Association between Smoking and Noise-Induced Hearing Loss: A Meta-Analysis of Observational Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Quality Assessment
2.4. Statistical Analysis
3. Results
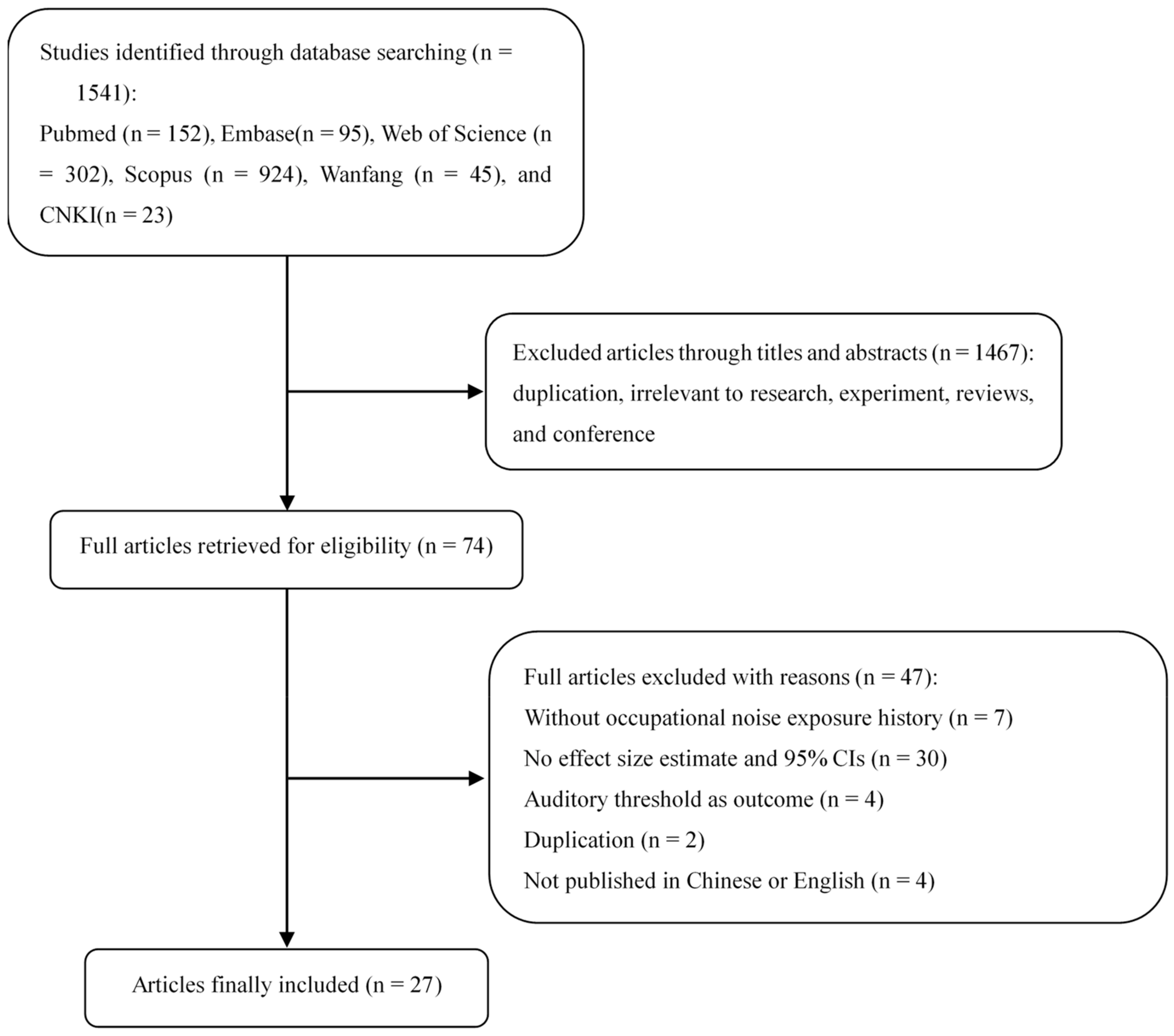
3.1. Literature Search
