Occupational Exposures and Environmental Health Hazards of Military Personnel
Abstract
1. Introduction
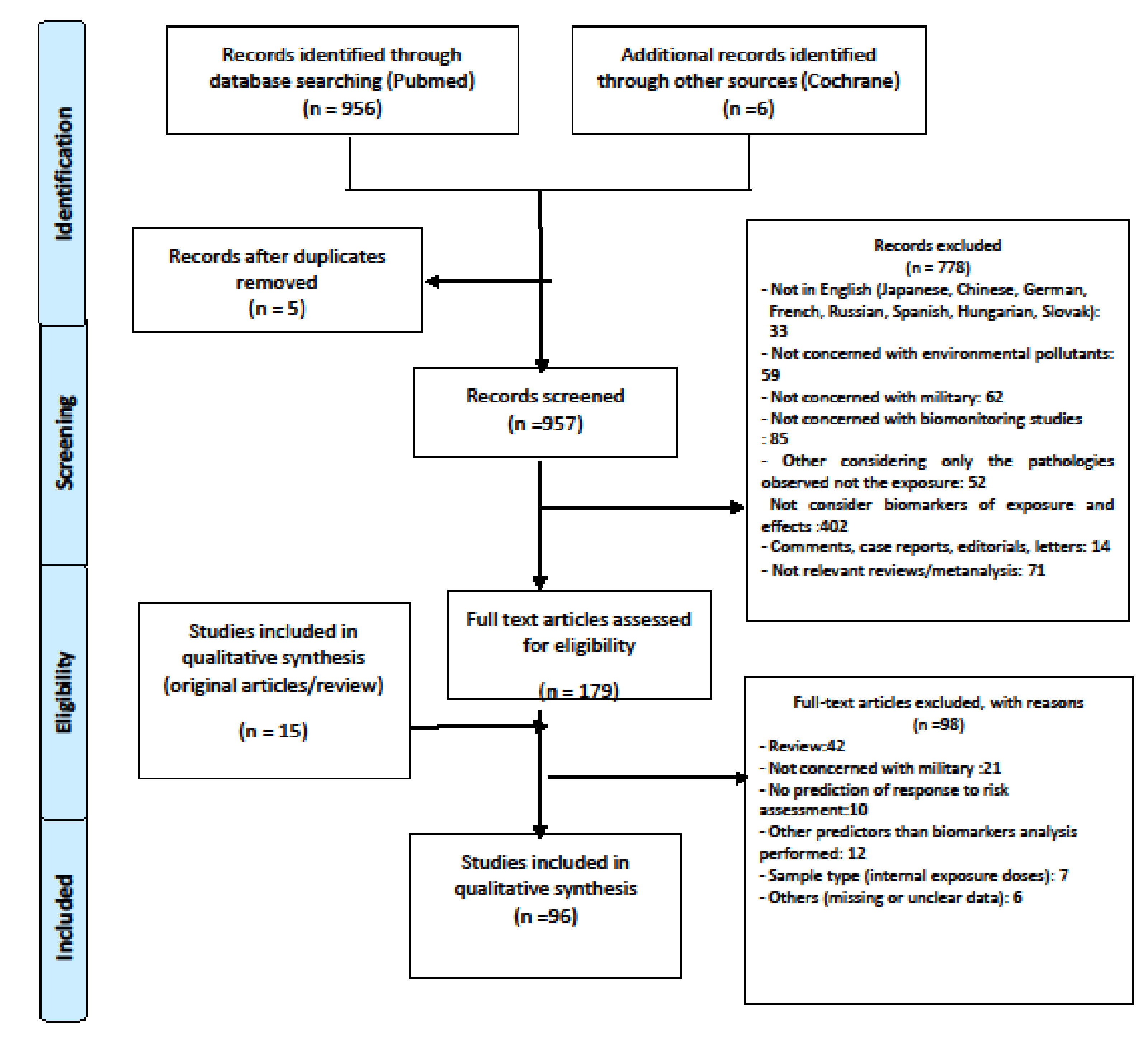
2. Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Identification and Selection of Studies
3. Sulfur Mustard
4. Sarin and Cyclosarin
5. Organochlorinates
5.1. Herbicides
5.2. Pesticides
5.3. Cs Gas (Tear Gas)
6. Combustion Products
6.1. Oil Combustion
6.2. Cooking Oil Fumes
6.3. Open Pit Burning
7. Fuel
7.1. Jet Fuel
7.2. Submarine Fuel
8. Firing Ranges
9. Sunlight Exposure
10. Electromagnetic Fields
11. Ionizing Radiations
12. Discussion
13. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Halverson, M.A.J.K.M.; Lewis, J.A.; Jackson, D.A.; Chen, J.; Dennis, W.; Brennan, L.; Baer, C.; Permenter, M.; Krakauer, T. Environmental Hazard In Vitro Biomarker Discovery Tools. Report 2007. Available online: https://pdfs.semanticscholar.org/ab35/f7390ee57d5395e02ece96cde33e72516373.pdf (accessed on 16 May 2021).
- Wallace, M.A.; Kormos, T.M.; Pleil, J.D. Blood-borne biomarkers and bioindicators for linking exposure to health effects in environmental health science. J. Toxicol. Environ. Health B Crit. Rev. 2016, 19, 380–409. [Google Scholar] [CrossRef]
- Bolognesi, C.; Migliore, L.; Lista, F.; Caroli, S.; Patriarca, M.; De Angelis, R.; Capocaccia, R. Biological monitoring of Italian soldiers deployed in Iraq. Results of the SIGNUM project. Int. J. Hyg. Environ. Health 2016, 219, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Skabelund, A.J.; Rawlins, F.A., 3rd; McCann, E.T.; Lospinoso, J.A.; Burroughs, L.; Gallup, R.A.; Morris, M. Pulmonary Function and Respiratory Health of Military Personnel Before Southwest Asia Deployment. Respir. Care 2017, 62, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.J.; Skabelund, A.J.; Rawlins, F.A., 3rd; Gallup, R.A.; Aden, J.K.; Holley, A.B. Study of Active Duty Military Personnel for Environmental Deployment Exposures: Pre- and Post-Deployment Spirometry (STAMPEDE II). Respir. Care 2019, 64, 536–544. [Google Scholar] [CrossRef] [PubMed]
- May, L.M.; Weese, C.; Ashley, D.L.; Trump, D.H.; Bowling, C.M.; Lee, A.P. The recommended role of exposure biomarkers for the surveillance of environmental and occupational chemical exposures in military deployments: Policy considerations. Mil. Med. 2004, 169, 761–767. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Patterson, S.L.; Dancy, B.C.R.; Ippolito, D.L.; Stallings, J.D. Potential and Actual Health Hazards in the Dense Urban Operational Environment: Critical Gaps and Solutions for Military Occupational Health. J. Occup. Environ. Med. 2017, 59, e197–e203. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.I.; Mallon, C.T.; Hopke, P.K.; Uppal, K.; Go, Y.M.; Rohrbeck, P. Deployment-Associated Exposure Surveillance With High-Resolution Metabolomics. J. Occup. Environ. Med. 2016, 58, S12–S21. [Google Scholar] [CrossRef]
- Izzotti, A. Detection of modified DNA nucleotides by postlabeling procedures. Toxicol. Meth 1998, 8, 175–205. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Hefazi, M. Comparison of early and late toxic effects of sulfur mustard in Iranian veterans. Basic Clin. Pharm. Toxicol. 2008, 99, 273–282. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Mousavi, S.H.; Balali-Mood, B. Chronic health effects of Sulphur mustard exposure with special reference to Iranian veterans. Emerg. Health Threat. J. 2008, 1, e7. [Google Scholar]
- Kehe, K.; Thiermann, H.; Balszuweit, F.; Eyer, F.; Steinritz, D.; Zilker, T. Acute effects of sulfur mustard injury—Munich experiences. Toxicology 2009, 263, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Harchegani, A.B.; Tahmasbpour, E.; Borna, H.; Imamy, A.; Ghanei, M.; Shahriary, A. Free radical production and oxidative stress in lung tissue of patients exposed to sulfur mustard: An overview of cellular molecular mechanisms. Chem. Res. Toxicol. 2018, 31, 211–222. [Google Scholar] [CrossRef] [PubMed]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Chemical Agents and Related Occupations. Lyon (FR): International Agency for Research on Cancer; 2012. (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 100F.). Available online: https://www.ncbi.nlm.nih.gov/books/NBK304416/ (accessed on 16 May 2021).
