Toxicity of Microplastics and Nanoplastics in Mammalian Systems
Abstract
1. Introduction
2. Toxicity of MPs/NPs in Fishes
3. Toxicity of MPs/NPs in Mouse Models
4. Toxicity of MPs/NPs in Human Cells
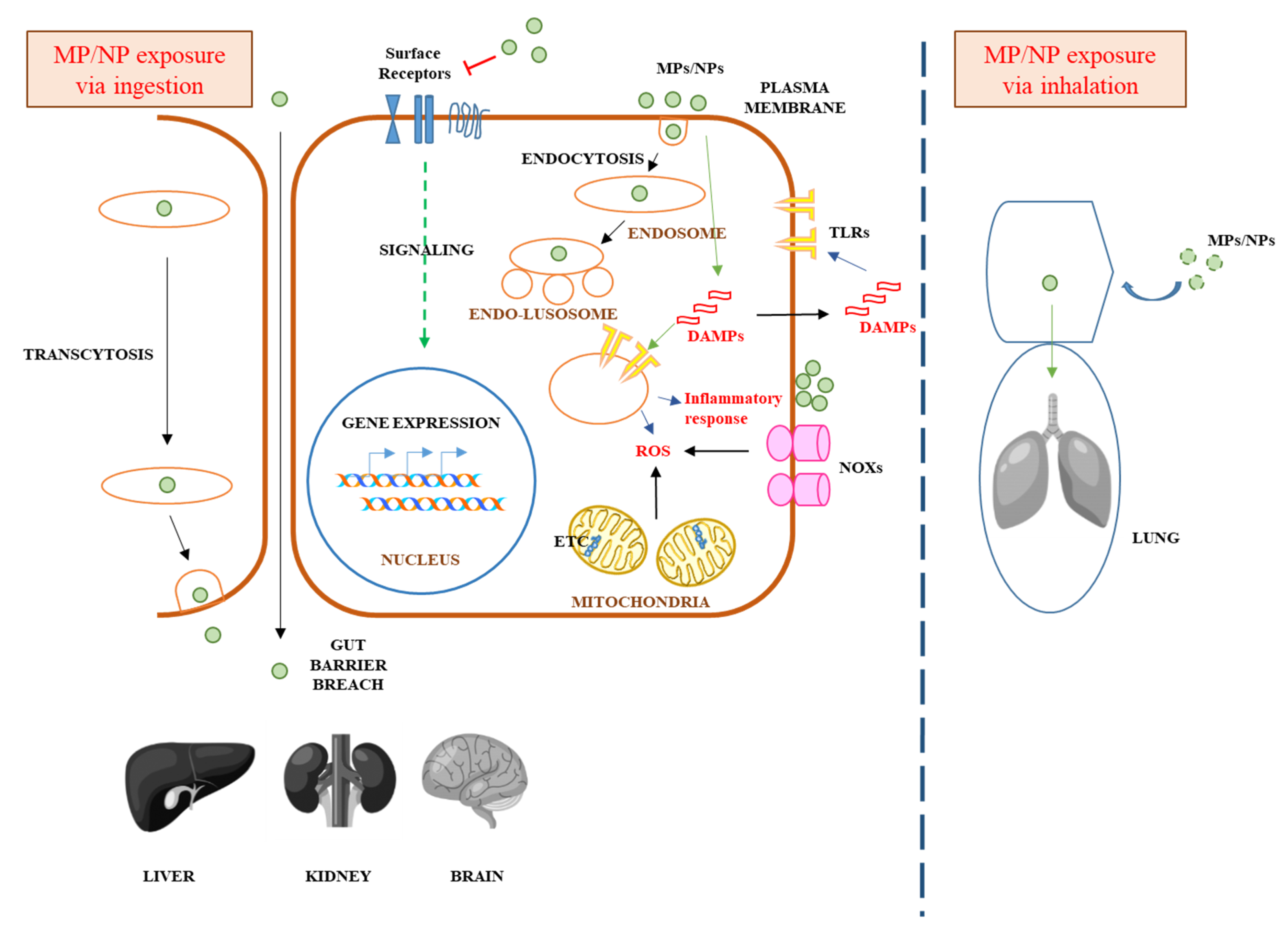
5. Mechanisms Underlying MPs/NPs’ Acute or Chronic Toxicity in Mammalian Cells
6. MPs/NPs’ Potential Systemic Effect in Humans
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zalasiewicz, J.; Waters, C.; Ivar do Sul, J.; Corcoran, P.; Barnosky, A.; Cearreta, A.; Edgeworth, M.; Gałuszkah, A.; Jeandel, C.; Leinfelder, R.; et al. The geological cycle of plastics and their use as a stratigraphic indicator of the Anthropocene. Anthropocene 2016, 13, 4–17. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Gündoğdu, S.; Yeşilyurt, İ.N.; Erbaş, C. Potential interaction between plastic litter and green turtle Chelonia mydas during nesting in an extremely polluted beach. Mar. Pollut. Bull. 2019, 140, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Chapron, L.; Peru, E.; Engler, A.; Ghiglione, J.F.; Meistertzheim, A.L.; Pruski, A.M.; Purser, A.; Vétion, G.; Galand, P.E.; Lartaud, F. Macro- and microplastics affect cold-water corals growth, feeding and behaviour. Sci. Rep. 2018, 8, 15299. [Google Scholar] [CrossRef] [PubMed]
- Akdogan, Z.; Guven, B. Microplastics in the environment: A critical review of current understanding and identification of future research needs. Environ. Pollut. 2019, 254, 113011. [Google Scholar] [CrossRef]
- de Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an emerging threat to terrestrial ecosystems. Glob. Chang. Biol. 2018, 24, 1405–1416. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef]
- Galloway, T.S.; Cole, M.; Lewis, C. Interactions of microplastic debris throughout the marine ecosystem. Nat. Ecol. Evol. 2017, 1, 116. [Google Scholar] [CrossRef]
- da Costa, J.P.; Santos, P.S.M.; Duarte, A.C.; Rocha-Santos, T. (Nano)plastics in the environment—Sources, fates and effects. Sci. Total Environ. 2016, 566–567, 15–26. [Google Scholar] [CrossRef]
- Gigault, J.; Halle, A.T.; Baudrimont, M.; Pascal, P.Y.; Gauffre, F.; Phi, T.L.; El Hadri, H.; Grassl, B.; Reynaud, S. Current opinion: What is a nanoplastic? Environ. Pollut. 2018, 235, 1030–1034. [Google Scholar] [CrossRef]
- de Sá, L.C.; Oliveira, M.; Ribeiro, F.; Rocha, T.L.; Futter, M.N. Studies of the effects of microplastics on aquatic organisms: What do we know and where should we focus our efforts in the future? Sci. Total Environ. 2018, 645, 1029–1039. [Google Scholar] [CrossRef] [PubMed]
- Alimba, C.G.; Faggio, C. Microplastics in the marine environment: Current trends in environmental pollution and mechanisms of toxicological profile. Environ. Toxicol. Pharmacol. 2019, 68, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Lambert, S.; Scherer, C.; Wagner, M. Ecotoxicity testing of microplastics: Considering the heterogeneity of physicochemical properties. Integr. Environ. Assess. Manag. 2017, 13, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Adam, V.; Yang, T.; Nowack, B. Toward an ecotoxicological risk assessment of microplastics: Comparison of available hazard and exposure data in freshwaters. Environ. Toxicol. Chem. 2019, 38, 436–447. [Google Scholar] [CrossRef]
- Hartmann, N.B.; Rist, S.; Bodin, J.; Jensen, L.H.; Schmidt, S.N.; Mayer, P.; Meibom, A.; Baun, A. Microplastics as vectors for environmental contaminants: Exploring sorption, desorption, and transfer to biota. Integr. Environ. Assess. Manag. 2017, 13, 488–493. [Google Scholar] [CrossRef]
- Liu, J.; Ma, Y.; Zhu, D.; Xia, T.; Qi, Y.; Yao, Y.; Guo, X.; Ji, R.; Chen, W. Polystyrene nanoplastics-enhanced contaminant transport: Role of irreversible adsorption in glassy polymeric domain. Environ. Sci. Technol. 2018, 52, 2677–2685. [Google Scholar] [CrossRef]
- Caruso, G. Microplastics as vectors of contaminants. Mar. Pollut. Bull. 2019, 146, 921–924. [Google Scholar] [CrossRef]
- Sgier, L.; Freimann, R.; Zupanic, A.; Kroll, A. Flow cytometry combined with viSNE for the analysis of microbial biofilms and detection of microplastics. Nat. Commun. 2016, 7, 11587. [Google Scholar] [CrossRef]
- Wu, X.; Pan, J.; Li, M.; Li, Y.; Bartlam, M.; Wang, Y. Selective enrichment of bacterial pathogens by microplastic biofilm. Water Res. 2019, 165, 114979. [Google Scholar] [CrossRef]
- Sussarellu, R.; Suquet, M.; Thomas, Y.; Lambert, C.; Fabioux, C.; Pernet, M.E.J.; Le Goïc, N.; Quillien, V.; Mingant, C.; Epelboin, Y.; et al. Oyster reproduction is affected by exposure to polystyrene microplastics. Proc. Natl. Acad. Sci. USA 2016, 113, 2430–2435. [Google Scholar] [CrossRef]
- Cui, R.; Kim, S.W.; An, Y.J. Polystyrene nanoplastics inhibit reproduction and induce abnormal embryonic development in the freshwater crustacean Daphnia galeata. Sci. Rep. 2017, 7, 12095. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.D.; Weinstein, J.E. Size and shape dependent effects of microplastic particles on adult daggerblade glass shrimp, Palaemonetes pugio. Environ. Toxicol. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Bergami, E.; Pugnalini, S.; Vannuccini, M.L.; Manfra, L.; Faleri, C.; Savorelli, F.; Dawson, K.A.; Corsi, I. Long-term toxicity of surface-charged polystyrene nanoplastics to marine planktonic species Dunaliella tertiolecta and Artemia franciscana. Aquat. Toxicol. 2017, 189, 159–169. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Environmentally relevant concentrations of polyethylene microplastics negatively impact the survival, growth and emergence of sediment-dwelling invertebrates. Environ. Pollut. 2018, 236, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Bour, A.; Haarr, A.; Keiter, S.; Hylland, K. Environmentally relevant microplastic exposure affects sediment-dwelling bivalves. Environ. Pollut. 2018, 236, 652–660. [Google Scholar] [CrossRef]
- Tang, J.; Ni, X.; Zhou, Z.; Wang, L.; Lin, S. Acute microplastic exposure raises stress response and suppresses detoxification and immune capacities in the scleractinian coral Pocillopora damicornis. Environ. Pollut. 2018, 243, 66–74. [Google Scholar] [CrossRef]
- Marques-Santos, L.F.; Grassi, G.; Bergami, E.; Faleri, C.; Balbi, T.; Salis, A.; Damonte, G.; Canesi, L.; Corsi, I. Cationic polystyrene nanoparticle and the sea urchin immune system: Biocorona formation, cell toxicity, and multixenobiotic resistance phenotype. Nanotoxicology 2018. [Google Scholar] [CrossRef]
