Role of Type and Volume of Recreational Physical Activity on Heart Rate Variability in Men
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
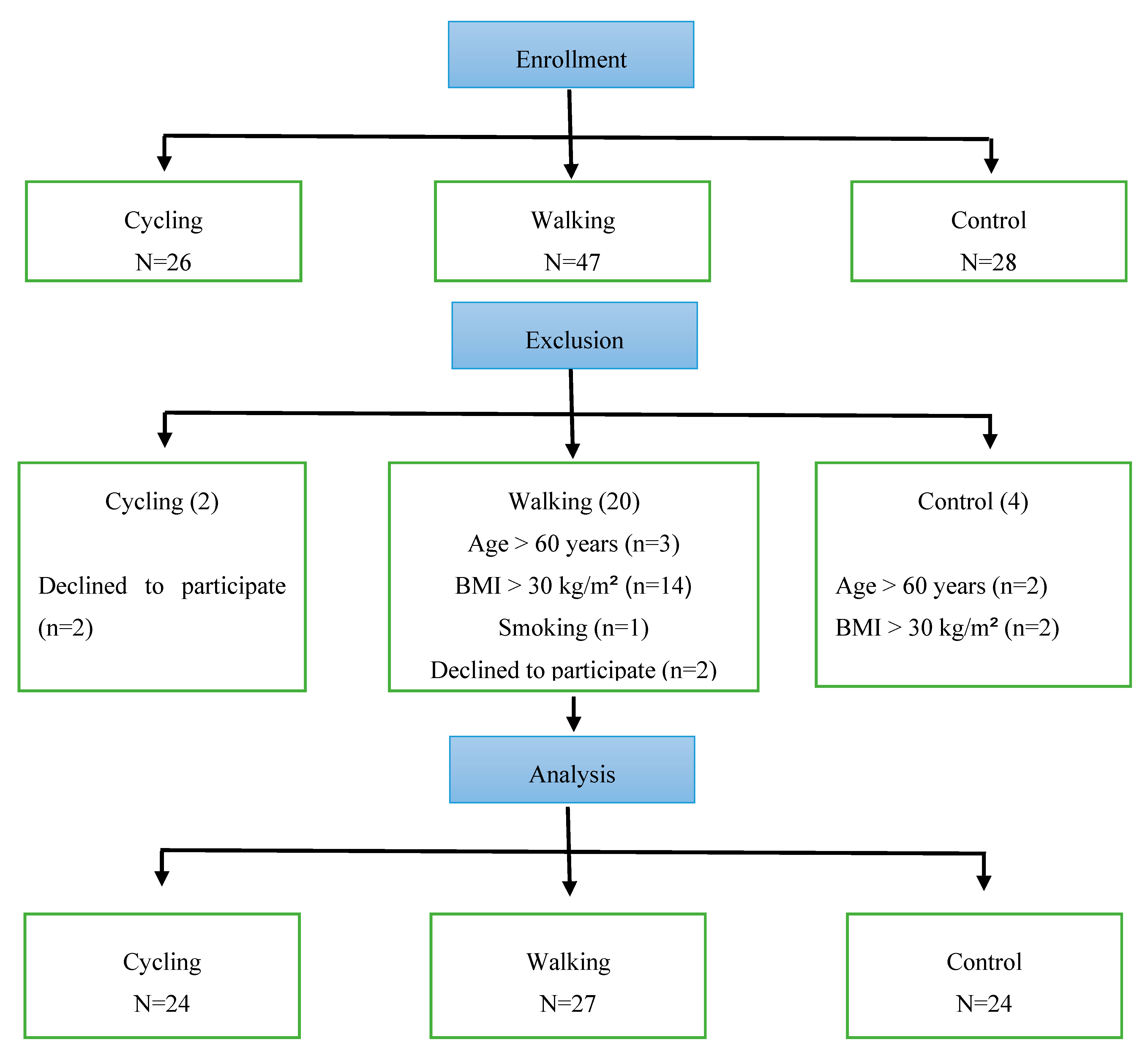
2.2. Participants
2.3. Pre-Screening Procedures
2.4. Study Variable Measures
2.4.1. Body Composition
2.4.2. Heart Rate Variability
2.4.3. Volume of Physical Activity for Walking and Cycling Participants
2.5. Statistical Analysis
3. Results
4. Discussion
Ethical Approval
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aubert, A.; Seps, B.; Beckers, F. Heart rate variability in athletes. Sports Med. 2003, 33, 889–919. [Google Scholar] [CrossRef] [PubMed]
- Sondermeijer, H.P.; van Marle, A.G.; Kamen, P.; Krum, H. Acute effects of caffeine on heart rate variability. Am. J. Cardiol. 2002, 90, 906–907. [Google Scholar] [CrossRef]
- Jarczok, M.N.; Koenig, J.; Wittling, A.; Fischer, J.E.; Thayer, J.F. First evaluation of an index of low vagally-mediated heart rate variability as a marker of health risks in human adults: Proof of concept. J. Clin. Med. 2019, 8, 1940. [Google Scholar] [CrossRef] [PubMed]
- Force, T. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. task force of the European society of cardiology the north American society of pacing electrophysiology. Circulation 1996, 93, 1043–1065. [Google Scholar]
- Yeragani, V.K.; Krishnan, S.; Engels, H.J.; Gretebeck, R. Effects of caffeine on linear and nonlinear measures of heart rate variability before and after exercise. Depress. Anxiety 2005, 21, 130–134. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American college of sports medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Bonato, M.; Meloni, A.; Merati, G.; La Torre, A.; Agnello, L.; Vernillo, G. Effect of repeated-sprints on the reliability of short-term parasympathetic reactivation. PLoS ONE 2018, 13, e0192231. [Google Scholar] [CrossRef]
- Fronchetti, L.; Nakamura, F.; De-Oliveira, F.; Lima-Silva, A.; De Lima, J. Effects of high-intensity interval training on heart rate variability during exercise. J. Exerc. Physiol. Online 2007, 10, 1–9. [Google Scholar]
- Boutcher, S.H.; Stein, P. Association between heart rate variability and training response in sedentary middle-aged men. Eur. J. Appl. Physiol. Occup. Physiol. 1995, 70, 75–80. [Google Scholar] [CrossRef]
- Perini, R.; Fisher, N.; Veicsteinas, A.; Pendergast, D. Aerobic training and cardiovascular responses at rest and during exercise in older men and women. Med. Sci. Sports Exerc. 2002, 34, 700–708. [Google Scholar]
