Bisphenols as Environmental Triggers of Thyroid Dysfunction: Clues and Evidence
Abstract
1. Introduction
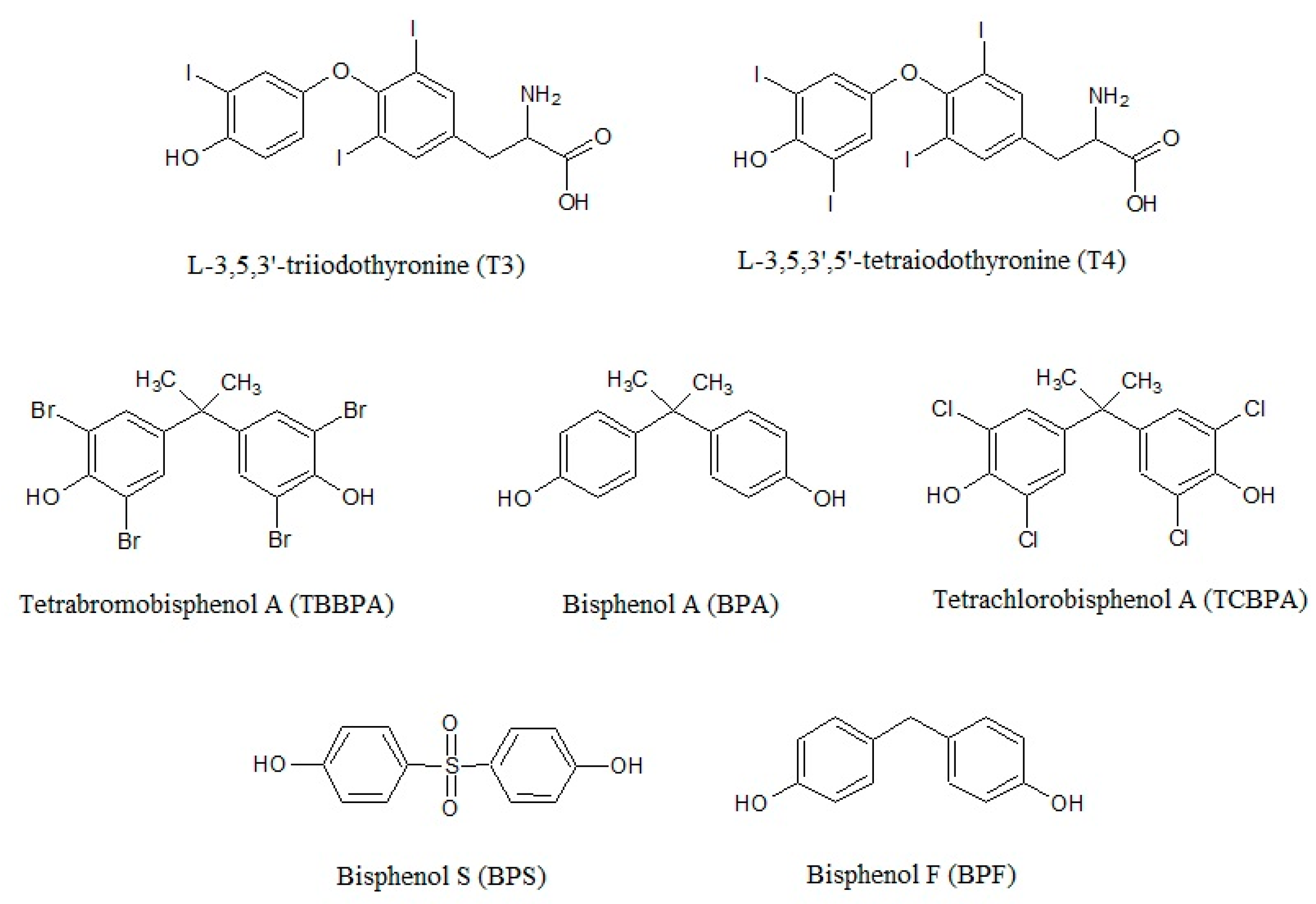
2. Bisphenols in the Environment and Humans
3. Thyroid Disrupting Properties of BPs: in Vitro Studies
3.1. Interference with T3 Transcriptional Activity
3.2. Cell Proliferation
3.3. Cytotoxicity
3.4. Competitive Binding with Thyroid Hormone Binding Proteins
3.5. Perturbation of Thyroid Hormone Uptake
3.6. Dysregulation of Gene Expression
4. Thyroid Disrupting Properties of BPs: in Vivo Studies
4.1. Rodents
4.2. Sheep
4.3. Zebrafish
5. Thyroid Disrupting Properties of BPs: Human Studies
5.1. Effects on Serum TH Levels
5.2. Association with Thyroid Diseases
6. Discussion
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 3,3′-Cl2BPA | 3,3′-dichlorobisphenol A |
| 3,5-Cl2BPA | 3,5-dichlorobisphenol A |
| 95% CI | 95% confidence interval |
| 96 h-LC50 | 96 h median lethal concentration |
| AM | Amiodarone |
| BMI | Body mass index |
| BPA | Bisphenol A; 2,2-bis(4-hydroxyphenyl)propane) |
| BPAF | Bisphenol AF; 1,1,1,3,3,3-hexafluoro-2,2-bis(4-hydroxyphenyl)propane |
| BPAP | Bisphenol AP (4,4′-(1-phenylethylidene)bisphenol |
| BPB | Bisphenol B (2,2-bis(4-hydroxyphenyl)butane) |
| BPF | Bisphenol F; 4,4′-dihydroxydiphenylmethane |
| BPM | Bisphenol M; 4,4′-(1,3-phenylenediisopropylidene)bisphenol) |
| BPP | Bisphenol P (2,2-bis(4-hydroxyphenyl)butane) |
| BPS | Bisphenol S; 4,4′-sulfonyldiphenol |
| BPZ | Bisphenol Z; 1,1-bis(4-hydroxyphenyl)-cyclohexane |
| BPs | Bisphenols |
| BrdU | 5′-bromo-2′-deoxyuridine |
| BSA | Body surface area |
| BW | Body weight |
| CHAMACOS | Center for the Health Assessment of Mothers and Children of Salinas |
| ClBPA | 3-chlorobisphenol A |
| ClxBPA | Sum of ClBPA, 3,5-Cl2BPA, and 3,3′-Cl2BPA |
| cr | Creatinine |
| Crh | Corticotrophin-releasing hormone |
| c-Src/PI3K | Proto-oncogene tyrosine-protein/phosphatidylinositol 3-kinase |
| D1 | Type 1 iodothyronine deiodinase |
| D2 | Type 2 iodothyronine deiodinase |
| DHPN | N-bis (2-hydroxypropyl) nitrosamine |
| Dio1 | Deiodinase 1 |
| Dio2 | Deiodinase 2 |
| Dio3 | Deiodinase 3 |
| dpf | Days post-fertilization |
| DTC | Differentiated thyroid cancer |
| EC50 | Median effective concentration |
| EDC(s) | Endocrine disrupting chemical(s) |
| EGFP | Excitation and emission of green fluorescent proteins |
| ELISA | Enzyme linked immunosorbent assay |
| ERs | Estrogen receptors |
| ERα | Estrogen receptor α |
| E2 | 17-β estradiol |
| ERK | Extracellular-signal-regulated kinase |
| Foxe1 | Forkhead box E1 |
| FT3 | Free triidothyronine |
| FT4 | Free thyroxine |
| GD | Gestation day |
| GFP | Green fluorescent protein |
| GH | Growth hormone |
| GM | Geometric mean |
| GPR30 | G protein-coupled receptor 30 |
| Hhex | Hematopoietically expressed homeobox |
| HOME | Health Outcomes and Measures of the Environment |
| hpf | hours post fertilization |
| HPT | Hypothalamic–pituitary–thyroid |
| HT | Hashimoto’s thyroiditis |
| IC10 | Concentration at which 10% of the inhibition was observed |
| IC50 | Concentration at which 50% of the inhibition was observed |
| ICI | Fulvestrant |
| IQR | Interquartile range |
| JNK | c-Jun NH2-terminal kinase |
| KI | Potassium iodine |
| LC50 | Median lethal concentration |
| LOAEL | Low observed adverse effect level |
| LOD | Limit of detection |
| LOEC | Lowest observed effect concentration |
| LOW | Limit of quantification |
| MAPK | Mitogen-activated protein kinase |
| MCT | Monocarboxylate transporter |
| MEK | Mitogen-activated protein kinase/ERK kinase |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NaI | Sodium iodide |
| N-CoR | Nuclear receptor co-repressor |
| n.d. | Not detected |
| NG | Nodular goiter |
| NHANES | National Health and Nutrition Examination Survey |
| NHES IV | Thai National Health Examination Survey |
| Nkx2-1 | NK2 homeobox 1 |
| OATP | Organic anion transport protein |
| Pax8 | Paired box 8 |
| PND | Postnatal day |
| PTC | Papillary thyroid cancer |
| qRT-PCR | Quantitative real time polymerase chain reaction |
| ROS | Reactive oxygen species |
| SAPK | Stress-activated proteins kinases |
| Slc5a5 | Gene encoded for sodium iodide symporter |
| SMRT | Silencing mediator for retinoid and thyroid |
| SRC | Steroid receptor coactivator |
| Sult-st1/2/3/5 | Sulfotransferase 1/2/3/5 |
| T3 | Triiodothyronine |
| T4 | Thyroxine |
| TAM | Tamoxifen |
| TBBPA | Tetrabromobisphenol A |
| TBBPS | Tetrabromobisphenol S |
| TBG | Thyroxine binding globulin |
| TC | Thyroid cancer |
| TCBPA | Tetrachlorobisphenol A |
| Tg | Thyroglobulin |
| TgAb | Thyroglobulin antibody |
| TH(s) | Thyroid hormone(s) |
| TN(s) | Thyroid nodule(s) |
| Tpo | Thyroid peroxidase |
| TPOAb | Thyroid peroxidase antibody |
| TR(s) | Thyroid receptor(s) |
| TRAb | Thyroid receptor antibody |
| Trα | Thyroid receptor α |
| Trβ | Thyroid receptor β |
| TRH | Thyrotropin-releasing hormone |
| Trh-R | Thyrotropin-releasing hormone receptor |
| TSH | Thyroid stimulating hormone |
| Tshβ | TSH-specific β subunit |
| Tsh-r | Thyroid stimulating hormone-receptor |
| TTR | Transthyretin |
| TT3 | Total triiodothyronine |
| TT4 | Total thyroxine |
| Ugt | Uridine diphosphate -glucuronosyltransferase |
References
- Calsolaro, V.; Pasqualetti, G.; Niccolai, F.; Caraccio, N.; Monzani, F. Thyroid Disrupting Chemicals. Int. J. Mol. Sci. 2017, 18, 2583. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, K.; Tagami, T.; Akamizu, T.; Usui, T.; Saijo, M.; Kanamoto, N.; Hataya, Y.; Shimatsu, A.; Kuzuya, H.; Nakao, K. Thyroid hormone action is disrupted by bisphenol A as an antagonist. J. Clin. Endocrinol. Metab. 2002, 87, 5185–5190. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Devesa, S.S.; Sosa, J.A.; Check, D.; Kitahara, C.M. Trends in Thyroid Cancer Incidence and Mortality in the United States, 1974-2013. JAMA 2017, 317, 1338. [Google Scholar] [CrossRef] [PubMed]
- Nettore, I.C.; Colao, A.; Macchia, P.E. Nutritional and Environmental Factors in Thyroid Carcinogenesis. Int. J. Environ. Res. Public Health 2018, 15, 1735. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.C.; Lin, H.T.; Lin, S.F.; Kuo, C.F.; Chung, T.T.; Yu, H.P. Nationwide cohort study on the epidemiology and survival outcomes of thyroid cancer. Oncotarget 2017, 8, 78429–78451. [Google Scholar] [CrossRef][Green Version]
- Pellegriti, G.; Frasca, F.; Regalbuto, C.; Squatrito, S.; Vigneri, R. Worldwide increasing incidence of thyroid cancer: Update on epidemiology and risk factors. J. Cancer Epidemiol. 2013, 2013, 965212. [Google Scholar] [CrossRef]
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R.; Lee, D.H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and endocrine-disrupting chemicals: Low-dose effects and nonmonotonic dose responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef]
- Gorini, F.; Iervasi, G.; Coi, A.; Pitto, L.; Bianchi, F. The Role of Polybrominated Diphenyl Ethers in Thyroid Carcinogenesis: Is It a Weak Hypothesis or a Hidden Reality? From Facts to New Perspectives. Int. J. Environ. Res. Public Health 2018, 15, 1834. [Google Scholar] [CrossRef]
- Kitamura, S.; Kato, T.; Iida, M.; Jinno, N.; Suzuki, T.; Ohta, S.; Fujimoto, N.; Hanada, H.; Kashiwagi, K.; Kashiwagi, A. Anti-thyroid hormonal activity of tetrabromobisphenol A, a flame retardant, and related compounds: Affinity to the mammalian thyroid hormone receptor, and effect on tadpole metamorphosis. Life Sci. 2015, 76, 1589–1601. [Google Scholar] [CrossRef]
- Welshons, W.V.; Nagel, S.C.; vom Saal, F.S. Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 2006, 147, S56–S69. [Google Scholar] [CrossRef] [PubMed]
- Rubin, B.S. Bisphenol A: An endocrine disruptor with widespread exposure and multiple effects. J. Steroid Biochem. Mol. Biol. 2011, 127, 27–34. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Maffini, M.V.; Sonnenschein, C.; Rubin, B.S.; Soto, A.M. Bisphenol A and the great divide: A review of controversies in the field of endocrine disruption. Endocr. Rev. 2009, 30, 75–95. [Google Scholar] [CrossRef]
- Kim, Y.S.; Hwang, K.A.; Hyun, S.H.; Nam, K.H.; Lee, C.K.; Choi, K.C. Bisphenol A and nonylphenol have the potential to stimulate the migration of ovarian cancer cells by inducing epithelial–mesenchymal transition via an estrogen receptor dependent pathway. Chem. Res. Toxicol. 2015, 28, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Ryu, J.H.; Jeon, R.; Kang, D.; Yoo, K.Y. Effects of bisphenol A on breast cancer and its risk factors. Arch. Toxicol. 2009, 83, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Mínguez-Alarcón, L.; Hauser, R.; Gaskins, A.J. Effects of bisphenol A on male and couple reproductive health: A review. Fertil. Steril. 2016, 106, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Mustieles, V.; Pérez-Lobato, R.; Olea, N.; Fernández, M.F. Bisphenol A: Human exposure and neurobehavior. Neurotoxicology 2015, 49, 174–184. [Google Scholar] [CrossRef]
- Han, C.; Hong, Y.C. Bisphenol A, Hypertension, and Cardiovascular Diseases: Epidemiological, Laboratory, and Clinical Trial Evidence. Curr. Hypertens. Rep. 2016, 18, 11. [Google Scholar] [CrossRef]
- Bertoli, S.; Leone, A.; Battezzati, A. Human Bisphenol A Exposure and the “Diabesity Phenotype”. Dose Response 2015, 13, 155932581559917. [Google Scholar] [CrossRef]
- Carwile, J.L.; Michels, K.B. Urinary bisphenol A and obesity: NHANES 2003–2006. Environ. Res. 2011, 111, 825–830. [Google Scholar] [CrossRef]
- Xu, L.C.; Sun, H.; Chen, J.F.; Bian, Q.; Qian, J.; Song, L.; Wang, X.R. Evaluation of androgen receptor transcriptional activities of bisphenol A, octylphenol and nonylphenol in vitro. Toxicology 2005, 216, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.A.; Birnbaum, L.S.; Farabollini, F.; Newbold, R.R.; Rubin, B.S.; Talsness, C.E.; Vandenbergh, J.G.; Walser-Kuntz, D.R.; Vom Saal, F.S. In vivo effects of bisphenol A in laboratory rodent studies. Reprod. Toxicol. 2007, 24, 199–224. [Google Scholar] [CrossRef] [PubMed]
- Li, D.K.; Zhou, Z.; Miao, M.; He, Y.; Wang, J.; Ferber, J.; Herrinton, L.J.; Gao, E.; Yuan, W. Urine bisphenol-A (BPA) level in relation to semen quality. Fertil. Steril. 2011, 95, 625–630. [Google Scholar] [CrossRef]
- Calafat, A.M.; Ye, X.; Wong, L.Y.; Reidy, J.A.; Needham, L.L. Exposure of the U.S. Population to Bisphenol A and 4- tertiary -Octylphenol: 2003–2004. Environ. Health Perspect. 2008, 116, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Michałowicz, J. Bisphenol A – sources, toxicity and biotransformation. Environ. Toxicol. Pharmacol. 2014, 37, 738–758. [Google Scholar] [CrossRef] [PubMed]
- Flint, S.; Markle, T.; Thompson, S.; Wallace, E. Bisphenol A exposure, effects, and policy: A wildlife perspective. J. Environ. Manag. 2012, 104, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Colin, A.; Bach, C.; Rosin, C.; Munoz, J.F.; Dauchy, X. Is drinking water a major route of human exposure to alkylphenol and bisphenol contaminants in France? Arch. Environ. Contam. Toxicol. 2014, 66, 86–99. [Google Scholar] [CrossRef]
- Canesi, L.; Fabbri, E. Environmental Effects of BPA: Focus on Aquatic Species. Dose Response 2015, 13, 1559325815598304. [Google Scholar] [CrossRef]
- Corrales, J.; Kristofco, L.A.; Steele, W.B.; Yates, B.S.; Breed, C.S.; Williams, E.S.; Brooks, B.W. Global Assessment of Bisphenol A in the Environment: Review and Analysis of Its Occurrence and Bioaccumulation. Dose Response 2015, 13, 155932581559830. [Google Scholar] [CrossRef]
- Vogel, S.A. The politics of plastics: The making and unmaking of bisphenol a “safety”. Am. J. Public Health 2009, 99, S559–S566. [Google Scholar] [CrossRef]
- EFSA, European Food Safety Authority. Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA Panel on Food Contact Materials E Flavourings and Processing Aids (CEF). EFSA J. 2015, 13, 3978. [Google Scholar] [CrossRef]
- Commission Directive 2011/8/EU of 28 January 2011 amending Directive 2002/72/EC as regards the restriction of use of Bisphenol A in plastic infant feeding bottles (Text with EEA relevance 4). Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2011:026:0011:0014:EN:PDF (accessed on 14 February 2020).