- Ghazanfari, T.; Faghihzadeh, S.; Aragizadeh, H.; Soroush, M.R.; Yaraee, R.; Hassan, Z.M. Sardasht-Iran Cohort Study of Chemical Warfare Victims: Design and Methods. Arch. Iran. Med. 2009, 12, 5–14. [Google Scholar] [PubMed]
- Ghazanfari, T.; Yaraee, R.; Kariminia, A.; Ebtekar, M.; Faghihzadeh, S.; Vaez-Mahdavi, M.R. Alterations in the serum levels of chemokines 20 years after sulfur mustard exposure: Sardasht-Iran Cohort Study. Int. Immunopharmacol. 2009, 9, 1471–1476. [Google Scholar] [CrossRef] [PubMed]
- Balali-Mood, M. First report of delayed toxic effects of Yperite poisoning in Iranian fighters. In Proceedings of the Second World Congress on New Compounds in Biological and Chemical Warfare: Toxicological Evaluation; Heyndrickx, B., Ed.; Ghent University Press: Ghent, Belgium, 2017; pp. 489–495. [Google Scholar]
- Khateri, S.; Balali-Mood, M.; Blain, P.; Williams, F.; Jowsey, P.; Soroush, M.R. DNA damage and repair proteins in cellular response to sulfur mustard in Iranian veterans more than two decades after exposure. Toxicol. Lett. 2018, 293, 67–72. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Hefazi, M.; Mahmoudi, M.; Jalali, E.; Attaran, D.; Maleki, M. Long-term complications of sulphur mustard poisoning in severely intoxicated Iranian veterans. Fundam. Clin. Pharm. 2005, 19, 713–721. [Google Scholar] [CrossRef]
- Namazi, S.; Niknahad, H.; Razmkhah, H. Long-term complications of Sulphur mustard poisoning in intoxicated Iranian veterans. J. Med. Toxicol. 2009, 5, 191–195. [Google Scholar] [CrossRef]
- Zojaji, R.; Balali-Mood, M.; Mirzadeh, M.; Saffari, A.; Maleki, M. Delayed head and neck complications of sulfur mustard poisoning in Iranian veterans. J. Laryngol. Otol. 2009, 123, 1150–1154. [Google Scholar] [CrossRef]
- Darchini-Maragheh, E.; Balali-Mood, M.; Malaknezhad, M.; Mousavi, S.R. Progressive delayed respiratory complications of sulfur mustard poisoning in 43 Iranian veterans, three decades after exposure. Hum. Exp. Toxicol. 2018, 37, 175–184. [Google Scholar] [CrossRef]
- Hefazi, M.; Attaran, D.; Mahmoudi, M.; Balali-Mood, M. Late respiratory complications of mustard gas poisoning in Iranian veterans. Inhal. Toxicol. 2005, 17, 587–592. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Afshari, R.; Zojaji, R.; Kahrom, H.; Kamrani, M.; Attaran, D. Delayed toxic effects of sulfur mustard on respiratory tract of Iranian veterans. Hum. Exp. Toxicol. 2011, 30, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, H.; Javadi, M.A.; Ardestani, S.K.; Mahmoudi, M.; Pourfarzam, S.; Mahdavi, M.R.V. Alteration in inflammatory mediators in seriously eye-injured war veterans, long-term after sulfur mustard exposure. Int. Immunopharmacol. 2020, 80, 105897. [Google Scholar] [CrossRef] [PubMed]
- Ghazanfari, T.; Mostafaie, A.; Talebi, F.; Mahdavi, M.R.V.; Yarmohammadi, M.E.; Askari, N. Alteration in serum levels of immunoglobulins in seriously eye-injured long-term following sulfur-mustard exposure. Int. Immunopharmacol. 2020, 80, 105895. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Hefazi, M.; Rastin, M.; Balali-Mood, M. Long-term hematological and immunological complications of sulfur mustard poisoning in Iranian veterans. Int. Immunopharmacol. 2005, 5, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Azizi, T.; Moghadam, M.R.; Amin, G.; Parvin, S.; Sahebkar, A. Oral health status among Iranian veterans exposed to sulfur mustard: A case-control study. J. Clin. Exp. Dent. 2015, 7, e192–e196. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tahmasbpour, E.; Ghanei, M.; Qazvini, A.; Vahedi, E.; Panahi, Y. Gene expression profile of oxidative stress and antioxidant defense in lung tissue of patients exposed to sulfur mustard. Mutat. Res. 2016, 800–801, 12–21. [Google Scholar] [CrossRef]
- Tahmasbpour Marzony, E.; Ghanei, M.; Panahi, Y. Oxidative stress and altered expression of peroxiredoxin genes family (PRDXS) and sulfiredoxin-1 (SRXN1) in human lung tissue following exposure to sulfur mustard. Exp. Lung Res. 2016, 42, 217–226. [Google Scholar] [CrossRef]
- Tahmasbpour, M.E.; Nejad-Moghadam, A.; Ghanei, M.; Panahi, Y. Sulfur mustard causes oxidants/antioxidants imbalance through the overexpression of free radical producing-related genes in human mustard lungs. Environ. Toxicol. Pharmacol. 2016, 45, 187–192. [Google Scholar] [CrossRef]
- Kaboudanian, A.S.; Taravati, A.; Kianmehr, Z.; Hajizadeh Dastjerdi, A.; Pourfarzam, S.; Soroush, M.R. Altered levels of GST activity, Vit C, TPX and Cu in individuals with long-term sulfur mustard-induced lung complications. Inhal Toxicol. 2018, 30, 483–491. [Google Scholar] [CrossRef]
- Behboudi, H.; Noureini, S.K.; Ghazanfari, T.; Ardestani, S.K. DNA damage and telomere length shortening in the peripheral blood leukocytes of 20 years SM-exposed veterans. Int. Immunopharmacol. 2018, 61, 37–44. [Google Scholar] [CrossRef]
- Behravan, E.; Moallem, S.A.; Kalalinia, F.; Ahmadimanesh, M.; Blain, P.; Jowsey, P.; Khateri, S.; Forghanifard, M.M.; Balali-Mood, M. Telomere shortening associated with increased levels of oxidative stress in sulfur mustard-exposed Iranian veterans. Mutat. Res. Gen. Tox. En. 2018, 834, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Gharbi, S.; Khateri, S.; Soroush, M.R.; Shamsara, M.; Naeli, P.; Najafi, A. MicroRNA expression in serum samples of sulfur mustard veterans as a diagnostic gateway to improve care. PLoS ONE 2018, 13, e0194530. [Google Scholar] [CrossRef]
- Khateri, S.; Ghanei, M.; Keshavarz, S.; Soroush, M.; Haines, D. Incidence of lung, eye, and skin lesions as late complications in 34,000 Iranians with wartime exposure to mustard agent. Occup. Environ. Med. 2003, 45, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.C.; Smith, T.C.; Knoke, J.D.; Heller, J.M. The postwar hospitalization experience of Gulf War Veterans possibly exposed to chemical munitions destruction at Khamisiyah, Iraq. Am. J. Epidemiol. 1999, 150, 532–540. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kang, H.K.; Bullman, T.A. Mortality among US veterans of the Persian Gulf War: 7-year follow-up. Am. J. Epidemiol. 2001, 154, 399–405. [Google Scholar] [CrossRef]
- Bullman, T.A.; Mahan, C.M.; Kang, H.K.; Page, W.F. Mortality in US Army Gulf War veterans exposed to 1991 Khamisiyah chemical munitions destruction. Am. J. Public Health 2005, 95, 1382–1388. [Google Scholar] [CrossRef]
- Chao, L.L.; Rothlind, J.C.; Cardenas, V.A.; Meyerhoff, D.J.; Weiner, M.W. Effects of low-level exposure to sarin and cyclosarin during the 1991 Gulf War on brain function and brain structure in US veterans. Neurotoxicology 2010, 31, 493–501. [Google Scholar] [CrossRef]
- Chao, L.L.; Abadjian, L.; Hlavin, J.; Meyerhoff, D.J.; Weiner, M.W. Effects of low-level sarin and cyclosarin exposure and Gulf War Illness on Brain Structure and Function: A study at 4 T. Neurotoxicology 2011, 32, 814–822. [Google Scholar] [CrossRef]
- Chao, L.L.; Zhang, Y.; Buckley, S. Effects of low-level sarin and cyclosarin exposure on white matter integrity in Gulf War Veterans. Neurotoxicology 2015, 48, 239–248. [Google Scholar] [CrossRef]