- Jaikumar, G.; Brun, N.R.; Vijver, M.G.; Bosker, T. Reproductive toxicity of primary and secondary microplastics to three cladocerans during chronic exposure. Environ. Pollut. 2019, 249, 638–646. [Google Scholar] [CrossRef]
- Eltemsah, Y.S.; Bøhn, T. Acute and chronic effects of polystyrene microplastics on juvenile and adult Daphnia magna. Environ. Pollut. 2019, 254, 112919. [Google Scholar] [CrossRef]
- Jiang, X.; Chen, H.; Liao, Y.; Ye, Z.; Li, M.; Klobučar, G. Ecotoxicity and genotoxicity of polystyrene microplastics on higher plant Vicia faba. Environ. Pollut. 2019, 250, 831–838. [Google Scholar] [CrossRef]
- Chae, Y.; An, Y.J. Effects of micro- and nanoplastics on aquatic ecosystems: Current research trends and perspectives. Mar. Pollut. Bull. 2017, 124, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Chatterjee, S. Microplastic pollution, a threat to marine ecosystem and human health: A short review. Environ. Sci. Pollut. Res. Int. 2017, 24, 21530–21547. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Gao, H.; Jin, S.; Li, R.; Na, G. The ecotoxicological effects of microplastics on aquatic food web, from primary producer to human: A review. Ecotoxicol. Environ. Saf. 2019, 173, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.; Nelson, K. Trophic level transfer of microplastic: Mytilus edulis (L.) to Carcinus maenas (L.). Environ. Pollut. 2013, 177, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, K.; Ekvall, M.T.; Hansson, L.A.; Linse, S.; Malmendal, A.; Cedervall, T. Altered behavior, physiology, and metabolism in fish exposed to polystyrene nanoparticles. Environ. Sci. Technol. 2015, 49, 553–561. [Google Scholar] [CrossRef]
- Chae, Y.; Kim, D.; Kim, S.W.; An, Y.J. Trophic transfer and individual impact of nano-sized polystyrene in a four-species freshwater food chain. Sci. Rep. 2018, 8, 284. [Google Scholar] [CrossRef]
- Velzeboer, I.; Kwadijk, C.J.A.F.; Koelmans, A.A. Strong sorption of PCBs to nanoplastics, microplastics, carbon nanotubes, and fullerenes. Environ. Sci. Technol. 2014, 48, 4869–4876. [Google Scholar] [CrossRef]
- Hüffer, T.; Hofmann, T. Sorption of non-polar organic compounds by micro-sized plastic particles in aqueous solution. Environ. Pollut. 2016, 214, 194–201. [Google Scholar] [CrossRef]
- Hüffer, T.; Weniger, A.K.; Hofmann, T. Sorption of organic compounds by aged polystyrene microplastic particles. Environ. Pollut. 2018, 236, 218–225. [Google Scholar] [CrossRef]
- Fang, S.; Yu, W.; Li, C.; Liu, Y.; Qiu, J.; Kong, F. Adsorption behavior of three triazole fungicides on polystyrene microplastics. Sci. Total Environ. 2019, 691, 1119–1126. [Google Scholar] [CrossRef]
- Tourinho, P.S.; Kočí, V.; Loureiro, S.; van Gestel, C.A.M. Partitioning of chemical contaminants to microplastics: Sorption mechanisms, environmental distribution and effects on toxicity and bioaccumulation. Environ. Pollut. 2019, 252, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Chua, E.M.; Shimeta, J.; Nugegoda, D.; Morrison, P.D.; Clarke, B.O. Assimilation of polybrominated diphenyl ethers from microplastics by the marine amphipod, Allorchestes compressa. Environ. Sci. Technol. 2014, 48, 8127–8134. [Google Scholar] [CrossRef] [PubMed]
- Batel, A.; Linti, F.; Scherer, M.; Erdinger, L.; Braunbeck, T. Transfer of benzo[a]pyrene from microplastics to Artemia nauplii and further to zebrafish via a trophic food web experiment: CYP1A induction and visual tracking of persistent organic pollutants. Environ. Toxicol. Chem. 2016, 35, 1656–1666. [Google Scholar] [CrossRef] [PubMed]
- Gandara E Silva, P.P.; Nobre, C.R.; Resaffe, P.; Pereira, C.D.S.; Gusmão, F. Leachate from microplastics impairs larval development in brown mussels. Water Res. 2016, 106, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yin, D.; Jia, Y.; Schiwy, S.; Legradi, J.; Yang, S.; Hollert, H. Enhanced uptake of BPA in the presence of nanoplastics can lead to neurotoxic effects in adult zebrafish. Sci. Total Environ. 2017, 609, 1312–1321. [Google Scholar] [CrossRef]
- Lu, K.; Qiao, R.; An, H.; Zhang, Y. Influence of microplastics on the accumulation and chronic toxic effects of cadmium in zebrafish (Danio rerio). Chemosphere 2018, 202, 514–520. [Google Scholar] [CrossRef]
- Banaee, M.; Soltanian, S.; Sureda, A.; Gholamhosseini, A.; Haghi, B.N.; Akhlaghi, M.; Derikvandy, A. Evaluation of single and combined effects of cadmium and micro-plastic particles on biochemical and immunological parameters of common carp (Cyprinus carpio). Chemosphere 2019, 236, 124335. [Google Scholar] [CrossRef]
- Miranda, T.; Vieira, L.R.; Guilhermino, L. Neurotoxicity, behavior, and lethal effects of Cadmium, microplastics, and their mixtures on Pomatoschistus microps juveniles from two wild populations exposed under laboratory conditions-Implications to environmental and human risk assessment. Int. J. Environ. Res. Public Health 2019, 16, 2857. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Vieira, L.R.; Branco, V.; Figueiredo, N.; Carvalho, F.; Carvalho, C.; Guilhermino, L. Microplastics cause neurotoxicity, oxidative damage and energy-related changes and interact with the bioaccumulation of mercury in the European seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat. Toxicol. 2018, 195, 49–57. [Google Scholar] [CrossRef]
- Bradney, L.; Wijesekara, H.; Palansooriya, K.N.; Obadamudalige, N.; Bolan, N.S.; Ok, Y.S.; Rinklebe, J.; Kim, K.H.; Kirkham, M.B. Particulate plastics as a vector for toxic trace-element uptake by aquatic and terrestrial organisms and human health risk. Environ. Int. 2019, 131, 104937. [Google Scholar] [CrossRef]
- Wright, S.L.; Kelly, F.J. Plastic and human health: A micro issue? Environ. Sci. Technol. 2017, 51, 6634–6647. [Google Scholar] [CrossRef] [PubMed]
- Rubio, L.; Marcos, R.; Hernández, A. Potential adverse health effects of ingested micro- and nanoplastics on humans. Lessons learned from in vivo and in vitro mammalian models. J. Toxicol. Environ. Health B Crit. Rev. 2020, 23, 51–68. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Love, D.C.; Rochman, C.M.; Neff, R.A. Microplastics in seafood and the implications for human health. Curr. Environ. Health Rep. 2018, 5, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Lee, H.J.; Kim, S.K.; Kim, H.J. Global pattern of microplastics (MPs) in commercial food-grade salts: Sea salt as an indicator of seawater MP pollution. Environ. Sci. Technol. 2018, 52, 12819–12828. [Google Scholar] [CrossRef]
