Efficient Removal of Cd(II) Using SiO2-Mg(OH)2 Nanocomposites Derived from Sepiolite
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of the SiO2-Mg(OH)2 and Characterization Methods
2.3. Batch Adsorption Experiments
2.3.1. Adsorption Equilibrium and Kinetic Experiments
2.3.2. Effect of Adsorbent Dosage and pH-Value
2.3.3. Adsorption Isotherms
3. Results and Discussion
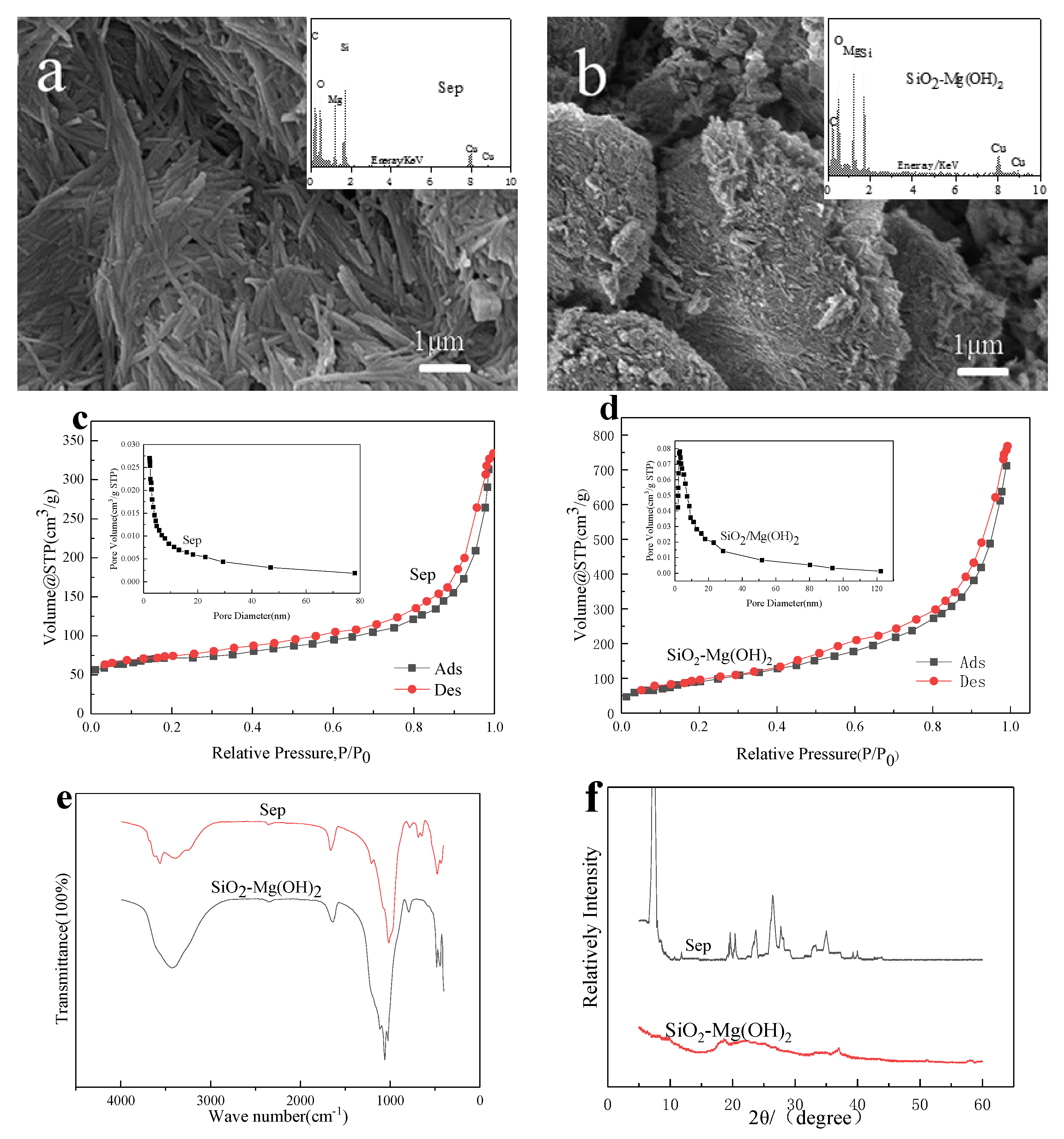
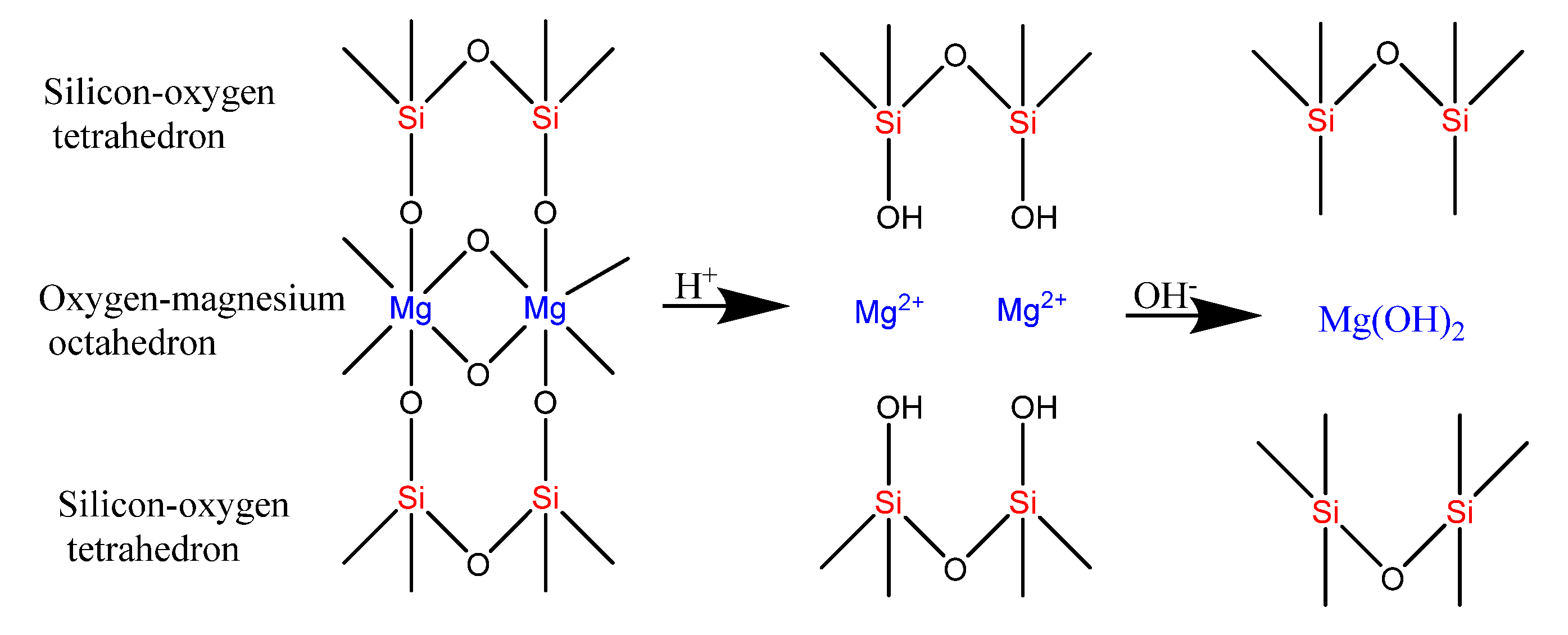
3.1. Characterization of SiO2-Mg(OH)2 Nanocomposites
3.1.1. SEM-EDS Analysis
3.1.2. N2 Adsorption-Desorption Isotherm and Pore Size Distribution
3.1.3. FTIR Analysis
3.1.4. PXRD Analysis
3.2. Adsorption Kinetics
3.3. Effect of SiO2-Mg (OH)2 Dosage and pH on Cd (II) Removal
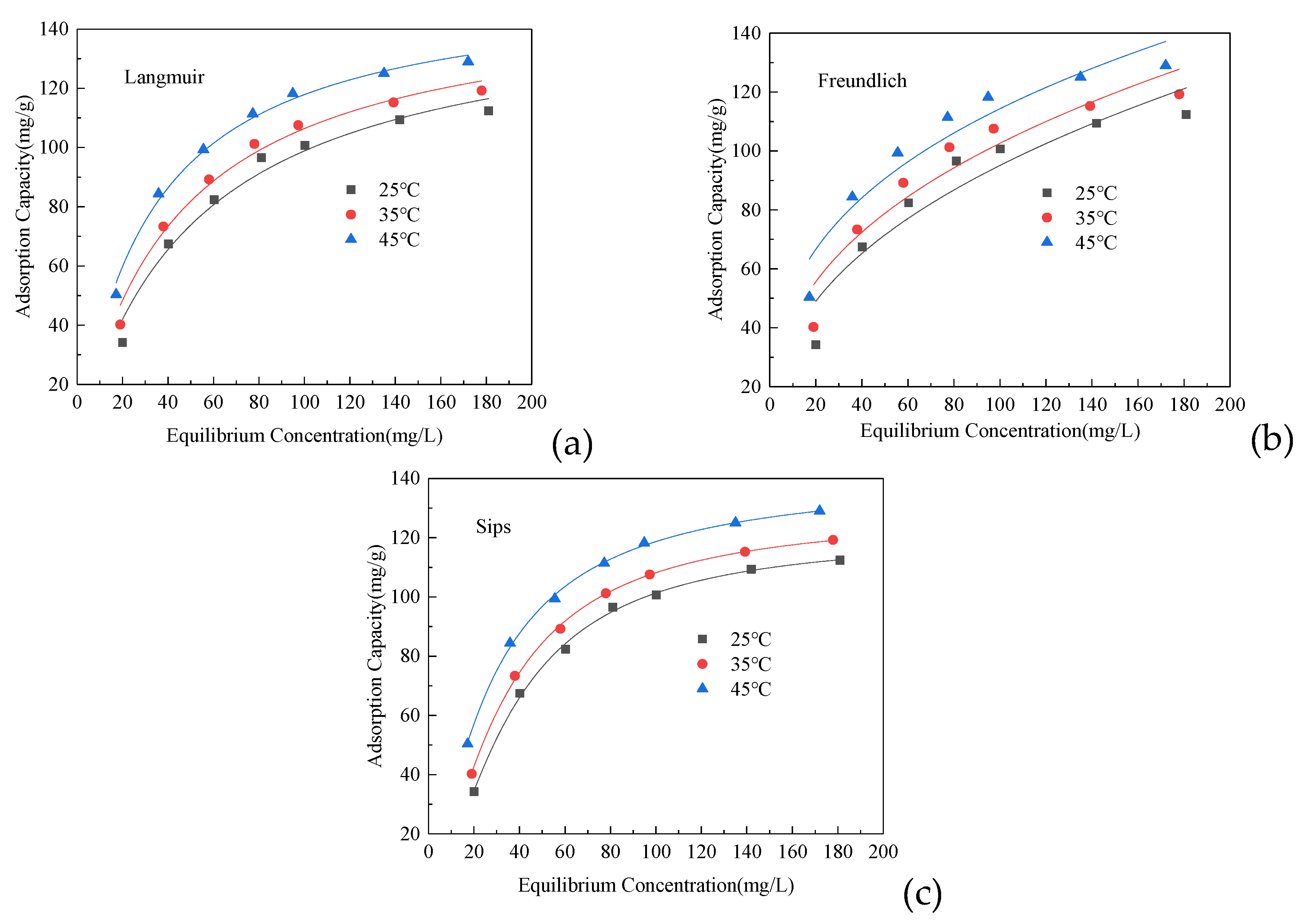
3.4. Adsorption Isotherm
3.5. Thermodynamic Parameters
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jiang, F.; Ren, B.; Hursthouse, A.; Deng, R.; Wang, Z. Distribution, source identification, and ecological-health risks of potentially toxic elements (PTEs) in soil of thallium mine area (southwestern Guizhou, China). Environ. Sci. Pollut. Res. 2019, 26, 16556–16567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ren, B.; Hursthouse, A.; Deng, R.; Hou, B. Leaching and Releasing Characteristics and Regularities of Sb and As from Antimony Mining Waste Rocks. Pol. J. Environ. Stud. 2019, 28, 4017–4025. [Google Scholar] [CrossRef]