- Chao, L.L.; Zhang, Y. Effects of low-level sarin and cyclosarin exposure on hippocampal microstructure in Gulf War Veterans. Neurotoxicol. Teratol. 2018, 68, 36–46. [Google Scholar] [CrossRef]
- Heaton, K.J.; Palumbo, C.L.; Proctor, S.P.; Killiany, R.J.; Yurgelun-Todd, D.A.; White, R.F. Quantitative magnetic resonance brain imaging in US army veterans of the 1991 Gulf War potentially exposed to sarin and cyclosarin. Neurotoxicology 2007, 28, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Proctor, S.P.; Heaton, K.J.; Heeren, T.; White, R.F. Effects of sarin and cyclosarin exposure during the 1991 Gulf War on neurobehavioral functioning in US army veterans. Neurotoxicology 2006, 27, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Fulco, C.E.; Liverman, C.T.; Sox, H.C. Gulf War and Health. Depleted Uranium, Sarin, Pyridostigmine Bromide, Vaccines. In Institute of Medicine (US) Committee on Health Effects Associated with Exposures During the Gulf War; National Academies Press (US): Washington, DC, USA, 2000; Volume 1. [Google Scholar]
- Tochigi, M.; Umekage, T.; Otani, T.; Kato, T.; Iwanami, A.; Asukai, N. Serum cholesterol, uric acid and cholinesterase in victims of the Tokyo subway sarin poisoning: A relation with post-traumatic stress disorder. Neurosci. Res. 2002, 44, 267–272. [Google Scholar] [CrossRef]
- Lurker, P.A.; Berman, F.; Clapp, R.W.; Stellman, J.M. Post-Vietnam military herbicide exposures in UC-123 Agent Orange spray aircraft. Environ. Res. 2014, 130, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.H.; Hewitt, A.; Armitage, J.; Solomon, K.; Watkins, D.K.; Ginevan, M.E. Exposure to TCDD from base perimeter application of Agent Orange in Vietnam. Sci. Total Environ. 2015, 511, 82–90. [Google Scholar] [CrossRef]
- Cypel, Y.; Kang, H. Mortality patterns of army chemical corps veterans who were occupationally exposed to herbicides in Vietnam. AEP 2010, 20, 339–346. [Google Scholar] [CrossRef][Green Version]
- Yi, S.W.; Hong, J.S.; Ohrr, H.; Yi, J.J. Agent Orange exposure and disease prevalence in Korean Vietnam veterans: The Korean veterans’ health study. Environ. Res. 2014, 133, 56–65. [Google Scholar] [CrossRef]
- Yi, S.W.; Ryu, S.Y.; Ohrr, H.; Hong, J.S. Agent Orange exposure and risk of death in Korean Vietnam veterans: Korean Veterans Health Study. Int. J. Epidemiol. 2014, 43, 1825–1834. [Google Scholar] [CrossRef]
- Patterson, A.T.; Kaffenberger, B.H.; Keller, R.; Elston, D. Skin diseases associated with Agent Orange and other organochlorine exposures. J. Am. Acad. Derm. 2016, 74, 143–170. [Google Scholar] [CrossRef]
- Pham, N.T.; Nishijo, M.; Pham, T.T.; Tran, N.N.; Le, V.Q.; Tran, H.A. Perinatal dioxin exposure and neurodevelopment of 2-year-old Vietnamese children in the most contaminated area from Agent Orange in Vietnam. Sci. Total Environ. 2019, 678, 217–226. [Google Scholar] [CrossRef]
- Appel, K.E.; Gundert-Remy, U.; Fischer, H.; Faulde, M.; Mross, K.G.; Letzel, S. Risk assessment of Bundeswehr (German Federal Armed Forces) permethrin-impregnated battle dress uniforms (BDU). Int. J. Hyg. Environ. Health 2018, 211, 88–104. [Google Scholar] [CrossRef] [PubMed]
- Rossbach, B.; Appel, K.E.; Mross, K.G.; Letzel, S. Uptake of permethrin from impregnated clothing. Toxicol. Lett. 2010, 192, 50–55. [Google Scholar] [CrossRef]
- Kegel, P.; Letzel, S.; Rossbach, B. Biomonitoring in wearers of permethrin impregnated battle dress uniforms in Afghanistan and Germany. Occup. Environ. Med. 2014, 71, 112–117. [Google Scholar] [CrossRef]
- DeBeer, B.B.; Davidson, D.; Meyer, E.C.; Kimbrel, N.A.; Gulliver, S.B.; Morissette, S.B. The Association between Toxic Exposures and Chronic Multisymptom Illness in Veterans of the Wars of Iraq and Afghanistan. J. Occup. Environ. Med. 2017, 59, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.; Krengel, M.; Bradford, W.; Stone, C.; Thompson, T.A.; Heeren, T.; White, R.F. Neuropsychological functioning in military pesticide applicators from the Gulf War: Effects on information processing speed, attention and visual memory. Neurotoxicol. Teratol. 2018, 65, 1–13. [Google Scholar] [CrossRef]
- Buchanan, M.A.; Stubner, A.; Hout, J.J.; Brueggemeyer, M.; Bragg, W. Association between exposure to o-chlorobenzylidene malononitrile (CS riot control agent) and urinary metabolite 2-chlorohippuric acid in U.S. Army Mask Confidence Training. J. Occup. Environ. Hyg. 2017, 14, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Hout, J.J.; Kluchinsky, T.; LaPuma, P.T.; White, D.W. Evaluation of CS (o-chlorobenzylidene malononitrile) concentrations during U.S. Army mask confidence training. J. Environ. Health 2011, 74, 18–21. [Google Scholar]
- Hout, J.J.; White, D.W.; Stubner, A.; Stevens, M.; Knapik, J.J. O-chlorobenzylidene malononitrile (CS riot control agent) exposure in a U.S. Army basic combat training cohort. J. Environ. Health 2014, 77, 14–21. [Google Scholar]
- Hout, J.J.; White, D.W.; Artino, A.R.; Knapik, J.J. O-chlorobenzylidene malononitrile (CS riot control agent) associated acute respiratory illnesses in a U.S. Army basic combat training cohort. Mil. Med. 2014, 179, 793. [Google Scholar] [CrossRef]
- Petruccelli, B.P.; Goldenbaum, M.; Scott, B.; Lachiver, R.; Kanjarpane, D.; Elliott, E.; Francis, M.; McDiarmid, M.A.; Deeter, D. Health effects of the 1991 Kuwait oil fires: A survey of US army troops. J. Occup. Environ. Med. 1999, 41, 433–439. [Google Scholar] [CrossRef]
- Coombe, M.D.; Drysdale, S.F. Assessment of the effects of atmospheric oil pollution in post war Kuwait. J. R. Army Med. Corps. 1993, 139, 95–97. [Google Scholar] [CrossRef]
- Lange, J.L.; Schwartz, D.A.; Doebbeling, B.N.; Heller, J.K.; Thorne, P.S. Exposures to the Kuwait oil fires and their association with asthma and bronchitis among gulf war veterans. Environ. Health Perspect. 2002, 110, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Heller, J.M. Oil well fires of Operation Desert Storm—Defining troop exposures and determining health risks. Mil. Med. 2011, 176, 46–51. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Etzel, R.A.; Ashley, D.L. Volatile organic compounds in the blood of persons in Kuwait during the oil fires. Int. Arch. Occup. Environ. Health 1994, 66, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Poirier, M.C.; Weston, A.; Schoket, B.; Shamkhani, H.; Pan, C.F.; McDiarmid, M.A. Biomonitoring of United States Army soldiers serving in Kuwait in 1991. Cancer Epidemiol. Biomark. Prev. 1998, 7, 545–551. [Google Scholar]
- Deeter, D.P. The Kuwait Oil Fire Health Risk Assessment Biological Surveillance Initiative. Mil. Med. 2011, 176, 52–55. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lai, C.H.; Jaakkola, J.J.K.; Chuang, C.Y.; Liou, S.H.; Lung, S.C.; Loh, C.H. Exposure to cooking oil fumes and oxidative damages: A longitudinal study in Chinese military cooks. J. Exp. Sci. Environ. Epidemiol. 2013, 23, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.A.; Smith, B.; Granado, N.S.; Boyko, E.J.; Gackstetter, G.D.; Ryan, M.A. Millennium Cohort Study Team. Newly reported lupus and rheumatoid arthritis in relation to deployment within proximity to a documented open-air burn pit in Iraq. J. Occup. Environ. Med. 2012, 54, 698–707. [Google Scholar] [CrossRef]