- Pivokonsky, M.; Cermakova, L.; Novotna, K.; Peer, P.; Cajthaml, T.; Janda, V. Occurrence of microplastics in raw and treated drinking water. Sci. Total Environ. 2018, 643, 1644–1651. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Mohamed Nor, N.H.; Hermsen, E.; Kooi, M.; Mintenig, S.M.; De France, J. Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res. 2019, 155, 410–422. [Google Scholar] [CrossRef]
- Oßmann, B.E.; Sarau, G.; Holtmannspötter, H.; Pischetsrieder, M.; Christiansen, S.H.; Dicke, W. Small-sized microplastics and pigmented particles in bottled mineral water. Water Res. 2018, 141, 307–316. [Google Scholar] [CrossRef]
- Zuccarello, P.; Ferrante, M.; Cristaldi, A.; Copat, C.; Grasso, A.; Sangregorio, D.; Fiore, M.; Oliveri Conti, G. Exposure to microplastics (<10 μm) associated to plastic bottles mineral water consumption: The first quantitative study. Water Res. 2019, 157, 365–371. [Google Scholar]
- Hernandez, L.M.; Xu, E.G.; Larsson, H.C.E.; Tahara, R.; Maisuria, V.B.; Tufenkji, N. Plastic teabags release billions of microparticles and nanoparticles into Tea. Environ. Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Prata, J.C. Airborne microplastics: Consequences to human health? Environ. Pollut. 2018, 234, 115–126. [Google Scholar] [CrossRef]
- Schwabl, P.; Köppel, S.; Königshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of various microplastics in human stool: A prospective case series. Ann. Intern. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Microplastics in Drinking-Water. 2019. Available online: https://www.who.int/water_sanitation_health/publications/microplastics-in-drinking-water/en/ (accessed on 26 February 2020).
- Foley, C.J.; Feiner, Z.S.; Malinich, T.D.; Höök, T.O. A meta-analysis of the effects of exposure to microplastics on fish and aquatic invertebrates. Sci. Total Environ. 2018, 631–632, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ge, J.; Yu, X. Bioavailability and toxicity of microplastics to fish species: A review. Ecotoxicol. Environ. Saf. 2020, 189, 109913. [Google Scholar] [CrossRef] [PubMed]
- Ašmonaitė, G.; Sundh, H.; Asker, N.; Carney Almroth, B. Rainbow Trout Maintain Intestinal Transport and Barrier Functions Following Exposure to Polystyrene Microplastics. Environ. Sci. Technol. 2018, 52, 14392–14401. [Google Scholar] [CrossRef]
- Ašmonaitė, G.; Larsson, K.; Undeland, I.; Sturve, J.; Carney Almroth, B. Size matters: Ingestion of relatively large microplastics contaminated with environmental pollutants posed little risk for fish health and fillet quality. Environ. Sci. Technol. 2018, 52, 14381–14391. [Google Scholar] [CrossRef]
- Jacob, H.; Gilson, A.; Lanctôt, C.; Besson, M.; Metian, M.; Lecchini, D. No effect of polystyrene microplastics on foraging activity and survival in a post-larvae coral-reef fish, Acanthurus triostegus. Bull. Environ. Contam. Toxicol. 2019, 102, 457–461. [Google Scholar] [CrossRef]
- Mattsson, K.; Johnson, E.V.; Malmendal, A.; Linse, S.; Hansson, L.A.; Cedervall, T. Brain damage and behavioural disorders in fish induced by plastic nanoparticles delivered through the food chain. Sci. Rep. 2017, 7, 11452. [Google Scholar] [CrossRef]
- Chen, Q.; Gundlach, M.; Yang, S.; Jiang, J.; Velki, M.; Yin, D.; Hollert, H. Quantitative investigation of the mechanisms of microplastics and nanoplastics toward zebrafish larvae locomotor activity. Sci. Total Environ. 2017, 584–585, 1022–1031. [Google Scholar] [CrossRef]
- Pitt, J.A.; Kozal, J.S.; Jayasundara, N.; Massarsky, A.; Trevisan, R.; Geitner, N.; Wiesner, M.; Levin, E.D.; Di Giulio, R.T. Uptake, tissue distribution, and toxicity of polystyrene nanoparticles in developing zebrafish (Danio rerio). Aquat. Toxicol. 2018, 194, 185–194. [Google Scholar] [CrossRef]
- Yin, L.; Liu, H.; Cui, H.; Chen, B.; Li, L.; Wu, F. Impacts of polystyrene microplastics on the behavior and metabolism in a marine demersal teleost, black rockfish (Sebastes schlegelii). J. Hazard. Mater. 2019, 380, 120861. [Google Scholar] [CrossRef]
- Mak, C.W.; Yeung, K.C.-F.; Chan, K.M. Acute toxic effects of polyethylene microplastic on adult zebrafish. Ecotoxicol. Environ. Saf. 2019, 182, 109442. [Google Scholar] [CrossRef] [PubMed]
- Pannetier, P.; Morin, B.; Le Bihanic, F.; Dubreil, L.; Clérandeau, C.; Chouvellon, F.; Van Arkel, K.; Danion, M.; Cachot, J. Environmental samples of microplastics induce significant toxic effects in fish larvae. Environ. Int. 2020, 134, 105047. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Xiong, H.; Mi, K.; Xue, W.; Wei, W.; Zhang, Y. Toxicity comparison of nano-sized and micron-sized microplastics to Goldfish Carassius auratus Larvae. J. Hazard. Mater. 2020, 388, 122058. [Google Scholar] [CrossRef] [PubMed]
- Karami, A.; Romano, N.; Galloway, T.; Hamzah, H. Virgin microplastics cause toxicity and modulate the impacts of phenanthrene on biomarker responses in African catfish (Clarias gariepinus). Environ. Res. 2016, 151, 58–70. [Google Scholar] [CrossRef]
- Cong, Y.; Jin, F.; Tian, M.; Wang, J.; Shi, H.; Wang, Y.; Mu, J. Ingestion, egestion and post-exposure effects of polystyrene microspheres on marine medaka (Oryzias melastigma). Chemosphere 2019, 228, 93–100. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Lu, L.; Zheng, M.; Zhang, X.; Tian, H.; Wang, W.; Ru, S. Polystyrene microplastics cause tissue damages, sex-specific reproductive disruption and transgenerational effects in marine medaka (Oryzias melastigma). Environ. Pollut. 2019, 254, 113024. [Google Scholar] [CrossRef]
- Pitt, J.A.; Trevisan, R.; Massarsky, A.; Kozal, J.S.; Levin, E.D.; Di Giulio, R.T. Maternal transfer of nanoplastics to offspring in zebrafish (Danio rerio): A case study with nanopolystyrene. Sci. Total Environ. 2018, 643, 324–334. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, Y.; Deng, Y.; Jiang, W.; Zhao, Y.; Geng, J.; Ding, L.; Ren, H. Uptake and accumulation of polystyrene microplastics in Zebrafish (Danio rerio) and toxic effects in liver. Environ. Sci. Technol. 2016, 50, 4054–4060. [Google Scholar] [CrossRef]
- LeMoine, C.M.R.; Kelleher, B.M.; Lagarde, R.; Northam, C.; Elebute, O.O.; Cassone, B.J. Transcriptional effects of polyethylene microplastics ingestion in developing zebrafish (Danio rerio). Environ. Pollut. 2018, 243, 591–600. [Google Scholar] [CrossRef]
- Ding, J.; Zhang, S.; Razanajatovo, R.M.; Zou, H.; Zhu, W. Accumulation, tissue distribution, and biochemical effects of polystyrene microplastics in the freshwater fish red tilapia (Oreochromis niloticus). Environ. Pollut. 2018, 238, 1–9. [Google Scholar] [CrossRef]