- Jiang, F.; Ren, B.; Hursthouse, A.S.; Zhou, Y. Trace Metal Pollution in Topsoil Surrounding the Xiangtan Manganese Mine Area (South-Central China): Source Identification, Spatial Distribution and Assessment of Potential Ecological Risks. Int. J. Environ. Res. Public Health 2018, 15, 2412. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.Y.; Wang, Y.Y.; Lyu, H.H.; Liu, Y.X.; Yang, R.Q.; Yang, S.M. Cadmium adsorption by biochar prepared from pyrolysis of silk waste at different temperatures. J. Appl. Ecol. 2018, 29, 1328–1338. [Google Scholar]
- Duan, Y.; Duan, J.; Feng, Y.; Huang, X.; Fan, W.; Wang, K.; Ouyang, P.; Deng, Y.; Du, Z.; Chen, D.; et al. Hepatoprotective Activity of Vitamin E and Metallothionein in Cadmium-Induced Liver Injury in Ctenopharyngodon idellus. Oxid. Med. Cell. Longev. 2018, 2018, 9506543. [Google Scholar] [CrossRef]
- Kau, D.; Snukiškis, J.; Gefenien, A. Kinetics of cadmium(II) sorption by an iminodiacetic ion exchanger in the presence of a nonionic surfactant. Desalination 2003, 154, 67–77. [Google Scholar]
- Zhou, Q.; Liao, B.; Lin, L.; Qiu, W.; Song, Z. Adsorption of Cu(II) and Cd(II) from aqueous solutions by ferromanganese binary oxide-biochar composites. Sci. Total Environ. 2018, 615, 115–122. [Google Scholar] [CrossRef]
- Baek, K.; Yang, J.W. Humic-Substance-Enhanced Ultrafiltration for Removal of Heavy Metals. Sep. Sci. Technol. 2005, 40, 699–708. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Krukowska, J.; Thomas, P. Comparison of Sorption and Desorption Studies of Heavy Metal Ions from Biochar and Commercial Active Carbon. Chem. Eng. J. 2016, 307, 353–363. [Google Scholar] [CrossRef]
- Alshameri, A.; He, H.; Zhu, J.; Xi, Y.; Zhu, R.; Ma, L.; Tao, Q. Adsorption of ammonium by different natural clay minerals: Characterization, kinetics and adsorption isotherms. Appl. Clay Sci. 2018, 159, 83–93. [Google Scholar] [CrossRef]