3.2. Characteristics of the Included Studies
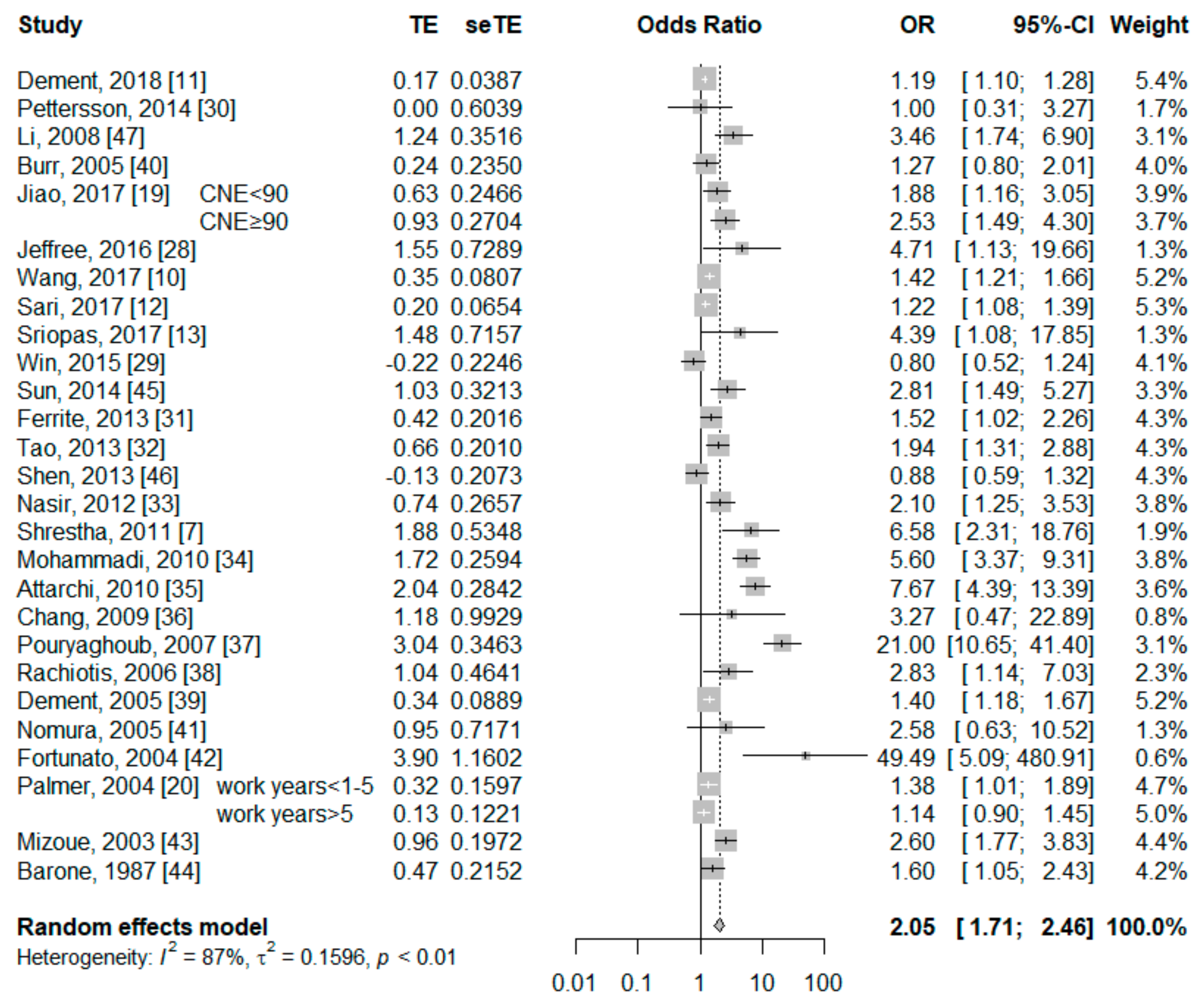
3.3. Association between Current Smokers and Risk of NIHL
3.4. Association between Former Smokers and Risk of NIHL
3.5. Subgroup Analyses
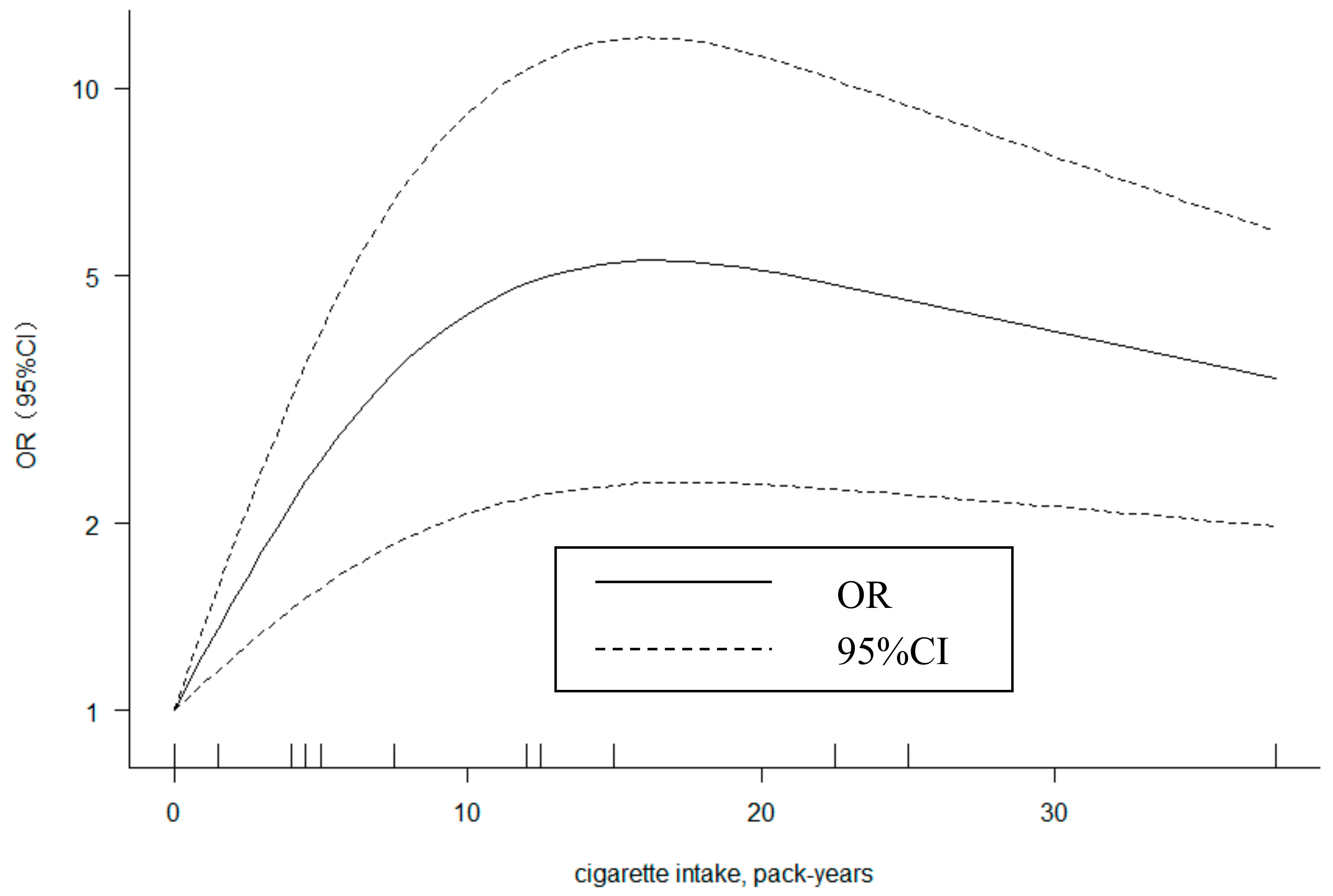
3.6. Dose-Response Analysis
3.7. Sensitivity Analysis and Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Deafness and Hearing Loss. Available online: https://www.who.int/en/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 21 December 2019).