- Powell, T.M.; Smith, T.C.; Jacobson, I.G.; Boyko, E.J.; Hooper, T.I.; Gackstetter, G.D. Millennium Cohort Study Team. Prospective assessment of chronic multisymptom illness reporting possibly associated with open-air burn pit smoke exposure in Iraq. J. Occup. Environ. Med. 2012, 54, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.; Wong, C.A.; Boyko, E.J.; Phillips, C.J.; Gackstetter, G.D.; Ryan, M.A. Millennium Cohort Study Team. The effects of exposure to documented open-air burn pits on respiratory health among deployers of the Millennium Cohort Study. J. Occup. Environ. Med. 2012, 54, 708–716. [Google Scholar] [CrossRef]
- Masiol, M.; Mallon, T.; Haines, K.M., Jr.; Utell, M.J.; Hopke, P.K. Airborne Dioxins, Furans and Polycyclic Aromatic Hydrocarbons Exposure to military personnel in Iraq. J. Occup. Environ. Med. 2016, 58, S22–S30. [Google Scholar] [CrossRef]
- Masiol, M.; Mallon, T.; Haines, K.M., Jr.; Utell, M.J.; Hopke, P.K. Source apportionment of airborne dioxins, furans and polycyclic aromatic hydrocarbons at a U.S. forward operating air base during the Iraq war. J. Occup. Environ. Med. 2016, 58, S31–S37. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rohrbeck, P.; Hu, Z.; Mallon, T.M. Assessing health outcomes after environmental exposures associated with open pit burning in deployed U.S. service members. J. Occup. Environ. Med. 2016, 58, S104–S110. [Google Scholar] [CrossRef] [PubMed]
- Abraham, J.H.; Eick-Cost, A.; Clark, L.L.; Hu, Z.; Baird, C.P.; DeFraites, R.; Tobler, S.K. A retrospective cohort study of military deployment and postdeployment medical encounters for respiratory conditions. Mil. Med. 2014, 179, 5–540. [Google Scholar] [CrossRef] [PubMed]
- Woeller, C.F.; Thatcher, T.H.; Van Twisk, D.; Pollock, S.J.; Croadsdell, A.; Hopke, P.K. MicroRNAs as novel biomarkers of deployment status and exposure to polychlorinated dibenzo-p-dioxins/dibenzofurans. J. Occup. Environ. Med. 2016, 58, S89–S96. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Carroll-Haddad, A.; Brown, N.; Utell, M.J.; Mallon, T.; Hopke, P.K. Polycyclic aromatic hydrocarbons and polychlorinated dibenzo-p-dioxins/dibenzofurans in microliter samples of human serum as exposure indicators. J. Occup. Environ. Med. 2016, 58, S72–S79. [Google Scholar] [CrossRef]
- Dalgard, C.L.; Polston, K.F.; Sukumar, G.; Mallon, C.T.; Wilkerson, M.D.; Pollard, H.B. MicroRNA expression profiling of the armed forces health surveillance branch cohort for identification of “Enviro-miRs” associated with deployment-based environmental exposure. J. Occup. Environ. Med. 2016, 58, S97–S103. [Google Scholar] [CrossRef]
- Bukowinski, A.T.; DeSciciolo, C.; Conlin, A.M.; Ryan, K.M.A.; Sevick, C.J.; Smith, T. Birth defects in infants born in 1998-2004 to men and women serving in the U.S. military during the 1990–1991 Gulf War era. Birth Defects Res. A Clin. Mol. Teratol. 2012, 94, 721–728. [Google Scholar] [CrossRef]
- Conlin, A.M.; DeScisciolo, C.; Sevick, C.J.; Bukowinski, A.T.; Phillips, C.J.; Smith, T.C. Birth outcomes among military personnel after exposure to documented open-air burn pits before and during pregnancy. J. Occup. Environ. Med. 2012, 54, 689–697. [Google Scholar] [CrossRef]
- Falvo, M.J.; Helmer, D.A.; Klein, J.C.; Osinubi, O.Y.; Ndirangu, D.; Patrick-DeLuca, L.A. Isolated diffusing capacity reduction is a common clinical presentation in deployed Iraq and Afghanistan veterans with deployment-related environmental exposures. Clin. Respir. J. 2018, 12, 795–798. [Google Scholar] [CrossRef]
- Tekbas, F.; Vaizoğlu, S.A.; Guleç, M.; Hasde, M.; Güler, C. Smoking prevalence in military men, and factors affecting this. Mil. Med. 2002, 167, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.; Occhiolini, L.; Di Mascio, R.; Vellante, P.; Colagrande, V.; Ballone, E. Smoking habits in a sample of young Italian soldiers. Mil. Med. 2006, 171, 69–73. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lemasters, G.K.; Livingston, G.K.; Lockey, J.E.; Olsen, D.M.; Shukla, R.; New, G. Genotoxic changes after low-level solvent and fuel exposure on aircraft maintenance personnel. Mutagenesis 1997, 12, 237–243. [Google Scholar] [CrossRef][Green Version]
- Proctor, S.P.; Heaton, K.J.; Smith, K.W.; Rodrigues, E.R.; Widing, D.E.; Herrick, R. The Occupational JP8 Exposure Neuroepidemiology Study (OJENES): Repeated workday exposure and central nervous system functioning among US Air Force personnel. Neurooxicology 2011, 32, 799–808. [Google Scholar] [CrossRef]
- Smith, K.W.; Proctor, S.P.; Ozonoff, A.L.; McClean, M.D. Urinary biomarkers of occupational jet fuel exposure among Air Force personnel. J. Expo. Sci. Environ. Epidemiol. 2012, 22, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Merchant-Borna, K.M.; Rodrigues, E.G.; Smith, K.W.; Proctor, S.P.; McClean, M.D. Characterization of inhalation exposure to jet fuel among U.S. Air Force personnel. Ann. Occup. Hyg. 2012, 56, 736–745. [Google Scholar]
- Rodrigues, E.G.; Smith, K.; Maule, A.L.; Sjodin, A.; Li, Z.; Romanoff, L. Urinary polycyclic aromatic hydrocarbon (OH-PAH) metabolite concentrations and the effect of GST polymorphisms among US Air Force personnel exposed to jet fuel. J. Occup. Environ. Med. 2014, 56, 465–471. [Google Scholar] [CrossRef][Green Version]
- Maule, A.L.; Proctor, S.P.; Blount, B.C.; Chambers, D.M.; McClean, D. Volatile Organic Compounds in blood as biomarker of exposure to JP-8 jet fuel among US Air Force Personnel. J. Occup. Environ. Med. 2016, 58, 24–29. [Google Scholar] [CrossRef]
- Heaton, K.J.; Maule, A.L.; Smith, K.W.; Rodrigues, E.G.; McClean, M.D.; Proctor, S.P. JP8 exposure and neurocognitive performance among US Air Force personnel. Neurotoxicology 2017, 62, 170–180. [Google Scholar] [CrossRef]
- Fuente, A.; Hickson, L.; Morata, T.C.; Williams, W.; Khan, A.; Fuentes-Lopez, E. Jet fuel exposure and auditory outcomes in Australian air force personnel. BMC Public Health 2019, 19, 675. [Google Scholar] [CrossRef]
- Duplessis, C.A.; Gumpert, B. Inhalational diesel exhaust exposure in submariners: Observational study. Mil. Med. 2008, 173, 671–676. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dean, M.R. Chronic myeloid leukaemia and occupational exposure to benzene in a Royal Navy submariner. J. R. Nav. Med. Serv. Spring 1996, 82, 28–33. [Google Scholar]
- Greenberg, N.; Frimer, R.; Meyer, R.; Derazne, E.; Chodick, G. Lead exposure in military outdoor firing ranges. Mil. Med. 2016, 181, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Park, W.J.; Lee, S.H.; Lee, S.H.; Yoon, H.S.; Moon, J.D. Occupational lead exposure from indoor firing ranges in Korea. J. Korean Med. Sci. 2016, 31, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, L.; Borraccia, V.; Corfiati, M.; Mantineo, G.A.; Caciari, T.; Marino, M. Lead exposure in firearms instructors of the Italian State Police. Med. Lav. 2010, 101, 30–37. [Google Scholar] [PubMed]