- Myriam, M.; Suarez, M.; Martin-Pozas, J.M. Structural and Textural Modifications of Palygorskite and Sepiolite under Acid Treatment. Clays Clay Miner. 1998, 46, 225–231. [Google Scholar] [CrossRef]
- Yavas, B.H.; Tanrıver, N.; Benli, B.; Kizilcan, N. In Situ Polymerization of Sepiolite Modified Polysulfone. Proced. Soc. Behav. Sci. 2015, 195, 2206–2209. [Google Scholar] [CrossRef]
- Kocaoba, S. Adsorption of Cd(II), Cr(III) and Mn(II) on natural sepiolite. Desalination 2009, 244, 24–30. [Google Scholar] [CrossRef]
- Wu, J.; Wang, Y.; Wu, Z.; Gao, Y.; Li, X. Adsorption properties and mechanism of sepiolite modified by anionic and cationic surfactants on oxytetracycline from aqueous solutions. Sci. Total Environ. 2020, 708, 134409. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Xiong, S.; Xiong, L.; Zhou, S.; Cao, M.; Tu, S. Adsorption Efficiency of Cadmium and Arsenic by Iron-modified Sepiolite and Its Influencing Factors. Technol. Water Treat. 2019, 45, 73–77. [Google Scholar]
- Wang, Z.; Liao, L.; Hursthouse, A.; Song, N.; Ren, B. Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China. Int. J. Environ. Res. Public Health 2018, 15, 1653. [Google Scholar] [CrossRef]
- Xu, Y.M.; Liang, X.F.; Sun, G.H.; Sun, Y.; Qin, X.; Wang, L.; Dai, X.H. Effects of acid and heating treatment on the structure of sepiolite and its adsorption of Lead and Cadmium. Environ. Sci. 2010, 31, 1560–1567. [Google Scholar]
- Lazarević, S.; Janković-Častvan, I.; Jovanović, D.; Milonjić, S.; Janaćković, D.; Petrović, R. Adsorption of Pb2+, Cd2+ and Sr2+ ions onto natural and acid-activated sepiolites. Appl. Clay Sci. 2007, 37, 57. [Google Scholar] [CrossRef]