- Food and Drug Administration (FDA). Food Additive Regulations Amended to No Longer Provide for the Use of BPA-Based Materials in Baby Bottles, Sippy Cups, and Infant Formula Packaging. In 2012, 77 Fed. Reg. 41,899. Available online: https://www.federalregister.gov/documents/2012/07/17/2012-17366/indirect-food-additives-polymers (accessed on 14 February 2020).
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef] [PubMed]
- CDC (Centers for Disease Control and Prevention). Fourth National Report on Human Exposure to Environmental Chemicals; CDC: Atlanta, GA, USA, 2013; pp. 1–770. [Google Scholar]
- Zhang, T.; Sun, H.; Kannan, K. Blood and urinary bisphenol A concentrations in children, adults, and pregnant women from china: Partitioning between blood and urine and maternal and fetal cord blood. Environ. Sci. Technol. 2013, 47, 4686–4694. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, C.; Shin, H.; Kho, Y.; Choi, K. 2019 Comparison of thyroid hormone disruption potentials by bisphenols A, S, F, and Z in embryo-larval zebrafish. Chemosphere 2013, 221, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Tian, X.; Fang, X.; Ji, F. Waterborne exposure to bisphenol F causes thyroid endocrine disruption in zebrafish larvae. Chemosphere 2016, 147, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef]
- Cacho, J.I.; Campillo, N.; Viñas, P.; Hernández-Córdoba, M. Stir bar sorptive extraction coupled to gas chromatography–mass spectrometry for the determination of bisphenols in canned beverages and filling liquids of canned vegetables. J. Chromatogr. A 2012, 1247, 146–153. [Google Scholar] [CrossRef]
- Liao, C.; Liu, F.; Moon, H.B.; Yamashita, N.; Yun, S.; Kannan, K. Bisphenol analogues in sediments from industrialized areas in the United States, Japan, and Korea: Spatial and temporal distributions. Environ. Sci. Technol. 2012, 46, 11558–11565. [Google Scholar] [CrossRef]
- Zhou, X.; Kramer, J.; Calafat, A.M.; Ye, X. Automated on-line column-switching high performance liquid chromatography isotope dilution tandem mass spectrometry method for the quantification of bisphenol A, bisphenol F, bisphenol S, and 11 other phenols in urine. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 944, 152–156. [Google Scholar] [CrossRef]
- Office of Chemical Safety & Pollution Prevention, EPA. Doc. No.740-R1-4004, TSCA Work Plan Chemical Problem Formulation and Initial Assessment: Tetrabromobisphenol A and Related Chemicals Cluster Flame Retardants 10, 2015. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/tbbpa_problem_formulation_august_2015.pdf (accessed on 14 February 2020).
- Yin, N.; Liang, S.; Liang, S.; Yang, R.; Hu, B.; Qin, Z.; Liu, A.; Faiola, F. TBBPA and Its Alternatives Disturb the Early Stages of Neural Development by Interfering with the NOTCH and WNT Pathways. Environ. Sci. Technol. 2018, 52, 5459–5468. [Google Scholar] [CrossRef]
- Malkoske, T.; Tang, Y.; Xu, W.; Yu, S.; Wang, H. A review of the environmental distribution, fate, and control of tetrabromobisphenol A released from sources. Sci. Total Environ. 2016, 569–570, 1608–1617. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, Y.; Kang, D.; Wang, J.; Zhang, Y.; Du, D.; Pan, B.; Lin, Z.; Huang, C.; Dong, Q. Tetrabromobisphenol A and heavy metal exposure via dust ingestion in an e-waste recycling region in Southeast China. Sci. Total Environ. 2016, 541, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, E.; Yamashita, N.; Taniyasu, S.; Lam, J.; Lam, P.K.S.; Moon, H.B.; Jeong, Y.; Kannan, P.; Achyuthan, H.; Munuswamy, N.; et al. Bisphenol A and other bisphenol analogues including BPS and BPF in surface water samples from Japan, China, Korea and India. Ecotoxicol. Environ. Saf. 2015, 122, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Liu, F.; Guo, Y.; Moon, H.B.; Nakata, H.; Wu, Q.; Kannan, K. Occurrence of eight bisphenol analogues in indoor dust from the United States and several Asian countries: Implications for human exposure. Environ. Sci. Technol. 2012, 46, 9138–9145. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.; Kawamura, K. Ubiquity of bisphenol A in the atmosphere. Environ. Pollut. 2010, 158, 3138–3143. [Google Scholar] [CrossRef]
- Yamamoto, T.; Yasuhara, A.; Shiraishi, H.; Nakasugi, O. Bisphenol A in hazardous waste landfill leachates. Chemosphere 2001, 42, 415–418. [Google Scholar] [CrossRef]
- Geens, T.; Neels, H.; Covaci, A. Distribution of bisphenol-A, triclosan and n-nonylphenol in human adipose tissue, liver and brain. Chemosphere 2012, 87, 796–802. [Google Scholar] [CrossRef]
- Sun, Y.; Irie, M.; Kishikawa, N.; Wada, M.; Kuroda, N.; Nakashima, K. Determination of bisphenol A in human breast milk by HPLC with column-switching and fluorescence detection. Biomed. Chromatogr. 2004, 18, 501–507. [Google Scholar] [CrossRef]
- World Health Organization & Food and Agriculture Organization of the United Nations (2011). In Proceedings of the Joint FAO/WHO Expert Meeting to Review Toxicological and Health Aspects of Bisphenol A: Final Report, Including Report of Stakeholder Meeting on Bisphenol A, Ottawa, ON, Canada, 1–5 November 2010. Available online: https://apps.who.int/iris/bitstream/handle/10665/44624/97892141564274_eng.pdf?sequence=1&isAllowed=y (accessed on 14 February 2020).
- Liao, C.; Liu, F.; Alomirah, H.; Loi, V.D.; Mohd, M.A.; Moon, H.B.; Nakata, H.; Kannan, K. Bisphenol S in urine from the United States and seven Asian countries: Occurrence and human exposures. Environ. Sci. Technol. 2012, 46, 6860–6866. [Google Scholar] [CrossRef]
- Liu, K.; Li, J.; Yan, S.; Zhang, W.; Li, Y.; Han, D. A review of status of tetrabromobisphenol A (TBBPA) in China. Chemosphere 2016, 148, 8–20. [Google Scholar] [CrossRef]
- Yang, S.; Wang, S.; Liu, H.; Yan, Z. Tetrabromobisphenol A: Tissue distribution in fish, and seasonal variation in water and sediment of Lake Chaohu, China. Environ. Sci. Pollut. Res. Int. 2012, 19, 4090–4096. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.C.; Chen, S.J.; Zheng, J.; Tian, M.; Feng, A.H.; Luo, X.J.; Mai, B.X. Occurrence of brominated flame retardants (BFRs), organochlorine pesticides (OCPs), and polychlorinated biphenyls (PCBs) in agricultural soils in a BFR-manufacturing region of North China. Sci. Total Environ. 2014, 481, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Cariou, R.; Antignac, J.P.; Zalko, D.; Berrebi, A.; Cravedi, J.P.; Maume, D.; Marchand, P.; Monteau, F.; Riu, A.; Andre, F.; et al. Exposure assessment of French women and their newborns to tetrabromobisphenol-A: Occurrence measurements in maternal adipose tissue, serum, breast milk and cord serum. Chemosphere 2008, 73, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on Tetrabromobisphenol A (TBBPA) and its derivatives in food: TBBPA and its derivatives in food. EFSA J. 2011, 9, 2477. Available online: https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2011.2477 (accessed on 14 February 2020). [CrossRef]
- Harvey, C.B.; Bassett, J.H.D.; Maruvada, P.; Yen, P.M.; Williams, G.R. The rat thyroid hormone receptor (TR) Δβ3 displays cell-, TR isoform-, and thyroid hormone response element-specific actions. Endocrinology 2007, 148, 1764–1773. [Google Scholar] [CrossRef]
- Kublaoui, B.; Levine, M.A. Receptor transduction pathways mediating hormone action. In Pediatric Endocrinology, 4th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2014; pp. 34–89. [Google Scholar]
- Fattori, J.; Campos, J.L.; Doratioto, T.R.; Assis, L.M.; Vitorino, M.T.; Polikarpov, I.; Xavier-Neto, J.; Figueira, A.C. RXR agonist modulates TR: Corepressor dissociation upon 9-cis retinoic acid treatment. Mol. Endocrinol. 2015, 29, 258–273. [Google Scholar] [CrossRef]
- Aagaard, M.M.; Siersbæk, R.; Mandrup, S. Molecular basis for gene-specific transactivation by nuclear receptors. Biochim. Biophys. Acta 2011, 1812, 824–835. [Google Scholar] [CrossRef]
- Guyot, R.; Chatonnet, F.; Gillet, B.; Hughes, S.; Flamant, F. Toxicogenomic analysis of the ability of brominated flame retardants TBBPA and BDE-209 to disrupt thyroid hormone signaling in neural cells. Toxicology 2014, 325, 125–132. [Google Scholar] [CrossRef]
- Jugan, M.L.; Lévy-Bimbot, M.; Pomérance, M.; Tamisier-Karolak, S.; Blondeau, J.P.; Lévi, Y. A new bioluminescent cellular assay to measure the transcriptional effects of chemicals that modulate the alpha-1 thyroid hormone receptor. Toxicol. In Vitro 2007, 21, 1197–1205. [Google Scholar] [CrossRef]
- Iwamuro, S.; Yamada, M.; Kato, M.; Kikuyama, S. Effects of bisphenol A on thyroid hormone-dependent up-regulation of thyroid hormone receptor α and β and down-regulation of retinoid X receptor γ in Xenopus tail culture. Life Sci. 2006, 79, 2165–2171. [Google Scholar] [CrossRef]
- Seiwa, C.; Nakahara, J.; Komiyama, T.; Katsu, Y.; Iguchi, T.; Asou, H. Bisphenol A exerts thyroid-hormone-like effects on mouse oligodendrocyte precursor cells. Neuroendocrinology 2004, 80, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhan, T.; Ma, M.; Xu, C.; Wang, J.; Zhang, C.; Liu, W.; Zhuang, S. Thyroid Disruption by Bisphenol S Analogues via Thyroid Hormone Receptor β: In Vitro, in Vivo, and Molecular Dynamics Simulation Study. Environ. Sci. Technol. 2018, 52, 6617–6625. [Google Scholar] [CrossRef] [PubMed]
- Terasaki, M.; Kosaka, K.; Kunikane, S.; Makino, M.; Shiraishi, F. Assessment of thyroid hormone activity of halogentaed bisphenol A using a yeast two-hybrid assay. Chemosphere 2011, 84, 1527–1530. [Google Scholar] [CrossRef] [PubMed]
- Kudo, Y.; Yamauchi, K.; Fukazawa, H.; Terao, Y. In vitro and in vivo analysis of the thyroid system–disrupting activities of brominated phenolic and phenol compounds in Xenopus laevis. Toxicol. Sci. 2006, 92, 87–95. [Google Scholar] [CrossRef]
- Kitamura, S.; Jinno, N.; Ohta, S.; Kuroki, H.; Fujimoto, N. Thyroid hormonal activity of the flame retardants tetrabromobisphenol A and tetrachlorobisphenol A. Biochem. Biophys. Res. Commun. 2002, 293, 554–559. [Google Scholar] [CrossRef]
- Hofmann, P.J.; Schomburg, L.; Köhrle, J. Interference of Endocrine Disrupters with Thyroid Hormone Receptor–Dependent Transactivation. Toxicol. Sci. 2009, 110, 125–137. [Google Scholar] [CrossRef]
- Sun, H.; Shen, O.X.; Wang, X.R.; Zhou, L.; Zhen, S.; Chen, X. Anti-thyroid hormone activity of bisphenol A, tetrabromobisphenol A and tetrachlorobisphenol A in an improved reporter gene assay. Toxicol. In Vitro 2009, 23, 950–954. [Google Scholar] [CrossRef]
- Otsuka, S.; Ishihara, A.; Yamauchi, K. Ioxynil and tetrabromobisphenol a suppress thyroid-hormone-induced activation of transcriptional elongation mediated by histone modifications and RNA polymerase II phosphorylation. Toxicol. Sci. 2014, 138, 290–299. [Google Scholar] [CrossRef]
- Yang, J.; Chan, K.M. Evaluation of the toxic effects of brominated compounds (BDE-47, 99, 209, TBBPA) and bisphenol A (BPA) using a zebrafish liver cell line, ZFL. Aquat. Toxicol. 2015, 159, 138–147. [Google Scholar] [CrossRef]
- Freitas, J.; Cano, P.; Craig-Veit, C.; Goodson, M.L.; David Furlow, J.; Murk, A.J. Detection of thyroid hormone receptor disruptors by a novel stable in vitro reporter gene assay. Toxicol. In Vitro 2011, 250, 257–266. [Google Scholar] [CrossRef]