- Marshall, T.M.; Dardia, G.P.; Colvin, K.L.; Nevin, R.; Macrellis, J. Neurotoxicity Associated with Traumatic Brain Injury, Blast, Chemical, Heavy Metal and Quinoline Drug Exposure. Altern. Ther. Health Med. 2019, 25, 28–34. [Google Scholar] [PubMed]
- Ramani, M.L.; Bennett, R.G. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J. Am. Acad. Derm. 1993, 28, 733–737. [Google Scholar] [CrossRef]
- Maceachern, W. Jillson of A Practical Sunscreen—Red Vet Pet. Arch. Dermatol. 1964, 89, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Page, W.F.; Whiteman, D.; Murphy, M. A comparison of melanoma mortality among WWII veterans of the Pacific and European theaters. Ann. Epidemiol. 2000, 10, 192–195. [Google Scholar] [CrossRef]
- Garland, F.C.; White, M.R.; Garland, C.F.; Shaw, E.; Gorham, E.D. Occupational sunlight exposure and melanoma in the U.S. Navy. Arch. Environ. Health 1990, 45, 261–267. [Google Scholar] [CrossRef]
- Henning, J.S.; Firoz, B.F. Combat dermatology: The prevalence of skin disease in a deployed dermatology clinic in Iraq. J. Drugs Derm. 2010, 9, 210–214. [Google Scholar]
- Lee, T.; Taubman, S.B.; Williams, V.F. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005–2014. MSMR 2016, 23, 2–6. [Google Scholar] [PubMed]
- Brundage, J.F.; Williams, V.F.; Stahlman, S.; McNellis, M.G. Incidence rates of malignant melanoma in relation to years of military service overall and in selected military occupational groups, active component, U.S. Armed Forces, 2001–2015. MSMR 2017, 24, 8–14. [Google Scholar] [PubMed]
- Lea, C.S.; Efird, J.T.; Toland, A.E.; Lewis, D.R.; Phillips, C.J. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000–2007. Mil. Med. 2014, 179, 247–253. [Google Scholar] [CrossRef]
- Zhou, J.; Enewold, L.; Zahm, S.H.; Devesa, S.S.; Anderson, W.F.; Potter, J.F. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol. Biomark. Prev. 2011, 20, 318–323. [Google Scholar] [CrossRef]
- Strand, L.A.; Martinsen, J.I.; Borud, E.K. Cancer incidence and all-cause mortality in a cohort of 21,582 Norwegian military peacekeepers deployed to Lebanon during 1978–1998. Cancer Epidemiol. 2015, 39, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kapoor, N. Occupational EMF exposure from radar at X and Ku frequency band and plasma catecholamine levels. Bioelectromagnetics 2015, 36, 444–450. [Google Scholar] [CrossRef]
- Sobiech, J.; Kieliszek, J.; Puta, R.; Bartczak, D.; Stankiewicz, W. Occupational exposure to electromagnetic fields in the Polish Armed Forces. Int. J. Occup. Med. Environ. Health 2017, 30, 565–577. [Google Scholar] [CrossRef]
- Dabouis, V.; Arvers, P.; Debouzy, J.C.; Sebbah, C.; Crouzier, D.; Perrin, A. First epidemiological study on occupational radar exposure in the French Navy: A 26-year cohort study. Int. J. Environ. Health Res. 2016, 26, 131–144. [Google Scholar] [CrossRef]
- Hietanen, M. Establishing the health risks of exposure to radiofrequency fields requires multidisciplinary research. Scand. J. Work Environ. Health 2006, 32, 169–170. [Google Scholar] [CrossRef]
- Foster, K.R. Thermal and nonthermal mechanisms of interaction of radio-frequency energy with biological systems. IEEE Trans. Plasma Sci. 2000, 28, 15–23. [Google Scholar] [CrossRef]
- Mortazavi, S.A.; Taeb, S.; Mortazavi, S.M.; Zarei, S.; Haghani, M.; Habibzadeh, P. The Fundamental Reasons Why Laptop Computers should not be Used on Your Lap. J. Biomed. Phys. Eng. 2016, 6, 279–284. [Google Scholar]
- Kamali, K.; Atarod, M.; Sarhadi, S.; Nikbakht, J.; Emami, M.; Maghsoudi, R. Effects of electromagnetic waves emitted from 3G+wi-fi modems on human semen analysis. Urologia 2017, 84, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Baste, V.; Riise, T.; Moen, B.E. Radiofrequency electromagnetic fields; male infertility and sex ratio of offspring. Eur. J. Epidemiol. 2008, 23, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Knave, B. Long-term exposure to electric-fields—Cross-sectional epidemiologic investigation of occupationally exposed workers in high-voltage substations. Scand. J. Work Environ. Health 1979, 5, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, S. Reproductive hazards among workers at high voltage substations. Bioelectromagnetics 1983, 4, 91–101. [Google Scholar] [CrossRef]
- Saadat, M. Offspring sex ratio in men exposed to electromagnetic fields. J. Epidemiol. Community Health 2005, 59, 339. [Google Scholar] [CrossRef][Green Version]
- Takahashi, S.; Imai, N.; Nabae, K.; Wake, K.; Kawai, H.; Wang, J. Lack of adverse effects of whole-body exposure to a mobile telecommunication electromagnetic field on the rat fetus. Radiat. Res 2010, 173, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Gathiram, P.; Kistnasamy, B.; Lalloo, U. Effects of a unique electromagnetic field system on the fertility of rats. Arch. Environ. Occup. Health 2009, 64, 93–100. [Google Scholar] [CrossRef]
- Ohnishi, Y.; Mizuno, F.; Sato, T.; Yasui, M.; Kikuchi, T.; Ogawa, M. Effects of power frequency alternating magnetic fields on reproduction and pre-natal development of mice. J. Toxicol. Sci. 2002, 27, 131–138. [Google Scholar] [CrossRef][Green Version]
- Li, J.H.; Jiang, D.P.; Wang, Y.F.; Yan, J.J.; Guo, Q.Y.; Miao, X.; Lang, H.Y.; Xu, S.L.; Liu, J.Y.; Guo, G.Z. Influence of electromagnetic pulse on the offspring sex ratio of male BALB/c mice. Environ. Toxicol. Pharm. 2017, 54, 155–161. [Google Scholar] [CrossRef] [PubMed]
- McDiarmid, M.A.; Albertini, R.J.; Tucker, J.D.; Vacek, P.M.; Carter, E.W.; Bakhmutsky, M.V. Measures of genotoxicity in Gulf war I veterans exposed to depleted uranium. Environ. Mol. Mutagen 2011, 52, 569–581. [Google Scholar] [CrossRef]
- Bakhmutsky, M.V.; Oliver, M.S.; McDiarmid, M.A.; Squibb, K.S.; Tucker, J.D. Long term depleted uranium exposure in Gulf War I veterans does not cause elevated numbers of micronuclei in peripheral blood lymphocytes. Mutat. Res. 2011, 720, 53–57. [Google Scholar] [CrossRef]
- Albertini, R.J.; Vacek, P.M.; Carter, E.W.; Nicklas, J.A.; Squibb, K.S.; Gucer, P.W. Mutagenicity monitoring following battlefield exposures: Longitudinal study of HPRT mutations in Gulf War I veterans exposed to depleted uranium. Environ. Mol. Mutagen 2015, 56, 581–593. [Google Scholar] [CrossRef] [PubMed]
- McDiarmid, M.A.; Gaitens, J.M.; Hines, S.; Condon, M.; Roth, T.; Oliver, M. Biologic monitoring and surveillance results for the department of veterans’ affairs’ depleted uranium cohort: Lessons learned from sustained exposure over two decades. Am. J. Ind. Med. 2015, 58, 583–594. [Google Scholar] [CrossRef]