- Yu, S. Preparation of Nano-mineral Ecomaterials and Their Applications in Heavy Metals Treatment. Ph.D. Thesis, University of Science and Technology of China, Hefei, China, 2016. [Google Scholar]
- Zhang, M.; Song, W.; Chen, Q.; Miao, B.; He, W. One-pot synthesis of magnetic Ni@Mg(OH)2 core-shell nanocomposites as a recyclable removal agent for heavy metals. ACS Appl. Mater. Interfaces 2015, 7, 1533–1540. [Google Scholar] [CrossRef]
- Hu, J.; Liu, M.; Chen, C. Synthesis of Magnetite/Graphene Oxide Composite and Application for Cobalt(II) Removal. J. Phys. Chem. C 2011, 115, 25234–25240. [Google Scholar]
- Nieto-Suarez, M.; Palmisano, G.; Ferrer, M.L.; Gutierrez, M.C.; Yurdakal, S.; Augugliaro, V.; Pagliaro, M.; Del Monte, F. Self-assembled titania–silica–sepiolite based nanocomposites for water decontamination. J. Mater. Chem. 2009, 19, 2070. [Google Scholar] [CrossRef]
- Yuan, P.; Fan, M.; Yang, D.; He, H.; Liu, N.; Yuan, A.; Zhu, J.; Tianhu, C. Montmorillonite-supported magnetite nanoparticles for the removal of hexavalent chromium [Cr(VI)] from aqueous solutions. J. Hazard. Mater. 2009, 166, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Liu, D.; Fan, M.; Yang, D.; Zhu, R.; Ge, F.; Zhu, J.; He, H. Removal of hexavalent chromium [Cr(VI)] from aqueous solutions by the diatomite-supported/unsupported magnetite nanoparticles. J. Hazard. Mater. 2010, 173, 614–621. [Google Scholar] [CrossRef] [PubMed]
- CChen, L.; Zhou, C.; Fiore, S.; Tong, D.S.; Zhang, H.; Li, C.S.; Ji, S.F.; Yu, W.H. Functional magnetic nanoparticle/clay mineral nanocomposites: Preparation, magnetism and versatile applications. Appl. Clay Sci. 2016, 127, 143–163. [Google Scholar] [CrossRef]