- Sheng, Z.G.; Tang, Y.; Liu, Y.X.; Yuan, Y.; Zhao, B.Q.; Chao, X.J.; Zhu, B.Z. Low concentrations of bisphenol a suppress thyroid hormone receptor transcription through a nongenomic mechanism. Toxicol. Appl. Pharmacol. 2012, 259, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, M.; Lei, J.; Ingbar, D.H. Nongenomic actions of L-thyroxine and 3,5,3′-triiodo-L-thyronine. Focus on “L-Thyroxine vs. 3,5,3′-triiodo-L-thyronine and cell proliferation: Activation of mitogen-activated protein kinase and phosphatidylinositol 3-kinase”. Am. J. Physiol. Cell Physiol. 2009, 296, C977–C979. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.; Wang, C.; Ren, F.; Liu, Y.; Zhu, B. Molecular mechanism of endocrine-disruptive effects induced by Bisphenol A: The role of transmembrane G-protein estrogen receptor 1 and integrin αvβ3. J. Environ. Sci. (China) 2019, 75, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.F.; Ren, X.M.; Li, Y.Y.; Yao, X.F.; Li, C.H.; Qin, Z.F.; Guo, L.H. Bisphenol A alternatives bisphenol S and bisphenol F interfere with thyroid hormone signaling pathway in vitro and in vivo. Environ. Pollut. 2018, 237, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Ghisari, M.; Bonefeld-Jorgensen, E.C. Impact of environmental chemicals on the thyroid hormone function in pituitary rat GH3 cells. Mol. Cell. Endocrinol. 2005, 244, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Schriks, M.; Vrabie, C.M.; Gutleb, A.C.; Faassen, E.J.; Rietjens, I.M.C.M.; Murk, A.J. T-screen to quantify functional potentiating, antagonistic and thyroid hormone-like activities of poly halogenated aromatic hydrocarbons (PHAHs). Toxicol. In Vitro 2006, 20, 490–498. [Google Scholar] [CrossRef]
- Kitamura, S. Comparative study of the endocrine-disrupting activity of bisphenol a and 19 related compounds. Toxicol. Sci. 2005, 84, 249–259. [Google Scholar] [CrossRef]
- Hamers, T.; Kamstra, J.H.; Sonneveld, E.; Murk, A.J.; Kester, M.H.A.; Andersson, P.L.; Legler, J.; Brouwer, A. In vitro profiling of the endocrine-disrupting potency of brominated flame retardants. Toxicol. Sci. 2006, 92, 157–173. [Google Scholar] [CrossRef]
- Lee, J.; Kim, S.; Choi, K.; Ji, K. Effects of bisphenol analogs on thyroid endocrine system and possible interaction with 17β-estradiol using GH3 cells. Toxicol. In Vitro 2018, 53, 107–113. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Wei, F.; Zhang, J.; Hao, L.; Jiang, J.; Dang, L.; Mei, D.; Fan, S.; Yu, Y.; Jiang, L. Bisphenol A and estrogen induce proliferation of human thyroid tumor cells via an estrogen-receptor-dependent pathway. Arch. Biochem. Biophys. 2017, 633, 29–39. [Google Scholar] [CrossRef]
- Strack, S.; Detzel, T.; Wahl, M.; Kuch, B.; Krug, H.F. Cytotoxicity of TBBPA and effects on proliferation, cell cycle and MAPK pathways in mammalian cells. Chemosphere 2007, 67, S405–S411. [Google Scholar] [CrossRef] [PubMed]
- English, J.; Pearson, G.; Wilsbacher, J.; Swantek, J.; Karandikar, M.; Xu, S.; Cobb, M.H. New insights into the control of MAP kinase pathways. Exp. Cell Res. 1999, 253, 255–270. [Google Scholar] [CrossRef]
- Gentilcore, D.; Porreca, I.; Rizzo, F.; Ganbaatar, E.; Carchia, E.; Mallardo, M.; De Felice, M.; Ambrosino, C. Bisphenol A interferes with thyroid specific gene expression. Toxicology 2013, 304, 21–31. [Google Scholar] [CrossRef]
- Cao, J.; Guo, L.-H.; Wan, B.; Wei, Y. In vitro fluorescence displacement investigation of thyroxine transport disruption by bisphenol A. J. Environ. Sci. (China) 2011, 23, 315–321. [Google Scholar] [CrossRef]
- Ishihara, A.; Sawatsubashi, S.; Yamauchi, K. Endocrine disrupting chemicals: Interference of thyroid hormone binding to transthyretins and to thyroid hormone receptors. Mol. Cell. Endocrinol. 2003, 199, 105–117. [Google Scholar] [CrossRef]
- Meerts, I.A.T.M. Potent competitive interactions of some brominated flame retardants and related compounds with human transthyretin in vitro. Toxicol. Sci. 2000, 56, 95–104. [Google Scholar] [CrossRef]
- Marchesini, G.R.; Meimaridou, A.; Haasnoot, W.; Meulenberg, E.; Albertus, F.; Mizuguchi, M.; Takeuchi, M.; Irth, H.; Murk, A.J. Biosensor discovery of thyroxine transport disrupting chemicals. Toxicol. Appl. Pharmacol. 2008, 232, 150–160. [Google Scholar] [CrossRef]
- Dong, H.; Wade, M.G. Application of a nonradioactive assay for high throughput screening for inhibition of thyroid hormone uptake via the transmembrane transporter MCT8. Toxicol. In Vitro 2017, 40, 234–242. [Google Scholar] [CrossRef]
- Friesema, E.C.; Kuiper, G.G.; Jansen, J.; Visser, T.J.; Kester, M.H. Thyroid hormone transport by the human monocarboxylate transporter 8 and its rate-limiting role in intracellular metabolism. Mol. Endocrinol. 2006, 20, 2761–2772. [Google Scholar] [CrossRef]
- Strømme, P.; Groeneweg, S.; Lima de Souza, E.C.; Zevenbergen, C.; Torgersbråten, A.; Holmgren, A.; Gurcan, E.; Meima, M.E.; Peeters, R.; Visser, W.E.; et al. Mutated Thyroid Hormone Transporter OATP1C1 Associates with Severe Brain Hypometabolism and Juvenile Neurodegeneration. Thyroid 2018, 28, 1406–1415. [Google Scholar] [CrossRef]
- Trajkovic, M.; Visser, T.J.; Mittag, J.; Horn, S.; Lukas, J.; Darras, V.M.; Raivich, G.; Bauer, K.; Heuer, H. Abnormal thyroid hormone metabolism in mice lacking the monocarboxylate transporter 8. J. Clin. Investig. 2007, 117, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Johannes, J.; Jayarama-Naidu, R.; Meyer, F.; Wirth, E.K.; Schweizer, U.; Schomburg, L.; Köhrle, J.; Renko, K. Silychristin, a Flavonolignan Derived From the Milk Thistle, Is a Potent Inhibitor of the Thyroid Hormone Transporter MCT8. Endocrinology 2016, 157, 1694–1701. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, C.; Youn, H.; Choi, K. Thyroid hormone disrupting potentials of bisphenol A and its analogues - in vitro comparison study employing rat pituitary (GH3) and thyroid follicular (FRTL-5) cells. Toxicol. In Vitro 2017, 40, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Beland, F.A.; Fang, J.L. Effect of triclosan, triclocarban, 2,2′,4,4′-tetrabromodiphenyl ether, and bisphenol A on the iodide uptake, thyroid peroxidase activity, and expression of genes involved in thyroid hormone synthesis. Toxicol. In Vitro 2016, 32, 310–319. [Google Scholar] [CrossRef]
- Porreca, I.; Ulloa Severino, L.; D’Angelo, F.; Cuomo, D.; Ceccarelli, M.; Altucci, L.; Amendola, E.; Nebbioso, A.; Mallardo, M.; De Felice, M.; et al. “Stockpile” of Slight Transcriptomic Changes Determines the Indirect Genotoxicity of Low-Dose BPA in Thyroid Cells. PLoS ONE 2016, 11, e0151618. [Google Scholar] [CrossRef]
- da Silva, M.M.; Gonçalves, C.F.L.; Miranda-Alves, L.; Fortunato, R.S.; Carvalho, D.P.; Ferreira, A.C.F. Inhibition of Type 1 Iodothyronine Deiodinase by Bisphenol A. Horm. Metab. Res. 2019, 51, 671–677. [Google Scholar] [CrossRef]
- Chan, S.; Kachilele, S.; Hobbs, E.; Bulmer, J.N.; Boelaert, K.; McCabe, C.J.; Driver, P.M.; Bradwell, A.R.; Kester, M.; Visser, T.J.; et al. Placental iodothyronine deiodinase expression in normal and growth-restricted human pregnancies. J. Clin. Endocrinol. Metab. 2003, 88, 4488–4495. [Google Scholar] [CrossRef]
- Zoeller, R.T.; Bansal, R.; Parris, C. Bisphenol-A, an environmental contaminant that acts as a thyroid hormone receptor antagonist in vitro, increases serum thyroxine, and alters RC3/neurogranin expression in the developing rat brain. Endocrinology 2005, 146, 607–612. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X.; Li, Y.; Zhou, Z.; Wu, C.; Liu, Z.; Hao, L.; Fan, S.; Jiang, F.; Xie, Y.; et al. Low dose of Bisphenol A enhance the susceptibility of thyroid carcinoma stimulated by DHPN and iodine ecxcess in F344 rats. Oncotarget 2017, 8, 69874–69887. [Google Scholar]
- Silva, M.M.D.; Xavier, L.L.F.; Gonçalves, C.F.L.; Santos-Silva, A.P.; Paiva-Melo, F.D.; Freitas, M.L.D.; Fortunato, R.S.; Alves, L.M.; Ferreira, A.C.F. Bisphenol A increases hydrogen peroxide generation by thyrocytes both in vivo and in vitro. Endocr. Connect. 2018, 7, 1198–1207. [Google Scholar] [CrossRef]
- Saegusa, Y.; Fujimoto, H.; Woo, G.H.; Inoue, K.; Takahashi, M.; Mitsumori, K.; Hirose, M.; Nishikawa, A.; Shibutani, M. Developmental toxicity of brominated flame retardants, tetrabromobisphenol A and 1,2,5,6,9,10-hexabromocyclododecane, in rat offspring after maternal exposure from mid-gestation through lactation. Reprod. Toxicol. 2009, 2, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Miyagawa, M.; Wang, R.S.; Suda, M.; Sekiguchi, S.; Honma, T. Effects of in utero and lactational exposure to bisphenol A on thyroid status in F1 rat offspring. Ind. Health 2005, 43, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Zoeller, R.T. CLARITY-BPA: Bisphenol A or Propylthiouracil on Thyroid Function and Effects in the Developing Male and Female Rat Brain. Endocrinology 2019, 160, 1771–1785. [Google Scholar] [CrossRef] [PubMed]
- Osimitz, T.G.; Droege, W.; Hayes, A.W. Subchronic toxicology of tetrabromobisphenol A in rats. Hum. Exp. Toxicol. 2016, 35, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Fan, S.; Guo, Y.; Tan, R.; Zhang, J.; Zhang, W.; Pan, B.X.; Kato, N. The effects of perinatal bisphenol A exposure on thyroid hormone homeostasis and glucose metabolism in the prefrontal cortex and hippocampus of rats. Brain Behav. 2019, 9, e01225. [Google Scholar] [CrossRef]
- Xu, X.; Liu, Y.; Sadamatsu, M.; Tsutsumi, S.; Akaike, M.; Ushijima, H.; Kato, N. Perinatal bisphenol A affects the behavior and SRC-1 expression of male pups but does not influence on the thyroid hormone receptors and its responsive gene. Neurosci. Res. 2007, 58, 149–155. [Google Scholar] [CrossRef]
- Jiang, W.; Cao, L.; Wang, F.; Ge, H.; Wu, P.C.; Li, X.W.; Chen, G.H. Accelerated reduction of serum thyroxine and hippocampal histone acetylation links to exacerbation of spatial memory impairment in aged CD-1 mice pubertally exposed to bisphenol A. Age (Dordr) 2016, 38, 405–418. [Google Scholar] [CrossRef]
- Van der Ven, L.T.M.; Van de Kuil, T.; Verhoef, A.; Verwer, C.M.; Lilienthal, H.; Leonards, P.E.G.; Schauer, U.M.D.; Cantón, R.F.; Litens, S.; De Jong, F.H.; et al. Endocrine effects of tetrabromobisphenol-A (TBBPA) in Wistar rats as tested in a one-generation reproduction study and a subacute toxicity study. Toxicology 2008, 245, 76–89. [Google Scholar] [CrossRef]
- Silva, B.S.; Bertasso, I.M.; Pietrobon, C.B.; Lopes, B.P.; Santos, T.R.; Peixoto-Silva, N.; Carvalho, J.C.; Claudio-Neto, S.; Manhães, A.C.; Cabral, S.S.; et al. Effects of maternal bisphenol A on behavior, sex steroid and thyroid hormones levels in the adult rat offspring. Life Sci. 2019, 218, 253–264. [Google Scholar] [CrossRef]
- Ahmed, R.G.; Walaa, G.H.; Asmaa, F.S. Suppressive effects of neonatal bisphenol A on the neuroendocrine system. Toxicol. Ind. Health 2018, 34, 397–407. [Google Scholar] [CrossRef]
- Fernandez, M.O.; Bourguignon, N.S.; Arocena, P.; Rosa, M.; Libertun, C.; Lux-Lantos, V. Neonatal exposure to bisphenol A alters the hypothalamic-pituitary-thyroid axis in female rats. Toxicol. Lett. 2018, 285, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Delclos, K.B.; Camacho, L.; Lewis, S.M.; Vanlandingham, M.M.; Latendresse, J.R.; Olson, G.R.; Davis, K.J.; Patton, R.E.; da Costa, G.G.; Woodling, K.A.; et al. Toxicity Evaluation of Bisphenol A Administered by Gavage to Sprague Dawley Rats From Gestation Day 6 Through Postnatal Day 90. Toxicol. Sci. 2014, 139, 174–197. [Google Scholar] [CrossRef] [PubMed]
- Matos, L.P.L.; Penha, R.C.C.; Cardoso-Weide, L.C.; Freitas, M.L.; Silva, D.L.S.G.; Ferreira, A.C.F. Regulation of thyroid sodium-iodide symporter in different stages of goiter: Possible involvement of reactive oxygen species. Clin. Exp. Pharmacol. Physiol. 2018, 45, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, R.S.; Lima de Souza, E.C.; Ameziane-el Hassani, R.; Boufraqech, M.; Weyemi, U.; Talbot, M.; Lagente-Chevallier, O.; de Carvalho, D.P.; Bidart, J.M.; Schlumberger, M.; et al. Functional consequences of dual oxidase-thyroperoxidase interaction at the plasma membrane. J. Clin. Endocrinol. Metab. 2010, 95, 5403–5411. [Google Scholar] [CrossRef] [PubMed]
