Frequent Karaoke Training Improves Frontal Executive Cognitive Skills, Tongue Pressure, and Respiratory Function in Elderly People: Pilot Study from a Randomized Controlled Trial
Abstract
1. Introduction
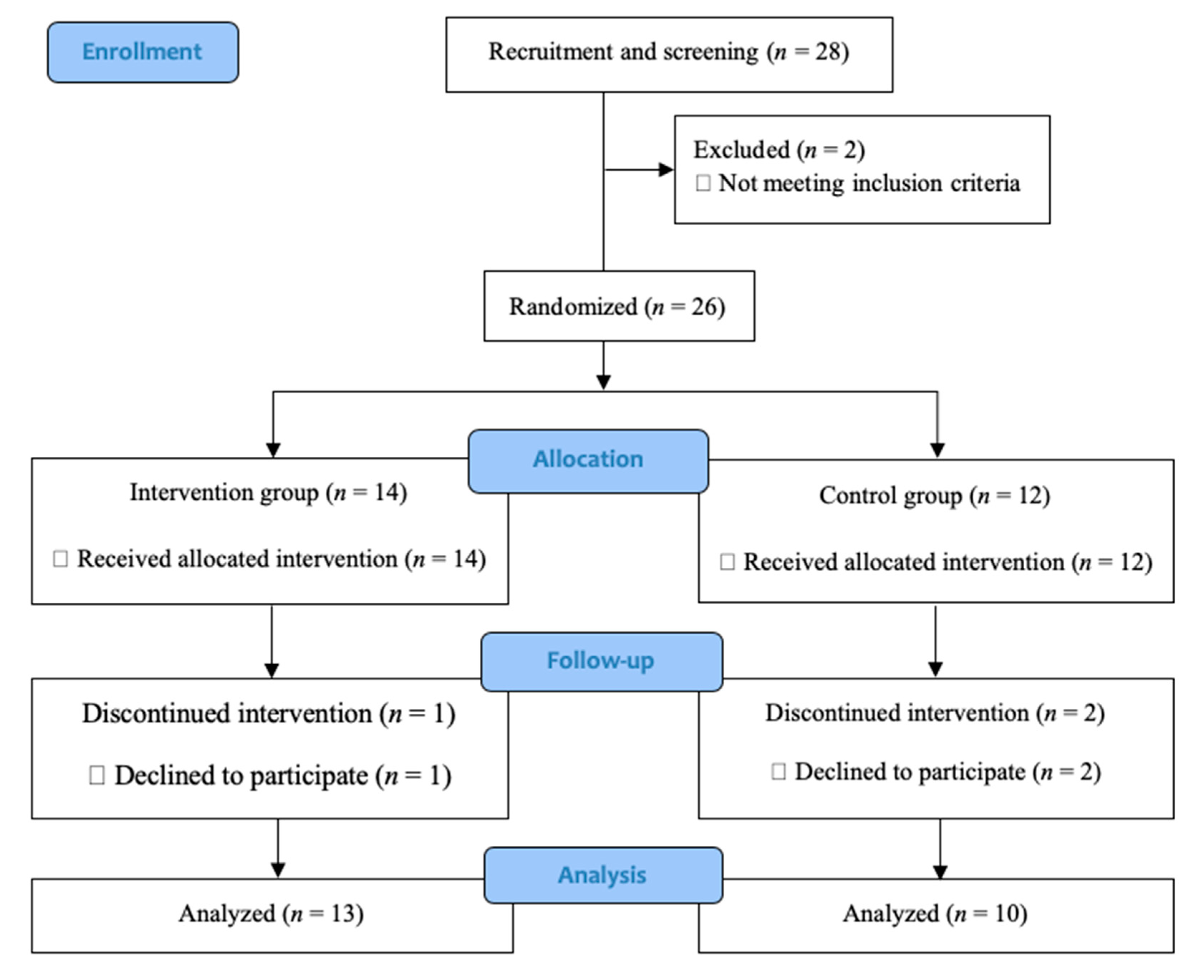
2. Materials and Methods
2.1. Trial Design and Setting
2.2. Participants
2.3. Sample Size
2.4. Overview of Interventions
2.5. Karaoke Training (Intervention Group)
2.6. Scratch Art (Active Control Group)
2.7. Cognitive Function Measures
2.8. Physical Function Measures
2.9. Psychological Measures
2.10. Other and Sarcopenia Measures
2.11. Statistical Analyses
3. Results
3.1. Background Characteristics
3.2. Cognitive Function
3.3. Physical Function
3.4. Psychological Measures
3.5. Summary
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parikh, P.K.; Troyer, A.K.; Maione, A.M.; Murphy, K.J. The impact of memory change on daily life in normal aging and mild cognitive impairment. Gerontologist 2016, 56. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R., Jr.; Knopman, D.S.; Jagust, W.J.; Shaw, L.M.; Aisen, P.S.; Weiner, M.W.; Petersen, R.C.; Trojanowski, J.Q. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 2010, 9, 119–128. [Google Scholar] [CrossRef]
- Mosconi, L.; Tsui, W.H.; Herholz, K.; Pupi, A.; Drzezga, A.; Lucignani, G.; Reiman, E.M.; Holthoff, V.; Kalbe, E.; Sorbi, S.; et al. Multicenter standardized 18F-FDG PET diagnosis of mild cognitive impairment, Alzheimer’s disease, and other dementias. J. Nucl. Med. 2008, 49, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Easterling, C.S.; Robbins, E. Dementia and dysphagia. Geriatr. Nurs. 2008, 29, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Heyn, P.; Abreu, B.C.; Ottenbacher, K.J. The effects of exercise training on elderly persons with cognitive impairment and dementia: A meta-analysis. Arch. Phys. Med. Rehabil. 2004, 85, 1694–1704. [Google Scholar]
- Larson, E.B.; Wang, L.; Bowen, J.D.; McCormick, W.C.; Teri, L.; Crane, P.; Kukull, W. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann. Intern. Med. 2006, 144, 73–81. [Google Scholar] [CrossRef]
- Shumaker, N.L.; Ball, A.L.; Neils-Strunjas, J.; Smith, R.; Weiler, E.; Krikorian, R. Using memory strategies to improve 24-hour dietary recalls among older adults. J. Allied Health 2003, 32, 196–201. [Google Scholar]
- Hohenfeld, C.; Nellessen, N.; Dogan, I.; Kuhn, H.; Müller, C.; Papa, F.; Ketteler, S.; Goebel, R.; Heinecke, A.; Shah, N.J.; et al. Cognitive improvement and brain changes after real-time functional MRI neurofeedback training in healthy elderly and prodromal Alzheimer’s disease. Front. Neurol. 2017, 8, 384. [Google Scholar] [CrossRef]
- Kawashima, R.; Okita, K.; Yamazaki, R.; Tajima, N.; Yoshida, H.; Taira, M.; Iwata, K.; Sasaki, T.; Maeyama, K.; Usui, N.; et al. Reading aloud and arithmetic calculation improve frontal function of people with dementia. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 380–384. [Google Scholar] [CrossRef]
- Moro, V.; Condoleo, M.T.; Valbusa, V.; Broggio, E.; Moretto, G.; Gambina, G. Cognitive stimulation of executive functions in mild cognitive impairment: Specific efficacy and impact in memory. Am. J. Alzheimers Dis. Other Dement. 2015, 30, 153–164. [Google Scholar] [CrossRef]