- Yao, Q.Z.; Yu, S.H.; Zhao, T.L.; Qian, F.J.; Li, H.; Zhou, G.T.; Fu, S.Q. Enhanced Potential Toxic Metal Removal Using a Novel Hierarchical SiO2–Mg(OH)2 Nanocomposite Derived from Sepiolite. Minerals 2019, 9, 298. [Google Scholar] [CrossRef]
- Tuler, F.; Portela, R.; Avila, P.; Bortolozzi, J.P.; Miró, E.E.; Milt, V.G. Development of sepiolite/SiC porous catalytic filters for diesel soot abatement. Microporous Mesoporous Mater. 2016, 230, 11–19. [Google Scholar] [CrossRef]
- Xie, S.; Xu, Y.M.; Yan, C.X.; Luo, W.W.; Sun, Y.B. Substructure Characteristics of Combined Acid-base Modified Sepiolite and Its Adsorption for Cd(II). Environ. Sci. 2020, 41, 293. [Google Scholar]
- Ma, Y.; Wu, X.; Zhang, G. Core-shell Ag@Pt nanoparticles supported on sepiolite nanofibers for the catalytic reduction of nitrophenols in water: Enhanced catalytic performance and DFT study. Appl. Catal. B Environ. 2017, 205, 262–270. [Google Scholar] [CrossRef]
- Addy, M.; Losey, B.; Mohseni, R.; Zlotnikov, E.; Vasiliev, A. Adsorption of heavy metal ions on mesoporous silica-modified montmorillonite containing a grafted chelate ligand. Appl. Clay Sci. 2012, 59, 115–120. [Google Scholar] [CrossRef]
- Ji, Q.; Kamiya, S.; Jung, J.-H.; Shimizu, T. Self-assembly of glycolipids on silica nanotube templates yielding hybrid nanotubes with concentric organic and inorganic layers. J. Mater. Chem. 2005, 15, 743. [Google Scholar] [CrossRef]
- Shi, J.Y.; Yao, Q.Z.; Li, X.M.; Zhou, G.T.; Fu, S.Q. Formation of Asymmetrical Structured Silica Controlled by a Phase Separation Process and Implication for Biosilicification. PLoS ONE 2013, 8, 61164–61172. [Google Scholar] [CrossRef] [PubMed]
