Shall We Dance? Dancing Modulates Executive Functions and Spatial Memory
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Materials
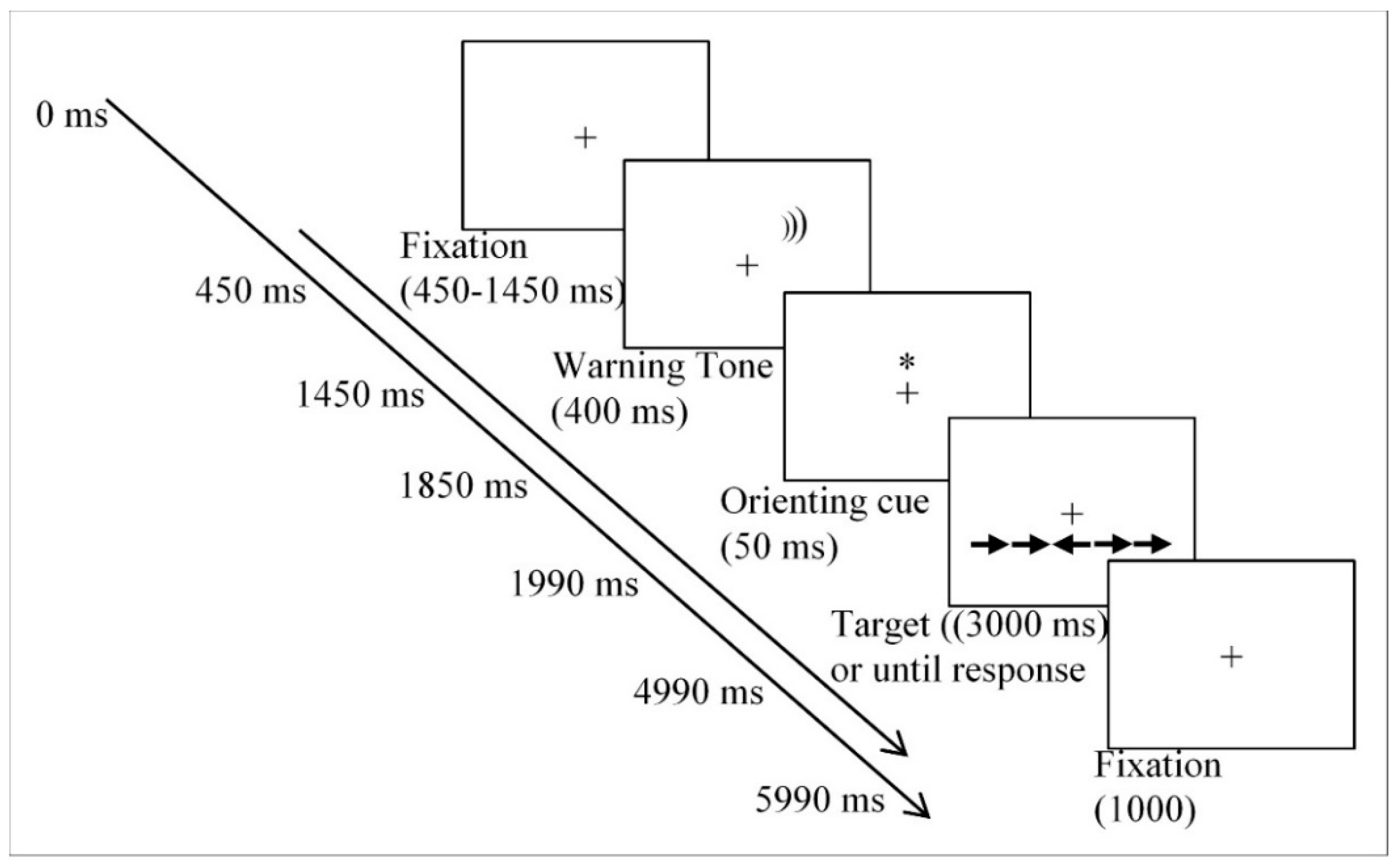
2.3. Procedure
2.4. Statistical Analysis
3. Results
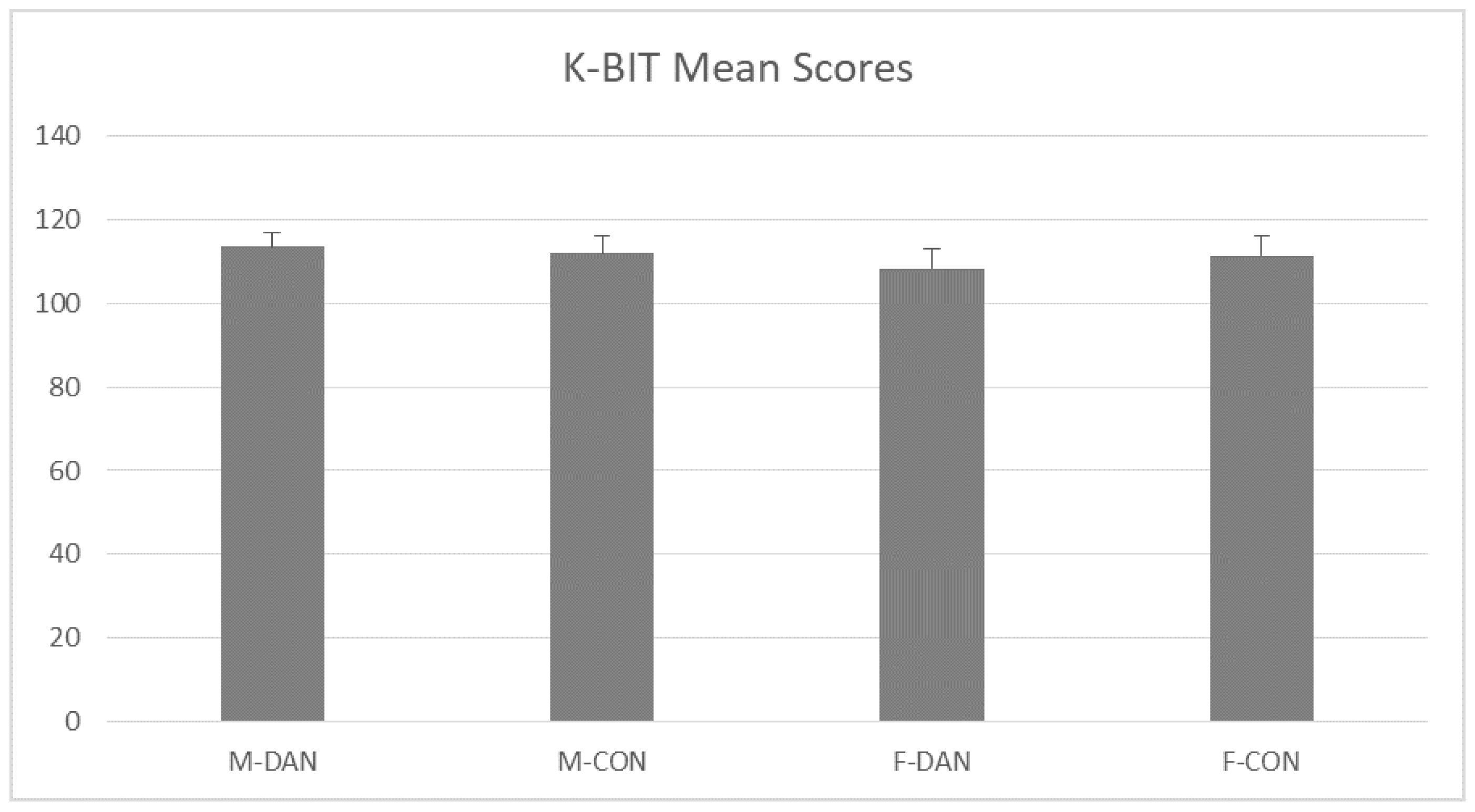
3.1. K-BIT (General Intelligence Measurement)
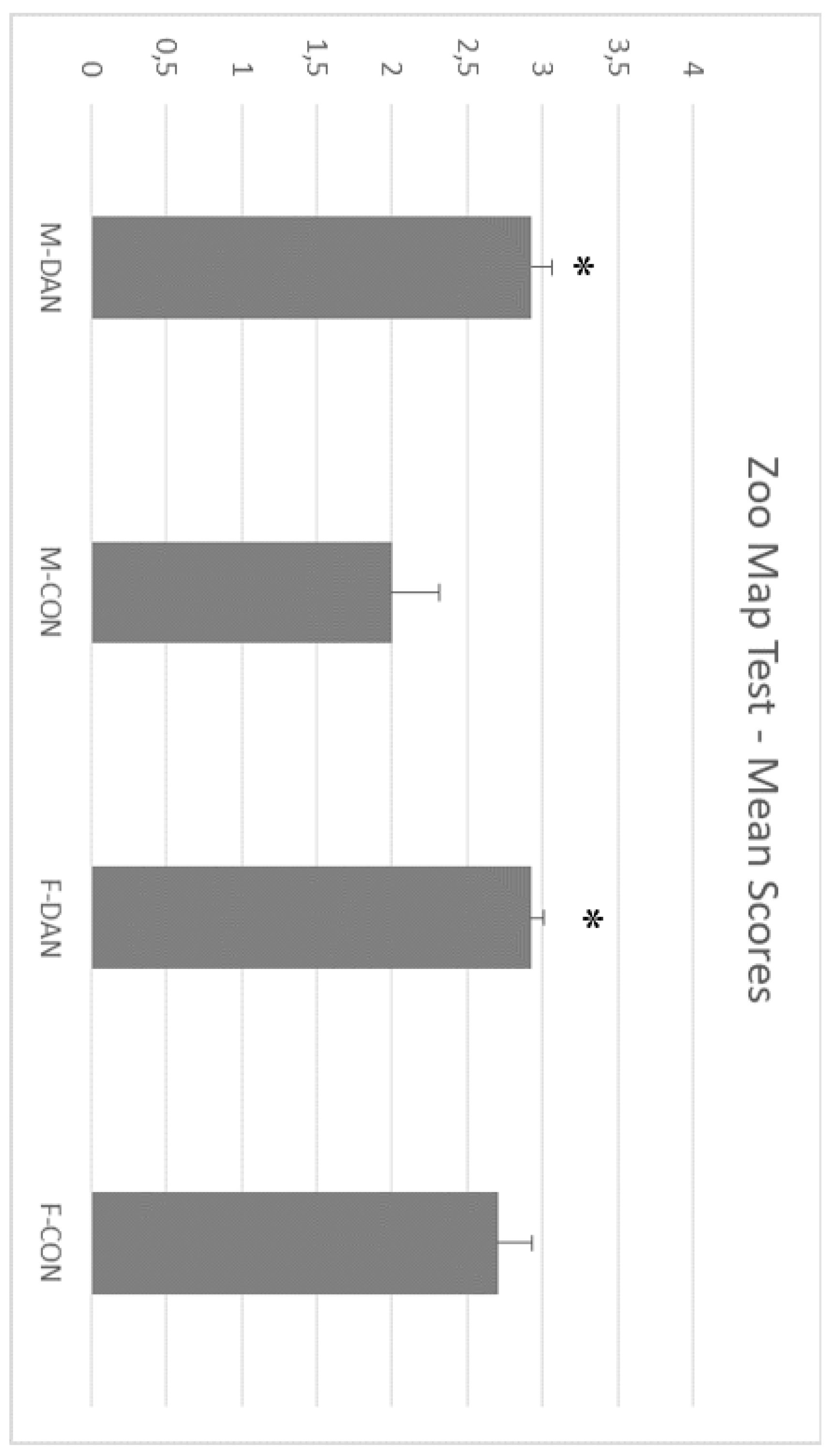
3.2. Zoo Test
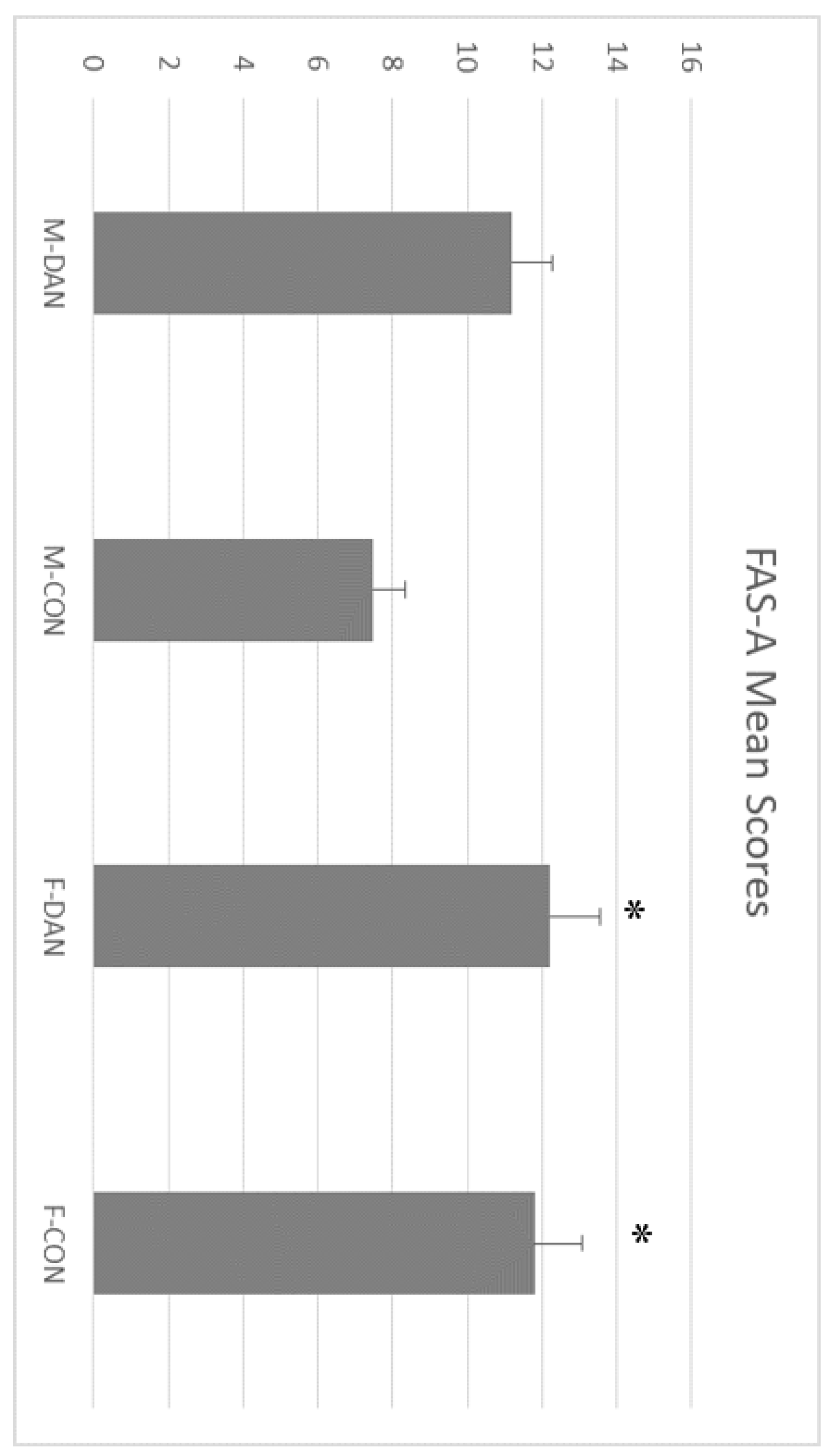
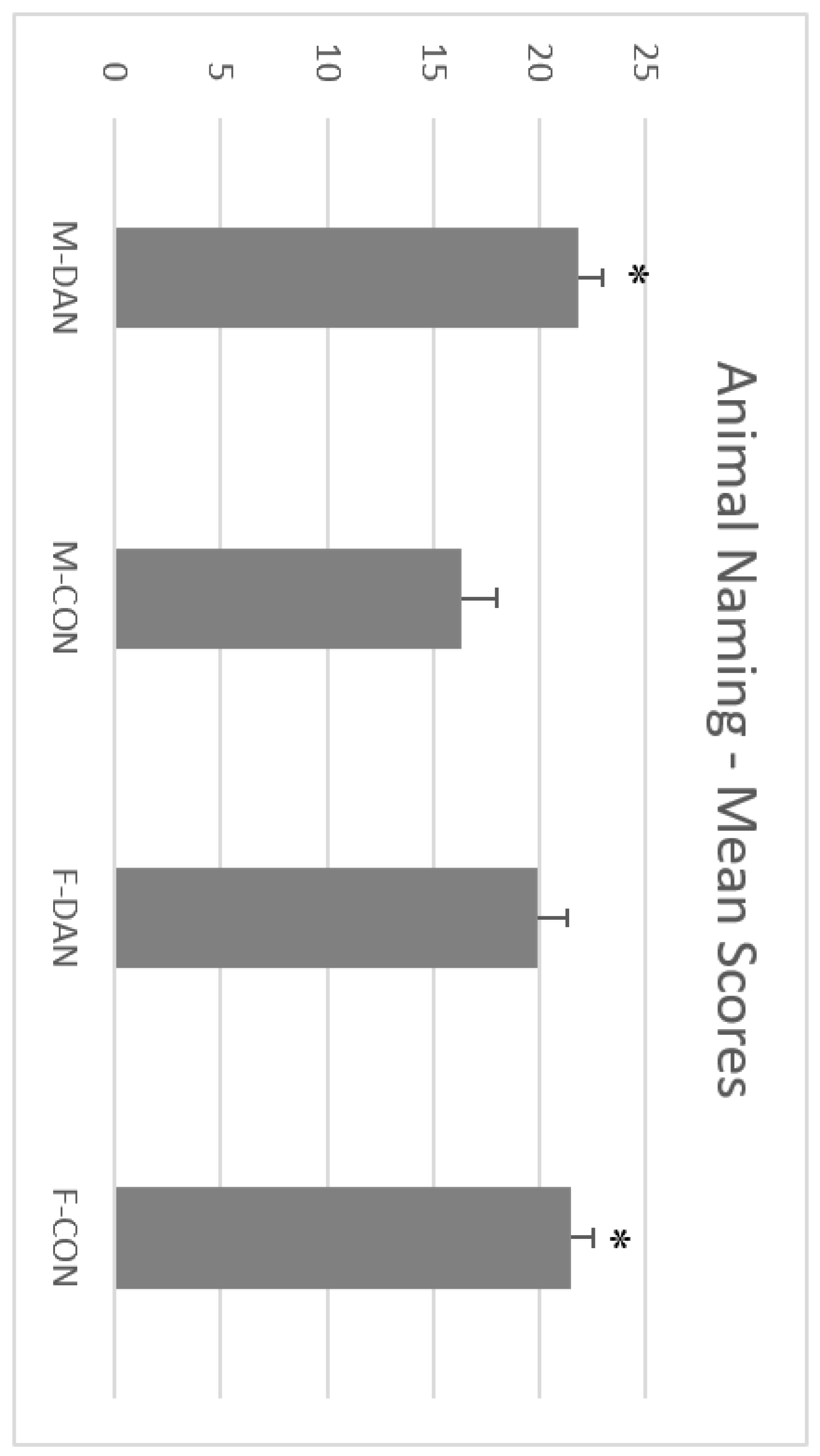
3.3. FAS-A Test and Animal Naming
3.4. The ANT-I Task
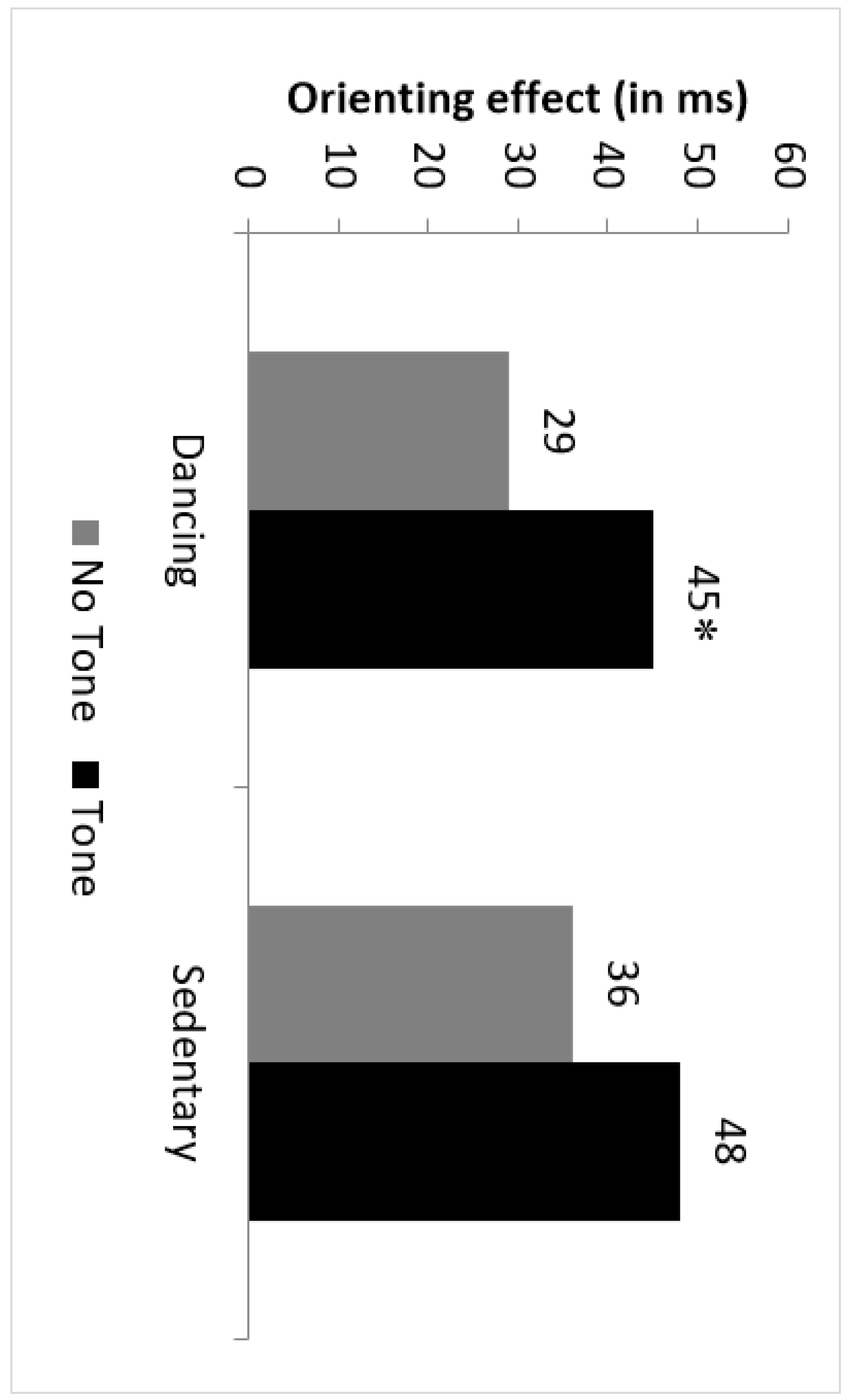
3.4.1. Latencies
3.4.2. Errors
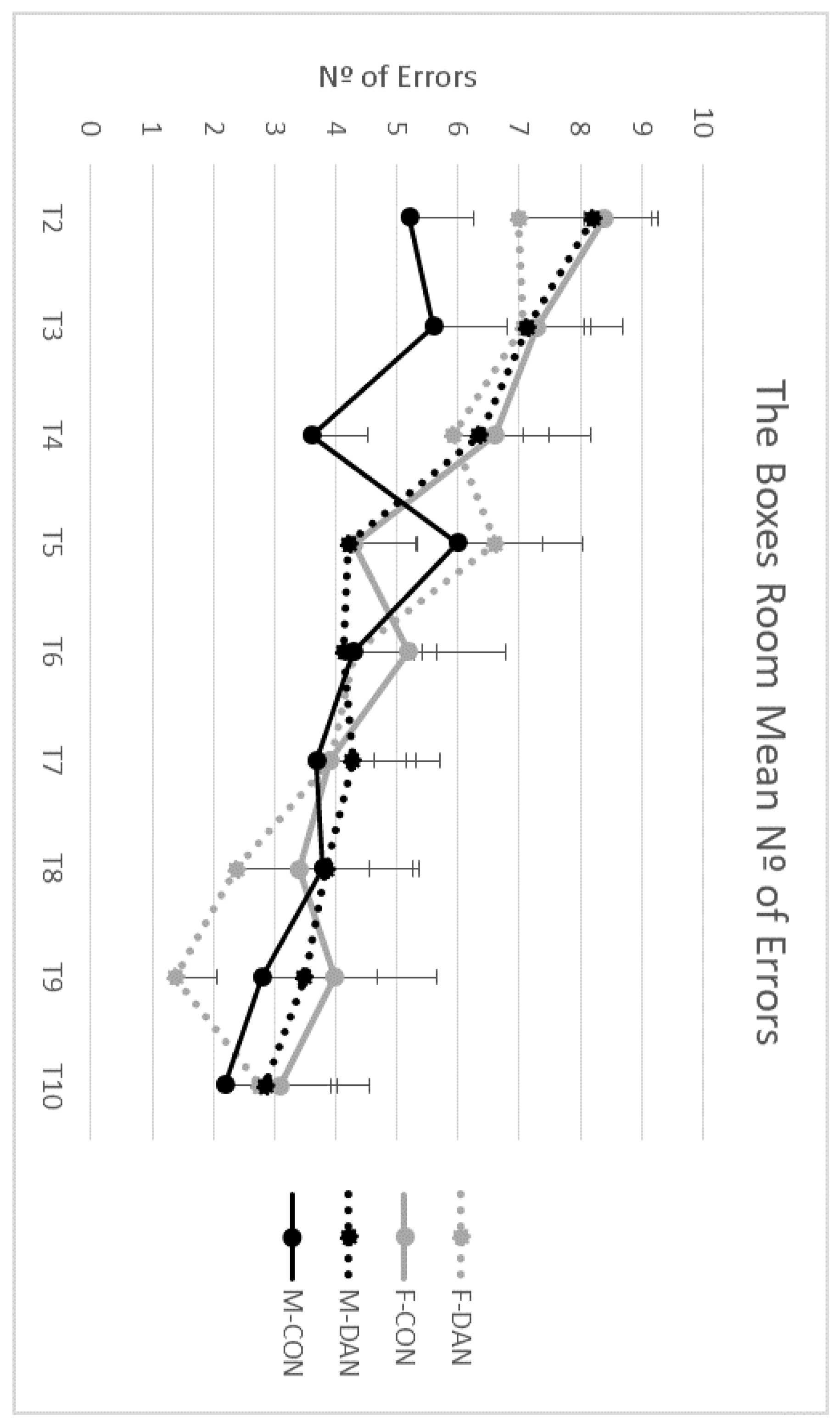
3.5. The Boxes Room Task
Errors
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lutz, W.; Warren, S.; Scherboy, S. The coming acceleration of global population ageing. Nature 2008, 451, 716–719. [Google Scholar] [CrossRef]
- Crespo, D.; Fernández, C. Cambios cerebrales en el envejecimiento normal y patológico. Rev. Neuropsicol. Neuropsiquiatr. Neurocienc. 2012, 21, 21–36. [Google Scholar]
- Beason-Held, L.L.; Hohman, T.J.; Venkatraman, V.; An, Y.; Resnick, S.M. Brain network changes and memory decline in aging. Brain Imaging Behav. 2017, 11, 859–873. [Google Scholar] [CrossRef] [PubMed]
- Nyberg, L. Functional brain imaging of episodic memory decline in ageing. J. Intern. Med. 2017, 281, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Kessels, R.P.C.; De Haan, E.H.F.; Kappelle, L.J.; Postma, A. Varieties of human spatial memory: A meta-analysis on the effects of hippocampal lesions. Brain Res. Brain Res. Rev. 2001, 35, 295–303. [Google Scholar] [CrossRef]
- Antonova, E.; Parslow, D.; Brammer, M.; Simmons, A.; Williams, S.; Dawson, G.R.; Morris, R. Scopolamine disrupts hippocampal activity during allocentric spatial memory in humans: An fMRI study using a virtual reality analogue of the Morris Water Maze. J. Psychopharmacol. 2011, 5, 1256–1265. [Google Scholar] [CrossRef] [PubMed]
- Astur, R.S.; Ortiz, M.L.; Sutherland, R.J. A characterization of performance by men and women in a virtual Morris water task: A large and reliable sex difference. Behav. Brain Res. 1998, 93, 185–190. [Google Scholar] [CrossRef]
- Cánovas, R.; Espínola, M.; Iribarne, L.; Cimadevilla, J.M. A new virtual task to evaluate human place learning. Behav. Brain Res. 2008, 190, 112–118. [Google Scholar] [CrossRef]
