Indicators of Genotoxicity in Farmers and Laborers of Ecological and Conventional Banana Plantations in Ecuador
Abstract
1. Introduction
2. Material and Methods
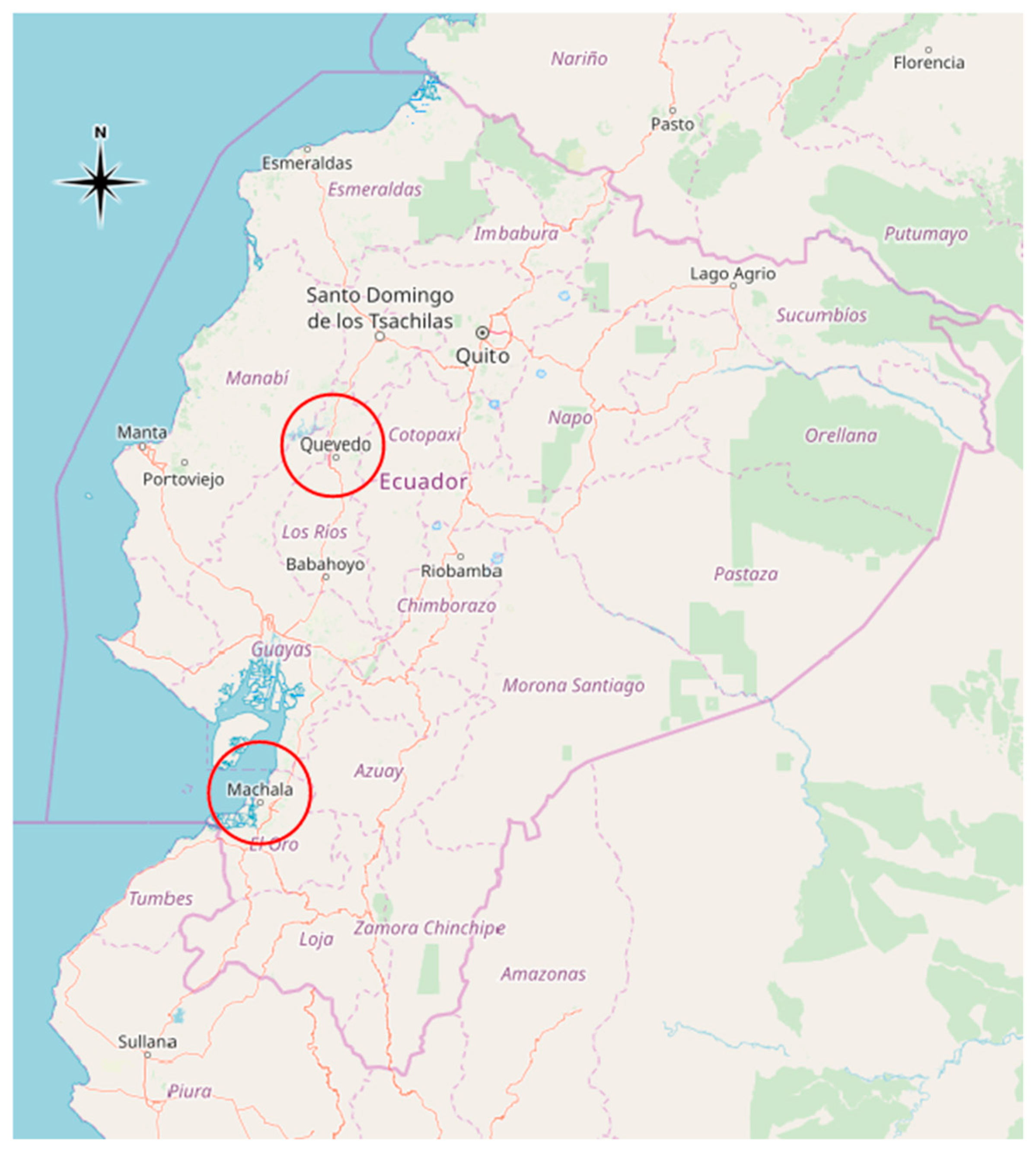
2.1. Study Area and Subjects
2.2. Examination Procedure
2.3. Buccal Cells Micronucleus Assay (BMCA)
2.3.1. Buccal Cells Sampling and Staining
2.3.2. Evaluation of Nuclear Anomalies
2.3.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT 2019. Domain: Production—Crops. Countries: Ecuador; Elements: Production Quantity; Items: Bananas. 2017. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 21 November 2019).