- Bonato, M.; Agnello, L.; Galasso, L.; Montaruli, A.; Roveda, E.; Merati, G.; La Torre, A.; Vitale, J.A. Acute modification of cardiac autonomic function of high-intensity interval training in collegiate male soccer players with different chronotype: A cross-over study. J. Sports Sci. Med. 2017, 16, 286. [Google Scholar] [PubMed]
- Vitale, J.A.; Bonato, M.; La Torre, A.; Banfi, G. Heart rate variability in sport performance: Do time of day and chronotype play a role? J. Clin. Med. 2019, 8, 723. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Subramanian, S.K.; Radhakrishnan, K.; Rajendran, R.; Ravindran, B.S.; Arunachalam, V. Comparison of structured and unstructured physical activity training on predicted VO2max and heart rate variability in adolescents–a randomized control trial. J. Basic Clin. Physiol. Pharmacol. 2017, 28, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.D.; Steptoe, A.; Chambers, J.C.; Kooner, J.S. Ethnic and gender differences in the relationship between hostility and metabolic and autonomic risk factors for coronary heart disease. Psychosom. Med. 2011, 73, 53–58. [Google Scholar] [CrossRef]
- Hill, L.K.; Hu, D.D.; Koenig, J.; Sollers III, J.J.; Kapuku, G.; Wang, X.; Snieder, H.; Thayer, J.F. Ethnic differences in resting heart rate variability: A systematic review and meta-analysis. Psychosom. Med. 2015, 77, 16. [Google Scholar] [CrossRef]
- Al-Fehaid, A.F.; Alkahtani, S.A.; Al-Sunni, A.A.; Yar, T. Role of the work-to-rest ratio in high-intensity interval exercise on heart rate variability and blood pressure in sedentary obese men. Saudi J. Health Sci. 2018, 7, 83. [Google Scholar]
- Loimaala, A.; Huikuri, H.; Oja, P.; Pasanen, M.; Vuori, I. Controlled 5-mo aerobic training improves heart rate but not heart rate variability or baroreflex sensitivity. J. Appl. Physiol. 2000, 89, 1825–1829. [Google Scholar] [CrossRef]
- Migliaro, E.; Contreras, P.; Bech, S.; Etxagibel, A.; Castro, M.; Ricca, R.; Vicente, K. Relative influence of age, resting heart rate and sedentary life style in short-term analysis of heart rate variability. Braz. J. Med Biol. Res. 2001, 34, 493–500. [Google Scholar] [CrossRef]
- Melanson, E.L. Resting heart rate variability in men varying in habitual physical activity. Med. Sci. Sports Exerc. 2000, 32, 1894–1901. [Google Scholar] [CrossRef]
- Buchheit, M.; Simon, C.; Charloux, A.; Doutreleau, S.; Piquard, F.; Brandenberger, G. Heart rate variability and intensity of habitual physical activity in middle-aged persons. Med. Sci. Sports Exerc. 2005, 37, 1530–1534. [Google Scholar] [CrossRef]
- Soares-Miranda, L.; Sandercock, G.; Valente, H.; Vale, S.; Santos, R.; Mota, J. Vigorous physical activity and vagal modulation in young adults. Eur. J. Cardiovasc. Prev. Rehabil. 2009, 16, 705–711. [Google Scholar] [CrossRef]