- Maguire, E.A.; Nannery, R.; Spiers, H.J. Navigation around London by a taxi driver with bilateral hippocampal lesions. Brain 2006, 129, 2894–2907. [Google Scholar] [CrossRef]
- Matheis, R.J.; Schultheis, M.T.; Tiersky, L.A.; DeLuca, J.; Millis, S.R.; Rizzo, A.A. Is learning and memory different in a virtual environment? Clin. Neuropsychol. 2007, 21, 146–161. [Google Scholar] [CrossRef]
- Piccardi, L.; Iaria, G.; Bianchini, F.; Zompanti, L.; Guariglia, C. Dissociated deficits of visuo-spatial memory in near space and navigational space: Evidence from brain-damaged patients and healthy older participants. Neuropsychol. Dev. Cognit. B Aging Neuropsychol. Cognit. 2011, 18, 362–384. [Google Scholar] [CrossRef] [PubMed]
- Cimadevilla, J.M.; Lizana, J.R.; Roldán, M.D.; Cánovas, R.; Rodríguez, E. Spatial memory alterations in children with epilepsy of genetic origin or unknown cause. Epilept. Disord. 2014, 16, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.A.; Moodley, K.K.; Lever, C.; Minati, L.; Chan, D. Allocentric spatial memory testing predicts conversion from mild cognitive impairment to dementia: An initial proof-of-concept study. Front. Neurol. 2016, 7, 215. [Google Scholar] [CrossRef] [PubMed]
- Fjell, A.M.; Sneve, M.H.; Storsve, A.B.; Grydeland, H.; Yendiki, A.; Walhovd, K.B. Brain events underlying episodic memory changes in aging: A longitudinal investigation of structural and functional connectivity. Cereb. Cortex 2016, 26, 1272–1286. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; McCandliss, B.D.; Sommer, T.; Raz, A.; Posner, M. Testing the efficiency and independence of attentional networks. J. Cognit. Neurosci. 2002, 14, 340–347. [Google Scholar] [CrossRef]
- Eriksen, B.A.; Eriksen, C.W. Effects of noise letters upon the identification of a target letter in a nonsearch task. Percept. Psychophys. 1974, 16, 143–149. [Google Scholar] [CrossRef]
- Posner, M.I. Orienting of attention. Q. J. Exp. Psychol. 1980, 32, 3–25. [Google Scholar] [CrossRef]
- Posner, M.I.; Petersen, S.E. The attention system of the human brain. Annu. Rev. Neurosci. 1990, 13, 25–42. [Google Scholar] [CrossRef]
- Petersen, S.E.; Posner, M.I. The attention system of the human brain: 20 years after. Annu. Rev. Neurosci. 2012, 35, 73–89. [Google Scholar] [CrossRef]