- Wan, Z.; Wang, C.; Zhou, J.; Shen, M.; Wang, X.; Fu, Z.; Jin, Y. Effects of polystyrene microplastics on the composition of the microbiome and metabolism in larval zebrafish. Chemosphere 2019, 217, 646–658. [Google Scholar] [CrossRef] [PubMed]
- Qiao, R.; Sheng, C.; Lu, Y.; Zhang, Y.; Ren, H.; Lemos, B. Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Sci. Total Environ. 2019, 662, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Wu, S.; Lu, S.; Liu, M.; Song, Y.; Fu, Z.; Shi, H.; Raley-Susman, K.M.; He, D. Microplastic particles cause intestinal damage and other adverse effects in zebrafish Danio rerio and nematode Caenorhabditis elegans. Sci. Total Environ. 2018, 619–620, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Sun, M.; Zhou, M.; Chang, Z.; Li, L. Polyvinyl chloride microplastics induce growth inhibition and oxidative stress in Cyprinus carpio var. larvae. Sci. Total Environ. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hu, C.; Lok-Shun Lai, N.; Zhang, W.; Hua, J.; Lam, P.K.S.; Lam, J.C.W.; Zhou, B. Acute exposure to PBDEs at an environmentally realistic concentration causes abrupt changes in the gut microbiota and host health of zebrafish. Environ. Pollut. 2018, 240, 17–26. [Google Scholar] [CrossRef]
- Brun, N.R.; van Hage, P.; Hunting, E.R.; Haramis, A.P.G.; Vink, S.C.; Vijver, M.G.; Schaaf, M.J.M.; Tudorache, C. Polystyrene nanoplastics disrupt glucose metabolism and cortisol levels with a possible link to behavioural changes in larval zebrafish. Commun. Biol. 2019, 2, 382. [Google Scholar] [CrossRef]
- Greven, A.C.; Merk, T.; Karagöz, F.; Mohr, K.; Klapper, M.; Jovanović, B.; Palić, D. Polycarbonate and polystyrene nanoplastic particles act as stressors to the innate immune system of fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2016, 35, 3093–3100. [Google Scholar] [CrossRef]
- Espinosa, C.; García Beltrán, J.M.; Esteban, M.A.; Cuesta, A. In vitro effects of virgin microplastics on fish head-kidney leucocyte activities. Environ. Pollut. 2018, 235, 30–38. [Google Scholar] [CrossRef]
- Lu, L.; Wan, Z.; Luo, T.; Fu, Z.; Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 2018, 631–632, 449–458. [Google Scholar] [CrossRef]
- Limonta, G.; Mancia, A.; Benkhalqui, A.; Bertolucci, C.; Abelli, L.; Fossi, M.C.; Panti, C. Microplastics induce transcriptional changes, immune response and behavioral alterations in adult zebrafish. Sci. Rep. 2019, 9, 15775. [Google Scholar] [CrossRef]
- Choi, J.S.; Hong, S.H.; Park, J.W. Evaluation of microplastic toxicity in accordance with different sizes and exposure times in the marine copepod Tigriopus japonicus. Mar. Environ. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Chernick, M.; Rittschof, D.; Hinton, D.E. Chronic dietary exposure to polystyrene microplastics in maturing Japanese medaka (Oryzias latipes). Aquat. Toxicol. 2019, 220, 105396. [Google Scholar] [CrossRef] [PubMed]
- Malafaia, G.; de Souza, A.M.; Pereira, A.C.; Gonçalves, S.; da Costa Araújo, A.P.; Ribeiro, R.X.; Rocha, T.L. Developmental toxicity in zebrafish exposed to polyethylene microplastics under static and semi-static aquatic systems. Sci. Total Environ. 2020, 700, 134867. [Google Scholar] [CrossRef] [PubMed]
- Barboza, L.G.A.; Lopes, C.; Oliveira, P.; Bessa, F.; Otero, V.; Henriques, B.; Raimundo, J.; Caetano, M.; Vale, C.; Guilhermino, L. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 2019. [Google Scholar] [CrossRef]
- Pannetier, P.; Cachot, J.; Clérandeau, C.; Faure, F.; Van Arkel, K.; de Alencastro, L.F.; Levasseur, C.; Sciacca, F.; Bourgeois, J.P.; Morin, B. Toxicity assessment of pollutants sorbed on environmental sample microplastics collected on beaches: Part I-adverse effects on fish cell line. Environ. Pollut. 2019. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Martins, M.A.; Soares, A.M.V.; Cuesta, A.; Oliveira, M. Polystyrene nanoplastics alter the cytotoxicity of human pharmaceuticals on marine fish cell lines. Environ. Toxicol. Pharmacol. 2019, 69, 57–65. [Google Scholar] [CrossRef]
- Rainieri, S.; Conlledo, N.; Larsen, B.K.; Granby, K.; Barranco, A. Combined effects of microplastics and chemical contaminants on the organ toxicity of zebrafish (Danio rerio). Environ. Res. 2018, 162, 135–143. [Google Scholar] [CrossRef]
- Lee, W.S.; Cho, H.J.; Kim, E.; Huh, Y.H.; Kim, H.J.; Kim, B.; Kang, T.; Lee, J.S.; Jeong, J. Bioaccumulation of polystyrene nanoplastics and their effect on the toxicity of Au ions in zebrafish embryos. Nanoscale 2019, 11, 3173–3185. [Google Scholar] [CrossRef]
- Zhang, S.; Ding, J.; Razanajatovo, R.M.; Jiang, H.; Zou, H.; Zhu, W. Interactive effects of polystyrene microplastics and roxithromycin on bioaccumulation and biochemical status in the freshwater fish red tilapia (Oreochromis niloticus). Sci. Total Environ. 2019, 648, 1431–1439. [Google Scholar] [CrossRef]
- Trevisan, R.; Voy, C.; Chen, S.; Di Giulio, R.T. Nanoplastics Decrease the Toxicity of a Complex PAH Mixture but Impair Mitochondrial Energy Production in Developing Zebrafish. Environ. Sci. Technol. 2019, 53, 8405–8415. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, Y.; Lemos, B.; Ren, H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci. Rep. 2017, 7, 46687. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, Y.; Qiao, R.; Bonilla, M.M.; Yang, X.; Ren, H.; Lemos, B. Evidence that microplastics aggravate the toxicity of organophosphorus flame retardants in mice (Mus musculus). J. Hazard. Mater. 2018, 357, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Lu, L.; Tu, W.; Luo, T.; Fu, Z. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Sci. Total Environ. 2018, 649, 308–317. [Google Scholar] [CrossRef]
- Yang, Y.F.; Chen, C.Y.; Lu, T.H.; Liao, C.M. Toxicity-based toxicokinetic/toxicodynamic assessment for bioaccumulation of polystyrene microplastics in mice. J. Hazard. Mater. 2019, 366, 703–713. [Google Scholar] [CrossRef]
- Luo, T.; Wang, C.; Pan, Z.; Jin, C.; Fu, Z.; Jin, Y. Maternal polystyrene microplastic exposure during gestation and lactation altered metabolic homeostasis in the dams and their F1 and F2 offspring. Environ. Sci. Technol. 2019, 53, 10978–10992. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Ding, Y.; Cheng, X.; Sheng, D.; Xu, Z.; Rong, Q.; Wu, Y.; Zhao, H.; Ji, X.; Zhang, Y. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 2019, 244, 125492. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Zhang, Y.; Wang, C.; Wang, X.; Zhou, J.; Shen, M.; Zhao, Y.; Fu, Z.; Jin, Y. Maternal exposure to different sizes of polystyrene microplastics during gestation causes metabolic disorders in their offspring. Environ. Pollut. 2019, 255, 113122. [Google Scholar] [CrossRef]