- Brisbois, B. Bananas, pesticides and health in southwestern Ecuador: A scalar narrative approach to targeting public health responses. Soc. Sci. Med. 2016, 150, 184–191. [Google Scholar] [CrossRef]
- Rodríguez, T.; Younglove, L.; Lu, C.; Funez, A.; Weppner, S.; Barr, D.B.; Fenske, R.A. Biological monitoring of pesticide exposures among applicators and their children in Nicaragua. Int. J. Occup. Environ. Health 2006, 12, 312–320. [Google Scholar] [CrossRef]
- Mendez, A.; Castillo, L.E.; Ruepert, C.; Hungerbuehler, K.; Ng, C.A. Tracking pesticide fate in conventional banana cultivation in Costa Rica: A disconnect between protecting ecosystems and consumer health. Sci. Total Environ. 2018, 613–614, 1250–1262. [Google Scholar] [CrossRef]
- Barraza, D.; Jansen, K.; van Wendel de Joode, B.; Wesseling, C. Pesticide use in banana and plantain production and risk perception among local actors in Talamanca, Costa Rica. Environ. Res. 2011, 111, 708–717. [Google Scholar] [CrossRef] [PubMed]
- van Maele-Fabry, G.; Hoet, P.; Vilain, F.; Lison, D. Occupational exposure to pesticides and Parkinson’s disease: A systematic review and meta-analysis of cohort studies. Environ. Int. 2012, 46, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.Z.; Loomis, D.; Grosse, Y.; El Ghissassi, F.; Benbrahim-Tallaa, L.; Guha, N.; Scoccianti, C.; Mattock, H.; Straif, K.; International Agency for Research on Cancer Monograph Working Group, IARC, Lyon, France. Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet Oncol. 2015, 16, 490–491. [Google Scholar] [CrossRef]
- Alavanja, M.C.R.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef] [PubMed]
- Claudio, S.R.; Simas, J.M.M.; Souza, A.C.F.; De Alencar, M.D.C.B.; Yamauchi, L.Y.; Ribeiro, D.A. Genomic Instability and Cytotoxicity in Buccal Mucosal Cells of Workers in Banana Farming Evaluated by Micronucleus Test. Anticancer Res. 2019, 39, 1283–1286. [Google Scholar] [CrossRef] [PubMed]
- Hutter, H.-P.; Khan, A.W.; Lemmerer, K.; Wallner, P.; Kundi, M.; Moshammer, H. Cytotoxic and Genotoxic Effects of Pesticide Exposure in Male Coffee Farmworkers of the Jarabacoa Region, Dominican Republic. Int. J. Environ. Res. Public Health 2018, 15, 1641. [Google Scholar] [CrossRef]
- Hutter, H.-P.; Kundi, M.; Lemmerer, K.; Poteser, M.; Weitensfelder, L.; Wallner, P.; Moshammer, H. Subjective Symptoms of Male Workers Linked to Occupational Pesticide Exposure on Coffee Plantations in the Jarabacoa Region, Dominican Republic. Int. J. Environ. Res. Public Health 2018, 15, 2099. [Google Scholar] [CrossRef]
- Moshammer, H.; Khan, A.W.; Wallner, P.; Poteser, M.; Kundi, M.; Hutter, H.P. Validity of reported indicators of pesticide exposure and relevance for cytotoxic and genotoxic effects on buccal cells. Mutagenesis 2019, 34, 147–152. [Google Scholar] [CrossRef] [PubMed]
- FAOSTAT 2019. Domain: Inputs—Pesticides Use. [Countries: Ecuador; Elements: Agricultural Use; Items: Pesticides (Total); Years: 2017]. Available online: http://www.fao.org/faostat/en/#data/RP (accessed on 21 November 2019).