- Callejas, A.; Lupiañez, J.; Tudela, P. The three attentional networks: On their Independence and interaction. Brain Cognit. 2004, 54, 225–227. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Gu, X.; Guise, K.G.; Liu, X.; Fossella, J.; Wangc, H.; Posner, M.I. Testing the behavioral interaction and integration of attentional networks. Brain Cognit. 2009, 70, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Weinbach, N.; Henik, A. The relationship between alertness and executive control. J. Exp. Psychol. Hum. Percept. Perform. 2012, 38, 1530–1540. [Google Scholar] [CrossRef]
- Rueda, M.R.; Fan, J.; McCandliss, B.; Halparin, J.D.; Gruber, D.B.; Pappert, L.; Posner, M.I. Development of attentional networks in childhood. Neuropsychologia 2004, 42, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Íñiguez, C.; Fuentes, L.J.; Martínez-Sánchez, F.; Campoy, G.; Montoro, P.R.; Palmero, F. Emotional cuing to test attentional network functioning in trait anxiety. Psicológica 2014, 35, 309–329. [Google Scholar] [CrossRef]
- Pacheco-Unguetti, A.P.; Acosta, A.; Marqués, E.; Lupiáñez, J. Alterations of the attentional networks in patients with anxiety disorders. J. Anxiety Disord. 2011, 25, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Fossella, J.; Sommer, T.; Fan, J.; Wu, Y.; Swanson, J.M.; Pfaff, D.W.; Posner, M.I. Assessing the molecular genetics of attention networks. BMC Neurosci. 2002, 3, 14. [Google Scholar] [CrossRef]
- Fan, J.; McCandliss, B.D.; Fossella, J.; Flombaum, J.I.; Posner, M.I. The activation of attentional networks. NeuroImage 2005, 26, 471–479. [Google Scholar] [CrossRef]
- Jha, A.P.; Krompinger, J.; Baime, M.J. Mindfulness training modifies subsystems of attention. Cognit. Affect. Behav. Neurosci. 2007, 7, 109–119. [Google Scholar] [CrossRef]
- Huertas, F.; Zahonero, J.; Sanabria, D.; Lupiañez, J. Functioning of the attentional networks at rest vs during acute bouts of aerobic exercise. J. Sport Exerc. Psychol. 2011, 33, 649–665. [Google Scholar] [CrossRef]
- Noguera, C.; Sánchez-Horcajo, R.; Álvarez-Cazorla, D.; Cimadevilla, J.M. Ten years younger: Practice of chronic aerobic exercise improves attention and spatial memory functions in ageing. Exp. Gerontol. 