- Habish, A.; Lazarevic, S.; Janković-Častvan, I.; Potkonjak, B.; Janaćković, D.; Petrović, R. The effect of salinity on the sorption of cadmium ions from aqueous medium on Fe(III)-sepiolite. Chem. Ind. Chem. Eng. Q. 2015, 21, 295–303. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Febrianto, J.; Kosasih, A.N.; Sunarso, J.; Ju, Y.H.; Indraswati, N.; Ismadji, S. Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: A summary of recent studies. J. Hazard. Mater. 2009, 162, 616–645. [Google Scholar] [CrossRef] [PubMed]
- Klapiszewski, Ł.; Siwińska-Stefańska, K.; Kołodyńska, D. Development of lignin based multifunctional hybrid materials for Cu(II) and Cd(II) removal from the aqueous system. Chem. Eng. J. 2017, 330, 518–530. [Google Scholar] [CrossRef]
- Benavente, M.; Moreno, L.; Martinez, J. Sorption of heavy metals from gold mining wastewater using chitosan. J. Taiwan Inst. Chem. Eng. 2011, 42, 976–988. [Google Scholar] [CrossRef]
- Tzabar, N.; ter Brake, H.J.M. Adsorption isotherms and Sips models of nitrogen, methane, ethane, and propane on commercial activated carbons and polyvinylidene chloride. Adsorption 2016, 22, 901–914. [Google Scholar] [CrossRef]
- Awual, M.R. A facile composite material for enhanced cadmium(II) ion capturing from wastewater. J. Environ. Chem. Eng. 2019, 7, 103378. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, S.; Kou, Z.; Zhai, S.; Ma, W.; Yang, Y. Removal of cadmium(II) from aqueous solutions by chemically modified maize straw. Carbohydr. Polym. 2015, 115, 177–185. [Google Scholar] [CrossRef]