- Rosen, A.C.; Sugiura, L.; Kramer, J.H.; Whitfield-Gabrieli, S.; Gabrieli, J.D. Cognitive training changes hippocampal function in mild cognitive impairment: A pilot study. J. Alzheimers Dis. 2011, 26, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Nouchi, R.; Taki, Y.; Takeuchi, H.; Nozawa, T.; Sekiguchi, A.; Kawashima, R. Reading aloud and solving simple arithmetic calculation intervention (learning therapy) improves inhibition, verbal episodic memory, focus attention and processing speed in healthy elderly people: Evidence from a randomized controlled trial. Front. Hum. Neurosci. 2016, 10, 217. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S. Kawashima, R. Reading and solving arithmetic problems improves cognitive functions of normal aged people: A randomized controlled study. Age 2008, 30, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Kulason, K.; Nouchi, R.; Hoshikawa, Y.; Noda, M.; Okada, Y.; Kawashima, R. The beneficial effects of cognitive training with Simple Calculation and Reading Aloud (SCRA) in the elderly postoperative population: A pilot randomized controlled trial. Front. Aging Neurosci. 2018, 10, 68. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Sapienza, C.M. Implications of expiratory muscle strength training for rehabilitation of the elderly: Tutorial. J. Rehabil. Res. Dev. 2005, 42, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, I.; Fujiu-Kurachi, M.; Arai, H.; Hyodo, M.; Kagaya, H.; Maeda, K.; Mori, T.; Nishioka, S.; Oshima, F.; Ogawa, S.; et al. Sarcopenia and dysphagia: Position paper by four professional organizations. Geriatr. Gerontol. Int. 2019, 19, 91–97. [Google Scholar] [CrossRef]
- Maeda, K.; Takaki, M.; Akagi, J. Decreased skeletal muscle mass and risk factors of sarcopenic dysphagia: A prospective observational cohort study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 72, 1290–1294. [Google Scholar] [CrossRef]
- Machida, N.; Tohara, H.; Hara, K.; Kumakura, A.; Wakasugi, Y.; Nakane, A.; Minakuchi, S. Effects of aging and sarcopenia on tongue pressure and jaw opening force. Geriatr. Gerontol. Int. 2017, 17, 295–301. [Google Scholar] [CrossRef]
- Nicosia, M.A.; Hind, J.A.; Roecker, E.B.; Carnes, M.; Doyle, J.; Dengel, G.A.; Robbins, J. Age effects on the temporal evolution of isometric and swallowing pressure. J. Gerontol. A. Biol. Sci. Med. Sci. 2000, 55, M634–M640. [Google Scholar] [CrossRef]
- Yoshida, M.; Kikutani, T.; Tsuga, K.; Utanohara, Y.; Hayashi, R.; Akagawa. Decreased tongue pressure reflects symptom of dysphagia. Dysphagia 2006, 21, 61–65. [Google Scholar] [CrossRef]
- Youmans, S.R.; Youmans, G.L.; Stierwalt, J.A. Differences in tongue strength across age and gender: Is there a diminished strength reserve? Dysphagia 2009, 24, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Youmans, S.R.; Stierwalt, J.A. Measures of tongue function related to normal swallowing. Dysphagia 2006, 21, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Minakuchi, S.; Tsuga, K.; Ikebe, K.; Ueda, T.; Tamura, F.; Nagao, K.; Furuya, J.; Matsuo, K.; Yamamoto, K.; Kanazawa, M.; et al. Oral hypofunction in the older population: Position paper of the Japanese Society of Gerodontology in 2016. Gerodontology 2008, 35, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Akagi, J. Decreased tongue pressure is associated with sarcopenia and sarcopenic dysphagia in the elderly. Dysphagia 2015, 30, 80–87. [Google Scholar]
