| Parameters | Fecundability | |||||||
|---|---|---|---|---|---|---|---|---|
| Age (y) Group | n | First Quartile | Median | Third Quartile | Mean | Variance | ||
| All | 525 | 0.82 | 4.87 | 0.04 | 0.10 | 0.21 | 0.14 | 0.02 |
| 24 or younger | 31 | 0.93 | 2.16 | 0.11 | 0.25 | 0.45 | 0.30 | 0.05 |
| 25–29 | 223 | 1.65 | 10.25 | 0.06 | 0.12 | 0.19 | 0.14 | 0.01 |
| 30–34 | 183 | 0.77 | 4.44 | 0.04 | 0.10 | 0.22 | 0.15 | 0.02 |
| 35–39 | 76 | 0.26 | 1.87 | 0.00 | 0.03 | 0.17 | 0.12 | 0.03 |
| 40+ | 12 | 1.19 | 147.43 | 0.00 | 0.01 | 0.01 | 0.01 | 0.00 |
References
- Bongaarts, J.; Potter, R.G. Fertility, Biology, and Behavior. In An Analysis of the Proximate Determinants; Academic Press: New York, NY, USA, 1983. [Google Scholar]
- Wood, J.W. Dynamics of Human Reproduction; Aldine de Gruyter: New York, NY, USA, 1994. [Google Scholar]
- Gini, C. Decline in the birth-rate and the “fecundability” of woman. Eugen. Rev. 1926, 17, 258–274. [Google Scholar] [PubMed]
- Bendel, J.P.; Hua, C. An estimate of the natural fecundability ratio curve. Soc. Biol. 1978, 25, 210–227. [Google Scholar] [CrossRef]
- Schwartz, D.; Mayauz, M.J. Female fecundity as a function of age. N. Engl. J. Med. 1982, 306, 404–406. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J.; Wise, L.A.; Sørensen, H.T.; Riis, A.H.; Mikkelsen, E.M.; Hatch, E.E. Volitional determinants and age-related decline in fecundability: A general population prospective cohort study in Denmark. Fertil. Steril. 2013, 99, 1958–1964. [Google Scholar] [CrossRef] [PubMed]
- Konishi, S.; Sakata, S.; Oba, S.M.; O’Connor, K.A. Age and time to pregnancy for the first child among couples in Japan. J. Popul. Stud. 2018, 54, 1–18. [Google Scholar]
- Weinberg, C.R.; Gladen, B.C. The Beta-Geometric Distribution Applied to Comparative Fecundability Studies. Biometrics 1986, 42, 547–560. [Google Scholar] [CrossRef]
- Wood, J.W.; Holman, D.J.; Yashin, A.I.; Peterson, R.J.; Weinstein, M.; Chang, M.-C. A Multistate Model of Fecundability and Sterility. Demography 1994, 31, 403–426. [Google Scholar] [CrossRef] [PubMed]
- Dunson, D.B.; Baird, D.D.; Colombo, B. Increased infertility with age in men and women. Obstet. Gynecol. 2004, 103, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Ecochard, R. Heterogeneity in fecundability studies: Issues and modelling. Stat. Methods Med. Res. 2006, 15, 141–160. [Google Scholar] [CrossRef]
- Dyer, S.; Chambers, G.M.; de Mouzon, J.; Nygren, K.G.; Zegers-Hochschild, F.; Mansour, R.; Ishihara, O.; Banker, M.; Adamson, G.D. International Committee for Monitoring Assisted Reproductive Technologies world report: Assisted Reproductive Technology 2008, 2009 and 2010. Hum. Reprod. 2016, 31, 1588–1609. [Google Scholar] [CrossRef]
- Steiner, A.Z.; Jukic, A.M.Z. Impact of female age and nulligravidity on fecundity in an older reproductive age cohort. Fertil. Steril. 2016, 105, 1584–1588. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, S.; Macchiarelli, G.; Micara, G.; Linari, A.; Boninsegna, C.; Aragona, C.; Rossi, G.; Cecconi, S.; Nottola, S.A. Ultrastructural markers of quality are impaired in human metaphase II aged oocytes: A comparison between reproductive and in vitro aging. J. Assist. Reprod. Genet. 2015, 32, 1343–1358. [Google Scholar] [CrossRef] [PubMed]
- Navot, D.; Bergh, R.A.; Williams, M.A.; Garrisi, G.J.; Guzman, I.; Sandler, B.; Grunfeld, L. Poor oocyte quality rather than implantation failure as a cause of age-related decline in female fertility. Lancet 1991, 337, 1375–1377. [Google Scholar] [CrossRef]