- Yafune, A.; Taniai, E.; Morita, R.; Akane, H.; Kimura, M.; Mitsumori, K.; Shibutani, M. Immunohistochemical cellular distribution of proteins related to M phase regulation in early proliferative lesions induced by tumor promotion in rat two-stage carcinogenesis models. Exp. Toxicol. Pathol. 2014, 66, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Viguié, C.; Collet, S.H.; Gayrard, V.; Picard-Hagen, N.; Puel, S.; Roques, B.B.; Toutain, P.L.; Lacroix, M.Z. Maternal and fetal exposure to bisphenol A is associated with alterations of thyroid function in pregnant ewes and their newborn lambs. Endocrinology 2013, 154, 521–528. [Google Scholar] [CrossRef]
- Guignard, D.; Gayrard, V.; Lacroix, M.Z.; Puel, S.; Picard-Hagen, N.; Viguié, C. Evidence for bisphenol A-induced disruption of maternal thyroid homeostasis in the pregnant ewe at low level representative of human exposure. Chemosphere 2017, 182, 458–467. [Google Scholar] [CrossRef]
- Fisher, D.A.; Polk, D.H.; Wu, S. Fetal thyroid metabolism: A pluralistic system. Thyroid 1994, 4, 367–371. [Google Scholar] [CrossRef]
- Marelli, F.; Persani, L. How zebrafish research has helped in understanding thyroid diseases. F1000Res. 2017, 6, 2137. [Google Scholar] [CrossRef]
- Terrien, X.; Fini, J.B.; Demeneix, B.A.; Schramm, K.W.; Prunet, P. Generation of fluorescent zebrafish to study endocrine disruption and potential crosstalk between thyroid hormone and corticosteroids. Aquat. Toxicol. 2011, 105, 13–20. [Google Scholar] [CrossRef]
- Raldúa, D.; Thienpont, B.; Babin, P.J. Zebrafish eleutheroembryos as an alternative system for screening chemicals disrupting the mammalian thyroid gland morphogenesis and function. Reprod. Toxicol. 2012, 33, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Zhao, G.; Yang, L.; Zhou, B. Tetrabromobisphenol A caused neurodevelopmental toxicity via disrupting thyroid hormones in zebrafish larvae. Chemosphere 2018, 197, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhou, E.; Yang, Z. Waterborne exposure to BPS causes thyroid endocrine disruption in zebrafish larvae. PLoS ONE 2017, 12, e0176927. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Yang, Y.; Chen, Y.; Tang, W.; Wang, F.; Diao, X. Thyroid Disruption in Zebrafish Larvae by Short-Term Exposure to Bisphenol AF. Int. J. Environ. Res. Public Health 2015, 12, 13069–13084. [Google Scholar] [CrossRef] [PubMed]
- Naderi, M.; Wong, M.Y.L.; Gholami, F. Developmental exposure of zebrafish (Danio rerio) to bisphenol-S impairs subsequent reproduction potential and hormonal balance in adults. Aquat. Toxicol. 2014, 148, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Baumann, L.; Ros, A.; Rehberger, K.; Neuhauss, S.C.F.; Segner, H. Thyroid disruption in zebrafish (Danio rerio) larvae: Different molecular response patterns lead to impaired eye development and visual functions. Aquat. Toxicol. 2016, 172, 44–55. [Google Scholar] [CrossRef]
- Chan, W.K.; Chan, K.M. Disruption of the hypothalamic-pituitary-thyroid axis in zebrafish embryo–larvae following waterborne exposure to BDE-47, TBBPA and BPA. Aquat. Toxicol. 2012, 108, 106–111. [Google Scholar] [CrossRef]
- Kwon, B.; Kho, Y.; Kim, P.G.; Ji, K. Thyroid endocrine disruption in male zebrafish following exposure to binary mixture of bisphenol AF and sulfamethoxazole. Environ. Toxicol. Pharmacol. 2016, 48, 168–174. [Google Scholar] [CrossRef]
- Berto-Júnior, C.; Santos-Silva, A.P.; Ferreira, A.C.F.; Graceli, J.B.; de Carvalho, D.P.; Soares, P.; Romeiro, N.C.; Miranda-Alves, L. Unraveling molecular targets of bisphenol A and S in the thyroid gland. Environ. Sci. Pollut. Res. Int. 2018, 25, 26916–26926. [Google Scholar] [CrossRef]
- Liang, Y.Q.; Huang, G.Y.; Ying, G.G.; Liu, S.S.; Jiang, Y.X.; Liu, S. Progesterone and norgestrel alter transcriptional expression of genes along the hypothalamic–pituitary–thyroid axis in zebrafish embryos-larvae. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2015, 167, 101–107. [Google Scholar] [CrossRef]
- Bianco, A.C.; Salvatore, D.; Gereben, B.Z.; Berry, M.J.; Larsen, P.R. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocr. Rev. 2002, 23, 38–89. [Google Scholar] [CrossRef] [PubMed]
- Lévy-Bimbot, M.; Major, G.; Courilleau, D.; Blondeau, J.P.; Lévi, Y. Tetrabromobisphenol-A disrupts thyroid hormone receptor alpha function in vitro: Use of fluorescence polarization to assay corepressor and coactivator peptide binding. Chemosphere 2012, 87, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Aung, M.T.; Johns, L.E.; Ferguson, K.K.; Mukherjee, B.; McElrath, T.F.; Meeker, J.D. Thyroid hormone parameters during pregnancy in relation to urinary bisphenol A concentrations: A repeated measures study. Environ. Int. 2017, 104, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Kim, U.J.; Oh, J.E. Tetrabromobisphenol A and hexabromocyclododecane flame retardants in infant–mother paired serum samples, and their relationships with thyroid hormones and environmental factors. Environ. Pollut. 2014, 184, 193–200. [Google Scholar] [CrossRef]
- Wang, F.; Hua, J.; Chen, M.; Xia, Y.; Zhang, Q.; Zhao, R.; Zhou, W.; Zhang, Z.; Wang, B. High urinary bisphenol A concentrations in workers and possible laboratory abnormalities. Occup. Environ. Med. 2012, 69, 679–684. [Google Scholar] [CrossRef]
- Przybyla, J.; Geldhof, G.J.; Smit, E.; Kile, M.L. A cross sectional study of urinary phthalates, phenols and perchlorate on thyroid hormones in US adults using structural equation models (NHANES 2007–2008). Environ. Res. 2018, 163, 26–35. [Google Scholar] [CrossRef]
- Chevrier, J.; Gunier, R.B.; Bradman, A.; Holland, N.T.; Calafat, A.M.; Eskenazi, B.; Harley, K.G. Maternal urinary bisphenol A during pregnancy and maternal and neonatal thyroid function in the CHAMACOS study. Environ. Health Perspect. 2013, 121, 138–144. [Google Scholar] [CrossRef]
- Sriphrapradang, C.; Chailurkit, L.; Aekplakorn, W.; Ongphiphadhanakul, B. Association between bisphenol A and abnormal free thyroxine level in men. Endocrine 2013, 44, 441–447. [Google Scholar] [CrossRef]
- Meeker, J.D.; Ferguson, K.K. Relationship between urinary phthalate and bisphenol A concentrations and serum thyroid measures in U.S. adults and adolescents from the National Health and Nutrition Examination Survey (NHANES) 2007–2008. Environ. Health Perspect. 2011, 119, 1396–1402. [Google Scholar] [CrossRef]
- Minatoya, M.; Sasaki, S.; Araki, A.; Miyashita, C.; Itoh, S.; Yamamoto, J.; Matsumura, T.; Mitsui, T.; Moriya, K.; Cho, K.; et al. Cord Blood Bisphenol A Levels and Reproductive and Thyroid Hormone Levels of Neonates. Epidemiology 2017, 28, S3–S9. [Google Scholar] [CrossRef]
- Andrianou, X.D.; Gängler, S.; Piciu, A.; Charisiadis, P.; Zira, C.; Aristidou, K.; Piciu, D.; Hauser, R.; Makris, K.C. Human Exposures to Bisphenol A, Bisphenol F and Chlorinated Bisphenol A Derivatives and Thyroid Function. PLoS ONE 2016, 11, e0155237. [Google Scholar] [CrossRef] [PubMed]
- Romano, M.E.; Webster, G.M.; Vuong, A.M.; Thomas Zoeller, R.; Chen, A.; Hoofnagle, A.N.; Calafat, A.M.; Karagas, M.R.; Yolton, K.; Lanphear, B.P.; et al. Gestational urinary bisphenol A and maternal and newborn thyroid hormone concentrations: The HOME study. Environ. Res. 2015, 138, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Lu, J.; Xu, M.; Xu, Y.; Li, M.; Liu, Y.; Tian, X.; Chen, Y.; Dai, M.; Wang, W.; et al. Urinary bisphenol A concentration and thyroid function in Chinese adults. Epidemiology 2013, 24, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Calafat, A.M.; Hauser, R. Urinary Bisphenol A Concentrations in relation to serum thyroid and reproductive hormone levels in men from an infertility clinic. Environ. Sci. Technol. 2010, 44, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Sugiura-Ogasawara, M.; Ozaki, Y.; Sonta, S.; Makino, T.; Suzumori, K. Exposure to bisphenol A is associated with recurrent miscarriage. Hum. Reprod. 2005, 20, 2325–2329. [Google Scholar] [CrossRef] [PubMed]
- Geens, T.; Dirtu, A.C.; Dirinck, E.; Malarvannan, G.; Van Gaal, L.; Jorens, P.G.; Covaci, A. Daily intake of bisphenol A and triclosan and their association with anthropometric data, thyroid hormones and weight loss in overweight and obese individuals. Environ. Int. 2015, 76, 98–105. [Google Scholar] [CrossRef]
- Takeuchi, T.; Tsutsumi, O.; Nakamura, N.; Ikezuki, Y.; Takai, Y.; Yano, T.; Taketani, Y. Gender difference in serum bisphenol A levels may be caused by liver UDP-glucuronosyltransferase activity in rats. Biochem. Biophys. Res. Commun. 2004, 325, 549–554. [Google Scholar] [CrossRef]
- Takeuchi, T.; Tsutsumi, O. Serum bisphenol A concentrations showed gender differences, possibly linked to androgen levels. Biochem. Biophys. Res. Commun. 2002, 291, 76–78. [Google Scholar] [CrossRef]
- Sur, U.; Erkekoglu, P.; Bulus, A.D.; Andiran, N.; Kocer-Gumusel, B. Oxidative stress markers, trace elements and endocrine disrupting chemicals in children with Hashimoto’s thyroiditis. Toxicol. Mech. Methods 2019, 29, 1–30. [Google Scholar] [CrossRef]
- Chailurkit, L.; Aekplakorn, W.; Ongphiphadhanakul, B. The Association of Serum Bisphenol A with Thyroid Autoimmunity. Int. J. Environ. Res. Public Health 2016, 13, 1153. [Google Scholar] [CrossRef]
- American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer; Cooper, D.S.; Doherty, G.M.; Haugen, B.R.; Kloos, R.T.; Lee, S.L.; Mandel, S.J.; Mazzaferri, E.L.; McIver, B.; Pacini, F.; et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009, 19, 1167–1214. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zhou, Y.; Fu, C.; Wang, H.; Huang, P.; Wang, B.; Su, M.; Jiang, F.; Fang, H.; Zhao, Q.; et al. Influence of Bisphenol A on Thyroid Volume and Structure Independent of Iodine in School Children. PLoS ONE 2015, 10, e0141248. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ying, Y.; Zhang, C.; Wang, W.; Li, Y.; Feng, Y.; Liang, J.; Song, H.; Wang, Y. Bisphenol A exposure and risk of thyroid nodules in Chinese women: A case-control study. Environ. Int. 2019, 126, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, J.; Jiang, F.; Xie, Y.; Zhang, X.; Jiang, L. Higher urinary bisphenol A concentration and excessive iodine intake are associated with nodular goiter and papillary thyroid carcinoma. Biosci. Rep. 2017, 37, BSR20170678. [Google Scholar] [CrossRef]
- Marotta, V.; Russo, G.; Gambardella, C.; Grasso, M.; La Sala, D.; Chiofalo, M.G.; D’Anna, R.; Puzziello, A.; Docimo, G.; Masone, S.; et al. Human exposure to bisphenol AF and diethylhexylphthalate increases susceptibility to develop differentiated thyroid cancer in patients with thyroid nodules. Chemosphere 2019, 218, 885–894. [Google Scholar] [CrossRef]
- Puzianowska-Kuznicka, M.; Nauman, A.; Madej, A.; Tanski, Z.; Cheng, S.; Nauman, J. Expression of thyroid hormone receptors is disturbed in human renal clear cell carcinoma. Cancer Lett. 2000, 155, 145–152. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, L.; Yang, L.; Zhou, B. Bioconcentration and metabolism of decabromodiphenyl ether (BDE-209) result in thyroid endocrine disruption in zebrafish larvae. Aquat. Toxicol. 2012, 110–111, 141–148. [Google Scholar] [CrossRef]
- Jia, P.P.; Ma, Y.B.; Lu, C.J.; Mirza, Z.; Zhang, W.; Jia, Y.F.; Li, W.G.; Pei, D.S. The Effects of Disturbance on Hypothalamus-Pituitary-Thyroid (HPT) Axis in Zebrafish Larvae after Exposure to DEHP. PLoS ONE 2016, 11, e0155762. [Google Scholar] [CrossRef]
- Arnaldi, L.A.; Borra, R.C.; Maciel, R.M.; Cerutti, J.M. Gene expression profiles reveal that DCN, DIO1, and DIO2 are underexpressed in benign and malignant thyroid tumors. Thyroid 2005, 15, 210–221. [Google Scholar] [CrossRef]
- Li, C.G.; Nyman, J.E.; Braithwaite, A.W.; Eccles, M.R. PAX8 promotes tumor cell growth by transcriptionally regulating E2F1 and stabilizing RB protein. Oncogene 2011, 30, 4824–4834. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, H.; Wang, W.; Ru, S. Exposure to monocrotophos pesticide causes disruption of the hypothalamic–pituitary–thyroid axis in adult male goldfish (Carassius auratus). Gen. Comp. Endocrinol. 2013, 193, 158–166. [Google Scholar] [CrossRef]