- McDiarmid, M.A.; Gaitens, J.M.; Hines, S.; Condon, M.; Roth, T.; Oliver, M. The, U.S. Department of Veterans’ Affairs depleted uranium exposed cohort at 25 years: Longitudinal surveillance results. Environ. Res. 2017, 152, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Shiu, J.; Gaitens, J.; Squibb, K.S.; Gucer, P.W.; McDiarmid, M.A.; Gaspari, A.A. Significance of dermatologic findings in a cohort of depleted uranium-exposed veterans of Iraqi conflicts. Dermatitis 2015, 26, 142–147. [Google Scholar] [CrossRef]
- Ough, E.A.; Lewis, B.J.; Andrews, W.S.; Bennett, L.G.; Hancock, R.G.; Scott, K. An examination of uranium levels in Canadian forces personnel who served the Gulf War and Kosovo. Health Phys. 2002, 82, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, P.; Talbäck, M.; Lundin, A.; Lagercrantz, B.; Gyllestad, P.E.; Fornell, L. Incidence of cancer among Swedish military and civil personnel involved in UN missions in the Balkans 1989–1999. Occup. Environ. Med. 2004, 61, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Bland, D.; Rona, R.; Coggon, D.; Anderson, J.; Greenberg, N.; Hull, L. Urinary isotopic analysis in the UK Armed Forces: No evidence of depleted uranium absorption in combat and other personnel in Iraq. Occup. Environ. Med. 2007, 64, 834–838. [Google Scholar] [CrossRef]
- Storm, H.H.; Jørgensen, H.O.; Kejs, A.M.; Engholm, G. Depleted uranium and cancer in Danish Balkan veterans deployed 1992-2001. Eur. J. Cancer 2006, 42, 2355–2358. [Google Scholar] [CrossRef] [PubMed]
- Bogers, R.P.; van Leeuwen, F.E.; Grievink, L.; Schouten, L.J.; Kiemeney, L.A.; Schram-Bijkerk, D. Cancer incidence in Dutch Balkan veterans. Cancer Epidemiol. 2013, 37, 550–555. [Google Scholar] [CrossRef]
- Capocaccia, R.; Biselli, R.; Ruggeri, R.; Tesei, C.; Grande, E.; Martina, L. Mortality in Italian veterans deployed in Bosnia-Herzegovina and Kosovo. Eur. J. Public Health 2016, 26, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Holtgrewe, M.; Knaus, A.; Hildebrand, G.; Pantel, J.T.; Santos, M.R.L.; Neveling, K.; Goldmann, J.; Schubach, M.; Jäger, M.; Coutelier, M.; et al. Multisite de novo mutations in human offspring after paternal exposure to ionizing radiation. Sci. Rep. 2018, 8, 14611. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Neri, M.; Vecchio, D.; Puntoni, R. Molecular epidemiology in cancer research. Int. J. Oncol. 1997, 11, 1053–1069. [Google Scholar]
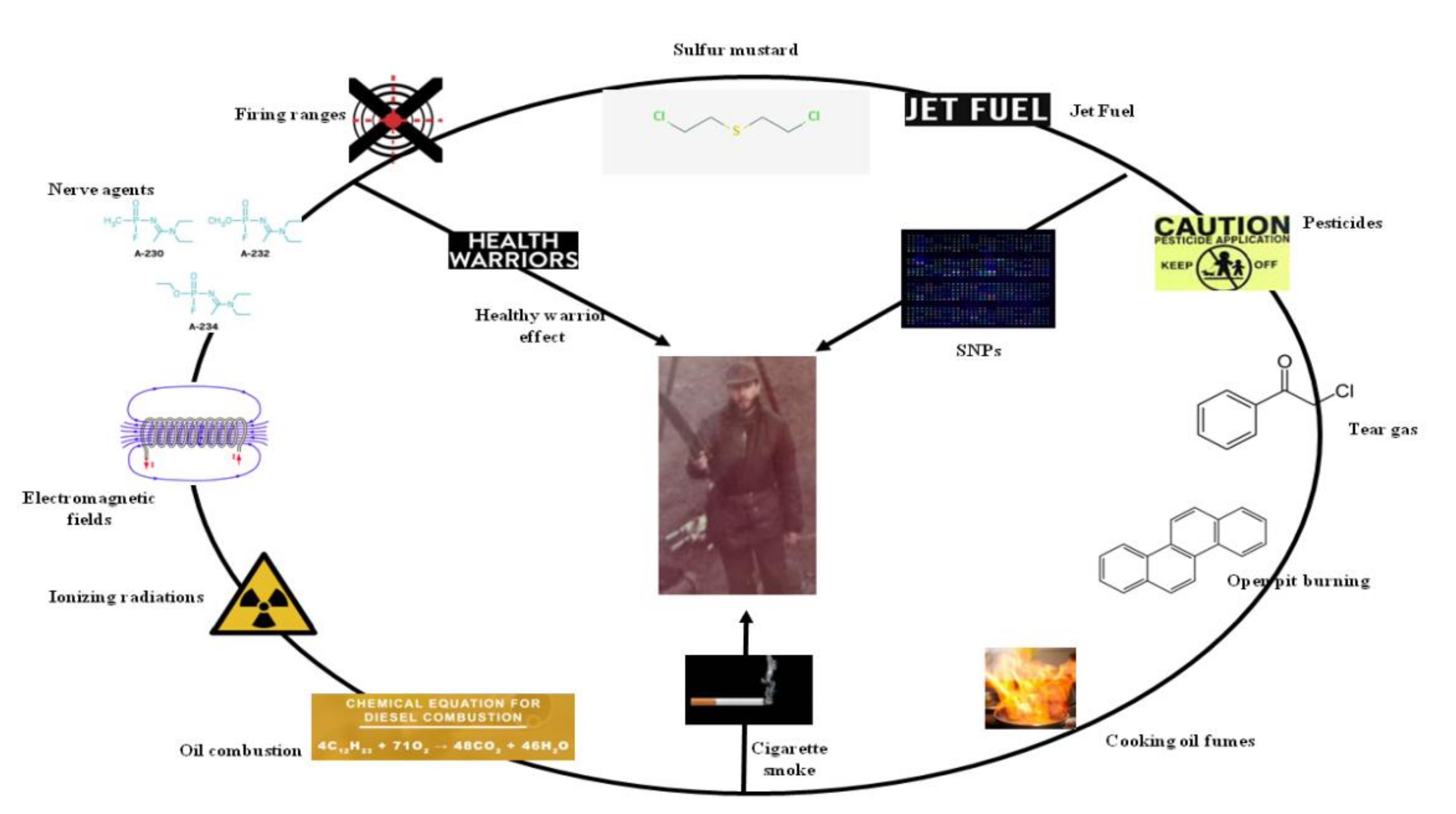
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sulfur Mustard | 236 | 2–28 months | Iran | Iran/Iraq border | - | - | - | Respiratory tract, central nervous system, skin, eyes. | - | [17] | ||
| 75 | 16–20 years | Iran | Khorasan (Iran) | - | Immunity | WBC, RBC, hematocrit, IgM, C3, monocytes, and CD3+ lymphocytes CD16+. | - | Complications in the lungs, peripheral nerves, skin, and eyes. | [19] | |||
| 134 | 17–22 years | Iran | Fars (Iran) | - | - | - | - | Complications in lung, skin, and eyes. | [20] | |||
| 43 | 12–17 years | Iran | Khorasan (Iran) | - | - | - | - | Complications in lungs, peripheral nerves, skin, eyes, head, and neck. | [21] | |||
| 12 | 6–8 days | Iran | Majnoun Island and Persian Gulf | - | - | - | Eye, throat, lung, and gastro-intestinal symptoms | - | [12] | |||
| 372 | 20 years | Iran | Sardasht (Azerbaijan) | - | Immunity | Chemokines, citokines, and inflammatory markers | - | Respiratory function | [15,16] | |||
| 40 | 16–20 years | Iran | Khorasan (Iran) | - | - | - | - | COPD, bronchiectasis, asthma, large airway narrowing, pulmonary fibrosis, simple chronic bronchitis. | [23] | |||
| 43 | 20–25 years | Iran | Iran/Iraq border | - | - | - | - | Dysphonia, post-nasal discharge, lower larynx position, limitation of vocal cords, inflammation of larynx mucosa, COPD, asthma, large-airway narrowing, pulmonary fibrosis, simple chronic bronchitis. | [24] | |||
| 43 | 25 years | Iran | Iran/Iraq border | - | - | - | - | Bronchiectasis, pulmonary fibrosis, and ground-glass attenuation. | [22] | |||
| 75 | 16–20 years | Iran | Khorasan (Iran) | - | Immunity | WBC, RBC, Hct, monocytes, CD3+T-lymphocytes, CD16+56 positive cells, IgM and C3 levels. | - | Increased risk of infections and tumors. | [27] | |||
| 200 | - | Iran | Iran | - | - | - | - | Increased prevalence of oral candidiasis. | [28] | |||
| 11 | 25 years | Iran | Iran | - | Gene expression | Peroxiredoxins (PRDXS) and sulfiredoxin-1 (SRXN1), oxidative stress responsive kinase-1 (OXSR1), forkhead box M1 (FOXM1), glutathione peroxidase-2 (GPX2), aldehyde oxidase 1 (AOX1), myeloperoxidase (MPO), dual oxidase 1 and 2 (DUOX1, DUOX2), thyroid peroxidase (TPO), eosinophil peroxidase (EPO), metallothionein-3 (MT3), and glutathione reductase. | - | - | [29,30,31] | |||
| 268 | 25 years | Iran | Iran | - | Oxidative stress, gene expression, cellular senescence | Lipid peroxidation derivative malondialdehyde (MDA), 8-oxo-dG, OGG1, and p16INK4a mRNA. | - | DNA damage and immune system subjected to cellular senescence. | [33] | |||
| 80 | - | Iran | Iran | - | Oxidative stress | Prostaglandin-like compound 8-isoprostane F2- alpha | - | - | [34] | |||
| 75 | 25 years | Iran | Khorasan (Iran) | - | DNA damage | DNA repair proteins (MRE11, NBS1, RAD51, and XPA), phosphor-H2AX. | - | Long-term health problems. | [18] | |||
| 81 | - | Iran | Iran | - | microRNA expression | miR-589-3p, miR-365a-3p, miR-143-3p, miR-200a-3p, miR-663a. | - | - | [35] | |||
| 34,000 | 18-23 years | Iran | Iran | - | - | - | Lesions of the lungs, eyes and skin | [36] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Sarin and Cyclosarin | 349,291 | - | United States | Khamisiyah (Iraq) | - | - | - | - | Suffering postwar morbidity. | [37] |
| 1,368,150 | 7 years | United States | Khamisiyah (Iraq) | - | - | - | - | Cause-specific mortality. | [38] | |
| 80 | 11-16 years | United States | Khamisiyah (Iraq) | - | - | - | - | Reduced total gray matter volume. | [40] | |
| 128 | 14–19 years | United States | Khamisiyah (Iraq) | - | - | - | - | Reduced gray matter and white matter volumes. | [41] | |
| 118 | 14–19 years | United States | Khamisiyah (Iraq) | - | - | - | - | Increased axial diffusivity throughout the brain. | [42] | |
| 170 | 23–26 years | United States | Khamisiyah (Iraq) | - | - | - | - | Smaller hippocampal volumes. | [43] | |
| 351,041 | 10 years | United States | Khamisiyah (Iraq) | - | - | - | - | Increased risk of brain cancer death. | [39] | |
| 26 | - | United States | Khamisiyah (Iraq) | - | - | - | - | White matter reduction and ventricles volume increase. | [44] | |
| 140 | 4–5 years | United States | Khamisiyah (Iraq) | High exposure: 0.072 mg min/m3; Moderate exposure: from 0.035 to 0.144 mg min/m3 but no more than 0.072 mg min/m3 | - | - | - | Neuropsychological task performances for psycho-motor dexterity and visuospatial abilities. | [45] |
| Pollutant. | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Agent Orange | - | - | United States | Vietnam | Inner aircraft concentrations ranged from 11.49 to 13.2–27.0 pg/m3 | - | - | - | - | [48] |
| - | - | United States | Vietnam | Exposure estimated using a software | - | - | - | - | [49] | |
| 5609 | - | United States | Vietnam/ Non-Vietnam | - | - | - | - | COPD mortality. | [50] | |