- Nelson, D.I.; Nelson, R.Y.; Concha-Barrientos, M.; Fingerhut, M. The global burden of occupational noise-induced hearing loss. Am. J. Ind. Med. 2005, 48, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Daniel, E. Noise and hearing loss: A review. J. Sch. Health 2007, 77, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Hormozi, M.; Ansari-Moghaddam, A.; Mirzaei, R.; Dehghan Haghighi, J.; Eftekharian, F. The risk of hearing loss associated with occupational exposure to organic solvents mixture with and without concurrent noise exposure: A systematic review and meta-analysis. Int. J. Occup. Med. Environ. Health 2017, 30, 521–535. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.F.; Chen, G.S.; Jiao, J.; Gu, G.Z.; Zhang, H.L.; Wang, X.M.; Zhou, W.H.; Wu, H.; Li, Y.H.; Zheng, Y.X. A cohort study on occupational noise induced hearing loss in workers at an iron and steel plant. Zhonghua Yu Fang Yi Xue Za Zhi 2017, 51, 13–19. (In Chinese) [Google Scholar] [CrossRef]
- Tikka, C.; Verbeek, J.H.; Kateman, E.; Morata, T.C.; Dreschler, W.A.; Ferrite, S. Interventions to prevent occupational noise-induced hearing loss. Cochrane Database Syst. Rev. 2017, 7, CD006396. [Google Scholar] [CrossRef]
- Shrestha, I.; Shrestha, B.L.; Pokharel, M.; Amatya, R.C.; Karki, D.R. Prevalence of noise induced hearing loss among traffic police personnel of Kathmandu Metropolitan City. Kathmandu Univ. Med. J. 2011, 9, 274–278. [Google Scholar] [CrossRef]
- Sliwinska-Kowalska, M.; Pawelczyk, M. Contribution of genetic factors to noise-induced hearing loss: A human studies review. Mutat. Res. 2013, 752, 61–65. [Google Scholar] [CrossRef]
- Torre, P.; Cruickshanks, K.J.; Klein, B.E.K.; Klein, R.; Nondahl, D.M. The association between cardiovascular disease and cochlear function in older adults. J. Speech Lang. Hear. Res. 2005, 48, 473–481. [Google Scholar] [CrossRef]
- Wang, D.M.; Wang, Z.C.; Zhou, M.; Li, W.Z.; He, M.A.; Zhang, X.M.; Guo, H.; Yuan, J.; Zhan, Y.; Zhang, K.; et al. The combined effect of cigarette smoking and occupational noise exposure on hearing loss: Evidence from the Dongfeng-Tongji Cohort Study. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Dement, J.; Welch, L.S.; Ringen, K.; Cranford, K.; Quinn, P. Hearing loss among older construction workers: Updated analyses. Am. J. Ind. Med. 2018, 61, 326–335. [Google Scholar] [CrossRef]
- Sari, M.A.; Adnan, A.; Munir, D.; Eyanoer, P.C. The correlation of smoking and noise induced hearing loss on workers at a palm oil factory X in Medan-Indonesia. Bali Med. J. 2017, 6, 637–640. [Google Scholar] [CrossRef]
- Sriopas, A.; Chapman, R.S.; Sutammasa, S.; Siriwong, W. Occupational noise-induced hearing loss in auto part factory workers in welding units in Thailand. J. Occup. Health 2017, 59, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Tobacco in China. Available online: https://www.who.int/china/health-topics/tobacco (accessed on 21 December 2019).
- Nomura, K.; Nakao, M.; Morimoto, T. Effect of smoking on hearing loss: Quality assessment and meta-analysis. Prev. Med. 2005, 40, 138–144. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. Ann. Intern. Med. 2009, 151, W65–W94. [Google Scholar] [CrossRef]
- Wells, G.; Shea, B.; O’Connell, D. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 2010. [Google Scholar]
- Rostom, A.; Dubé, C.; Cranney, A.; Saloojee, N.; Sy, R.; Garritty, C.; Sampson, M.; Zhang, L.; Yazdi, F.; Mamaladze, V.; et al. Evidence Reports/Technology Assessments; Appendix Quality Assessment; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2004.