- Latina, A.; Gullo, D.; Trimarchi, F.; Benvenga, S. Hashimoto’s thyroiditis: Similar and dissimilar characteristics in neighboring areas. Possible implications for the epidemiology of thyroid cancer. PLoS ONE 2013, 8, e55450. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.; Xia, Y.; Zhang, B.; Li, J.; Jiang, Y. A meta-analysis of Hashimoto’s thyroiditis and papillary thyroid carcinoma risk. Oncotarget 2017, 8, 62414–62424. [Google Scholar] [CrossRef] [PubMed]
- Ginabreda, M.G.P.; Cardoso, L.C.; Nobrega, F.M.; Ferreira, A.C.F.; Gonçalves, M.D.C.; Vaisman, M.; Carvalho, D.P. Negative correlation between thyroperoxidase and dual oxidase H2O2-generating activities in thyroid nodular lesions. Eur. J. Endocrinol. 2008, 158, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, Y.; Wang, L.; Wang, X.; Sun, C.; Xing, M.; Zhao, W. Strong association of high urinary iodine with thyroid nodule and papillary thyroid cancer. Tumour Biol. 2014, 35, 11375–11379. [Google Scholar] [CrossRef]
- Guan, H.; Ji, M.; Bao, R.; Yu, H.; Wang, Y.; Hou, P.; Zhang, Y.; Shan, Z.; Teng, W.; Xing, M. Association of High Iodine Intake with the T1799A BRAF Mutation in Papillary Thyroid Cancer. J. Clin. Endocrinol. Metab. 2009, 94, 1612–1617. [Google Scholar] [CrossRef]
- Filetti, S.; Bidart, J.; Arturi, F.; Caillou, B.; Russo, D.; Schlumberger, M. Sodium/iodide symporter: A key transport system in thyroid cancer cell metabolism. Eur. J. Endocrin. 1999, 141, 443–457. [Google Scholar] [CrossRef]
| (a) Bisphenol A | ||
| Environmental Matrix | Concentration | Reference |
| Surface water | nd-1.95 µg/L | [47] |
| Sediments (industrialized areas) | nd-13,370 µg/kg dry weight | [41] |
| Soil | <0.01–1000 µg/kg | [29] |
| Indoor dust | nd-39.1 µg/g | [48] |
| Atmosphere | 10−3–1.74 ng/m3 | [49] |
| Landfill leachate (hazardous waste site) | Up to 17,200 µg/L | [50] |
| Human Body | Concentration | Reference |
| Brain | Mean: 0.91 ng/g | [51] |
| Liver | Mean: 1.30 ng/g | [5] |
| Adipose tissue | Mean: 3.78 ng/g | [51] |
| Breast milk | Mean: 0.61 µg/L | [52] |
| Blood (adults) | Mean: 0.20 µg/L | [36] |
| Cord blood | Mean: 0.13 µg/L | [36] |
| Urine (European adult population) | Geometric mean: 2.5–3.6 µg/L | [31] |
| Urine (North America children) | Geometric mean: 1.3–3.7 µg/L | [31] |
| Urine (North America adults) | Geometric mean: 1.0–2.6 µg/L | [31] |
| Age Group/Source of Exposure | Average External Exposure | Reference |
| Infants (0–3 month)/Formula fed from polycarbonate bottles | 2.4 µg/kg/day | [53] |
| Infants (0–6 month)/Formula fed from non- polycarbonate bottles | 0.03 µg/kg/day | [31] |
| Infants (6–12 month) and toddlers (12–36 month)/Diet | 0.375 µg/kg/day | [31] |
| Infants (6–12 month) and toddlers (12–36 month)/Oral dust and toys | 0.007–0.009 µg/kg/day | [31] |
| Infants (0–12 month) and toddlers (12–36 month)/Inhalation | 0.7 µg/kg/day | [31] |
| General population (>3 years)/Diet | 0.116–0.290 μg/kg/day | [31] |
| General population (>3 years)/Thermal paper | 0.059–0.094 µg/kg/day | [31] |
| General population (>3 years)/Cosmetics | 0.002 µg/kg/day | [31] |
| General population (>3 years)/Inhalation | 0.2–0.4 µg/kg/day | [31] |
| (b) Principal Bisphenol A Substitutes | ||
| Environmental Matrix | Concentration | Reference |
| Surface water (BPF) | nd-2.850 µg/L | [47] |
| Sediments in industrialized areas (BPF) | nd-9650 µg/kg dry weight | [41] |
| Indoor dust (sum of several bisphenols including BPF, BPS, BPZ) | 0.00083–26.6 μg/g | [49] |
| Human Body | Concentration | Reference |
| Urine (BPS: general population—USA/Asian countries) | Geometric mean: 0.030–1.18 µg/L | [54] |
| Age Group | Estimated Exposure | Reference |
| Children and adolescents (<20 years)—USA/Asian countries (BPS) | Median: 0.009 μg/kg/day | [54] |
| Adults (≥20 years) – USA/Asian countries (BPS) | Median: 0.004 μg/kg/day | [54] |
| (c) Tetrabromobisphenol A | ||
| Environmental Matrix | Concentration | Reference |
| Atmosphere (e-waste dismantling site) | 66.01–95.04 ng/m3 | [55] |
| Indoor dust | 42.21–46,191 ng/g dry weight | [46] |
| Sediments | Up to 518 ng/g | [56] |
| Soil (industrialized areas) | 1.64–7758 ng/g dry weight | [57] |
| Surface water | 0.85–4.87 μg/L | [56] |
| Human Body | Concentration | Reference |
| Breast milk | 4.110 ng/g lipid weight | [58] |
| Cord serum | Mean: 0.199 ng/g fresh weight | [58] |
| Age group/Source of Exposure | Average External Exposure | Reference |
| Infants/Breast-feeding | <0.00018–0.171 μg/kg/day | [59] |
| Infants/Dust ingestion (e-waste recycling site) | 0.00031–0.054 μg/kg/day | [46] |
| Adults/Dust ingestion (e-waste recycling site) | 0.00004–0.0075 μg/kg/day | [46] |
| Adults/High fish consumers | 0.00026 μg/kg/day | [59] |
| Species | Model | Method | Exposure Time | Doses Tested | Principal Results | Reference |
|---|---|---|---|---|---|---|
| Interference with T3 transcriptional Activity | ||||||
| Yeast (Saccharomyces cerevisiae Y190) | Yeast cells | Recombinant two-hybrid yeast assay | 2.5 h | 0.005 nM–50 μM BPA/BPS/TBBPA/TBBPS ±10−4 M T3 | Antagonistic activity of BPs toward TRβ in a dose-dependent manner, with TBBPS showing the strongest antagonistic activity. IC10 BPA = 884 ± 65.3 nM IC10 BPS = 312 ± 25.9 nM IC10 TBBPA = 21.1 ± 9.6 nM IC10 TBBPS = 10.1 ± 5.1 nM | [68] |
| Yeast (Saccharomyces cerevisiae Y190) | Yeast cells | Yeast two-hybrid assay | 4 h | 10−8–10−4 M BPA/TCBPA/TBBPA ± rat liver S9 preparation | Agonistic activity of TBBPA toward TRα with a dose-dependent response curve. After exposure to S9 metabolic activation, increase of agonistic activity of TBBPA and TCBPA. No activity of BPA. | [69] |
| 0/1.6*10−7/8*10−7/4*10−6/2*10−5 M BPA/TCBPA/TBBPA ±100 nM T3 ± rat liver S9 preparation | With T3, significant antagonistic activity of TBBPA and TCBPA toward TRα, enhanced by exposure to S9 metabolic activation at the same concentration. | |||||
| Zebrafish (Danio rerio) | Hepatocyte (ZFL cells) | Luciferase reporter gene assay | 24 h | 5/10/25/37.5% LC50 BPA/TBBPA | No induction of TR transcriptional activity by BPA or TBBPA alone. | [75] |
| 1/2.5/5/10/25% LC50 BPA/TBBPA +0.1 nM T3 | With T3, decrease of transcriptional activity by BPA. | |||||
| Amphibian (Xenopus laevis) | Tadpole tail culture | Semi-quantitative RT-PCR | 5 days | 10−7–10−5 M BPA | Inhibition of Trα and Trβ mRNA expression with a greater effect on the expression of Trβ than Trα. Moderate suppression of RXRγ mRNA expression. | [66] |
| 10−5 M ±2.5*10−8/10−7/4*10−7 T3 | T3 counteracts the inhibitory effects of BPA on Trα and Trβ mRNA expression, and, in the case of Trβ, dose-dependently. Dose-dependent suppressive effect of T3 on RXRγ gene expression independently of the presence of BPA. | |||||
| Amphibian (Xenopus laevis) | XL58-TRE-Luc cells | Luciferase reporter gene assay | 24 h | 10−8–10−6 M BPA/TBBPA ±2 nM T3 | With T3, inhibition of transcription in a dose-dependent manner. In the absence of T3, agonistic activity. | [70] |
| Amphibian (Xenopus laevis) | XL58-TRE-Luc cells | Luciferase reporter gene assay | 24 h | 0/10−8–10−6 TBBPA ±2 nM T3 | With T3, inhibition of transcription in a dose-dependent manner. | [74] |
| Chinese hamster | Ovary (CHO-K1 cells) | Luciferase reporter gene assay | 24 h | 10−10–10−4 M BPA/TBBPA/TCBPA | Suppression of transcription in cell transfected with TRα1 or TRβ1 by both TBBPA and TCBPA | [10] |
| 0/3.1/6.3/13/25/50/100 µM TBBPA/TCBPA +10 nM T3 | With T3, inhibition of transcriptional activities by TBBPA and TCBPA/ | |||||
| Mouse | Cerebellum (C17.α cells) | Luciferase reporter gene assay | 24 h | 0/10−9–10−5 M TBBPA ±0.1/1/10 nM T3 | Antagonistic effect at least in part independent from T3 concentration. | [64] |
| Mouse | Oligodendrocyte precursors cells | Stimulation/inhibition | 48 h | 0/10−5 M BPA ±100 nM T3 | No variation in TRα levels; TRβ1 levels significantly decreased compared to controls. | [67] |
| Rat | Adrenal medulla pheochromocytoma (PC12 cells) | Luciferase reporter gene assay | 16 h | 0/10/20/40/60/100 µM TBBPA ±1/100 nM T3 | Agonistic activity in the absence of T3. With 1 nM T3, antagonistic activity counteracted by large excess of T3. | [65] |
| Rat | Thyroid pituitary tumor (GH3 cells) | Luciferase reporter gene assay | 24 h | 0–500 µM BPA/0–100 µM TBBPA/TCBPA ±0.25 nM T3 | With T3, slight induction of luciferase activity at doses up to 1 µM. Without T3, no effect from TBBPA and TCBPA. | [76] |
| 0.25 nM T3 ±0/0.5/1/5/10/15 µM AM | AM induced T3-mediated response up to 1 µM. | |||||
| Rat | Thyroid pituitary tumor (GH3 cells) | Luciferase reporter gene assay | 24 h | 0/0.1/1/5/10/50 µM BPA/BPS/BPF ±1 nM T3 | Agonistic activity of all chemicals on TH signaling in the absence of T3 and of BPA and BPF in the presence of T3. | [80] |
| African green monkey (Cerchopitecus aethiops) | Kidney (CV-1 cells) | Luciferase reporter gene assay | 24 h | 10−6–10−4 M TBBPA/TCBPA/BPA +10 nM T3 | With T3, antagonistic activity in cells transfected with TRβ1. In the absence of T3, no effects of the three chemicals. | [73] |
| African green monkey (Cerchopitecus aethiops) | Kidney (CV-1 cells) | Luciferase reporter gene assay | n.d. | 10−9–10−7 M BPA ±0.1 nM T3 | With T3, suppression of TR-mediated transcription also in the presence of SRC1. No effects of BPA on transcription in the absence of T3. | [77] |
| Mammalian two-hybrid assay | n.d. | 10−8 M BPA ±0.1 nM T3 | No effects of BPA on T3-mediated binding of SRC1 to TRβ1. | |||
| 10−9–10−7 M BPA ±0.1 nM T3/±10 nM T4 | Transcription activated by increasing concentrations of BPA in the presence of both T3/T4 and NCor/SMRT1. | |||||
| 10−8 M BPA ±0.1 nM T3/±10 nM T4 | Overexpression of either β-integrin or c-Src reduced recruitment of N-CoR or SMRT to TR-β1 stimulated by BPA in the presence of T3/T4. | |||||
| Human | Hepatoblastoma (HpeG2 cells) | Luciferase reporter gene assay Mammalian two-hybrid assay | 24 h | 10−9/10−7/10−5 M BPA +10 nM T3 | With T3, dose-dependent inhibition of transcription mediated by native TRα1 and TRβ1. | [2] |
| n.d. | 10−9/10−7/10−5 M BPA ±1/3/6 nM T3 | Enhancement of interaction of TRs with N-CoR in a dose-dependent manner. | ||||
| Human | Hepatocarcinoma (HepG2 cells) | Luciferase reporter gene assay | 24 h | 10−11–10−5 M TBBPA ±1 nM T3 | Activation of expression in the absence of T3 and antagonistic effect with T3 at the same dose (10−4M). | [72] |
| qRT-PCR | 24 h | 10−5 M TBBPA ±1 nM T3 | Antagonistic effect on T3-induced DIO1 expression. | |||
| Human | Embryonic kidney (HEK293 cells) | Luciferase reporter gene assay | 24 h | 0, 10−9–10−5 M TBBPA ±0.1/1/10 nM T3 | Antagonistic effect stronger at lower T3 concentration. | [64] |
| Cell Proliferation | ||||||
| Rat | Thyroid pituitary tumor (GH3 cells) | WST-1 cell proliferation assay | 48/96 h | 10−9–10−6 M BPA/BPAF/BPAP/BPB /BPC/BPF/BPM/BPP/BPS/BPZ ±6.4*10−10 M T3-EC50 | All BPA analogues alone stimulated cell proliferation at the highest concentration. BPAF had the strongest effect. With T3, inhibition of cell proliferation at 48 h; agonistic effects at 96 h by high doses of BPA, BPAF, BPF, BPS, BPZ. | [85] |
| 10−9–10−6 M BPA/BPAF ±10−12 M E2/±6.4*10−10 M T3-EC50 | Additive like-effects of co-treatment with BPA analogues and E2. With T3, inhibition of analogues alone and enhancement of cell proliferation by co-treatment with E2. | |||||
| Rat | Thyroid pituitary tumor (GH3 cells) | WST-1 cell proliferation assay | 7 days | 10−8–10−4 M TTBPA/TCBPA ±0.1/1 nM T3 | Stimulation of cell growth and, with T3, no inhibition of induction of GH3 cell growth. | [71] |
| GH-production assay | 48 h | 10−8–10−4 M TTBPA/TCBPA | Stimulation of GH release from cells. | |||
| Rat | Thyroid pituitary tumor (GH3 cells) | GH production assay | 48 h | 10−8–10−5 M BPA/TTBPA/TCBPA | Stimulation of GH release from cells by only TTBPA and TCBPA. With T3, no inhibition of cell growth. | [83] |