- Buehring, B.; Hind, J.; Fidler, E.; Krueger, D.; Binkley, N.; Robbins, J. Tongue strength is associated with jumping mechanography performance and handgrip strength but not with classic functional tests in older adults. J. Am. Geriatr. Soc. 2013, 61, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Humbert, I.A.; Robbins, J. Dysphagia in the elderly. Phys. Med. Rehabil. Clin. N. Am. 2008, 9, 853–866. [Google Scholar] [CrossRef]
- Robbins, J.; Gangnon, R.E.; Theis, S.M.; Kays, S.A.; Hewitt, A.L.; Hind, J.A. The effects of lingual exercise on swallowing in older adults. J. Am. Geriatr. Soc. 2005, 53, 1483–1489. [Google Scholar] [CrossRef]
- Robbins, J.; Kays, S.A.; Gangnon, R.E.; Hind, J.A.; Hewitt, A.L.; Gentry, L.R.; Taylor, A.J. The effects of lingual exercise in stroke patients with dysphagia. Arch. Phys. Med. Rehabil. 2007, 88, 150–158. [Google Scholar] [CrossRef]
- Steele, C.M.; Bailey, G.L.; Molfenter, S.M.; Yeates, E.M.; Grace-Martin, K. Pressure profile similarities between tongue resistance training tasks and liquid swallows. J. Rehabil. Res. Dev. 2010, 47, 651–660. [Google Scholar] [CrossRef]
- Zaugg, M.; Lucchinetti, E. Respiratory function in the elderly. Anesthesiol. Clin. N. Am. 2000, 18, 47–58. [Google Scholar] [CrossRef]
- Sonies, B.C.; Stone, M.; Shawker, T. Speech and swallowing in the elderly. Gerodontology 1984, 3, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Martin-Harris, B. Clinical implications of respiratory-swallowing interactions. Curr. Opin. Otolaryngol. Head Neck Surg. 2008, 16, 194–199. [Google Scholar]
- Bahat, G.; Tufan, A.; Ozkaya, H.; Tufan, F.; Akpinar, T.S.; Akin, S.; Bahat, Z.; Kaya, Z.; Kiyan, E.; Erten, N.; et al. Relation between hand grip strength, respiratory muscle strength and spirometric measures in male nursing home residents. Aging Male 2014, 17, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.K.; Shin, M.J.; Kim, M.H.; Mok, J.H.; Kim, S.S.; Kim, B.H.; Kim, S.J.; Kim, Y.K.; Chang, J.H.; Shin, Y.B.; et al. Low pulmonary function is related with a high risk of sarcopenia in community-dwelling older adults: The Korea National Health and Nutrition Examination Survey (KNHANES) 2008–2011. Osteoporos. Int. 2015, 26, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Tamplin, J.; Baker, F.A.; Grocke, D.; Brazzale, D.J.; Pretto, J.J.; Ruehland, W.R.; Buttifant, M.; Brown, D.J.; Berlowitz, D.J. Effect of singing on respiratory function, voice, and mood after quadriplegia: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2013, 94, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.; Cave, P.; Stern, M.; Welch, L.; Taylor, K.; Russell, J.; Doyle, A.M.; Russell, A.M.; McKee, H.; Clift, S.; et al. Singing for Lung Health—a systematic review of the literature and consensus statement. NPJ Prim. Care Respir. Med. 2016, 26, 16080. [Google Scholar] [CrossRef] [PubMed]
- Eley, R.; Gorman, D. Didgeridoo playing and singing to support asthma management in Aboriginal Australians. J. Rural Health 2010, 26, 100–104. [Google Scholar] [CrossRef][Green Version]
- Gick, M.L.; Daugherty, C. Changes in spirometry, quality of life and well-being in persons with asthma following singing, diaphragmatic breathing, and singing and diaphragmatic breathing: A pilot study. Music Med. 2015, 7, 40–49. [Google Scholar]
- Skingley, A.; Page, S.; Clift, S.; Morrison, I.; Coulton, S.; Treadwell, P.; Vella-Burrows, T.; Salisbury, I.; Shipton, M. ‘Singing for Breathing’: Participants’ perceptions of a group singing program for people with COPD. Arts Health 2014, 6, 59–74. [Google Scholar] [CrossRef]