2019, 117, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.J.; Kramer, A.F. Fitness effects on the cognitive function of older adults: A meta-analytic study. Perspect. Psychol. Sci. 2003, 14, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Prakash, R.S.; Voss, M.W.; Chaddock, L.; Hu, L.; Morris, K.S.; White, S.M.; Wójcicki, T.R.; McAuley, E.; Kramer, A.F. Aerobic fitness is associated with hippocampal volume in elderly humans. Hippocampus 2009, 19, 1030–1039. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Horcajo, R.; Llamas-Alonso, J.; Cimadevilla, J.M. Practice of aerobic sports is associated with better spatial memory in adults and older men. Exp. Aging Res. 2015, 41, 193–203. [Google Scholar] [CrossRef]
- Colcombe, S.J.; Erickson, K.I.; Scalf, P.E.; Kim, J.S.; Prakash, R.; McAuley, E.; Elavsky, S.; Marquez, D.X.; Hu, L.; Kramer, A.F. Aerobic exercise training increases brain volume in aging humans. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 1166–1170. [Google Scholar] [CrossRef]
- Erickson, K.I.; Voss, M.W.; Prakash, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M.; et al. Exercise training increases size of the hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 2011, 108, 3017–3022. [Google Scholar] [CrossRef]
- Stern, Y. Cognitive reserve in ageing and Alzheimer’s disease. Lancet Neurol. 2012, 11, 1006–1012. [Google Scholar] [CrossRef]
- Gerber, M.; Brand, S.; Elliot, C.; Holsboer-Trachsler, E.; Púhse, U. Aerobic exercise, ball sports, dancing, and weight lifting as moderators of the relationship between stress and depressive symptoms: An exploratory cross-sectional study with swiss university students. Percept. Mot. Skills 2014, 119, 679–697. [Google Scholar] [CrossRef]
- Sevdalis, V.; Keller, P.E. Captured by motion: Dance, action understanding, and social cognition. Brain Cognit. 2011, 77, 231–236. [Google Scholar] [CrossRef]
- Kosmat, H.; Vranic, A. The efficacy of a dance intervention as cognitive training for the old-old. J. Aging Phys. Act. 2017, 25, 32–40. [Google Scholar] [CrossRef]
- Merom, D.; Grunseit, A.; Eramudugolla, R.; Jefferis, B.; Mcneill, J.; Anstey, K.J. Cognitive benefits of social dancing and walking in old age: The dancing mind randomized controlled trial. Front. Aging Neurosci. 2016, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Rehfeld, K.; Lüders, A.; Hökelmann, A.; Lessmann, V.; Kaufmann, J.; Brigadski, T.; Müller, N.G. Dance training is superior to repetitive physical exercise in inducing brain plasticity in the elderly. PLoS ONE 2018, 13, e0196636. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, A.; Kaufman, N. Kaufman Brief Intelligence Test; American Guidance Service: Circle Pines, MN, USA, 1990. [Google Scholar]