- Deknock, A.; De Troyer, N.; Houbraken, M.; Dominguez-Granda, L.; Nolivos, I.; Van Echelpoel, W.; Forio, M.A.E.; Spanoghe, P.; Goethals, P. Distribution of agricultural pesticides in the freshwater environment of the Guayas river basin (Ecuador). Sci. Total Environ. 2019, 646, 996–1008. [Google Scholar] [CrossRef] [PubMed]
- Vindas, R.; Ortiz, F.; Ramírez, V.; Cuenca, P. Genotoxicidad de tres plaguicidas utilizados en la actividad bananera de Costa Rica. Rev. Biol. Trop. 2004, 52, 601–609. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thomas, P.; Holland, N.; Bolognesi, C.; Kirsch-Volders, M.; Bonassi, S.; Zeiger, E.; Knasmueller, S.; Fenech, M. Buccal micronucleus cytome assay. Nat. Protoc. 2009, 4, 825–837. [Google Scholar] [CrossRef]
- Tolbert, P.E.; Shy, C.M.; Allen, J.W. Micronuclei and other nuclear anomalies in buccal smears: Methods development. Mutat. Res. 1992, 271, 69–77. [Google Scholar] [CrossRef]
- Biesterfeld, S.; Beckers, S.; Del Carmen Villa Cadenas, M.; Schramm, M. Feulgen staining remains the gold standard for precise DNA image cytometry. Anticancer Res. 2011, 31, 53–58. [Google Scholar]
- Bolognesi, C.; Fenech, M. Micronucleus assay in human cells: Lymphocytes and buccal cells. Methods Mol. Biol. 2013, 1044, 191–207. [Google Scholar] [CrossRef]
- Bloching, M.; Hofmann, A.; Lautenschläger, C.; Berghaus, A.; Grummt, T. Exfoliative cytology of normal buccal mucosa to predict the relative risk of cancer in the upper aerodigestive tract using the MN-assay. Oral Oncol. 2000, 36, 550–555. [Google Scholar] [CrossRef]
- Stich, H.F.; Rosin, M.P. Micronuclei in exfoliated human cells as a tool for studies in cancer risk and cancer intervention. Cancer Lett. 1984, 22, 241–253. [Google Scholar] [CrossRef]
- Cobanoglu, H.; Coskun, M.; Coskun, M.; Çayir, A. Results of buccal micronucleus cytome assay in pesticide-exposed and non-exposed group. Environ. Sci. Pollut. Res. Int. 2019, 26, 19676–19683. [Google Scholar] [CrossRef]
- Arévalo-Jaramillo, P.; Idrobo, A.; Salcedo, L.; Cabrera, A.; Vintimilla, A.; Carrión, M.; Bailon-Moscoso, N. Biochemical and genotoxic effects in women exposed to pesticides in Southern Ecuador. Environ. Sci. Pollut. Res. Int. 2019, 26, 24911–24921. [Google Scholar] [CrossRef] [PubMed]
- Bernieri, T.; Moraes, M.F.; Ardenghi, P.G.; Basso da Silva, L. Assessment of DNA damage and cholinesterase activity in soybean farmers in southern Brazil: High versus low pesticide exposure. J. Environ. Sci. Health B 2019, 23, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Barrón Cuenca, J.; Tirado, N.; Barral, J.; Ali, I.; Levi, M.; Stenius, U.; Berglund, M.; Dreij, K. Increased levels of genotoxic damage in a Bolivian agricultural population exposed to mixtures of pesticides. Sci. Total Environ. 2019, 695, 133942. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.C.; Campos, P.H.N.; Mattos, I.E.; Hajat, S.; Lacerda, E.M.; Ferreira, M.J.M. Environmental Exposure to Pesticides and Breast Cancer in a Region of Intensive Agribusiness Activity in Brazil: A Case-Control Study. Int. J. Environ. Res. Public Health 2019, 16, 3951. [Google Scholar] [CrossRef] [PubMed]