| Rat | Thyroid pituitary tumor (GH3 cells) | T-screen assay | n.d. | 0/0.1/1/5/10/50 µM BPA/BPS/BPF ±1 nM T3 ±2 µM AM | For all chemicals, induction of cell proliferation only with T3, and inhibition of growth in the absence of T3. Agonistic actions of BPs were antagonized by AM. | [80] |
| Rat | Thyroid pituitary tumor (GH3 cells) | T-screen assay | 6 days | 10−8–10−5 M BPA/BPA-DM/TBBPA ±0.5 nM T3 | Stimulation of growth by all tested chemicals. In presence of T3, potentiating effect on T3-induced growth. | [81] |
| 0.5/1 nM T3; 5*10−7–5*10−6 BPA/BPA-DM; 1*10−5/2.5*10−5 M TBBPA ±1 nM ICI | Suppression of induced cell proliferation by the antiestrogen ICI. None of the compounds able to counteract the inhibitory effects of ICI. | |||||
| Rat | Thyroid pituitary tumor (GH3 cells) | T-screen assay | 96 h | 10−7–10−5 M BPA/TBBPA/TCBPA ±0.25 nM T3 | No effects on growth for all compounds in the absence of T3. With T3, BPA stimulated growth with maximum potentiation at 10−6M, then cytotoxicity. | [82] |
| Rat | Thyroid pituitary tumor (GH3 cells) | T-screen assay | 96 h | 10−12–10−6 M TBBPA ±0.25 nM T3 | With T3, potentiation of T3-mediated cell growth. In the absence of T3, no effects on cell proliferation. | [84] |
| Human | PTC (BHP10-3 cells) | Cell Counting Kit-8 | 24/48/72 h | 10−8–10−3 M BPA 10−9–10−4 M E2 | Similar proliferative effects of BPA and E2 with non monotonic dose-response curve and progressive effects over time. | [86] |
| Cytotoxicity | ||||||
| Zebrafish (Danio rerio) | Hepatocyte (ZFL cells) | Fluorescence (AlamarBlueTM assay) | 24/96 h | 0/10–1000 µM BPA | 24 h LC50 of BPA: 367.1 µM 96 h LC50 of BPA: 357.6 µM | [75] |
| 0/3.16/10 µM TBBPA | 24 h LC50 of TBBPA: 4 µM 96 h LC50 of TBBPA: 4.2 µM | |||||
| Amphibian (Xenopus laevis) | XL58-TRE-Luc cells | Cell Count Reagent SF kit | 48 h | 0/0.5/1/2/4/8/16 µM BPA/TBBPA | Cell viability not compromised up to 4 µM for both chemicals. | [70] |
| Chinese hamster | Ovary (CHO-K1 cells) | EGFP fluorescence | 24 h | 0/3.1/6.3/13/25/50/100 µM TBBPA/TCBPA +10 nM T3 | Cytotoxicity at the highest concentrations tested for all chemicals. | [10] |
| African green monkey (Cerchopitecus aethiops | Kidney (CV−1 cells) | MTT assay | 24 h | 1/10/20/50/100 µM TBBPA/TCBPA/BPA ±10 nM T3 | Cytotoxicity of all chemicals at the highest concentration tested in the absence and in the presence of T3. | [73] |
| Rat | Adrenal medulla pheochromocytoma (PC12 cells) | MTT assay | 16 h | 100 µM TBBPA/TCBPA | No significant effects on cell viability. | [65] |
| Rat | Thyroid pituitary tumor (GH3 cells) | Fluorescence (AlamarBlueTM) assay | 4 h | 0–500 µM BPA/0–100 µM TBBPA/TCBPA ±0.25 nM T3 | At doses >10 µM TBBPA and TCBPA or 100 µM BPA visible cytotoxicity. | [76] |
| Rat | Immortalized thyroid follicular cells (FRTL-5 cells) | MTT-assay | 24/72 h | 10−9–10−4 M BPA | No effects on cell survival at any dose at either 1 day or 3 days of treatment. | [79] |
| Human | Thyroid anaplastic carcinoma (Cal-62 cells) | MTT assay (on each cell type) | 24 h | 0/25/50/75/100/150/200 µM BPA/TBBPA | TBBPA EC50 Cal 62 cells: 200 µM A549 cells: 168 µM NRK cells: 52 µM No toxic effects of BPA in the tested concentration range. | [87] |
| Rat | Kidney epithelial (NRK cells) | |||||
| Human | Epithelial alveolar type II-like lung (A549 cells) | DNA synthesis by BrdU-assay (on each cell type) | 24/48/72/ 96/120 h | 0/25/50/75/100/150/200 µM TBBPA ±10 µM U0126 | NRK cells the most sensitive to TBBPA (decreased growth at >10 µM). In Cal-62 cells, significant inhibition of growth after 24 exposure to TBBPA >100 µM or 10 µM U0126. | |
| Competitive Binding With Thyroid Hormone Binding Proteins | ||||||
| Amphibian (Xenopus laevis) | XL58-TRE-Luc cells | TTR and TR competitive binding assay | 1–1.5 h | 10−10–10−5 M BPA/TBBPA +0.1 nM 125I-T3 | Inhibition of T3 binding to TTR (TBBPA IC50 = 3.7+0.29 nM; BPA IC50 = 1670+30 nM) and, with weaker affinity, to TR. | [70] |
| Chinese hamster | Ovary (CHO-K1 cells) | Competitive binding assay | 40 min. | 10−7–10−4 M BPA/TTBPA/TCBPA +0.1 nM 125I-T3 | Inhibition of binding to TR (IC50TBBPA = 3.5 µM; IC50TCBPA = 9.5 µM). | [10] |
| Rat | Thyroid pituitary (MtT/E-2 cells) | Competitive binding assay | 40 min. | 10−7–10−4 M TTBPA/TCBPA; 10−5–10−4 M BPA +0.1 nM 125I-T3 | Inhibition of T3 binding to TR by TBBPA and TCBPA. Little effect by BPA. | [71] |
| Rat | Thyroid pituitary tumor (GH3 cells) | T4-TTR competition binding assay | Overnight | 1 nM–1 µM TBBPA + 55 nM (125 I-T4+T4) | Potent antagonism with T4 in TTR binding. IC50 = 0.031 µM. | [85] |
| Rat | Liver microsomes | T4-TTR competition binding assay | Overnight | 1.95–500 nM TBBPA/TCBPA + 55 nM (125I-T4+T4) | For TBBPA, maximum displacement (96.5%) of T4 from TTR at 500 nM. IC50 = 7.7 ± 0.9 nM | [92] |
| Perturbation of Thyroid Hormone Uptake | ||||||
| Dog | Kidney tubule (MDCK cells) | MCT8HTS assay | 10 min. | 2/4/8/18/32/62/125/250 µM BPA + 3.3 µM T3 | Inhibition of T3 uptake mediated by MCT8. | [94] |
| Mouse | Primary astrocytes | Nonradioactive uptake assay | 15 min. | 10 µM BPA + 10 µM T3 | Decrease in MCT8-mediated T3 uptake. | [98] |
| Dysregulation of Gene Expression | ||||||
| Zebrafish (Danio rerio) | Hepatocyte (ZFL cells) | qRT-PCR | 24 h | 10/25/50% LC50 BPA/TBBPA | Inhibition of expression of Dio1, Dio3, Trb, Sult1-st1, -st2, -st3, -st-5 and Ugt2a1, and induction of Ugt1ab genes by BPA. Inhibition of expression of Dio3, and up-regulation of Ttr, Sult1-st2, sult1-st5, and Ugt2a1 by TBBPA. | [75] |
| Amphibian (Xenopus laevis) | Tadpole tail culture | Semi-quantitative RT-PCR | 5 days | 10−7–10−5 M BPA ±2.5*10−8/10−7/4*10−7 T3 | Inhibition of Trα and Trβ mRNA expression. T3 counteracts the inhibitory effects of BPA on Trα and Trβ mRNA expression dose-dependently. | [66] |
| Rat | Liver and brown adipose tissue | Deiodinase 1 assay | 1 h | 0/0.005/0.05/0.5/5 mmol/L BPA | Inhibition of both hepatic DIO1 and brown adipose tissue DIO2 activities, with a greater effect on DIO1. IC50 (D1 activity) = 0.183 mmol/L IC50 (D2 activity) = 1.11 mmol | [102] |
| Deiodinase 2 assay | 3 h | |||||
| Rat | Thyroid pituitary tumor (GH3 cells) | qRT-PCR | 48 h | 10−9–10−6 M BPA/BPAF ±10−12 M E2 | Inhibition of transcription of Tshβ, Trα, and Trβ, Dio1 and Dio2. With E2, a more pronounced decrease of Trα, Trβ and Dio2, and increase of Tshβ transcription | [85] |
| Rat | Immortalized thyroid follicular cells (FRTL-5 cells) | qRT-PCR | 24 h | 0/1/10/100 mg/LBPA/BPB/BPF/BPS 0/0.1/1/10 mg/L BPAF/BPAP/BPC/BPM/BPP/BPZ | Stimulation of transcription of Tsh-r, Pax8, Nkx2-1, S lc5a5, Tg, Tpo by BPA, BPAF, BPAP, BPM, BPS. | [99] |
| Thyroid pituitary Tumor (GH3 cells) | 48 h | 0/0.01/0.1/1/10 mg/L BPA/BPAF/BPAP/BPB/BPC/ BPF/BPM/BPP/BPS/BPZ | Inhibition of transcription of Tshβ, Trα, and Trβ, and Dio2 by BPA, BPF, BPM, and BPZ. Some analogues but not BPA down-regulated Dio1. | |||
| Rat | Immortalized thyroid follicular cells (FRTL-5 cells) | qRT-PCR | 6/24/48 h | 0/10/30/100 µM BPA | Up-regulation of Tg, Pax-8, Foxe-1 and down-regulation of Slc5a5, Tpo, and Nkx2-1 transcripts at the highest dose. | [100] |
| Iodine uptake assay | 1 h | 10−7–10−4 M BPA +10 µM NaI | Concentration-dependent decrease of iodine uptake. | |||
| 24/48 h | 0/10/30/100 µM BPA | Significant decrease in iodine uptake at non-cytotoxic doses of BPA in the absence of NaI | ||||
| Rat | Immortalized thyroid follicular cells (FRTL-5 cells) | Microarray analysis/ qRT-PCR | 1/3/7 days | 10−9 M BPA | Deregulation of 372 and 1041 genes after 3 and 7 days, respectively. Most genes had a fold change >2 at both time points. Following exposure longer than 7 days, inhibition of genes involved in the DNA replication and repair network. | [101] |
| Alkaline comet assay/ TUNEL assay | 28 days + 5 days UV | 10−9 M BPA | After irradiation at 48 and 96 h, higher content of DNA damage in the BPA-treated cells. Until 120 h post irradiation, higher apoptotic levels in the BPA-treated cells. | |||
| Rat | Immortalized thyroid follicular cells (FRTL-5 cells) | Luciferase reporter gene assay | 24 h | 10−15–10−4 µM BPA ±1.78/3.44/6.87/13.7/27.5 ng/µl TSH | Enhancement of Tg-promoter activity also in the presence of the highest dose of TSH. | [89] |
| 10−9 M BPA ±1 µM ICI/TAM | No effect of the two antiestrogen on the activity of Tg-promoter induced by BPA. | |||||
| Human | Ovary cells (SVKO3 cells) | qRT-PCR | 24 h/72 h | 10−9–10−4 M BPA | Increase of transcription levels of Slc5a5, Tpo, Tsh-r, Tg, Pax8, Nkx2-1, Foxe 1. Increase of transcription of PAX8 also in SVKO3 cells. | |
| Species | Method | Window of Exposure | Doses Tested | Principal Results | Reference |
|---|---|---|---|---|---|
| Rodents | |||||
| Male and female CD-1 mice | ELISA | PND28-PND56 | 0/10/100 mg/L BPA in water | Significant reduction of FT4 but not FT3 levels at both doses. No effects of sex on FT3 and FT4 levels. | [113] |
| Pregnant female rats (Crj: CD (SD) IGS strain) | Chemiluminescence immunoassay | GD6-PND20 | 0/4/40 mg/kg BW BPA per day by diet | In male and female offspring no significant variations in T4 levels at 1, 3, 9 weeks of age. | [108] |
| TSH stimulation assay | Offspring at 9 weeks of age; 1 day treatment | TSH 25 mIU/5µL/g BW BPA intraperitoneally + 125 mIU 5µL/g BW BPA intramuscularly | In response to exogenous TSH, elevation of T4 levels, but any significant difference between control and BPA groups of both sexes. | ||
| Pregnant female Sprague Dawley rats | Radioimmunoassay | GD6-PND20 | 0/1/10/50 mg/kg BW BPA per day by diet | Increase of TT4 levels in both male and female pups only on PND15. No effects on serum TSH in male pups on PND15 among the different treated-groups. | [104] |
| Pregnant female Sprague Dawley rats | Electrochemiluminescence immunoassay | GD10-PND20 | 0/100/1000/10,000 ppm TBBPA by diet | In male pups on PND20, dose-unrelated, but statistically significant decrease of serum T3 levels whereas no significant variations of T4 and TSH. At PNW11, any changes of THs levels in any groups. | [107] |
| Necropsy | No significant changes in thyroid weights of treated dams and offspring compared with controls but dose-unrelated increased thyroid weight in all treated groups of dams. | ||||
| Histology | No significant increased incidence of diffuse thyroid follicular cell hypertrophy in dams at the highest dose. | ||||
| Pregnant female Sprague Dawley rats (strain code 23) | Radioimmunoassay | GD6-PND15 | 0/2.5/25/250/2500/25,000 μg/kg BW BPA per day by diet | No effects on T4 and TRH levels in male and females pups on PND15. | [109] |
| Pregnant female Sprague Dawley rats | ELISA | GD11-PND21 | 0/0.1/50 mg/L BPA per day in water | In dams, at 0.1 mg/L reduction of FT4 levels at delivery and on PND7. In male pups on PND7, increased FT4 levels at 0.1 mg/mL and decreased FT4 levels at 50 mg/L. In females no effects at each dose at all days. | [112] |
| Sprague Dawley rat dams | Radioimmunoassay | GD6-PND15 GD6-PND21 GD6-PND90 | 2.5–2700/100,000/300,000 μg/kg BW BPA per day by diet | No effects on THs levels, thyroid weight, or thyroid histology in the “low-dose” region. On PND15, elevation of T3 at both high doses. On PND90, increase of TSH in females at both high doses and of T4 in males at the highest dose. | [118] |
| Female Sprague Dawley rats | ELISA | GD11-PND21 | 0.1 mg/L BPA per day in water | In dams, after 10 days from exposure, significant decrease in TT4 and FT4 levels, but no variation of TT3 and FT3 levels. On PND21 but not on PND90, significant decrease of TT4, TT3, FT4, and FT3 levels in male pups. | [111] |
| Female Sprague Dawley rats | Radioimmunoassay/ELISA | PND1-PND10 | 0, 5 μg/50μL BPA (B5), 50 μg/50 μL BPA (B50), 500 μg/50 μL L BPA (B500) in castor oil | On PND13, no differences in TSH levels. In estrus, on PND90, increased TSH levels following exposure to B50, but not B5 or B500. In adult females, lower T4 levels at B5 and B500, but not B50. No significant differences of T3 among groups. | [117] |