- Tornberg, J.; Ikäheimo, T.M.; Kiviniemi, A.; Pyky, R.; Hautala, A.; Mäntysaari, M.; Jämsä, T.; Korpelainen, R. Physical activity is associated with cardiac autonomic function in adolescent men. PLoS ONE 2019, 14, e0222121. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Hwang, S.Y.; Choi, K.M.; Baik, S.H.; Lee, E.M.; Kim, E.J.; Rha, S.-W.; Park, C.G.; Oh, D.J.; Seo, H.S. Clinical implication of body size phenotype on heart rate variability. Metabolism 2016, 65, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.S.; Barker, A.R.; Wilkinson, K.M.; Abbott, R.A.; Williams, C.A. Is cardiac autonomic function associated with cardiorespiratory fitness and physical activity in children and adolescents? A systematic review of cross-sectional studies. Int. J. Cardiol. 2017, 236, 113–122. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, T.L.W.; Ostoli, T.L.V.d.P.; Sperandio, E.F.; Arantes, R.L.; Gagliardi, A.R.d.T.; Romiti, M.; da Silva, R.P.; Dourado, V.Z. Dose-response relationship between very vigorous physical activity and cardiovascular health assessed by heart rate variability in adults: Cross-sectional results from the EPIMOV study. PLoS ONE 2019, 14, e0210216. [Google Scholar] [CrossRef] [PubMed]
- Bravata, D.M.; Smith-Spangler, C.; Sundaram, V.; Gienger, A.L.; Lin, N.; Lewis, R.; Stave, C.D.; Olkin, I.; Sirard, J.R. Using pedometers to increase physical activity and improve health: A systematic review. JAMA 2007, 298, 2296–2304. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The physical activity guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Soares-Miranda, L.; Sattelmair, J.; Chaves, P.; Duncan, G.E.; Siscovick, D.S.; Stein, P.K.; Mozaffarian, D. Physical activity and heart rate variability in older adults: The Cardiovascular Health Study. Circulation 2014, 129, 2100–2110. [Google Scholar] [CrossRef]
- Rennie, K.L.; Hemingway, H.; Kumari, M.; Brunner, E.; Malik, M.; Marmot, M. Effects of moderate and vigorous physical activity on heart rate variability in a British study of civil servants. Am. J. Epidemiol. 2003, 158, 135–143. [Google Scholar] [CrossRef]
| Parameters | Walking | Cycling | Non-Active | Effect Size | P-Value |
|---|---|---|---|---|---|
| N | 27 | 24 | 24 | ||
| Age | 38.0 ± 8.4 | 39.3 ± 7.5 | 35.5 ± 4.4 | 0.047 | 0.180 |
| Fat (%) | 24.4 ± 6.1 | 22.4 ± 4.9 | 25.7 ± 4.6 | 0.061 | 0.105 |
| Ln-RR interval (sec) | 1.05(0.91–1.12) | 1.06 (1.0–1.1) | 0.99 (0.92–1.1) | 0.035 | 0.292 |
| Ln-SDNN (ms) | 53.3(31.1–69.9) | 52.6(35.7–73.8) | 45.8(37.9–54.9) | 0.017 | 0.563 |