- Stock, V.; Böhmert, L.; Lisicki, E.; Block, R.; Cara-Carmona, J.; Pack, L.K.; Selb, R.; Lichtenstein, D.; Voss, L.; Henderson, C.J.; et al. Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch. Toxicol. 2019, 93, 1817–1833. [Google Scholar] [CrossRef]
- Rafiee, M.; Dargahi, L.; Eslami, A.; Beirami, E.; Jahangiri-Rad, M.; Sabour, S.; Amereh, F. Neurobehavioral assessment of rats exposed to pristine polystyrene nanoplastics upon oral exposure. Chemosphere 2018, 193, 745–753. [Google Scholar] [CrossRef]
- Walczak, A.P.; Kramer, E.; Hendriksen, P.J.M.; Tromp, P.; Helsper, J.P.F.G.; van der Zande, M.; Rietjens, I.M.C.M.; Bouwmeester, H. Translocation of differently sized and charged polystyrene nanoparticles in in vitro intestinal cell models of increasing complexity. Nanotoxicology 2015, 9, 453–461. [Google Scholar] [CrossRef]
- Fiorentino, I.; Gualtieri, R.; Barbato, V.; Mollo, V.; Braun, S.; Angrisani, A.; Turano, M.; Furia, M.; Netti, P.A.; Guarnieri, D.; et al. Energy independent uptake and release of polystyrene nanoparticles in primary mammalian cell cultures. Exp. Cell Res. 2015, 330, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Magrì, D.; Sánchez-Moreno, P.; Caputo, G.; Gatto, F.; Veronesi, M.; Bardi, G.; Catelani, T.; Guarnieri, D.; Athanassiou, A.; Pompa, P.P.; et al. Laser ablation as a versatile tool to mimic polyethylene terephthalate nanoplastic pollutants: Characterization and toxicology assessment. ACS Nano 2018, 12, 7690–7700. [Google Scholar] [CrossRef] [PubMed]
- Hesler, M.; Aengenheister, L.; Ellinger, B.; Drexel, R.; Straskraba, S.; Jost, C.; Wagner, S.; Meier, F.; von Briesen, H.; Büchel, C.; et al. Multi-endpoint toxicological assessment of polystyrene nano- and microparticles in different biological models in vitro. Toxicol Vitro 2019, 61, 104610. [Google Scholar] [CrossRef] [PubMed]
- Prietl, B.; Meindl, C.; Roblegg, E.; Pieber, T.R.; Lanzer, G.; Fröhlich, E. Nano-sized and micro-sized polystyrene particles affect phagocyte function. Cell Biol. Toxicol. 2014, 30, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Schirinzi, G.F.; Pérez-Pomeda, I.; Sanchís, J.; Rossini, C.; Farré, M.; Barceló, D. Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ. Res. 2017, 159, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Wu, X.; Liu, S.; Wang, Z.; Chen, L. Size-dependent effects of polystyrene microplastics on cytotoxicity and efflux pump inhibition in human Caco-2 cells. Chemosphere 2019, 221, 333–341. [Google Scholar] [CrossRef]
- Hwang, J.; Choi, D.; Han, S.; Choi, J.; Hong, J. An assessment of the toxicity of polypropylene microplastics in human derived cells. Sci. Total Environ. 2019, 684, 657–669. [Google Scholar] [CrossRef]
- Poma, A.; Vecchiotti, G.; Colafarina, S.; Zarivi, O.; Aloisi, M.; Arrizza, L.; Chichiriccò, G.; Di Carlo, P. In vitro genotoxicity of polystyrene nanoparticles on the human fibroblast Hs27 cell line. ACS Appl. Nanomater 2019, 9, 1299. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.W.; Chen, Y.C.; Chen, H.H.; Lee, J.S.; Lin, C.H. Polystyrene microplastic particles: In vitro pulmonary toxicity assessment. J. Hazard. Mater. 2020, 385, 121575. [Google Scholar] [CrossRef]
- Xu, M.; Halimu, G.; Zhang, Q.; Song, Y.; Fu, X.; Li, Y.; Li, Y.; Zhang, H. Internalization and toxicity: A preliminary study of effects of nanoplastic particles on human lung epithelial cell. Sci. Total Environ. 2019, 694, 133794. [Google Scholar] [CrossRef]
- Lim, S.L.; Ng, C.T.; Zou, L.; Lu, Y.; Chen, J.; Bay, B.H.; Shen, H.M.; Ong, C.N. Targeted metabolomics reveals differential biological effects of nanoplastics and nanoZnO in human lung cells. Nanotoxicology 2019, 13, 1117–1132. [Google Scholar] [CrossRef] [PubMed]
- Oh, N.; Park, J.H. Endocytosis and exocytosis of nanoparticles in mammalian cells. Int. J. Nanomed. 2014, 9, 51–63. [Google Scholar]
- Zhang, S.; Gao, H.; Bao, G. Physical principles of nanoparticle cellular endocytosis. ACS Nano 2015, 9, 8655–8671. [Google Scholar] [CrossRef] [PubMed]
- Horstmann, H.; Ng, C.P.; Tang, B.L.; Hong, W. Ultrastructural characterization of endoplasmic reticulum--Golgi transport containers (EGTC). J. Cell Sci. 2002, 115, 4263–4273. [Google Scholar] [CrossRef] [PubMed]
- Treyer, A.; Pujato, M.; Pechuan, X.; Müsch, A. Iterative sorting of apical and basolateral cargo in Madin-Darby canine kidney cells. Mol. Biol. Cell. 2016, 27, 2259–2271. [Google Scholar] [CrossRef]
- Cordani, M.; Somoza, Á. Targeting autophagy using metallic nanoparticles: A promising strategy for cancer treatment. Cell Mol. Life Sci. 2019, 76, 1215–1242. [Google Scholar] [CrossRef]
- Besseling, E.; Wang, B.; Lürling, M.; Koelmans, A.A. Nanoplastic affects growth of S. obliquus and reproduction of D. magna. Environ. Sci. Technol. 2014, 48, 12336–12343. [Google Scholar] [CrossRef]
- Liu, Z.; Cai, M.; Yu, P.; Chen, M.; Wu, D.; Zhang, M.; Zhao, Y. Age-dependent survival, stress defense, and AMPK in Daphnia pulex after short-term exposure to a polystyrene nanoplastic. Aquat. Toxicol. 2018, 204, 1–8. [Google Scholar] [CrossRef]
- Jeong, J.; Choi, J. Adverse outcome pathways potentially related to hazard identification of microplastics based on toxicity mechanisms. Chemosphere 2019, 231, 249–255. [Google Scholar] [CrossRef]
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef]
- Riera Romo, M.; Pérez-Martínez, D.; Castillo Ferrer, C. Innate immunity in vertebrates: An overview. Immunology 2016, 148, 125–139. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, G.; Ferrari, S.; Cervone, F.; Okun, E. Extracellular DAMPs in plants and mammals: Immunity, tissue damage and repair. Trends Immunol. 2018, 39, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Gong, T.; Liu, L.; Jiang, W.; Zhou, R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Kreisel, D.; Goldstein, D.R. Processes of sterile inflammation. J. Immunol. 2013, 191, 2857–2863. [Google Scholar] [CrossRef] [PubMed]
- Plata, C.; Cruz, C.; Cervantes, L.G.; Ramírez, V. The gut microbiota and its relationship with chronic kidney disease. Int. Urol. Nephrol. 2019. [Google Scholar] [CrossRef]