- Guilford, J.P. The Nature of Human Intelligence; McGraw-Hill Education: New York, NY, USA, 1967. [Google Scholar] [CrossRef]
- Wilson, B.A.; Alderman, N.; Burgess, P.W.; Emslie, H.; Evans, J.J. The Behavioural Assessment of the Dysexecutive Syndrome; Thames Valley Test Company: Bury St Edmunds, UK, 1996. [Google Scholar] [CrossRef]
- Decety, J.; Grezes, J.; Costes, N.; Perani, D.; Jeannerod, M.; Procyk, E.; Grassi, F.; Fazio, F. Brain activity during observation of actions: Influence of action content and subject’s strategy. Brain 1997, 120, 1763–1777. [Google Scholar] [CrossRef] [PubMed]
- Frith, C.D.; Friston, K.; Liddle, P.F.; Frackowiak, R.S.J. Willed action and the prefrontal cortex in man: A study with PET. Proc. Biol. Sci. 1991, 244, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.F.; Gonzalez, J.; Cassavaugh, N. Development of attentional and oculomotor control. Dev. Neuropsychol. 2005, 41, 760–772. [Google Scholar] [CrossRef]
- Weinstein, A.M.; Voss, M.W.; Prakash, R.S.; Chaddock, L.; Szabo, A.; White, S.M.; Wojcicki, T.R.; Mailey, E.; McAuley, E.; Kramer, A.F.; et al. The association between aerobic fitness and executive function is mediated by prefrontal cortex volume. Brain Behav. Immun. 2012, 26, 811–819. [Google Scholar] [CrossRef]
- Colcombe, S.J.; Kramer, A.F.; Erickson, K.I.; Scalf, P.; McAuley, E.; Cohen, N.J.; Webb, A.; Jerome, G.J.; Marquez, D.X.; Elavsky, S. Cardiovascular fitness, cortical plasticity, and aging. Proc. Natl. Acad. Sci. USA 2004, 101, 3316–3321. [Google Scholar] [CrossRef]
- Wong, C.N.; Chaddock-Heyman, L.; Voss, M.W.; Burzynska, A.Z.; Basak, C.; Erickson, K.I.; Prakash, R.S.; Szabo-Reed, A.N.; Phillips, S.M.; Wojcicki, T.; et al. Brain activation during dual-task processing is associated with cardiorespiratory fitness and performance in older adults. Front. Aging Neurosci. 2015, 7, 154. [Google Scholar] [CrossRef]
- Burton, L.A.; Henninger, D.; Hafetz, J. Gender differences in relations of mental rotation, verbal fluency, and SAT scores to finger length ratios as hormonal indexes. Dev. Neuropsychol. 2005, 28, 493–505. [Google Scholar] [CrossRef]