- Lord, V.M.; Hume, V.J.; Kelly, J.L.; Cave, P.; Silver, J.; Waldman, M.; White, C.; Smith, C.; Tanner, R.; Sanchez, M.; et al. Singing classes for chronic obstructive pulmonary disease: A randomized controlled trial. BMC Pulm. Med. 2012, 12, 69. [Google Scholar] [CrossRef]
- Nistor, A.R.; Onac, I.; Ştefănescu, A.; Borda, I.M.; Ciortea, V.; Irsay, L.; Mureşan, A.; Ungur, R. The role of singing therapy in pulmonary rehabilitation. PM3. Civiliz. Sport 2015, 16, 158–163. [Google Scholar]
- Satoh, M.; Yuba, T.; Tabei, K.; Okubo, Y.; Kida, H.; Sakuma, H.; Tomimoto, H. Music therapy using singing training improves psychomotor speed in patients with Alzheimer’s disease: A neuropsychological and fMRI Study. Dement. Geriatr. Cognit. Disord. Extra 2015, 5, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Namiki, C.; Hara, K.; Tohara, H.; Kobayashi, K.; Chantaramanee, A.; Nakagawa, K.; Saitou, T.; Yamaguchi, K.; Yoshimi, K.; Nakane, A.; et al. Tongue-pressure resistance training improves tongue and suprahyoid muscle functions simultaneously. Clin. Interv. Aging 2019, 14, 601. [Google Scholar] [CrossRef] [PubMed]
- Bozek, A.; Krajewska, J.; Jarzab, J. The improvement of cognitive functions in patients with bronchial asthma after therapy. J. Asthma 2010, 47, 1148–1152. [Google Scholar] [CrossRef]
- Mori, T.; Yoshikawa, M.; Maruyama, M.; Hiraoka, A.; Nakamori, M.; Yoshida, M.; Tsuga, K. Development of a candy-sucking test for evaluating oral function in elderly patients with dementia: A pilot study. Geriatr. Gerontol. Int. 2017, 17, 1977–1981. [Google Scholar] [CrossRef]
- Sachdev, P.S.; Anstey, K.J.; Parslow, R.A.; Wen, W.; Maller, J.; Kumar, R.; Christensen, H.; Jorm, A.F. Pulmonary function, cognitive impairment and brain atrophy in a middle-aged community sample. Dement. Geriatr. Cognit. Disord. 2006, 21, 300–308. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bedirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; Group, C. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Yamada, T.; Chen, C.C.; Yamada, T.; Fahs, M.; Fukawa, T. Behavioral analysis of the choice of community-based formal home care, informal home care and nursing home care in Japan. Geneva Pap. Risk Insur.-Issues Pract. 2006, 31, 600–632. [Google Scholar] [CrossRef]
- Collin, C.; Wade, D.T.; Davies, S.; Horne, V. The Barthel ADL Index: A reliability study. Int. Disabil. Stud. 1988, 10, 61–63. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Freedland, K.E.; Mohr, D.C.; Davidson, K.W.; Schwartz, J.E. Usual and unusual care: Existing practice control groups in randomized controlled trials of behavioral interventions. Psychosom. Med. 2011, 73, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, L.H.; Knol, D.L.; Hol, E.M.; Swaab, D.F.; Scherder, E.J. Hand motor activity, cognition, mood, and the rest-activity rhythm in dementia: A clustered RCT. Behav. Brain Res. 2009, 196, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.H.; Van Lieshout, R.J.; Schmidt, L.A.; Dobson, K.G.; Buckley, N. Systematic review: Audiovisual interventions for reducing preoperative anxiety in children undergoing elective surgery. J. Pediatr. Psychol. 2016, 41, 182–203. [Google Scholar] [CrossRef] [PubMed]