| 111,726 | - | Korea | Vietnam | Exposure indexes based on the proximity of the veterans’ military unit to an Agent Orange-sprayed area. | - | - | - | Diabetes mellitus, thyroid, pituitary gland, and neurologic disorders. | [51] | |
| 180,639 | 30 years | Korea | Vietnam | Exposure indexes based on the proximity of the veterans’ military unit to an Agent Orange-sprayed area. | - | - | - | Cancers of the stomach, small intestine, liver, larynx, lung, bladder, and thyroid gland, chronic myeloid leukemia. | [52] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Pesticides | 187 | Day 0, 14, and 28 of the wearing period, and 28 days after termination | Germany | Germany | Permethrin concentration in uniforms: 1300 mg/m2 | Exposure biomarker in urines | Permethrin metabolites: cis-3-(2,2-dichlorovinyl)-2,2-dimethyl-(1-cyclopropane) carboxylic acid (cis-DCCA), trans-3-(2,2-dichlorovinyl)-2,2-dimethyl-(1-cyclopropane) carboxylic acid (trans-DCCA), and 3-phenoxybenzoic acid (3-PBA) | - | - | [55,56] |
| 549 (Study I); 195 (Study II) | Before wearing uniforms, after 14 days, after 28 days of wearing, and 28 days after cessation of wearing uniform | Germany | Two sub-cohorts in Germany and one in Afghanistan | Permethrin concentration in uniforms: 0.13 mg/cm2 | Exposure biomarker in urines | Permethrin metabolites: cis-DCCA, trans-DCCA 3-PBA | - | - | [57] | |
| 224 | - | United States | Iraq/Afghanistan | - | - | - | - | Chronic multisymptom illness. | [58] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Cs Gas (Tear Gas) | 87 | 2–8–24–30 h | United States | United States | 0.086–4.9 mg/m3 | Exposure biomarker in urines | 2-chlorohippuric acid | - | - | [60] |
| - | - | United States | United States | 2.33–3.29 mg/m3 | - | - | - | - | [61] | |
| 6730 | 5–15 min during exposure | United States | United States | 0.4–53.3 mg/m3 | Exposure biomarker | Personal air sampling | - | - | [62] | |
| 6723 | 7 days before CS exposure and 7 days after exposure | United States | United States | 0–2 mg/m3, 2–5 mg/m3, 5–10 mg/m3, >10 mg/m3 | - | - | - | Acute respiratory illnesses. | [63] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Oil Combustion | 1599 | 3 months | United States | Kuwait | - | - | - | Eye and upper respiratory tract irritation, shortness of breath, cough, rashes, and fatigue | - | [64] |
| 125 | During exposure: every fortnight for 5 months | United Kingdom | Kuwait | - | - | - | - | No lung function changes before and after deployment. | [65] | |
| 1560 | 5 years | United States | Kuwait | - | - | - | - | Asthma, bronchitis, injury and major depression. | [66] | |
| 61 | Sampling: before, during, and after deployment in Kuwait | Germany | Kuwait | - | DNA adducts and gene polymorphism | PAH-DNA adducts in blood; -OH-PG (1-hydroxypyrene-glucuronide) in urines; GSTM1 and GSTT1 gene polymorphisms | - | - | [68] | |
| 168 | - | United States | Kuwait city | - | Exposure biomarkers in blood | VOC: benzene, m-/p-xylene, o-xylene, styrene, and toluene | - | - | [68] | |
| 3440 | Sampling: before, during, and after deployment in Kuwait | Germany | Kuwait | - | - | 1-hydroxypyrene; VOCs; Sister chromatid exchange; DNA-PAH adducts. | - | - | [70] | |
| Cooking Oil Fumes | 98 | At the beginning and at the end of the weekly shift | Taiwan | Taiwan | PAH measured in kitchens and in offices | Exposure biomarker in urines and oxidative DNA damage biomarker | 1-hydroxypyreneand 8-oxodeoxyguanosine | - | Oxidative DNA damage. | [71] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Open Pit Burning | 20,000 | 1.3 years (mean years) | United States | Iraq and Afghanistan | - | - | - | - | Rheumatoid arthritis and lupus. | [72] |
| 21,000 | - | United States | Iraq | - | - | - | - | Chronic multisymptom illness. | [73] | |
| 22,844 | - | United States | Iraq | - | - | - | - | Respiratory outcomes. | [74] | |
| - | - | - | Iraq | PAHs, polychlorinated dibenzo-p-dioxins (PCDD), and polychlorinated dibenzofurans (PCDF) | - | - | - | [75,76] | ||
| 400 | - | United States | Iraq | - | - | - | - | Respiratory and cardiovascular disease. | [77] | |
| 179,914 | - | United States | Iraq, United States, and South Korea | - | - | - | - | Respiratory symptoms and asthma. | [78] | |
| 400 | - | United States | Iraq and Afghanistan | - | microRNA expression | PCDD/PCDF: let-7a-5p, let-7d-5p, miR-144-3p, miR-16-5p, miR-32-5p | - | - | [79] | |
| 400 | - | United States | Iraq and Afghanistan | - | Exposure biomarkers in serum | PAH and PCDD/PCDF | - | - | [80] | |
| 800 | - | - | - | - | microRNA expression | hsa-miR-26a, hsa-miR-30b, hsa-miR-103, hsa-miR-126, hsa-miR-766. | - | - | [81] | |
| 178,766 infants | - | - | - | - | - | - | - | Birth defects. | [82] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Jet Fuel | 130 | Prior to and after exposure, 15 and 30 weeks during exposure. | United States | United States | Jet fuel (JP-4), 1,1,1- trichloroethane, methyl ethyl ketone, xylenes, toluene, and methylene chloride. | Biomarkers of genotoxicity | Chromatid exchanges and micronuclei frequency | - | - | [87] |
| 74 | Post-shift on friday (Day 1) and during the following workweek (Days 2–6) | United States | United States | Personal breathing zone and work areas via active sampling methods. | Exposure biomarkers in urines, exhaled breath, and blood. Gene polymorphism | - Dermal exposure (total hydrocarbons, benzene, toluene, ethylbenzene, and xylene and naphthalene); - Exhaled breath (BTEX and naphthalene); -Urine samples (1- and 2-naphthol, 2-, 3-, and 9-hydroxyfluorene; 1-, 2-, 3-, and 4-hydro-xyphenanthrene; 1-hydroxypyrene and VOC mercapurates, including N-acetyl-S-(benzyl)-L-cysteine, N-ace-tyl-S-(phenyl)-L-cysteine. - Blood samples (benzene, ethylbenzene, m-/p-/o-xylenes, and toluene). - GSMT1 polymorphism and DNA pattern methylation | - | - | [88] | |
| 24 | During three consecutive workdays | United States | United States | Personal air samples: naphthalene | Biomarker of exposure in urines | 1- and 2-naphthol | - | - | [89] | |
| 73 | During four consecutive workdays | United States | United States | Personal air samples: benzene, ethylbenzene, toluene, xylene, total hydrocarbons, and naphthalene. | - | - | - | - | [90] | |
| 73 | During four consecutive workdays | United States | United States | Personal air samples analyzed for benzene, toluene, ethylbenzene, m-/p-xylene, o-xylene, and total hydrocarbons | Biomarker of exposure in urines. Gene polymorphism | 1- and 2-naphthol, 2-, 3-, and 9-hydroxyfluorene, 1-, 2-, 3-, and 4-hydroxyphenanthrene, and 1-hydroxypyrene; GSTM1 polymorphism and glutathione S-transferase theta-1 (GSTT1) | - | - | [91] | |
| 69 | Post-shift sampling | United States | United States | Personal air samples: total hydrocarbons | Biomarker of exposure in blood | Ethylbenzene, toluene, o-xylene, and m/p-xylene, and for the smoking biomarker, 2,5-dimethylfuran. | - | - | [92] | |