- Jin, M.; Qian, Z.; Yin, J.; Xu, W.; Zhou, X. The role of intestinal microbiota in cardiovascular disease. J. Cell Mol. Med. 2019, 23, 2343–2350. [Google Scholar] [CrossRef]
- Francescone, R.; Hou, V.; Grivennikov, S.I. Microbiome, inflammation, and cancer. Cancer J. 2014, 20, 181–189. [Google Scholar] [CrossRef]
- Ma, Q.; Xing, C.; Long, W.; Wang, H.Y.; Liu, Q.; Wang, R.F. Impact of microbiota on central nervous system and neurological diseases: The gut-brain axis. J. Neuroinflamm. 2019, 16, 53. [Google Scholar] [CrossRef]
- Mohr, K.; Sommer, M.; Baier, G.; Schöttler, S.; Okwieka, P.; Tenzer, S.; Landfester, K.; Mailänder, V.; Schmidt, M.; Meyer, R. Aggregation behavior of polystyrene-nanoparticles in human blood serum and its impact on the in vivo distribution in mice. J. Nanomed. Nanotechnol. 2014, 5. [Google Scholar] [CrossRef]
- Gopinath, P.M.; Saranya, V.; Vijayakumar, S.; Mythili Meera, M.; Ruprekha, S.; Kunal, R.; Pranay, A.; Thomas, J.; Mukherjee, A.; Chandrasekaran, N. Assessment on interactive prospectives of nanoplastics with plasma proteins and the toxicological impacts of virgin, coronated and environmentally released-nanoplastics. Sci. Rep. 2019, 9, 8860. [Google Scholar] [CrossRef]
- Pan, D.; Vargas-Morales, O.; Zern, B.; Anselmo, A.C.; Gupta, V.; Zakrewsky, M.; Mitragotri, S.; Muzykantov, V. The effect of polymeric nanoparticles on biocompatibility of carrier red blood cells. PLoS ONE 2016, 11, e0152074. [Google Scholar] [CrossRef] [PubMed]
| Fishes | Properties of MPs/NPs Used | Tissue Accumulation/ Invasion or Cellular Uptake | Notes on Toxicological, Pathological, or Behavioral Observations | References |
|---|---|---|---|---|
| Crucian Carp (Carassius carassius) | 24 and 27 nm polystyrene (PS) nanoparticles (NPs) (to fish through an aquatic food chain, from algae through Daphnia) | Trophic transfer to fish from algae through Daphnia | • Defects in feeding and shoaling behavior • Defects in metabolism • Changes in brain appearance and weight | Mattsson et al., 2015 [35] |
| Zebrafish (Danio rerio) | Virgin PS microplastic beads (5 µm) + cadmium (Cd) | • Increased Cd accumulation in livers, guts, and gills • Enhanced Cd toxicity • combined exposure caused oxidative damage and inflammation in tissues | Lu et al., 2018 [90] | |
| European seabass (Dicentrarchus labrax) | Fluorescence red polymer microspheres, (1–5 μm) and mercury individually and in combination | • Inhibition of brain acetylcholinesterase (AChE) activity and increase lipid oxidation in brain and muscle • Changes in activity of metabolic enzymes • Interactions and influences on mercury bioaccumulation | Barboza et al., 2018 [49] | |
| Crucian Carp (Carassius carassius) | Amino-modified positively charged PS nanoparticles (52 nm) | Trophic transfer to fish from algae through Daphnia. Nanoparticles found in fish brain | • Changes in feeding time • Changes in brain morphology (gyri sizes) | Mattsson et al., 2017 [68] |
| Zebrafish (Danio rerio) | PS NPs (50 nm, 1 mg/L) | Accumulation in zebrafish larvae | • Inhibited of larvae locomotion • Inhibited acetylcholinesterase activity • Upregulation of cytoskeletal markers | Chen et al., 2017 [69] |
| African catfish (Clarias gariepinus) | Virgin (50 or 500 µg/L) or phenanthrene-loaded (10 or 100 µg/L) low-density polyethylene (LDPE) fragments | • Liver and gill histopathology • Changes in blood biochemistry • Changes in the expression of reproductive axis genes | Karami et al., 2016 [75] | |
| Medaka (Oryzias melastigma) | PS microspheres (10–11 μm, 0.758 ± 0.217 × 105 particles/L) | Microplastics observed in observed in digestive tracts of larvae and dissected intestine of adults | • Increased mortality and decrease in average lengths and weights of larvae and adult fishes • Significant decrease in egg production by females | Cong et al., 2019 [76] |
| Zebrafish (Danio rerio) | PS NPs (mean 51 nm) | Uptake of the nanoparticles by embryos and larvae. Migrated to the gastrointestinal tract, gallbladder, liver, pancreas, heart, and brain throughout development | • Decreased heart rate • Altered larval behavior (swimming hypoactivity in exposed larvae) • Maternal-offspring transfer of PS nanoparticles • Delay/defect in swim bladder inflation by exposed F1 larvae | Pitt et al., 2018 [70,78] |
| Zebrafish (Danio rerio) | PS microspheres (70 nm, 5 μm, and 20 μm, 20 mg/L) | Accumulation in gills, gut, and liver (only the 5 μm particles) | • Liver histopathology (signs of inflammation and lipid accumulation) • Elevation of anti-oxidative stress enzymes • Changes in liver metabolomics profile | Lu et al., 2016 [79] |
| Zebrafish (Danio rerio) | PS MPs (10–45 µm, 20 mg/L) | Ingested microplastics observed in larvae gut | • Significant changes in transcriptome of zebrafish larvae after 2 days exposure • Downregulation of genes involved with neural development and function • Changes in genes associated with metabolism | LeMoine et al., 2018 [80] |
| Red tilapia (Oreochromis niloticus) | PS NPs (0.1 µm, at 1, 10, and 100 μg/L) | PS MPs found in gut and gills and to a lesser extent, liver and brain | • Inhibition of brain acetylcholinesterase (AChE) activity • Changes in liver enzyme markers | Ding et al., 2018 [81] |
| Zebrafish (Danio rerio) | Fluorescent and virgin PS MPs (5 and 50 µm) | Ingested microplastics observed in gut of larvae | • Changes in larval gut microbiota • Metabolomic alterations • Changes in the expression of genes associated with glucose and lipid metabolism • Significant reduction in the antioxidant GSH and the enzyme catalase | Wan et al., 2019 [82] |
| Zebrafish (Danio rerio) | PS microplastic beads (5-μm beads; 50 μg/L and 500 μg/L) | Accumulation of microplastics in zebrafish gut | • Induction of inflammation and oxidative stress of adult zebrafish gut • Significant alterations in the metabolome and microbiome of adult zebrafish gut. Alterations were associated with oxidative stress, inflammation, and lipid metabolism | Qiao et al., 2019 [83] |
| Zebrafish (Danio rerio) | Polyamides (PA), polyethylene (PE), polypropylene (PP), polyvinyl chloride (PVC) (~70 µm) and PS (0.1, 1, and 5 µm) particles | • Intestinal damage of adult fish gut | Lei et al., 2018 [84] | |
| Fathead minnow (Pimephales promelas) | PS (41.0 nm) and polycarbonate (PC) (158.7 nm) NPs | Neutrophil phagocytosis of PS nanoparticles. | • significant increases in innate immune response (in terms of degranulation of primary granules and neutrophil extracellular trap release) | Greven et al., 2016 [88] |