- Huth, M.M.; Broome, M.E.; Good, M. Imagery reduces children’s post-operative pain. Pain 2004, 110, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Slachevsky, A.; Litvan, I.; Pillon, B. The FAB: A frontal assessment battery at bedside. Neurology 2000, 55, 1621–1626. [Google Scholar] [CrossRef]
- Utanohara, Y.; Hayashi, R.; Yoshikawa, M.; Yoshida, M.; Tsuga, K.; Akagawa, Y. Standard values of maximum tongue pressure taken using newly developed disposable tongue pressure measurement device. Dysphagia 2008, 23, 286–290. [Google Scholar] [CrossRef]
- Brink, T.L.; Yesavage, J.A.; Lum, O.; Heersema, P.H.; Adey, M.; Rose, T.L. Screening tests for geriatric depression. Clin. Gerontol. 1982, 1, 37–43. [Google Scholar] [CrossRef]
- Koyano, W. Correlates of happiness in old age: Covariance structure model analysis of national survey date. Sociol. Theory Meth. 1993, 8, 111–125. (In Japanese) [Google Scholar] [CrossRef]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef]
- Chen, L.K.; Liu, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Bahyah, K.S.; Chou, M.Y.; Chen, L.Y.; Hsu, P.S.; Krairit, O.; et al. Sarcopenia in Asia: Consensus report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, B.; Torchiano, M.; Torchiano, M.M. Package ‘lmPerm’. Available online: ftp://ftp.math.ethz.ch/sfs/pub/Software/R-CRAN/web/packages/lmPerm/lmPerm.pdf (accessed on 22 February 2020).
- Nouchi, R.; Taki, Y.; Takeuchi, H.; Hashizume, H.; Nozawa, T.; Kambara, T.; Sekiguchi, A.; Miyauchi, C.M.; Kotozaki, Y.; Nouchi, H.; et al. Brain training game boosts executive functions, working memory and processing speed in the young adults: A randomized controlled trial. PLoS ONE 2013, 8, e55518. [Google Scholar] [CrossRef] [PubMed]
- Nouchi, R.; Kobayashi, A.; Nouchi, H.; Kawashima, R. Newly developed tv-based cognitive training games improve car driving skills, cognitive functions, and mood in healthy older adults: Evidence from a randomized controlled trial. Front. Aging Neurosci. 2019, 11, 99. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Routledge: Abingdon, UK, 1988. [Google Scholar]
- Moreno, S.; Farzan, F. Music training and inhibitory control: A multidimensional model. Ann. N. Y. Acad. Sci. 2015, 1337, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Schellenberg, E.G. Music lessons enhance IQ. Psychol. Sci. 2004, 15, 511–514. [Google Scholar] [CrossRef]
- The Japanese Neurological Association. Dementia Disease Clinical Practice Guidelines 2017; Igaku-Shoin Ltd.: Tokyo, Japan, 2017. (In Japanese) [Google Scholar]
- Cheng, S.T. Cognitive reserve and the prevention of dementia: The role of physical and cognitive activities. Curr. Psychiatry Rep. 2016, 18, 85. [Google Scholar] [CrossRef]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The physical activity guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Komatsu, R.; Okazaki, T.; Ebihara, S.; Kobayashi, M.; Tsukita, Y.; Nihei, M.; Sugiura, H.; Niu, K.; Ebihara, T.; Ichinose, M. Aspiration pneumonia induces muscle atrophy in the respiratory, skeletal, and swallowing systems. J. Cachexia Sarcopenia Muscle 2018, 9, 643–653. [Google Scholar] [CrossRef]
- Hixon, T.J.; Minifine, F.D.; Williams, F. Normal Aspects of Speech, Hearing, and Language; Prentice-Hall: Englewood Cliffs, NJ, USA, 1973. [Google Scholar]
- Batavia, A.I.; Batavia, M. Karaoke for quads: A new application of an old recreation with potential therapeutic benefits for people with disabilities. Disabil. Rehabil. 2003, 25, 297–300. [Google Scholar] [CrossRef]