- Curtis, K.M.; Savitz, D.A.; Arbuckle, T.E. Effects of Cigarette Smoking, Caffeine Consumption, and Alcohol Intake on Fecundability. Am. J. Epidemiol. 1997, 146, 32–41. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.A.; Holman, D.J.; Wood, J.W. Declining fecundity and ovarian ageing in natural fertility populations. Maturitas 1998, 30, 127–136. [Google Scholar] [CrossRef]
- McKinnon, C.J.; Hatch, E.E.; Rothman, K.J.; Mikkelsen, E.M.; Wesselink, A.K.; Hahn, K.A.; Wise, L.A. Body mass index, physical activity and fecundability in a North American preconception cohort study. Fertil. Steril. 2016, 106, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Wesselink, A.K.; Hatch, E.E.; Rothman, K.J.; Mikkelsen, E.M.; Aschengrau, A.; Wise, L.A. Prospective study of cigarette smoking and fecundability. Hum. Reprod. 2019, 34, 558–567. [Google Scholar] [CrossRef]
- Willis, S.K.; Hatch, E.E.; Wesselink, A.K.; Rothman, K.J.; Mikkelsen, E.M.; Wise, L.A. Female sleep patterns, shift work, and fecundability in a North American preconception cohort study. Fertil. Steril. 2019, 111, 1201–1210.e1. [Google Scholar] [CrossRef]
- Van Eekelen, R.; Scholten, I.; Tjon-Kon-Fat, R.I.; Van Der Steeg, J.W.; Steures, P.; Hompes, P.; Van Wely, M.; Van Der Veen, F.; Mol, B.W.; Eijkemans, M.J.; et al. Natural conception: Repeated predictions overtime. Hum. Reprod. 2017, 32, 346–353. [Google Scholar] [CrossRef]
- McLernon, D.J.; Lee, A.J.; Maheshwari, A.; Van Eekelen, R.; Van Geloven, N.; Putter, H.; Eijkemans, M.J.; Van Der Steeg, J.W.; Van Der Veen, F.; Steyerberg, E.W.; et al. Predicting the chances of having a baby with or without treatment at different time points in couples with unexplained subfertility. Hum. Reprod. 2019, 34, 1126–1138. [Google Scholar] [CrossRef]
- Bianchi, S.; Nottola, S.A.; Torge, D.; Palmerini, M.G.; Necozione, S.; Macchiarelli, G. Association between Female Reproductive Health and Mancozeb: Systematic Review of Experimental Models. Int. J. Environ. Res. Public Health 2020, 17, 2580. [Google Scholar] [CrossRef] [PubMed]
- Buck Louis, G.M.; Sundaram, R.; Schisterman, E.F.; Sweeney, A.M.; Lynch, C.D.; Gore-Langton, R.E.; Chen, Z.; Kim, S.; Caldwell, K.L.; Barr, D.B. Heavy metals and couple fecundity, the LIFE Study. Chemosphere 2012, 87, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Buck Louis, G.M.; Sundaram, R.; Sweeney, A.M.; Schisterman, E.F.; Maisog, J.; Kannan, K. Urinary bisphenol A, phthalates, and couple fecundity: The Longitudinal Investigation of Fertility and the Environment (LIFE) Study. Fertil. Steril. 2014, 101, 1359–1366. [Google Scholar] [CrossRef]
- Belli, M.; Palmerini, M.G.; Bianchi, S.; Bernardi, S.; Khalili, M.A.; Nottola, S.A.; Macchiarelli, G. Ultrastructure of mitochondria of human oocytes in different clinical conditions during assisted reproduction. Arch. Biochem. Biophys. 2021, 703, 108854. [Google Scholar] [CrossRef] [PubMed]
| Age (y) Group | Participants | All Conceptions | Natural Conceptions | TTP-All (Month) | TTP-Natural (Month) |
|---|---|---|---|---|---|
| All | 1264 | 952 | 834 | 10.3 (12.9) | 8.2 (10.4) |
| 24 or younger | 91 | 81 | 80 | 6.8 (9.7) | 6.5 (9.0) |
| 25–29 | 561 | 456 | 415 | 8.5 (11.2) | 7.0 (9.2) |
| 30–34 | 417 | 304 | 253 | 11.6 (14.0) | 9.5 (11.9) |
| 35–39 | 174 | 107 | 83 | 13.7 (15.0) | 9.8 (10.3) |
| 40+ | 21 | 4 | 3 | 19.7 (12.8) | 8.9 (7.9) |
| TTP-All | ||||||||
|---|---|---|---|---|---|---|---|---|
| Parameters | Fecundability | |||||||
| Age (y) Group | n | First Quartile | Median | Third Quartile | Mean | Variance | ||