| 74 | Post-shift on a Friday afternoon (Day1) and continued Monday morning through Friday morning of the following workweek (Days 2–6) | United States | United States | Personal air samples: naphthalene and total hydrocarbons. | Biomarkers of exposure in urines | 1-naphthol and 2-naphthol | - | - | [93] | |
| Submarine fuel | 38 | - | United States | United States | - | - | - | Reactive airways dysfunction syndrome (RADS) | - | [95] |
| 1 | - | United Kingdom | - | Median weighted levels of benzene exposure over 13-year period: 189 ug/m3 | - | - | - | Chronic myeloid leukemia. | [96] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Firing Ranges | 175 | During training period | Israel | Israel | Overall range of airborne lead levels in personal samples: 0.08–168.4 μg/m3 | Biomarkers of exposure in blood | Blood lead level | - | - | [97] |
| 120 | During working period | Korea | Korea | - | Biomarkers of exposure in blood | Blood lead level | - | - | [98] | |
| 546 | During working period | Italy | Italy | Environmental lead using personal samplers in 6 indoor and 6 outdoor firing ranges (<25 pg/m3) | Biomarkers of exposure in blood | Blood lead level | - | - | [99] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Sunlight exposure (Skin Cancer) | 154 | - | United States | Tropical/non-tropical theaters | - | - | - | - | Melanoma | [110] |
| 370 | - | - | Pacific theater (World War II) | - | - | - | - | Skin cancer. | [101] | |
| 5524 | - | - | Pacific theater (World War II) | - | - | - | - | Melanoma and colon cancer. | [103] | |
| 176 | During working period | United States | United States | - | - | - | - | Melanoma | [104] | |
| 300 | ||||||||||
| 2696 | - | - | Iraq | - | - | - | - | Eczematous dermatitis, benign neoplasms, and skin cancers. | [105] | |
| 6670 | - | United States | United States | - | - | - | - | Non-melanoma skin cancer. | [106] | |
| - | - | United States | United States | - | - | - | - | Melanoma. | [107] | |
| 2093,157 | - | United States | United States | - | - | - | - | Melanoma. | [108] | |
| 35,157 | - | Australia | Tropical locations | - | - | - | - | Melanoma. | [36] | |
| 21,582 | During exposure: from first day of service until date of death, emigration, or end of follow-up. | Norway | Lebanon | - | - | - | - | Cancer incidence and all-cause mortality | [111] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Electromagnetic fields | 166 | During exposure | India | India | Electromagnetic fields monitored at different locations. | Stress response | Catecholamine levels in blood | - | - | [111] |
| - | - | Poland | Poland | 204 military devices monitored | - | - | - | - | [112] | |
| 39,850 | - | France | France | - | - | - | - | Overall mortality and cancer mortality | [113] | |
| 10,495 | - | Norway | Norway | - | - | - | - | Male infertility and offspring sex ratio | [118] |
| Pollutant | Sample Size | Time after Exposure | Solider Nationality | Exposure Site (Operation Theater) | Environmental Monitoring (Exposure) | Biomarkers (Early Biological Effect) | Biomarker Description | Early Clinical Effect | Late Clinical Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Ionizing Radiations | 74 | 2 years | United States | Iraq | - | Biomarker of exposure in urines | Uranium concentration | - | - | [129] |
| 35 | 3 years | United States | Iraq | Biomarker of exposure in urines | Total and isotopic uranium concentrations. Analysis of metals. | - | - | [129] | ||
| 35 | 18 years | United States | Iraq | - | Biomarkers of exposure in urines. Gene mutations. | Total and isotopic uranium concentration; Peripheral blood cells: micronuclei (MN), chromosome aberrations, and mutations of hypoxanthine- guanine phosphoribosyl transferase (HPRT) and phosphatidylinositol glycan class-A (PIGA). | - | - | [126] | |
| 36 | 25 years | United States | Iraq | - | Biomarkers of exposure in urines | Total and isotopic uranium concentrations. Analysis of metals. | - | - | [130] | |
| 70 | - | United States | Iraq | - | Biomarkers of exposure in urines. Gene mutations | Total and isotopic uranium concentrations. Analysis of metals. Peripheral blood cells: hypoxanthine-guanine phosphoribosyl transferase (HPRT) mutations | - | - | [128] | |
| 35 | 18 years | United States | Iraq | - | Biomarker of exposure in urines and biomarkers of genotoxicity | Total uranium concentration Peripheral blood cells: Micronuclei | - | - | [127] | |
| 35 | 22 years | United States | Iraq | - | Biomarker of exposure in urines. | Total uranium concentration | - | chronic dermatological findings (dermatitis) | [131] | |
| 122 | - | Canada | Canada | - | Biomarker of exposure in urines. | Total uranium concentration Hair: isotopic uranium (238:235 U ratio) | - | - | [132] | |
| 9188 | - | Sweden | Balkans | - | - | - | - | Overall incidence of cancer | [133] | |
| 341 | - | United Kingdom | Iraq | - | Biomarker of exposure in urines. | Uranium concentration and 238 U/235 U isotopic ratio | - | - | [134] | |
| 14,012 | - | Denmark | Balkans | - | - | - | - | Overall incidence of cancer | [135] | |
| 153,530 | - | Holland | Balkans | - | - | - | - | incidence of all cancer | [136] | |
| 185,413 | 4–14 years | Italy | Balkans | - | - | - | - | General mortality and cancer mortality | [137] | |
| 981 | -Before the deployment (T0) -At the end of the deployment period (T1) - In the operative theater | Italy | Iraq | - | Biomarkers of exposure in urine and serum. Biomarkers of genotoxicity and DNA damage | - Urine and serum: As, Cd, Mo, Ni, Pb, U, V, W, and Zr, and Uranium isotopic ratio 238 U/235 U Peripheral blood cells: DNA adducts, micronuclei, GSTM1, XRCC1, and OGG1 polymorphisms; Oxidative DNA alterations (8-Hydroxy-2′-deoxyguanine) | - | - | [3] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geretto, M.; Ferrari, M.; De Angelis, R.; Crociata, F.; Sebastiani, N.; Pulliero, A.; Au, W.; Izzotti, A. Occupational Exposures and Environmental Health Hazards of Military Personnel. Int. J. Environ. Res. Public Health 2021, 18, 5395. https://doi.org/10.3390/ijerph18105395
Geretto M, Ferrari M, De Angelis R, Crociata F, Sebastiani N, Pulliero A, Au W, Izzotti A. Occupational Exposures and Environmental Health Hazards of Military Personnel. International Journal of Environmental Research and Public Health. 2021; 18(10):5395. https://doi.org/10.3390/ijerph18105395
Chicago/Turabian StyleGeretto, Marta, Marco Ferrari, Roberta De Angelis, Filippo Crociata, Nicola Sebastiani, Alessandra Pulliero, William Au, and Alberto Izzotti. 2021. "Occupational Exposures and Environmental Health Hazards of Military Personnel" International Journal of Environmental Research and Public Health 18, no. 10: 5395. https://doi.org/10.3390/ijerph18105395
APA StyleGeretto, M., Ferrari, M., De Angelis, R., Crociata, F., Sebastiani, N., Pulliero, A., Au, W., & Izzotti, A. (2021). Occupational Exposures and Environmental Health Hazards of Military Personnel. International Journal of Environmental Research and Public Health, 18(10), 5395. https://doi.org/10.3390/ijerph18105395