| Gilthead seabream (Sparus aurata) and European sea bass (Dicentrarchus labrax) | Virgin polyvinylchloride (PVC) and polyethylene (PE) (40–150 μm) | • Increased oxidative burst of in leukocytes of Sparus aurata • Upregulation of the redox regulator Nrf2 in leukocytes of Sparus aurata | Espinosa et al., 2018 [89] | |
| Carp (Cyprinus carpio) | MPs from a face and body scrub, mainly PE (250 and 500 μg/L), alone and + Cd | • Changes in plasma levels of various metabolic enzymes and immune markers • Combination of MP and Cd increased Cd toxicity | Banaee et al., 2019 [47] | |
| Black rockfish (Sebastes schlegelii) | PS MP/NPs (0.5 and 15 μm at 190 μg/L) | • Changes in behavior, including reduction in fish swimming speed and range of movement • Increased oxygen consumption and ammonia excretion, reduction of growth and energy reserve, with microparticles having greater effect than nanoparticles | Yin et al., 2019 [71] | |
| Zebrafish (Danio rerio) | PE MPs (10–600 μm at 2 mg/L) | MPs accumulation in gill and intestine | • Abnormal behaviors, including erratic movement, seizures, and morphological changes associated with MP feeding of adult fishes • Upregulation of intestinal Cytochrome P450 gene (cyp 1a) and liver vitellogenin 1 | Mak et al., 2019 [72] |
| Medaka (Oryzias melastigma) | PS nanoparticles (10 μm at 2–200 μg/L) | MPs accumulation in gill, intestine, and liver | • Oxidative stress and structural damages in tissues with MP accumulation • Reproductive endocrine disruption in a sex-dependent manner. • Prenatal exposure to MPs affected the early development of offspring | Wang et al., 2019 [77] |
| Zebrafish (Danio rerio) | PS nanoplastics, 25 nm | NP accumulation in intestine, pancreas, and gallbladder of exposed larvae | • Disruption of glucose homeostasis • Increase cortisol levels and hyperactivity | Brun et al., 2019 [87] |
| Zebrafish (Danio rerio) | PS and PE NPs (with size distribution indicated as 90% < 90 µm; 50% < 50 µm; 10% < 25 µm) | • Alterations in intestinal mucosa and gill epithelium with higher neutrophil infiltration • Changes in the expression of immune system genes, down-regulation of genes correlated with epithelium integrity and lipid metabolism • Changes in daily activity pattern | Limonta et al., 2019 [91] | |
| Japanese Medaka (Tigriopus japonicas) | PS MP/NPs, 50 nm and 10 μm | • Increase in ROS with corresponding changes in GSH and antioxidant enzyme activities | Choi et al., 2019 [92] | |
| Oryzias latipes | PS MPs, 10 μm | MP accumulation in gill and gut | • Dose-dependent decreases in egg number in mature females • Swollen enterocytes and histological alterations of buccal cavity, head kidney, and spleen | Zhu et al., 2019 [93] |
| Zebrafish (Danio rerio) | PE MPs, 38.26 ± 15.64 µm | • MPs induced significant changes in morphometric parameters of larvae • MPs cause lower larval survival rate after egg hatching. | Malafaia et al., 2019 [94] | |
| Japanese Medaka (Tigriopus japonicas) | Environmental MP samples collected from beaches | • Larvae ingestion of MPs decreased viability, decreased head/body ratios, increased Ethoxyresorufin-O-deethylase (EROD) activity, DNA breaks and altered swimming behavior • Juveniles exhibited no symptoms except for increase in DNA breaks | Pannetier et al., 2020 [73] | |
| Goldfish (Carassius auratus) | PS MP/NPs, 70 nm and 5 µm, at 10, 100 and 1000 μg/L | • MP/NPs inhibit fish larvae growth at high levels, increased larvae heart rate and decreased swimming speed • Observations of histopathological changes to intestine, liber and gill, and damages to skin and muscle • MP/NPs elevated oxidative stress markers and related enzymes | Yang et al., 2020 [74] | |
| Carp (Cyprinus carpio) | PVC MPs, ~100–200 μm, at 45.55, 91.1, and 136.65 μg/L | • MPs reduced weight and body length of carp larvae • Histopathological changes in liver • Elevated oxidative stress and related enzyme activities | Xia et al., 2020 [85] | |
| Non-laboratory feeding observations | ||||
| Wild fishes (Dicentrachus labrax, Trachurus trachurus, Scomber colias) sampled from North East Atlantic Ocean | MPs found in 49% of fishes | MPs found in gastrointestinal tract, gills. and dorsal muscle | • Fishes with MP have significantly higher lipid peroxidation levels in the brain, gills. and dorsal muscle and increased brain acetylcholinesterase activity | Barboza et al., 2019 [95] |
| Properties of MPs/NPs Used | Tissue Accumulation/ Invasion or Cellular Uptake | Notes on Toxicological, Pathological, or Behavioral Observations | References |
|---|---|---|---|
| Significant Toxicity/Pathology | |||
| Polystyrene (PS) microspheres 5 μm and 20, 0.01–0.5 mg/day | Accumulation in gut, liver, and kidney | • Signs of inflammation and lipid accumulation in liver • Altered lipid profile and impairment of energy metabolism (reduction in ATP levels) • Increased liver oxidative stress markers, decreased acetylcholinesterase | Deng et al., 2017 [102] |
| PS particles (0.5 and 50 µm) | • Decreased in body, liver, and lipid weights • Decreased mucus secretion in the gut • Alteration in gut microbiota • Changes in hepatic lipid profile and expression of some genes related to lipid metabolism | Lu et al., 2018 [90] | |
| PS and PE beads (0.5–1.0 μm) + organophosphorus flame retardants (OPFRs) | PS and PE beads detectable in gut and liver | • MPs enhanced OPFR-induced oxidative stress, neurotoxicity, and metabolic disorder compared to OFPR alone. | Deng et al., 2018 [103] |
| PS particles (5 µm, 100 and 1000 µg/L) | Accumulation in mouse gut | • Caused intestinal barrier dysfunction • Induced gut microbiota dysbiosis • Induced bile acids metabolism disorder | Jin et al., 2018 [104] |
| PS particles (5 and 20 µm) | Accumulation in mouse gut, liver, and kidney | • Toxicokinetic/toxicodynamic (TBTK/TD) modeling of organ-bioaccumulation and biomarker responses • Changes in oxidative stress markers and those of energy and lipid metabolism | Yang et al., 2019 [105] |
| PS MPs (5 µm) | • Noticeable liver histopathology and altered serum and hepatic markers • Changes in transcript of genes related to glycolipid metabolism • Metabolic disorder associated with gut microbiota dysbiosis and gut barrier dysfunction. • Maternal MPs exposure resulted in intergenerational effects and caused long-term metabolic consequences in the F1 and F2 generations. | Luo et al., 2019 [106] | |