| All ages | 1264 | 0.94 | 6.22 | 0.04 | 0.10 | 0.19 | 0.13 | 0.01 |
| 24 or younger | 91 | 1.21 | 4.48 | 0.09 | 0.18 | 0.31 | 0.21 | 0.03 |
| 25–29 | 561 | 1.44 | 8.76 | 0.06 | 0.12 | 0.20 | 0.14 | 0.01 |
| 30–34 | 417 | 0.82 | 5.91 | 0.03 | 0.09 | 0.18 | 0.12 | 0.01 |
| 35–39 | 174 | 0.61 | 5.98 | 0.02 | 0.05 | 0.13 | 0.09 | 0.01 |
| 40+ | 21 | 0.24 | 15.47 | 0.00 | 0.00 | 0.02 | 0.02 | 0.00 |
| TTP-Natural | ||||||||
| Parameters | Fecundability | |||||||
| Age (y) Group | n | First Quartile | Median | Third Quartile | Mean | Variance | ||
| All ages | 1264 | 0.84 | 5.44 | 0.04 | 0.10 | 0.19 | 0.13 | 0.02 |
| 24 or younger | 91 | 1.24 | 4.60 | 0.09 | 0.18 | 0.30 | 0.21 | 0.02 |
| 25–29 | 561 | 1.46 | 8.87 | 0.06 | 0.12 | 0.20 | 0.14 | 0.01 |
| 30–34 | 417 | 0.58 | 3.90 | 0.02 | 0.08 | 0.19 | 0.13 | 0.02 |
| 35–39 | 174 | 0.42 | 3.87 | 0.01 | 0.04 | 0.14 | 0.10 | 0.02 |
| 40+ | 21 | 0.31 | 15.23 | 0.00 | 0.01 | 0.02 | 0.02 | 0.00 |
| TTP-All | |||||||
|---|---|---|---|---|---|---|---|
| Parameters | Fecundability | ||||||
| Age (y) Group | n | q (Sterile) | First Quartile | Median | Third Quartile | ||
| All ages | 1264 | 1.01 | 6.60 | 0.02 | 0.04 | 0.10 | 0.19 |
| 24 or younger | 91 | 1.60 | 5.92 | 0.03 | 0.10 | 0.18 | 0.29 |
| 25–29 | 561 | 1.74 | 10.47 | 0.02 | 0.07 | 0.12 | 0.19 |
| 30–34 | 417 | 0.82 | 5.90 | 0.00 | 0.03 | 0.09 | 0.18 |
| 35–39 | 174 | 0.61 | 5.97 | 0.00 | 0.02 | 0.05 | 0.13 |
| 40+ | 21 | 0.29 | 20.22 | 0.00 | 0.00 | 0.00 | 0.02 |
| TTP-Natural | |||||||
| Parameters | Fecundability | ||||||
| Age (y) Group | n | q (Sterile) | First Quartile | Median | Third Quartile | ||
| All ages | 1264 | 1.88 | 10.73 | 0.14 | 0.05 | 0.11 | 0.19 |
| 24 or younger | 91 | 1.72 | 6.35 | 0.04 | 0.10 | 0.18 | 0.29 |
| 25–29 | 561 | 2.58 | 15.08 | 0.06 | 0.07 | 0.13 | 0.19 |
| 30–34 | 417 | 1.75 | 9.40 | 0.22 | 0.02 | 0.10 | 0.19 |
| 35–39 | 174 | 1.31 | 8.64 | 0.30 | 0.00 | 0.06 | 0.15 |
| 40+ | 21 | 0.31 | 15.21 | 0.00 | 0.00 | 0.01 | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konishi, S.; Kariya, F.; Hamasaki, K.; Takayasu, L.; Ohtsuki, H. Fecundability and Sterility by Age: Estimates Using Time to Pregnancy Data of Japanese Couples Trying to Conceive Their First Child with and without Fertility Treatment. Int. J. Environ. Res. Public Health 2021, 18, 5486. https://doi.org/10.3390/ijerph18105486
Konishi S, Kariya F, Hamasaki K, Takayasu L, Ohtsuki H. Fecundability and Sterility by Age: Estimates Using Time to Pregnancy Data of Japanese Couples Trying to Conceive Their First Child with and without Fertility Treatment. International Journal of Environmental Research and Public Health. 2021; 18(10):5486. https://doi.org/10.3390/ijerph18105486
Chicago/Turabian StyleKonishi, Shoko, Fumiko Kariya, Kisuke Hamasaki, Lena Takayasu, and Hisashi Ohtsuki. 2021. "Fecundability and Sterility by Age: Estimates Using Time to Pregnancy Data of Japanese Couples Trying to Conceive Their First Child with and without Fertility Treatment" International Journal of Environmental Research and Public Health 18, no. 10: 5486. https://doi.org/10.3390/ijerph18105486
APA StyleKonishi, S., Kariya, F., Hamasaki, K., Takayasu, L., & Ohtsuki, H. (2021). Fecundability and Sterility by Age: Estimates Using Time to Pregnancy Data of Japanese Couples Trying to Conceive Their First Child with and without Fertility Treatment. International Journal of Environmental Research and Public Health, 18(10), 5486. https://doi.org/10.3390/ijerph18105486