- Koike, N.; Ii, S.; Yoshinaga, T.; Nozaki, K.; Wada, S. Model-based inverse estimation for active contraction stresses of tongue muscles using 3D surface shape in speech production. J. Biomech. 2017, 64, 69–76. [Google Scholar] [CrossRef]
- Roussos, C. The Thorax: Disease Lung Biology in Health and Disease; CRC Press: New York, NY, USA, 1995. [Google Scholar]
- Tenório, L.H.S.; Santos, A.C.; Câmara Neto, J.B.; Amaral, F.J.; Passos, V.M.M.; Lima, A.M.J.; Brasileiro-Santos, M.D.S. The influence of inspiratory muscle training on diaphragmatic mobility, pulmonary function and maximum respiratory pressures in morbidly obese individuals: A pilot study. Disabil. Rehabil. 2013, 35, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Murry, T.; Tabaee, A.; Owczarzak, V.; Aviv, J.E. Respiratory retraining therapy and management of laryngopharyngeal reflux in the treatment of patients with cough and paradoxical vocal fold movement disorder. Ann. Otol. Rhinol. Laryngol. 2006, 115, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Akagi, J. Sarcopenia is an independent risk factor of dysphagia in hospitalized older people. Geriatr. Gerontol. Int. 2016, 16, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Butler, S.G.; Stuart, A.; Leng, X.; Wilhelm, E.; Rees, C.; Williamson, J.; Kritchevsky, S.B. The relationship of aspiration status with tongue and handgrip strength in healthy older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2011, 66, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.D.; Choi, J.B.; Yoo, S.J.; Chang, M.Y.; Lee, S.W.; Park, J.S. Tongue to palate resistance training improves tongue strength and oropharyngeal swallowing function in subacute stroke survivors with dysphagia. J. Oral Rehabil. 2017, 44, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Kent, R.D. Research on speech motor control and its disorders: A review and prospective. J. Commun. Disord. 2000, 33, 391–427. [Google Scholar] [CrossRef]
- Nishio, M.; Niimi, S. Changes in speaking fundamental frequency characteristics with aging. Folia Phoniatr. Logop. 2008, 60, 120–127. [Google Scholar] [CrossRef]
- Auyeung, T.W.; Kwok, T.; Lee, J.; Leung, P.C.; Leung, J.; Woo, J. Functional decline in cognitive impairment--the relationship between physical and cognitive function. Neuroepidemiology 2008, 31, 167–173. [Google Scholar] [CrossRef]
- Boyle, P.A.; Buchman, A.S.; Wilson, R.S.; Leurgans, S.E.; Bennett, D.A. Association of muscle strength with the risk of Alzheimer disease and the rate of cognitive decline in community-dwelling older persons. Arch. Neurol. 2009, 66, 1339–1344. [Google Scholar] [CrossRef]
- Loskutova, N.; Honea, R.A.; Vidoni, E.D.; Brooks, W.M.; Burns, J.M. Bone density and brain atrophy in early Alzheimer’s disease. J. Alzheimers Dis. 2009, 18, 777–785. [Google Scholar] [CrossRef]
- Burns, J.M.; Johnson, D.K.; Watts, A.; Swerdlow, R.H.; Brooks, W.M. Reduced lean mass in early Alzheimer disease and its association with brain atrophy. Arch. Neurol. 2010, 67, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Bramell-Risberg, E.; Jarnlo, G.B.; Elmstahl, S. Older women with dementia can perform fast alternating forearm movements and performance is correlated with tests of lower extremity function. Clin. Interv. Aging 2013, 8, 175–184. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tolea, M.I.; Galvin, J.E. Sarcopenia and impairment in cognitive and physical performance. Clin. Interv. Aging 2015, 10, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Tolea, M.I.; Morris, J.C.; Galvin, J.E. Longitudinal associations between physical and cognitive performance among community-dwelling older adults. PLoS ONE 2015, 10, e0122878. [Google Scholar] [CrossRef]