- Abu-Danso, E.; Peräniemi, S.; Leiviskä, T.; Bhatnagar, A. Synthesis of S-ligand tethered cellulose nanofibers for efficient removal of Pb(II) and Cd(II) ions from synthetic and industrial wastewater. Environ. Pollut. 2018, 242, 1988–1997. [Google Scholar] [CrossRef]
- Da Gama, B.M.V.; Nascimento, G.E.D.; Sales, D.C.S.; Rodríguez-Díaz, J.M.; Barbosa, C.M.M.D.B.; Duarte, M.M.M.B. Mono and binary component adsorption of phenol and cadmium using adsorbent derived from peanut shells. J. Clean. Prod. 2018, 201, 219–228. [Google Scholar] [CrossRef]
- Xie, J. Study on the Adsorption Characteristics of Sulfydryl-modified Sepiolite to Hg(II) and Cd(II). Master’s Thesis, Southwest University, Chongqing, China, 2016. [Google Scholar]
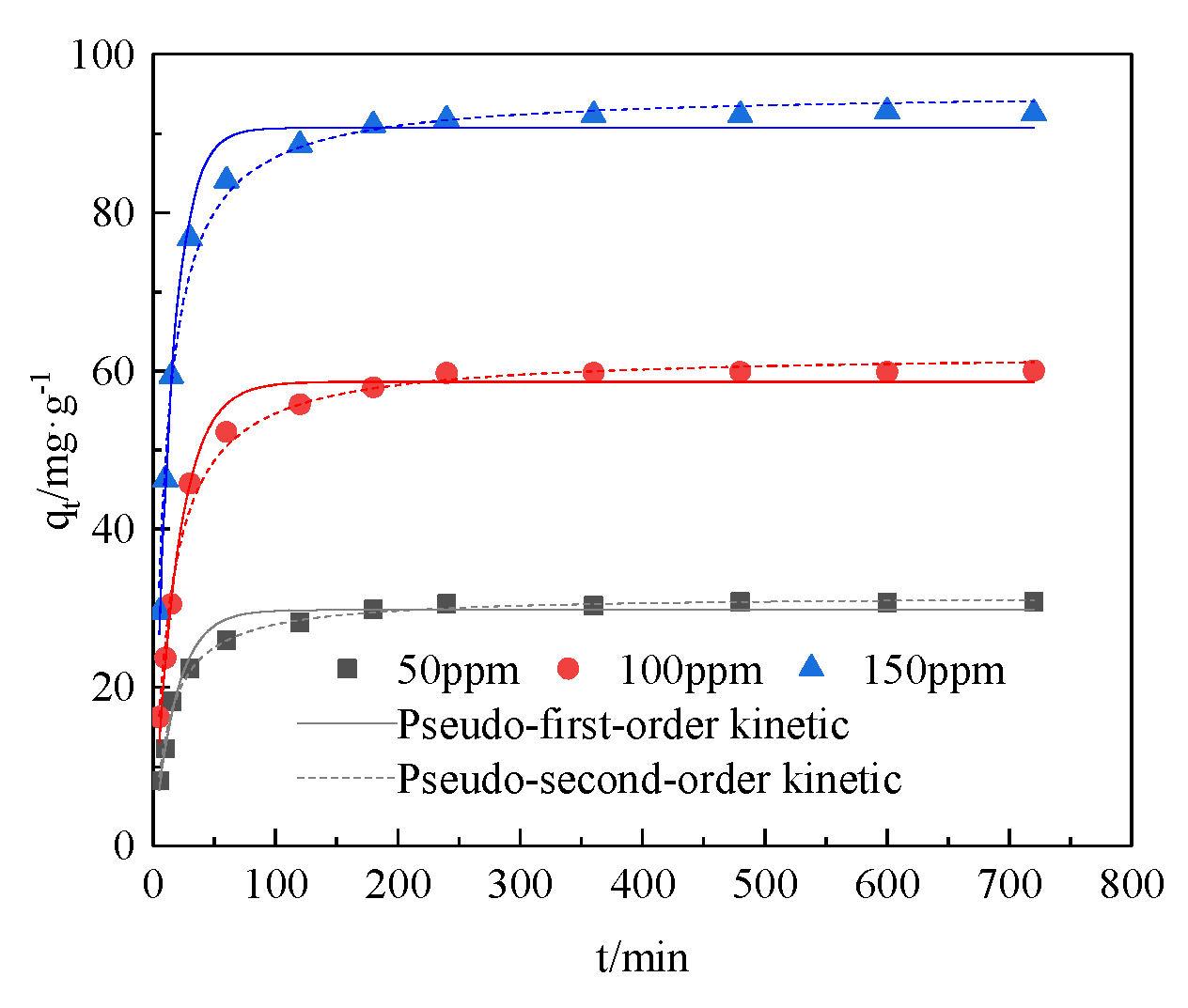

| C0/mg·L−1 | Pseudo-First-Order Kinetic Parameters | Pseudo-Second-Order Kinetic Parameters | ||||
|---|---|---|---|---|---|---|
| qe/mg·g−1 | K1/min−1 | R2 | qe/mg·g−1 | K2/min−1 | R2 | |
| 50 | 29.8092 | 0.0537 | 0.9717 | 31.6627 | 0.0024 | 0.9930 |
| 100 | 58.6236 | 0.0505 | 0.9863 | 62.2908 | 0.0012 | 0.9910 |
| 50 | 90.7180 | 0.0699 | 0.9876 | 95.3443 | 0.0010 | 0.9909 |
| T/K | Langmuir | Freundlich | Sips | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| KL/L·mg−1 | Qm/mg·g−1 | R2 | KF/L·mg−1 (L·mg−1)1/n | n | R2 | Ks/(L·mg1)β | Qm/mg·g−1 | β | R2 | |
| 298 | 0.0195 | 149.5368 | 0.9760 | 14.2767 | 2.4287 | 0.9024 | 0.0035 | 121.2328 | 1.5806 | 0.9982 |
| 308 | 0.0234 | 151.9684 | 0.9840 | 17.7933 | 2.6284 | 0.9103 | 0.0063 | 128.9022 | 1.4609 | 0.9992 |
| 318 | 0.03115 | 155.7178 | 0.9913 | 24.3692 | 2.9791 | 0.9228 | 0.0152 | 141.4869 | 1.2674 | 0.9990 |