- Jiao, J.; Gu, G.Z.; Chen, G.S.; Li, Y.H.; Zhang, H.L.; Yang, Q.Y.; Xu, X.R.; Zhou, W.H.; Wu, H.; He, L.H.; et al. Investigation into the relationship between mitochondrial 12 S rRNA gene, tRNA gene and cytochrome oxidase II gene variations and the risk of noise-induced hearing loss. Zhonghua Yu Fang Yi Xue Za Zhi 2017, 51, 34–40. (In Chinese) [Google Scholar] [PubMed]
- Palmer, K.T.; Griffin, M.J.; Syddall, H.E.; Coggon, D. Cigarette smoking, occupational exposure to noise, and self reported hearing difficulties. Occup. Environ. Med. 2004, 61, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Br. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Greenland, S.; Longnecker, M.P. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301–1309. [Google Scholar] [CrossRef]
- Orsini, N.; Li, R.; Wolk, A.; Khudyakov, P.; Spiegelman, D. Meta-analysis for linear and nonlinear dose-response relations: Examples, an evaluation of approximations, and software. Am. J. Epidemiol. 2012, 175, 66–73. [Google Scholar] [CrossRef]
- Harrell, F.E., Jr.; Lee, K.L.; Pollock, B.G. Regression models in clinical studies: Determining relationships between predictors and response. J. Natl. Cancer Inst. 1988, 80, 1198–1202. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Jeffree, M.S.; Ismail, N.; Lukman, K.A. Hearing impairment and contributing factors among fertilizer factory workers. J. Occup. Health 2016, 58, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Win, K.N.; Balalla, N.B.P.; Lwin, M.Z.; Lai, A. Noise-Induced Hearing Loss in the Police Force. Saf. Health Work 2015, 6, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, H.; Burstrom, L.; Hagberg, M.; Lundstrom, R.; Nilsson, T. Risk of Hearing Loss Among Workers with Vibration-Induced White Fingers. Am. J. Ind. Med. 2014, 57, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Ferrite, S.; Santana, V.S.; Marshall, S.W. Interaction between noise and cigarette smoking for the outcome of hearing loss among women: A population-based study. Am. J. Ind. Med. 2013, 56, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.Y.; Davis, R.; Heyer, N.; Yang, Q.L.; Qiu, W.; Zhu, L.L.; Li, N.; Zhang, H.; Zeng, L.; Zhao, Y.M. Effect of cigarette smoking on noise-induced hearing loss in workers exposed to occupational noise in China. Noise Health 2013, 15, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Nasir, H.M.; Rampal, K.G. Hearing loss and contributing factors among airport workers in Malaysia. Med. J. Malays. 2012, 67, 81–86. [Google Scholar]
- Mohammadi, S.; Mazhari, M.M.; Mehrparvar, A.H.; Attarchi, M.S. Effect of simultaneous exposure to occupational noise and cigarette smoke on binaural hearing impairment. Noise Health 2010, 12, 187–190. [Google Scholar] [CrossRef]
- Attarchi, M.S.; Labbafinejad, Y.; Mohammadi, S. Contemporary exposure to cigarette smoke and noise of automobile manufacturing company workers. J. Public Health 2009, 18, 245–249. [Google Scholar] [CrossRef]
- Chang, S.J.; Chang, C.K. Prevalence and risk factors of noise-induced hearing loss among liquefied petroleum gas (LPG) cylinder infusion workers in Taiwan. Ind. Health 2009, 47, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Pouryaghoub, G.; Mehrdad, R.; Mohammadi, S. Interaction of smoking and occupational noise exposure on hearing loss: A cross-sectional study. BMC Public Health 2007, 7, 137. [Google Scholar] [CrossRef] [PubMed]