- Polidoro, B.A.; Dahlquist, R.M.; Castillo, L.E.; Morra, M.J.; Somarriba, E.; Bosque-Pérez, N.A. Pesticide application practices, pest knowledge, and cost-benefits of plantain production in the Bribri-Cabécar Indigenous Territories, Costa Rica. Environ. Res. 2008, 108, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Okonya, J.S.; Petsakos, A.; Suarez, V.; Nduwayezu, A.; Kantungeko, D.; Blomme, G.; Legg, J.P.; Kroschel, J. Pesticide Use Practices in Root, Tuber, and Banana Crops by Smallholder Farmers in Rwanda and Burundi. Int. J. Environ. Res. Public Health 2019, 16, 400. [Google Scholar] [CrossRef] [PubMed]
- Torres-Bugarín, O.; Zavala-Cerna, M.G.; Nava, A.; Flores-García, A.; Ramos-Ibarra, M.L. Potential uses, limitations, and basic procedures of micronuclei and nuclear abnormalities in buccal cells. Dis. Markers 2014, 2014, 956835. [Google Scholar] [CrossRef]
- Kummermehr, J. Repopulation in normal tissues. Strahlenther. Onkol. 1988, 164, 657–661. [Google Scholar]
- Maidhof, R.; Hornstein, O.P. Autoradiographic study on some proliferative properties of human buccal mucosa. Arch. Dermatol. Res. 1979, 265, 165–172. [Google Scholar] [CrossRef]
- Brenner, D.E.; Normolle, D.P. Biomarkers for cancer risk, early detection, and prognosis: The validation conundrum. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 1918–1920. [Google Scholar] [CrossRef][Green Version]
- Verissimo, G.; Kos, T.R.; Garcia, T.R.; Ramos, J.A.D.S.; Souza, C.C.; Moreira, J.C.; Meyer, A. Pesticide exposure among students and their families in Nova Friburgo, Rio de Janeiro. Cienc. Saude Coletiva 2018, 23, 3903–3911. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.N.; Banda, V.R. Correlation of pesticide exposure from dietary intake and bio-monitoring: The different sex and socio-economic study of children. Ecotoxicol. Environ. Saf. 2018, 162, 170–177. [Google Scholar] [CrossRef] [PubMed]

| Group | No Education | Compulsory Education | Secondary Education |
|---|---|---|---|
| Conventional farming (n = 34) | 6 (17.6 %) | 22 (64.7 %) | 6 (17.6 %) |
| Organic farming (n = 37) | 1 (2.7 %) | 22 (59.5 %) | 14 (37.8 %) |
| Endpoint | Conventional Farmworkers (n = 31) | Ecological Farmworkers (n = 37) | p-Value 1 | ||
|---|---|---|---|---|---|
| Adj.Mean (95% CI) | Crude Mean (95% CI) | Adj.Mean (95% CI) | Crude Mean (95% CI) | ||
| Total Mni | 4.23 (3.74–4.79) | 4.19 (3.71–4.74) | 1.62 (1.36–1.94) | 1.65 (1.38–1.97) | <0.001 |
| MN | 3.30 (2.87–3.79) | 3.27 (2.85–3.76) | 1.31 (1.07–1.60) | 1.34 (1.10–1.63) | <0.001 |
| BUD | 5.28 (4.72–5.89) | 5.34 (4.79–5.95) | 2.92 (2.56–3.35) | 2.92 (2.55–3.34) | <0.001 |
| BN | 25.00 (23.27–26.86) | 25.03 (23.33–26.86) | 18.55 (17.20–20.01) | 18.54 (17.20–19.98) | <0.001 |
| KR | 45.46 (43.11–47.95) | 46.48 (44.14–48.95) | 35.16 (33.27–37.15) | 35.03 (33.17–36.99) | <0.001 |
| CC | 53.53 (50.97–56.22) | 54.77 (52.23–57.44) | 40.97 (38.93–43.12) | 40.86 (38.86–42.98) | <0.001 |