- Hyde, J.S.; Linn, M.C. Gender differences in verbal-ability: A meta-analysis. Psychol. Bull. 1988, 104, 53–69. [Google Scholar] [CrossRef]
- Fink, A.; Graif, B.; Neubauer, A.C. Brain correlates underlying creative thinking: EEG alpha activity in professional vs. novice dancers. Neuroimage 2009, 46, 854–862. [Google Scholar] [CrossRef]
- Posner, M.I. Measuring alertness. Ann. N. Y. Acad. Sci. 2008, 1129, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Corbetta, M.; Patel, G.; Shulman, G.L. The reorienting system of the human brain: From environment to theory of mind. Neuron 2008, 58, 306–324. [Google Scholar] [CrossRef] [PubMed]
- Guiney, H.; Machado, L. Benefits of regular aerobic exercise for executive functioning in healthy populations. Psychon. Bull. Rev. 2013, 20, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I.; Rothbart, M.K. Attentional mechanisms and conscious experience. In The Neuropsychology of Consciousness; Milner, A.D., Rugg, M.D., Eds.; Academic Press: New York, NY, USA, 1991; pp. 91–112. [Google Scholar] [CrossRef]
- León, I.; Tascón, L.; Ortells-Pareja, J.J.; Cimadevilla, J.M. Virtual reality assessment of walking and non-walking space in men and women with virtual reality-based tasks. PLoS ONE 2018, 13, e0204995. [Google Scholar] [CrossRef] [PubMed]
- Cánovas, R.; Cimadevilla, J.M. Sexual orientation and spatial memory. Psicothema 2011, 23, 752–758. [Google Scholar] [PubMed]
- Cánovas, R.; Fernández, R.; Cimadevilla, J.M. Effect of reference frames and number of cues available on the spatial orientation of males and females in a virtual memory task. Behav. Brain Res. 2011, 216, 116–121. [Google Scholar] [CrossRef]
- León, I.; Tascón, L.; Cimadevilla, J.M. Age and gender-related differences in a spatial memory task in humans. Behav. Brain Res. 2016, 306, 8–12. [Google Scholar] [CrossRef]
- Nori, R.; Piccardi, L.; Maialetti, A.; Goro, M.; Rossetti, A.; Argento, O.; Guariglia, C. No gender differences in egocentric and allocentric environmental transformation after compensating for male advantage by manipulating familiarity. Front. Neurosci. 2018, 12, 204. [Google Scholar] [CrossRef]
- Cánovas, R.; León, I.; Serrano, P.; Roldán, M.D.; Cimadevilla, J.M. Spatial navigation impairment in patients with refractory temporal lobe epilepsy: Evidence from a new virtual reality-based task. Epilepsy Behav. 2011, 22, 364–369. [Google Scholar] [CrossRef]