- Chang, K.V.; Hsu, T.H.; Wu, W.T.; Huang, K.C.; Han, D.S. Association between sarcopenia and cognitive Impairment: A systematic review and meta-Analysis. J. Am. Med. Dir. Assoc. 2016, 17, 1164.e7–1164.e15. [Google Scholar] [CrossRef]
- Van Dam, R.; Van Ancum, J.M.; Verlaan, S.; Scheerman, K.; Meskers, C.G.M.; Maier, A.B. Lower cognitive function in older patients with lower muscle strength and muscle mass. Dement. Geriatr. Cognit. Disord. 2018, 45, 243–250. [Google Scholar] [CrossRef]
- Logemann, J.A. The need for clinical trials in dysphagia. Dysphagia 1998, 13, 10–11. [Google Scholar]
- Marik, P.E.; Kaplan, D. Aspiration pneumonia and dysphagia in the elderly. Chest 2003, 124, 328–336. [Google Scholar] [CrossRef]
- Escolar Chua, R.L.; De Guzman, A.B. Effects of third age learning programs on the life satisfaction, self-esteem, and depression level among a select group of community dwelling Filipino elderly. Educ. Gerontol. 2014, 40, 77–90. [Google Scholar] [CrossRef]
- Hanna, G.; Perlstein, S. Creativity Matters: Arts and Aging in America. Available online: https://creativeaging.org/ (accessed on 22 February 2020).
- Hanna, G.P.; Noelker, L.S.; Bienvenu, B. The arts, health, and aging in America: 2005–2015. Gerontologist 2015, 55, 271–277. [Google Scholar] [CrossRef]
- Zeilig, H.; Killick, J.; Fox, C. The participative arts for people living with a dementia: A critical review. Int. J. Ageing Later Life 2014, 9, 7–34. [Google Scholar] [CrossRef]
| Criterion | Intervention Group | Control Group | T-Test p-Value | ||
|---|---|---|---|---|---|
| (n = 14) | (n = 12) | ||||
| Mean (SD) | Range | Mean (SD) | Range | ||
| Age, years | 80.00 (7.46) | 70 to 93 | 83.58 (7.18) | 69 to 93 | 0.23 |
| Sex (% female) | 71.43 | 83.33 | 0.49 | ||
| Education (% with >12 years) | 28.57 | 60.00 | 0.28 | ||
| ADL score | 84.11 (19.75) | 40 to 100 | 94.79 (8.36) | 75 to 100 | 0.09 |
| IADL score | 99.64 (1.34) | 95 to 100 | 96.67 (6.15) | 80 to 100 | 0.09 |
| MoCA score | 24.57 (3.39) | 19 to 30 | 24.25 (5.01) | 12 to 30 | 0.85 |
| FAB total score | 13.14 (3.42) | 6 to 17 | 12.92 (3.23) | 8 to 17 | 0.86 |
| GDS score | 4.36 (2.71) | 0 to 8 | 6.08 (3.12) | 1 to 12 | 0.14 |
| LSI-K score | 5.00 (1.84) | 3 to 8 | 3.83 (1.99) | 1 to 7 | 0.13 |
| Tongue pressure (kPa) | 27.12 (8.08) | 16.70 to 42.25 | 26.45 (6.78) | 13.10 to 34.30 | 0.82 |
| FVC (liter) | 1.85 (0.58) | 0.65 to 2.80 | 1.76 (0.58) | 0.66 to 2.66 | 0.68 |
| FEV1 (liter) | 1.42 (0.48) | 0.56 to 2.27 | 1.30 (0.29) | 0.81 to 1.88 | 0.48 |
| FIV1 (liter) | 0.49 (0.41) | 0.00 to 1.06 | 0.71 (0.46) | 0.00 to 1.25 | 0.20 |
| Body height (cm) | 155.00 (12.60) | 131.0 to 178.0 | 149.90 (8.01) | 140.0 to 169.0 | 0.24 |
| Body weight (kg) | 57.84 (15.81) | 33.6 to 86.7 | 49.54 (8.49) | 33.80 to 61.20 | 0.12 |
| BMI (kg/m2) | 23.73 (4.02) | 16.7 to 30.7 | 22.08 (3.77) | 16.30 to 29.90 | 0.29 |
| SMI (kg/m2) | 6.45 (1.40) | 4.1 to 9.4 | 5.81 (0.81) | 4.4 to 7.4 | 0.18 |