| KL | 77.26 (74.17–80.47) | 76.35 (73.34–79.49) | 62.31 (59.78–64.93) | 63.30 (60.78–65.91) | <0.001 |
| PY | 4.21 (3.53–5.01) | 4.19 (3.53–4.98) | 3.78 (3.20–4.47) | 3.97 (3.38–4.67) | 0.389 |
| BASAL | 8.35 (7.38–9.46) | 8.48 (7.52–9.57) | 8.14 (7.25–9.12) | 8.08 (7.22–9.05) | 0.764 |
| Exposure Parameter | Unit/Category | Mean (SD) or n (%) |
|---|---|---|
| Pesticide usage | Years | 12.94 (9.47) |
| Last spraying | Days | 36.23 (89.48) |
| Mixing of pesticides | Never | 21 (68 %) |
| Knowing which pesticides are used | No | 14 (45 %) |
| Mask usage | always or more than 50% | 3 (10 %) |
| Glove usage | always or more than 50% | 6 (19 %) |
| Washing hands at site | always or more than 50% | 25 (81 %) |
| Washing hands immediately at home | always or more than 50% | 11 (35 %) |
| Change clothes after spraying | Yes | 12 (39 %) |
| Endpoint | Change Clothes (No)(n = 19) | Change Clothes (yes)(n = 12) | p-Value | ||
|---|---|---|---|---|---|
| Mean | 95% CI | Mean | 95% CI | ||
| Total MNi | 4.40 | 0.35–3.77 | 3.86 | 0.41–3.13 | 0.336 |
| MN | 3.55 | 0.31–2.99 | 2.83 | 0.35–2.22 | 0.148 |
| BUD | 5.67 | 0.39–4.95 | 4.81 | 0.46–3.99 | 0.174 |
| BN | 25.65 | 1.18–23.43 | 23.74 | 1.45–21.07 | 0.321 |
| KR | 48.11 | 1.63–45.02 | 42.90 | 1.94–39.25 | 0.046 |
| CC | 55.78 | 1.75–52.45 | 52.02 | 2.14–47.99 | 0.185 |
| KL | 76.53 | 2.05–72.62 | 75.54 | 2.59–70.64 | 0.770 |
| PY | 3.86 | 0.46–3.06 | 4.20 | 0.61–3.16 | 0.656 |
| BASAL | 8.58 | 0.71–7.29 | 6.22 | 0.73–4.95 | 0.023 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hutter, H.-P.; Poteser, M.; Lemmerer, K.; Wallner, P.; Shahraki Sanavi, S.; Kundi, M.; Moshammer, H.; Weitensfelder, L. Indicators of Genotoxicity in Farmers and Laborers of Ecological and Conventional Banana Plantations in Ecuador. Int. J. Environ. Res. Public Health 2020, 17, 1435. https://doi.org/10.3390/ijerph17041435
Hutter H-P, Poteser M, Lemmerer K, Wallner P, Shahraki Sanavi S, Kundi M, Moshammer H, Weitensfelder L. Indicators of Genotoxicity in Farmers and Laborers of Ecological and Conventional Banana Plantations in Ecuador. International Journal of Environmental Research and Public Health. 2020; 17(4):1435. https://doi.org/10.3390/ijerph17041435
Chicago/Turabian StyleHutter, Hans-Peter, Michael Poteser, Kathrin Lemmerer, Peter Wallner, Shifra Shahraki Sanavi, Michael Kundi, Hanns Moshammer, and Lisbeth Weitensfelder. 2020. "Indicators of Genotoxicity in Farmers and Laborers of Ecological and Conventional Banana Plantations in Ecuador" International Journal of Environmental Research and Public Health 17, no. 4: 1435. https://doi.org/10.3390/ijerph17041435
APA StyleHutter, H.-P., Poteser, M., Lemmerer, K., Wallner, P., Shahraki Sanavi, S., Kundi, M., Moshammer, H., & Weitensfelder, L. (2020). Indicators of Genotoxicity in Farmers and Laborers of Ecological and Conventional Banana Plantations in Ecuador. International Journal of Environmental Research and Public Health, 17(4), 1435. https://doi.org/10.3390/ijerph17041435