| Ln-RMSSD (ms) | 47.5(26.9–71.2) | 39.4 (31.9–60.4) | 35.2 (22.1–46.5) | 0.039 | 0.250 |
| Ln-LF (ms2) | 918(246–2110) | 764(230–1838) | 594(414–1069) | 0.009 | 0.765 |
| Ln-HF (ms2) | 786(310–2444) | 609(345–1352) | 408(223–1937) | 0.038 | 0.317 |
| Ln-LF/HF ratio | 0.88(0.45–1.2) | 0.76 (0.53–1.3) | 1.07 (0.70–2.01) | 0.025 | 0.464 |
| Parameters | Volume of Physical Activity | Effect Size | P-Value | Adjusted for Age | |||
|---|---|---|---|---|---|---|---|
| Low | Moderate | High | Control | ||||
| N | 13 | 24 | 14 | 24 | |||
| Age | 33.3 ± 9.3 | 40.0 ± 7.4A | 41.1 ± 5.0A | 35.5 ± 4.4 | 0.174 | 0.003 | |
| Fat (%) | 23.4 ± 7.0 | 23.9 ± 4.9 | 22.6 ± 5.6 | 25.7 ± 4.6 | 0.054 | 0.349 | 0.279 |
| Ln-RR interval (sec) | 1.0 (0.9–1.1) | 1.08 (0.9–1.1) | 1.06 (1.01–1.12) | 0.99 (0.92–1.08) | 0.096 | 0.074 | 0.201 |
| Ln-SDNN (ms) | 41.9 (30.9–63.3) | 52.9 (32.2–69.9) | 67.3 (44.1–77.9)A | 45.8 (37.9–54.9) | 0.079 | 0.135 | 0.025 |
| Ln-RMSSD (ms) | 32.6(18.6–52.9) | 47.7 (35.3–69.6) | 44.0 (32.5–73.1) | 35.2 (22.1–46.5)B | 0.066 | 0.198 | 0.009 |
| Ln-LF (ms2) | 338.7(196.6–764) | 790 (203–2110) | 1759(922–4942)A | 594 (414–1069)C | 0.183 | 0.006 | < 0.001 |
| Ln-HF (ms2) | 481 (151.7–832) | 676 (365–4347)A | 838 (391–3109) | 406 (223–1189) | 0.163 | 0.014 | 0.004 |
| Ln-LF/HF ratio | 0.73 (0.46–1.18) | 0.75 (0.40–0.95) | 1.19 (0.59–2.1) | 1.07 (0.70–2.01) | 0.103 | 0.092 | 0.096 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alkahtani, S.; Flatt, A.A.; Kanas, J.; Aldyel, A.; Habib, S.S. Role of Type and Volume of Recreational Physical Activity on Heart Rate Variability in Men. Int. J. Environ. Res. Public Health 2020, 17, 2719. https://doi.org/10.3390/ijerph17082719
Alkahtani S, Flatt AA, Kanas J, Aldyel A, Habib SS. Role of Type and Volume of Recreational Physical Activity on Heart Rate Variability in Men. International Journal of Environmental Research and Public Health. 2020; 17(8):2719. https://doi.org/10.3390/ijerph17082719
Chicago/Turabian StyleAlkahtani, Shaea, Andrew A. Flatt, Jawad Kanas, Abdulaziz Aldyel, and Syed Shahid Habib. 2020. "Role of Type and Volume of Recreational Physical Activity on Heart Rate Variability in Men" International Journal of Environmental Research and Public Health 17, no. 8: 2719. https://doi.org/10.3390/ijerph17082719
APA StyleAlkahtani, S., Flatt, A. A., Kanas, J., Aldyel, A., & Habib, S. S. (2020). Role of Type and Volume of Recreational Physical Activity on Heart Rate Variability in Men. International Journal of Environmental Research and Public Health, 17(8), 2719. https://doi.org/10.3390/ijerph17082719