- Herting, M.M.; Nagel, B.J. Aerobic fitness relates to learning on a virtual Morris Water Task and hippocampal volume in adolescents. Behav. Brain Res. 2012, 233, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Kandola, A.; Hendrikse, J.; Lucassen, P.J.; Yucel, M. Aerobic exercise as a tool to improve hippocampal plasticity and function in humans: Practical implications for mental health treatment. Front. Hum. Neurosci. 2016, 10, 373. [Google Scholar] [CrossRef] [PubMed]
- León, I.; Garcia-Garcia, J.; Roldan-Tapia, L. Estimating cognitive reserve in healthy adults using the cognitive reserve scale. PLoS ONE 2014, 9, e0102632. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.B.; Lipton, R.B.; Sliwinski, M.; Katz, M.J.; Derby, C.A.; Verghese, J. Cognitive activities delay onset of memory decline in persons who develop dementia. Neurology 2009, 73, 356–361. [Google Scholar] [CrossRef]
- Stern, Y. Cognitive reserve: Implications for assessment and intervention. Folia Phoniatr. Logop. 2013, 65, 49–54. [Google Scholar] [CrossRef]
- Mass, A.; Duzzel, S.; Goerke, M.; Becke, A.; Sobieray, U.; Neumann, K.; Lövden, M.; Lindenberger, U.; Bäckman, L.; Braun-Dullaeus, R.; et al. Vascular hippocampal plasticity after aerobic exercise in older adults. Mol. Psychiatry 2015, 20, 585–593. [Google Scholar] [CrossRef]
- Krogh, J.; Rostrup, E.; Thomsem, C.; Elfving, B.; Videbech, P.; Nordemtoft, M. The effect of exercise on hippocampal volume and neurotrophines in patients with major depression-a randomized clinical trial. J. Affect. Disord. 2014, 165, 24–30. [Google Scholar] [CrossRef]
- Panjonk, F.G.; Wobrok, T.; Gruber, O.; Scherk, H.; Berner, D.; Kaizl, I.; Kierer, A.; Müller, S.; Oest, M.; Meyer, T.; et al. Hippocampal plasticity in response to exercise in schizophrenia. Arch. Gen. Psychiatry 2010, 67, 133–143. [Google Scholar] [CrossRef]
- Sacheli, L.M.; Zapparoli, L.; De Santis, C.; Preti, M.; Pelosi, C.; Ursino, N.; Zerbi, A.; Banfi, G.; Paulesu, E. Mental steps: Differential activation of internal pacemakers in motor imagery and in mental imitation of gait. Hum. Brain Mapp. 2017, 38, 5195–5216. [Google Scholar] [CrossRef]
- Pascual-Leone, A. The brain that plays music and is changed by it. Ann. N. Y. Acad. Sci. 2001, 930, 315–329. [Google Scholar] [CrossRef]