| PS MPs (0.5 and 5 µm) | • MP exposure caused changes in serum and liver metabolic markers • Maternal MPs exposure caused fatty acid metabolic disorder in the F1 offspring | Luo et al., 2019 [108] | |
| PS MPs (10–150 μm) | • MP exposure affected composition and diversity of gut microbiota • increased the secretion of IL-1α in serum, and decreased the Th17 and Treg cells among CD4+ cells • High-concentration of MPs induced inflammation of the small intestine | Li et al., 2019 [107] | |
| No Effect or Insignificant Effect | |||
| PS particles (25 and 50 nm) | • No significantly measurable neurobehavioral consequences in rats | Rafiee et al., 2018 [110] | |
| PS particles (1, 4 and 10 µm) | • No significant effect on body/organ weight and no pathological signs by histological examination • Reporter analyses did not reveal evidence for the occurrence of inflammation and/or oxidative stress • Very low number of particles taken up by intestinal tissue | Stock et al., 2019 [109] | |
| Human Cell Models | Properties of MPs/NPs Used | Cellular Uptake | Notes on Toxicological Observations | References |
|---|---|---|---|---|
| Significant Toxicity | ||||
| Human Peripheral blood monocytic cells (PBMCs) U937 (human monocytic cell line) THP-1 (human monocytic cell line) DMBM-2 (mouse macrophage cell line) | Carboxylated PS NPs (20–1000 nm) | 20 nm nanoparticles taken up passively, while larger ones taken up both actively and passively | • 20 nm NPs cytotoxic to U937 and THP-1 cells • 20 nm NPs stimulated IL-8 secretion in human monocytes and induced measurable oxidative burst in monocytes • 500 and 1000 nm NPs stimulated IL-6 and IL-8 secretion in monocytes and macrophages, chemotaxis and phagocytosis of bacteria by macrophages, and provoked an oxidative burst of granulocytes • At lower concentrations with no cytotoxicity, 20 nm NPs inhibited, while 500 and 1000 nm NPs increased phagocytosis of bacteria by DMBM-2 | Prietl et al., 2014 [115] |
| T98G (human glioblastoma cell line) HeLa (human cervical adenocarcinoma cell line) | PE microparticles (3–16 µm) PS particles (10 µm) | • Induced ROS generation • Cytotoxic effect, with PE having a higher EC50 value compared to PS in both T98G and HeLa cells | Schirinzi et al., 2017 [116] | |
| Caco-2 (human epithelial colorectal adenocarcinoma cell line) | PS particles (0.1 and 5 µm) | Cellular uptake of nanoparticles | • Low toxicity on cell viability, oxidative stress, and membrane integrity and fluidity • Disruption of mitochondrial membrane potential • Inhibition of plasma membrane ATP-binding cassette (ABC) transporter activity | Wu et al., 2019 [117] |
| Human dermal fibroblasts Peripheral blood mononuclear cells (PBMCs) HMC-1 (human mast cell line 1) RBL-2H3 (human basophilic leukemia cell line) RAW 264.7 (mouse macrophage cell line) | PP particles (~20 µm and 25–200 µm), either first dispersed in DMSO or used directly in culture media | • Some degree of cytotoxicity at high dosages of the smaller size 20 µm particles • Low degree of induction of proinflammatory cytokines IL-6 and TNF-α from PBMCs • Increased histamine release from HMC-1 and RBL-2H3 cells • Some degree of ROS induction at high dosages of the smaller size 20 µm particles | Hwang et al., 2019 [118] | |
| BEAS-2B (human lung epithelial cells) | PS MPs (4.06 ± 0.44 µm at 1–1000 μg/cm2 | • Cytotoxic effects • Oxidative stress and inflammatory responses • Disruption of epithelial layer | Dong et al., 2020 [120] | |
| A549 (Human alveolar type II epithelial cell line) | PS nanoparticles (25 and 70 nm) | Cellular uptake of nanoparticles | • Decreased viability and induced cell cycle arrest • Upregulation of transcripts for NF-κB and some pro-inflammatory cytokines • Alteration of cell cycle and apoptosis-regulation related protein expressions | Xu et al., 2019 [121] |
| BEAS-2B (Human bronchial epithelial cells) | PS nanoparticles | Cellular uptake of PS nanopaticles | • PS NPs only cytotoxic at very high concentrations • Metabolomics analyses revealed autophagic and endoplasmic reticulum (ER) stress-related metabolic changes | Lim et al., 2019 [122] |
| Hs27 (Human fibroblasts) | PS nanoparticles (100 nm at 5–75 µg/ml) | • Stimulation of ROS production • Genotoxic stress and DNA damage measured with the cytokinesis-block micronucleus (CBMN) assay | Poma et al., 2019 [119] | |
| No or Insignificant Effects | ||||
| Caco-2 | Polyethylene terephthalate (PET) NPs (laser ablated, ca. 100 nm) | Cellular uptake of NPs | • No apparent toxic effect • Nano-PET are internalized into endo-lysosomal compartments • Nano-PET has high propensity to cross the Caco-2 intestinal barrier model | Magri et al., 2018 [113] |
| Caco-2 THP-1 monocytic line | PS microparticles (1, 4, and 10 µm) | Cellular uptake of PS microparticles | • Low crossing of the cell monolayer on Transwells even by 1 µm microparticles • No pronounce loss of cell viability except only at very high dosage of 1 µm microparticles • Microparticles uptake did not affect macrophage differentiation or polarization | Stock et al., 2019 [109] |
| Caco-2 and HT29-MTX-E12 (human colon epithelial cell) co-culture BeWo b30 (Human placental trophoblast cell) | Carboxy-modified PS nanoparticles (50 nm and 0.5 μm, | Cellular uptake of PS nanoparticles | • No significant cytotoxicity unless at very high concentrations • No significant transport across the in vitro intestinal and placental “barriers” but intercellular distribution was observed | Hesler et al., 2019 [114] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yong, C.Q.Y.; Valiyaveettil, S.; Tang, B.L. Toxicity of Microplastics and Nanoplastics in Mammalian Systems. Int. J. Environ. Res. Public Health 2020, 17, 1509. https://doi.org/10.3390/ijerph17051509
Yong CQY, Valiyaveettil S, Tang BL. Toxicity of Microplastics and Nanoplastics in Mammalian Systems. International Journal of Environmental Research and Public Health. 2020; 17(5):1509. https://doi.org/10.3390/ijerph17051509
Chicago/Turabian StyleYong, Cheryl Qian Ying, Suresh Valiyaveettil, and Bor Luen Tang. 2020. "Toxicity of Microplastics and Nanoplastics in Mammalian Systems" International Journal of Environmental Research and Public Health 17, no. 5: 1509. https://doi.org/10.3390/ijerph17051509
APA StyleYong, C. Q. Y., Valiyaveettil, S., & Tang, B. L. (2020). Toxicity of Microplastics and Nanoplastics in Mammalian Systems. International Journal of Environmental Research and Public Health, 17(5), 1509. https://doi.org/10.3390/ijerph17051509