| Neonatal males R. norvegicus (Wistar strain) | ELISA | PND15-PND30 | 0/20/40 mg/kg BW/BPA per day by diet | On PND30, significant elevation of TSH levels, accompanied by a notable reduction of T3, T4, and GH levels in a dose-dependent manner. | [116] |
| Male and female CD® rats | Electrochemical luminescence immunoassay | 13 consecutive weeks. 6-week recovery period of animals for both control and 1000 mg/kg/ day groups. | 0/100/300/1000 mg/kg BW TBBPA per day by diet | No effects on TSH and T3 levels at any dose or time in both sexes. Decrease of T4 levels at all doses in both males and females. Following recovery period, T4 levels in males but not in females at 1000 mg/kg were comparable to controls. | [110] |
| Necropsy/Histology | No effects of treatment on thyroid weight and histopathology | ||||
| Wistar rats dams | Radioimmunoassay | GD1-PND21 | 0/10 (BPA10)/50 μg/kg BW per day (BPA50) by diet | On PND21, no change in THs levels in dams and in female pups, whereas in the BPA10 group of male pups lower T3 levels without any variation in T4 levels. On PND180, decrease of T3 levels in females and T4 levels in males among the BPA10 group. | [115] |
| Adult female Wistar rats | Radioimmunoassay | 15 days | 40 mg/kg BW per day BPA by diet | Higher T4 levels in BPA-exposed animals, whereas no variations of T3 levels. | [106] |
| Thyroid iodine uptake/TPO activity | Significant reduction of TPO and NIS activity. | ||||
| ROS generation | In BPA-treated animals, significant generation of H2O2 generation in the thyroid. | ||||
| qRT-PCR | Significant reduction of Tshβ mRNA levels in the pituitary of the treated rats. | ||||
| Adult male Wistar rats | RIA | 15 days | 0/40 mg/kg BW/BPA per day by diet | After 15 days of treatment, significant reduction of liver DIO1 activity, whereas no effects on brown adipose tissue DIO2 activity. Significant increase of TT4 levels but no variations of TT3 levels. Significant reduction of T3/T4 ratio. | [102] |
| Wistar rats (HsdCpb:WU) of both sexes | Radioimmunoassay | 70 or 14 days before mating – after mating (males) or PND21 (females) 28 days | 0/3/10/30/100/300/1000/3000 mg/kg BW TBBPA per day by diet (reproduction study) 0/30/100/300 mg/kg BW TBBPA per day by diet (subacute toxicity study) | In the reproduction study, decrease of T4 levels in pups of both sexes, and increase of T3 levels only in females. In the subacute toxicity study, significant decrease of T4 and increase of T3 levels in males, whereas in females parallel though not significant trends. | [114] |
| Necropsy | Dose-dependently increase of pituitary weight in male pups in the reproduction study. No effects in the subacute toxicity study. | ||||
| Histology | No changes observed in the histology of the pituitary gland both in the reproduction and in the subacute toxicity studies. | ||||
| Female F344 rats | Necropsy | 64 weeks | 250/1000 μg/kg BW per day BPA by diet ±2800 mg/kg sc. DHPN ±1000 μg/L KI in water | In the group exposed to DHPN, statistical significances among all groups, and the KI group had the heaviest thyroid weights. In the group not exposed, no significant differences were found among groups. | [105] |
| Histology | In the group exposed to DHPN + KI + 1000BPA, all thyroids had a tumor or focal hyperplasia. Significant difference in the total number of hyperplasia lesions among all groups of animals exposed to DPNA. | ||||
| Chemiluminescence immunoassay/ELISA | In the groups exposed to DHPN, TSH was significantly higher in the KI group than in the controls and the highest FT4 concentration was in the BPA1000 group. In the groups not exposed to DHPN, the highest concentration of TSH was in the controls whereas FT4 increased with increasing doses of BPA. | ||||
| Western blotting detection | In the groups exposed to DPNA, increased protein levels of ERα in the BPA250 and BPA1000 groups compared to the control. | ||||
| Sheep | |||||
| Lacaune ewes | Radioimmunoassay | GD28-GD128 | 0/0.5/50/5000 μg/kg per day sc. BPA | In mothers, no effects on TT4 and TSH levels, but significant decrease of FT4 (at the lowest dose) and TT3 (at the lowest and middle doses) throughout pregnancy. No changes in TT4 and FT4 levels in fetal jugular blood on GD132-GD134. | [123] |
| Lacaune ewes | Radioimmunoassay | GD28–GD145 | 0/5 mg/kg BW per day sc BPA | In pregnant ewes, reduction with time of TT4 but not of FT4. Decrease of jugular blood TT4 and FT4 concentration within the 1st hour of life and of TT4 concentration in cord blood. Slight decrease of TT3 in the 1st hour of life. At 2 months of life no changes in THs levels. | [122] |
| Zebrafish | |||||
| GFP-positive transgenic zebrafish embryos | Fluorescence | 24–48hpf/24–72 hpf | 10−7/5×10−7/10−6/5*10−6/10−5 M BPA ±10−8 M T3 | In the presence of T3, the two highest doses of BPA inhibited T3-induced transcriptional activity during 48 h exposure. No effects of BPA or T3 in the presence of T3 during 24h exposure. | [126] |
| Wild-type zebrafish (Danio rerio) embryos | qRT-PCR | 72 h | 0.01/0.1/1.0 μM BPA/BPS/TBBPA/TBBPS | At 0.1 μM, BPA and TBBPS up-regulated Trβ transcripts whereas BPS and TBBPS showed no significant effect. At 1.0 μM BPS and TBBPS up-regulated Trβ mRNA levels, while BPA had a down-regulating effect. | [68] |
| Wild-type zebrafish (Danio rerio) embryos | qRT-PCR | 24–48 hpf | 10−8–10−6 M BPA | Induction of Tg expression at 10−8 and 10−6 M. Up-regulation of Pax8 transcripts at low and high doses and of Pax2a and Tsh transcripts only at high dose. | [89] |
| Wild type zebrafish (Danio rerio) embryos | ELISA qRT-PCR | 2–168 hpf | 0/5/50/500 μg/L BPAF | Significant decrease of TT3, FT4 and TT4 levels at 50 and 500 μg/L. Reduction of FT3 levels in all treated groups. | [13]) |
| Up-regulation of Tshβ at 50 μg/L and significant decrease of transcription at the highest dose. Up-regulation of Tg and Dio2 at 50 μg/L, Dio1 at 50 and 500 μg/L, and Ttr at all doses. Down-regulation of transcription of Trβ at 50 μg/L, Slc5a5 and Trα at 50 and 500 μg/L. | |||||
| Wild type zebrafish (Danio rerio) embryos | ELISA | 75 dpf | 0/0.1/1/10 and 100 μg/L BPS | Lower levels of T3 and T4 in males exposed to 10 and 100 μg/L. Lower levels of T3 and T4 in females exposed to 100 μg/L. | [131] |
| Wild-type zebrafish (Danio rerio) embryos-larvae | ELISA | <4–120 hpf | 0.08/0.4/2 mg/L BPA/BPF; 2/10/50 mg/L BPS; 0.04/0.18/0.68 mg/L BPZ | Significant increase of T3 levels at 0.4 mg/L BPA and at 50 mg/L BPS (with a dose-response trend) and of T4 at 2.0 mg/L BPF (with a dose-response trend). | [37] |
| qRT-PCR | 0.4<72 mg/L BPA or BPF; 10/50 mg/L BPS; 0.08/0.4 mg/L BPZ | Up-regulation of Hhex, Tg, Ttr, Dio1, Ugt1ab genes by BPA. Up-regulation of Hhex, Ugt1ab genes by BPF, of Crh, Tshβ, Tsh-r, Hhex, Tpo, Ttr, Ugt1ab by BPS, and of Tshβ by BPZ. | |||
| Wild-type zebrafish (Danio rerio) embryos-larvae | ELISA | 2–144 hpf | 0.2/2/20/200 µg/L BPF | Dose-dependent decrease of TT4 levels in BPF treated groups. Significant elevation of TT3 at 200 µg/L and TSH at 20 and 200 µg/L BPF. | [38] |
| qRT-PCR | Up-regulation of Crh and Tg at 2,20, 200 µg/L. Induction of transcription of Slc5a5, Dio2, Ugt1ab at 20 and 200 µg/L. Down-regulation of Ttr at 20 and 200 µg/L. | ||||
| Wild-type zebrafish (Danio rerio) embryos-larvae | qRT-PCR | 0/1/5 dpf; exposure for 24/48h | 10−5 M BPA ±10−8 M T3 | Weak effects of BPA alone on gene expression compared with controls, except for the induction of TSH expression in the group 1day/48 h. With T3, reduction of Trα, Trβ, and Tsh expression in 1-day-old embryos at both 24 and 48 h, but up-regulation of Trα and Trβ, by 24-h exposure in 0-day-old embryos and of Tsh by 48-h exposure in 5-day-old larvae. | [126] |
| Wild-type zebrafish (Danio rerio) embryos-larvae | ELISA | 2–144 hpf | 50/100/200/400 μg/L TBBPA | Dose-dependent increase of T4 levels and dose-dependent decrease of T3 levels. | [128] |
| 200 μg/L TBBPA/20 μg/L T3/200 μg/L TBBPA+20 μg/L T3 | After exposure to T3, significant decrease of T4 and significant increase of T3 contents. After co-treatment TBBPA and T3, decrease of T4 and increase of T3 contents, which were significantly different from the control group (only for T3 increase) and the TBBPA treated group. | ||||
| qRT-PCR | 50/100/200/400 μg/L TBBPA | Up-regulation of Tsh and Tg and down-regulation of Ttr and Trβ transcript levels. | |||
| 200 μg/L TBBPA/20 μg/L T3/200 μg/L TBBPA+20 μg/L T3 | After exposure to T3 and TBBPA + T3 significant up-regulation of Tsh and Tg whereas no changes in Ttr and Trβ mRNA levels. | ||||
| Wild-type zebrafish (Danio rerio) embryos-larvae | ELISA | 2–168 hpf | 1/3/10/30 μg/L BPS | Significant decrease of TT4 levels at 10 and 30 μg/L and TT3 levels at the highest dose. Significant increase of TSH in the 10-, and 30 μg/L exposure groups. | [129] |
| qRT-PCR | Up-regulation of Crh, Tg, Dio1, and Ugt1ab at 10 and 30 μg/L. Induction of transcription of Pax8, Slc5a5, Tg, and Dio2 at 30 μg/L. Down-regulation of Ttr mRNA at 3, 10, and 30 μg/L. No effects on transcription of Trα, Trβ, and Dio3. | ||||
| Wild-type zebrafish (Danio rerio) embryos-larvae | qRT-PCR | 96–192 hpf | 0%/10%/50%/75% of the 96 h -LC50 BPA/TBBPA | Induction of Trα, Tshβ, and Ttr in larvae and of Trα and Ttr transcripts in embryos by TBBPA. Down-regulation of Trβ and Tshβ mRNA in embryos by TBBPA. Up-regulation of Tshβ mRNA in larvae by BPA. No effects on Slc5a5 and Tg expression in larvae neither on Tpo in embryos. | [133] |
| Wild type zebrafish (Danio rerio) larvae | qRT-PCR | 2–120 hpf | 0/100/200/300/400 μg/L TBBPA | Slight up-regulation of Dio1, Trβ, and Tsh, and significant up-regulation of Trα at low doses. Significant down-regulation of Tpo. No significant effects on expression of Dio2 and Dio3 transcripts. | [132] |
| Wild-type adult zebrafish (Danio rerio) | qRT-PCR | 72 h | 50 μg/L/100 μg/L BPA or BPS | After BPA exposure, increase of Slc5a5 mRNA levels at both doses and Tpo transcript levels at the lowest dose. No effects on Tg expression. After BPS exposure, increased expression of Slc5a5 and Tg transcripts at the highest dose and Tpo transcripts at the lowest dose. | [135] |
| Adult male zebrafish (Danio rerio) | ELISA | 21 days | 0/24.7 µg/L BPAF/5.6 µg/L SMX/24.7 µg/L BPAF + 5.6 µg/L SMX | Slight decrease of T4 levels in fish exposed to BPAF. Significant increase of T4 in fish exposed to the mixture BPAF + SMX. | [134] |
| qRT-PCR | Up-regulation of Trh, Trhr, Tshβ, Dio2 and down-regulation of Tpo, Slc5a5, Tg mRNA levels in fish exposed to BPAF. Up-regulation of Trα, Trβ, Dio1, Dio2, Tpo, Tg, Slc5a5 and down-regulation of Trh, Trhr, Tshβ transcript in fish exposure to mixture. | ||||
| Study Design | Country | Study Sample | Sample Size (N) | Age | Principal Results | BPA Concentration | Confounders | Reference |
|---|---|---|---|---|---|---|---|---|
| Prospective | Japan | Women with a history of three or more (3–11) first-trimester miscarriages. Blood samples collected 5–9 days after ovulation in at least two cycles. | 45 patients | 27–36 | No difference in serum BPA levels between patients with and without hypothyroidism. Serum TSH levels not estimated. | Patients with hypothyroidism: Mean (SD): 2.99 ± 3.04 µg/L Patients without hypothyroidism: Mean (SD): 2.50 ± 5.70 µg/L | - | [151] |
| Prospective | Belgium | Overweight and obese individuals. Lean individuals. For obese individuals, urine samples collected at baseline, before starting a weight loss program (N = 151) and after 3 (N = 95), 6 (N = 53), 12 months (N = 39). | 151 obese individuals 43 lean individuals | ≥18 | The obese group had higher urinary levels of BPA. Positive relationship of urinary BPA with serum TSH in lean subjects. | Obese individuals 0—median: 1.7 µg/L 3 months—median: 2.0 µg/L 6 months—median: 1.7 µg/L 12 months—median: 1.5 µg/L Lean individuals Median: 1.2 µg/L | [1,7,34] | [152] |