| Sorbents | Qm(mg/g) | Experimental Conditions | Refenrences |
|---|---|---|---|
| TiO2/lignin | 22.44 | pH 5.0; T 20 °C | [36] |
| Succinic anhydride modified maize straw | 196.1 | pH 5.8; T 20 °C | [40] |
| S-ligand tethered cellulose nanofibers | 92.17 | [41] | |
| Peanut shells | 55.42 | [42] | |
| SiO2-Mg(OH)2 | 121.23 | pH 7.0; T 25 °C | This work |
| Temperature (K) | lnK0 | ΔG° (KJ·mol−1) | ΔH° (KJ·mol−1) | ΔS° (KJ·mol−1·K−1) |
|---|---|---|---|---|
| 298 | 7.857 | −19.47 | 29.71 | 0.1647 |
| 308 | 8.154 | −20.88 | ||
| 318 | 8.613 | −22.77 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Z.; Ren, B.; Hursthouse, A.; Wang, Z. Efficient Removal of Cd(II) Using SiO2-Mg(OH)2 Nanocomposites Derived from Sepiolite. Int. J. Environ. Res. Public Health 2020, 17, 2223. https://doi.org/10.3390/ijerph17072223
He Z, Ren B, Hursthouse A, Wang Z. Efficient Removal of Cd(II) Using SiO2-Mg(OH)2 Nanocomposites Derived from Sepiolite. International Journal of Environmental Research and Public Health. 2020; 17(7):2223. https://doi.org/10.3390/ijerph17072223
Chicago/Turabian StyleHe, Zhendong, Bozhi Ren, Andrew Hursthouse, and Zhenghua Wang. 2020. "Efficient Removal of Cd(II) Using SiO2-Mg(OH)2 Nanocomposites Derived from Sepiolite" International Journal of Environmental Research and Public Health 17, no. 7: 2223. https://doi.org/10.3390/ijerph17072223
APA StyleHe, Z., Ren, B., Hursthouse, A., & Wang, Z. (2020). Efficient Removal of Cd(II) Using SiO2-Mg(OH)2 Nanocomposites Derived from Sepiolite. International Journal of Environmental Research and Public Health, 17(7), 2223. https://doi.org/10.3390/ijerph17072223