- Astur, R.; Taylor, L.B.; Mamelak, A.N.; Phipott, L.; Sutherland, R.J. Humans with hippocampus damage display severe spatial memory impairments in a virtual Morris water task. Behav. Brain Res. 2002, 132, 77–84. [Google Scholar] [CrossRef]
| Dancers | Sedentary | |||||||
|---|---|---|---|---|---|---|---|---|
| Men | Women | Men | Women | |||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Age | 57.1 | 5.8 | 54.9 | 4.35 | 61 | 5.1 | 54.2 | 3.5 |
| Educational Level 1–3 | 2 | 0.78 | 2.33 | 0.77 | 2 | 0.9 | 1.9 | 0.9 |
| Years Dancing | 9.1 | 7.9 | 7.4 | 7.8 | - | - | - | - |
| Videogame Experience 1–4 | 1.35 | 0.6 | 1.58 | 0.9 | 1.1 | 0.3 | 1.7 | 1.1 |
| Congruent | Incongruent | |||
|---|---|---|---|---|
| Dancers | Sedentary | Dancers | Sedentary | |
| No Tone | ||||
| Invalid | 634 | 684 | 736 | 797 |
| 21 | 24 | 19 | 22 | |
| No Cue | 637 | 694 | 736 | 791 |
| 19 | 22 | 19 | 22 | |
| Valid | 610 | 649 | 702 | 761 |
| 18 | 21 | 19 | 21 | |
| Tone | ||||
| Invalid | 613 | 671 | 735 | 785 |
| 18 | 21 | 19 | 22 | |
| No Cue | 587 | 644 | 703 | 772 |
| 18 | 20 | 19 | 21 | |
| Valid | 576 | 622 | 682 | 738 |
| 18 | 21 | 20 | 22 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noguera, C.; Carmona, D.; Rueda, A.; Fernández, R.; Cimadevilla, J.M. Shall We Dance? Dancing Modulates Executive Functions and Spatial Memory. Int. J. Environ. Res. Public Health 2020, 17, 1960. https://doi.org/10.3390/ijerph17061960
Noguera C, Carmona D, Rueda A, Fernández R, Cimadevilla JM. Shall We Dance? Dancing Modulates Executive Functions and Spatial Memory. International Journal of Environmental Research and Public Health. 2020; 17(6):1960. https://doi.org/10.3390/ijerph17061960
Chicago/Turabian StyleNoguera, Carmen, Dolores Carmona, Adrián Rueda, Rubén Fernández, and José Manuel Cimadevilla. 2020. "Shall We Dance? Dancing Modulates Executive Functions and Spatial Memory" International Journal of Environmental Research and Public Health 17, no. 6: 1960. https://doi.org/10.3390/ijerph17061960
APA StyleNoguera, C., Carmona, D., Rueda, A., Fernández, R., & Cimadevilla, J. M. (2020). Shall We Dance? Dancing Modulates Executive Functions and Spatial Memory. International Journal of Environmental Research and Public Health, 17(6), 1960. https://doi.org/10.3390/ijerph17061960