| Prospective birth cohort | USA | Pregnant women from the CHAMACOS study. Urine samples collected at 12 and 26 weeks of gestation. | 476 | ≥18 | In mothers, no association of urinary BPA (average) with FT4 and TSH levels, and negative association (BPA 26 week) with serum TT4. Inverse association between maternal BPA (average, 26 week) and TSH in male newborns but not in females. | LOD: 0.4 μg/L Mothers: GM: 1.3 µg/g cr Median: 1.2 µg/g cr | [1,2,4,8,10,12,13,14,15,16,17,18,19,20,21] | [143] |
| Prospective birth cohort | USA | Pregnant women from the HOME study. Urine samples collected at both 16 and 26 weeks of gestation (N = 237). | 249 | ≥18 | Neither association of maternal BPA (16 week) with maternal THs or TSH levels nor of maternal BPA (average, 16 week, or 26 week) with THs or TSH levels in newborns. Significant inverse association of maternal BPA with TSH levels (average and 26 week BPA) and slight positive association with TT3 levels (26 week BPA) in females. Stronger relationship of BPA–TSH among girls born from iodine-deficient mothers. | LOD: 0.4 μg/L 16 weeks GM: 2.0 (95%CI 1.8–2.2) µg/g cr 26 weeks GM: 2.3 (95%CI 2.1–2.5) µg/g cr | [1,2,8,9,10,15,16,17,19,20,22,23,24,25] | [148] |
| Prospective birth cohort | Japan | Pregnant women at 23–35 weeks of gestation (singleton babies). Cord blood obtained at delivery. | 283 | ≥18 | No association between BPA concentration in cord blood and TSH or FT4 levels in newborns of both sexes. | LOQ: 0.04 µg/L GM: 0.051 µg/L IQR: <LOQ–0.076 µg/L | [19,38] | [146] |
| Nested case-control | USA | Women who delivered preterm (< 37 weeks of gestation) and controls of women who delivered a singleton infant after 37 weeks of gestation. Urine samples collected at up to 4 visits (median for each visit: 9.64, 17.9, 26.0, and 35.1 weeks of gestation). | 116 cases 323 controls | ≥18 | IQR increase in BPA concentrations across study visits was significantly associated with lower TSH and higher FT4 levels. No effect on FT3, and inverse but not significant association of BPA with TT4 levels. No association of BPA with serum FT4 at visit 3. Significant inverse association of BPA with TSH levels at visits 3 and 4 and a slight increase of serum TT3 at visit 4. | Total: GM ± SD: 1.18 ± 2.82 µg/L 1 visit: GM ± SD: 1.33 ± 2.84 µg/L 2 visit: GM ± SD: 1.04 ± 2.85 µg/L 3 visit: GM ± SD: 1.22 ± 2.84 µg/L 4 visit: GM ± SD: 1.12 ± 2.70 µg/L | [2,3,4,8,17,19,25,35,36,37], | [139] |
| Case-control | Korea | Infants with congenital hypothyroidism and their mothers; healthy infants with their mothers. Blood samples collected. | 26 congenital hypothyroidism mother–infant pairs 12 normal mother–infant pairs | <24 months | TBBPA levels not significantly different in the two infant groups. No significant correlation between TBBPA and any of the THs levels in infants with congenital hypothyroidism. In their mothers, positive weak correlation of TBBPA with serum FT4 and thyroid stimulating immunoglobulin. | Normal group and their mothers TBBPA mothers: 10.93 ng/g lipid TBBPA infants: 77.65 ng/g lipid Congenital hypothyroidism group and their mothers TBBPA mothers: 8.89 ng/g lipid TBBPA infants: 83.4 ng/g lipid | [1,47,48,49] | [140] |
| Case-control | Cyprus (n = 122) Romania (n = 90) | Females with thyroid nodules (diameter >3mm). Females without nodules. Two spot urine samples collected in Cyprus (7 day apart from each other). In Romania one spot sample collected. | 212: 106 cases 106 controls | ≥18 | In the whole study population, median TSH and BPA levels were significantly lower in the cases. Significant positive association of BPA with TSH levels and TNs. Neither association of BPA, BPF, or ClxBPA with FT4 levels nor of BPF and ClxBPA with serum TSH. No association of BPF and ClxBPA with TNs. | LOD (BPA): 10 μg/L LOD (BPF): 13 μg/L LOD (ClxBPA): 10–17 μg/L Median: BPA: 2.25 (1.10–4.61) μg/L BPF: 0.46 (0.32–0.72) μg/L ClxBPA: 0.16 (0.15–0.19) μg/L BPA Cases: 1.75 (1.11–3.56) μg/L BPA Controls: 2.71 (1.08–5.91) μg/L | [1,3,10,11,39,40,41] | [147] |
| Case-control | Turkey | Children with HT. Children without HT. Spot urine samples collected. | 29 cases (25 females and 4 males) 29 controls | 8–16 | No significant difference in urinary BPA levels between the two groups. Significant negative correlation between BPA and FT4 levels in HT group. No correlation between urinary BPA concentration and TPOAb levels. | LOD: 0.5 ng/mL Mean (± standard error of the mean) Cases: 7.31 ± 1.46 µg/g cr Controls: 7.72 ± 1.74 µg/g cr | - | [155] |
| Case-control | China | Women with TNs. Women without nodules. First morning urine collected. | 1416: 705 cases 711 controls | ≥18 | Urinary BPA was significantly higher in cases than in controls. Increased prevalence of TNs with increasing urinary BPA quartiles. Significant association between urinary BPA and TNs in all age groups and in the thyroid autoantibody positive group. | LOD: 0.1 µg/L Median: Total: 3.08 (1.60–5.81) µg/g cr Cases: 3.27 (1.77–6.53) µg/g cr Controls: 2.92 (1.44–5.27) µg/g cr | [1,3,8,10,11,29,30,31,44,45,46] | [159] |
| Multicentre, cross-sectional | Italy | Patients with TNs. Patients with DTC. Blood collected from each patient. | 27 with TNs 28 with DTC | ≥18 | Significant correlation between urinary BPAF concentration and risk of DTC in patients with TNs. BPS and BPB concentrations higher in patients with TNs, as compared with DTCs. TSH levels higher in patients with DTCs and in subjects exposed to BPE and BPA. | Subjects with TNs (median ± SD) BPAF: 12.47 ± 0 ng BPS: 59.29 ± 50.29 ng BPB: 29.92 ± 35.60 ng Subjects with DTC (median ± SD) BPAF: 9.44 ± 5.41 ng BPS: 34.79 ± 41.86 ng BPB: 14.36 ± 15.31 ng | [50] | [161] |
| Cross-sectional | USA | Men who are partners of subfertile couples. Single spot urine (N = 167), second (N = 75), and third (N = 4) urine samples collected. | 167 | 18–55 | Inverse relationship of BPA with TSH levels. No effects on T3 and T4 levels. | LOD: 0.4 µg/L GM and median: 1.3 µg/L | [1,2,3,4,5,6] | [150] |
| Cross-sectional | USA | Adult and adolescents from the NHANES 2007–2008. Single spot urine collected. | 1346 adults 329 adolescents | ≥20 12–19 | In adults, inverse relationship of BPA with TT4 levels. Inverse trends between BPA quintiles and both TT4 and TSH. | GM: 2.03 µg/g cr GM: 1.88 µg/g cr | [1,2,3,7,8,9,10] | [145] |
| Cross-sectional | USA | Adults and adolescents from the NHANES 2007–2008. Urine sample collected. | 710 females; 850 males | 12–85 | Negative association between urinary multiple EDCs including BPA and TT4 levels in males but not in females. Positive but not statically significant association of EDCs with T3 levels in females. | LOD (BPA): 0.13 µg/L Males: GM: 2.19 (95% CI 1.94–2.47) µg/L Females: GM: 2.00 (95% CI 1.80–2.23) µg/L | [1,2,4,9,10,11,26] | [142] |
| Cross-sectional | China | Workers in two semiautomatic epoxy resin factories. Spot urine collected at the end of the shift on Friday. | 28 (21 males and 7 females) | 22–62 | The workers with the highest BPA concentrations (feeding position) had higher FT3, TT3, TT4 levels, and lower TSH levels. Urinary BPA significantly associated with higher FT3 levels (when office workers were excluded). Weak positive association between BPA and serum FT4. | Total workers: GM ± SD: 31.96 ± 4.42 μg/g cr Feeding operators: GM ± SD: 192.45 ± 2.78 μg/g cr Crushing and packing: GM ± SD: 17.08 ± 2.33 μg/g cr Office workers: GM ± SD: 11.60 ± 1.70 μg/g cr | [11] | [141] |
| Cross-sectional | China | Population-based study. Spot morning urine collected. | 3394: 1354 males 2040 females | ≥40 | Significant positive association of urinary BPA with serum FT3 and inverse association with TSH levels both in men and in women. Positive association between high thyroid function and high urinary BPA levels. | LOD: 0.3 μg/L Median: 0.81 µg/L IQR: 0.47–1.43 µg/L Overt and subclinical hypothyroidism subjects: GM: 0.63 (95% CI 0.52–0.77) µg/L Euthyroid subjects: GM: 0.81 (95% CI 0.78–0.83) µg/L Overt and subclinical hypothyroidism subjects: GM: 1.05 (95% CI 0.84–1.32) µg/L | [1,3,4,8,11,17,27] | [149] |
| Cross-sectional | China | Students of primary schools. First morning urine samples collected. | 718 | 9–11 | BPA levels similar among boys and girls but increased with age. Significant inverse association between urinary BPA and thyroid volume. Risk of TNs increased with age without any association with sex or urinary iodine level. BPA inversely associated with risk of multiple TNs. | Urinary iodine: Median: 159 μg/L Urinary BPA: Median (IQR): 2.45 (1.09–5.97) µg/g cr | [1,3,7,11,42,43] | [158] |
| Cross-sectional | China | Patients with NG. Patients with PTC. Healthy controls. Spot blood and urine samples collected in the morning. | 53 60 65 | ≥18 | Urinary BPA and urinary iodine levels in NG and PTC groups were significantly higher than those in controls. Urinary BPA levels in the females of NG group and both the male and female PTC groups were higher than those in the control group of the same sex. Urinary BPA concentration was significantly lower in the female PTC group than in the female NG group. Urinary iodine in the PTC or NG groups was higher than that of controls of the same sex. Significant correlation between urinary BPA and iodine concentrations in all groups. | LOQ for BPA in urine: 0.1 µg/L LOQ for BPA in serum: 0.2 µg/L LOD for iodine in urine: 3 µg/L Serum BPA GM: 7.42 (4.03–13.82) µg/L Urinary BPA: GM: 2.82 (0.016–59.78) µg/g cr Urinary iodine: GM: 335.05 (71.25–2995.74) µg/g cr BPA—Control group GM: 1.06 (0.015–27.88) µg/g cr BPA—NG group GM: 5.22 (0.022–50.78) µg/g cr BPA—PTC group GM: 4.68 (0.032–29.30) µg/g cr | [11] | [160] |
| Cross-sectional | Thailand | Subjects from the National survey NHES 2009. Serum sample collected. | 2340 | ≥15 | Significantly inverse association of serum BPA with FT4 levels in males but not in females after exclusion subjects of thyroid autoantibodies. No association with serum TSH in both sexes. | LOD: 0.3 μg/L Median: 0.33 (0–66.91) µg/L | [1,3,7] | [144] |
| Cross-sectional | Thailand | Subjects from the National survey NHES 2009. Serum sample collected. | 2361 | ≥15 | Significant association of increasing BPA quartiles with positivity for TgAb and TPOAb both in men and women but not for TRab. Age, sex, and BMI were independent predictors of TgAb and TPOAb positivity. | LOD: 0.3 μg/L Median: 0.32 (0–66.9) µg/L | - | [156] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorini, F.; Bustaffa, E.; Coi, A.; Iervasi, G.; Bianchi, F. Bisphenols as Environmental Triggers of Thyroid Dysfunction: Clues and Evidence. Int. J. Environ. Res. Public Health 2020, 17, 2654. https://doi.org/10.3390/ijerph17082654
Gorini F, Bustaffa E, Coi A, Iervasi G, Bianchi F. Bisphenols as Environmental Triggers of Thyroid Dysfunction: Clues and Evidence. International Journal of Environmental Research and Public Health. 2020; 17(8):2654. https://doi.org/10.3390/ijerph17082654
Chicago/Turabian StyleGorini, Francesca, Elisa Bustaffa, Alessio Coi, Giorgio Iervasi, and Fabrizio Bianchi. 2020. "Bisphenols as Environmental Triggers of Thyroid Dysfunction: Clues and Evidence" International Journal of Environmental Research and Public Health 17, no. 8: 2654. https://doi.org/10.3390/ijerph17082654
APA StyleGorini, F., Bustaffa, E., Coi, A., Iervasi, G., & Bianchi, F. (2020). Bisphenols as Environmental Triggers of Thyroid Dysfunction: Clues and Evidence. International Journal of Environmental Research and Public Health, 17(8), 2654. https://doi.org/10.3390/ijerph17082654