| Intervention Group (n = 13) | Control Group (n = 10) | ANCOVA p-Value | Permutation p-Value | Effect Size (η2) | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||||
| MoCA score | 0.92 | 1.30 | 0.00 | 3.02 | 0.523 | 0.667 | 0.02 |
| FAB total score | 0.77 | 1.97 | –1.80 | 2.44 | 0.011 | 0.013 * | 0.31 |
| GDS score | –0.77 | 2.91 | –2.00 | 3.19 | 0.965 | 1.000 | 0.00 |
| LSI-K score | 0.54 | 1.50 | 0.10 | 2.26 | 0.181 | 0.233 | 0.01 |
| Tongue pressure (kPa) | 3.71 | 5.96 | –1.55 | 3.40 | 0.050 | 0.040 * | 0.22 |
| FVC (liter) | 0.26 | 0.30 | –0.04 | 0.31 | 0.170 | 0.163 | 0.11 |
| FEV1 (liter) | 0.04 | 0.37 | 0.01 | 0.37 | 0.521 | 0.667 | 0.03 |
| FIV1 (liter) | 0.18 | 0.37 | –0.29 | 0.42 | 0.036 | 0.047 * | 0.25 |
| Intervention Group (n = 14) | Control Group (n = 12) | T-Test p-Value | |||
|---|---|---|---|---|---|
| Mean(SD) | Range | Mean(SD | Range | ||
| Similarities (conceptualization) | 1.57 (1.02) | 0 to 3 | 1.17 (0.83) | 0 to 2 | 0.280 |
| Lexical Fluency (mental flexibility) | 2.07 (0.92) | 1 to 3 | 2.50 (0.67) | 1 to 3 | 0.194 |
| Motor Series (programming) | 2.64 (0.63) | 1 to 3 | 2.92 (0.29) | 2 to 3 | 0.181 |
| Conflicting Instructions (sensitivity to interference) | 2.21 (0.80) | 1 to 3 | 1.92 (1.31) | 0 to 3 | 0.485 |
| Go No-Go (inhibitory control) | 1.64 (1.28) | 0 to 3 | 1.42 (1.31) | 0 to 3 | 0.661 |
| Prehension Behavior (environmental autonomy) | 3.00 (0.00) | 3 | 3.00 (0.00) | 3 | 1.000 |
| Intervention Group (n = 13) | Control Group (n = 10) | ANCOVA p-Value | Permutation p-Value | Effect Size (η2) | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||||
| Similarities (conceptualization) | −0.31 | 0.61 | 0.20 | 0.60 | 0.135 | 0.158 | 0.15 |
| Lexical Fluency (mental flexibility) | 0.31 | 0.46 | −0.50 | 1.20 | 0.103 | 0.144 | 0.13 |
| Motor Series (programming) | −0.31 | 0.82 | −1.30 | 0.90 | 0.113 | 0.108 | 0.13 |
| Conflicting Instructions (sensitivity to interference) | 0.46 | 0.84 | −0.30 | 1.10 | 0.003 | 0.001 * | 0.41 |
| Go No-Go (inhibitory control) | 0.69 | 1.07 | 0.10 | 1.30 | 0.013 | 0.015 * | 0.29 |
| Prehension Behavior (environmental autonomy) | −0.08 | 0.27 | 0.00 | 0.00 | 0.469 | 0.824 | 0.06 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyazaki, A.; Mori, H. Frequent Karaoke Training Improves Frontal Executive Cognitive Skills, Tongue Pressure, and Respiratory Function in Elderly People: Pilot Study from a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 1459. https://doi.org/10.3390/ijerph17041459
Miyazaki A, Mori H. Frequent Karaoke Training Improves Frontal Executive Cognitive Skills, Tongue Pressure, and Respiratory Function in Elderly People: Pilot Study from a Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2020; 17(4):1459. https://doi.org/10.3390/ijerph17041459
Chicago/Turabian StyleMiyazaki, Atsuko, and Hayato Mori. 2020. "Frequent Karaoke Training Improves Frontal Executive Cognitive Skills, Tongue Pressure, and Respiratory Function in Elderly People: Pilot Study from a Randomized Controlled Trial" International Journal of Environmental Research and Public Health 17, no. 4: 1459. https://doi.org/10.3390/ijerph17041459
APA StyleMiyazaki, A., & Mori, H. (2020). Frequent Karaoke Training Improves Frontal Executive Cognitive Skills, Tongue Pressure, and Respiratory Function in Elderly People: Pilot Study from a Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 17(4), 1459. https://doi.org/10.3390/ijerph17041459