- Rachiotis, G.; Alexopoulos, C.; Drivas, S. Occupational exposure to noise, and hearing function among electro production workers. Auris Nasus Larynx 2006, 33, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Dement, J.; Ringen, K.; Welch, L.; Bingham, E.; Quinn, P. Surveillance of hearing loss among older construction and trade workers at Department of Energy nuclear sites. Am. J. Ind. Med. 2005, 48, 348–358. [Google Scholar] [CrossRef]
- Burr, H.; Lund, S.P.; Sperling, B.B.; Kristensen, T.S.; Poulsen, O.M. Smoking and height as risk factors for prevalence and 5-year incidence of hearing loss. A questionnaire-based follow-up study of employees in Denmark aged 18-59 years exposed and unexposed to noise. Int. J. Audiol. 2005, 44, 531–539. [Google Scholar] [CrossRef]
- Nomura, K.; Nakao, M.; Yano, E. Hearing loss associated with smoking and occupational noise exposure in a Japanese metal working company. Int. Arch. Occup. Environ. Health 2005, 78, 178–184. [Google Scholar] [CrossRef]
- Fortunato, G.; Marciano, E.; Zarrilli, F.; Mazzaccara, C.; Intrieri, M.; Calcagno, G.; Vitale, D.F.; La Manna, P.; Saulino, C.; Marcelli, V.; et al. Paraoxonase and superoxide dismutase gene polymorphisms and noise-induced hearing loss. Clin. Chem. 2004, 50, 2012–2018. [Google Scholar] [CrossRef][Green Version]
- Mizoue, T.; Miyamoto, T.; Shimizu, T. Combined effect of smoking and occupational exposure to noise on hearing loss in steel factory workers. Occup. Environ. Med. 2003, 60, 56–59. [Google Scholar] [CrossRef]
- Barone, J.A.; Peters, J.M.; Garabrant, D.H.; Bernstein, L.; Krebsbach, R. Smoking as a risk factor in noise-induced hearing loss. J. Occup. Med. 1987, 29, 741–745. [Google Scholar]
- Sun, H.; Zhang, X. Influence of Smoking and Alcohol Drinking on Hearing Loss Among Workers Exposed to Industrial Noise. J. Prev. Med. Inf. 2014, 30, 391–394. (In Chinese) [Google Scholar] [CrossRef]
- Shen, H.; Shi, J. Influence of smoking and drinking on noise-induced hearing loss. Chin. J. Ind. Med. 2013, 26, 741–745. (In Chinese) [Google Scholar]
- Li, X.; Li, Y. Study of Interaction between Smoking and Occupational Noise Exposure on Hearing Loss. J. Trop. Med. 2008, 8, 441–444. (In Chinese) [Google Scholar]
- Browning, G.G.; Gatehouse, S.; Lowe, G.D. Blood viscosity as a factor in sensorineural hearing impairment. Lancet 1986, 1, 121–123. [Google Scholar] [CrossRef]
- Fechter, L.D.; Thorne, P.R.; Nuttall, A.L. Effects of carbon monoxide on cochlear electrophysiology and blood flow. Hear. Res. 1987, 27, 37–45. [Google Scholar] [CrossRef]
- Blanchet, C.; Erostegui, C.; Sugasawa, M.; Dulon, D. Acetylcholine-induced potassium current of guinea pig outer hair cells: Its dependence on a calcium influx through nicotinic-like receptors. J. Neurosci. 1996, 16, 2574–2584. [Google Scholar] [CrossRef]
- Attias, J.; Sapir, S.; Bresloff, I.; Reshef-Haran, I.; Ising, H. Reduction in noise-induced temporary threshold shift in humans following oral magnesium intake. Clin. Otolaryngol. Allied Sci. 2004, 29, 635–641. [Google Scholar] [CrossRef]
- Niskar, A.S.; Kieszak, S.M.; Holmes, A.E.; Esteban, E.; Rubin, C.; Brody, D.J. Estimated prevalence of noise-induced hearing threshold shifts among children 6 to 19 years of age: The Third National Health and Nutrition Examination Survey, 1988–1994, United States. Pediatrics 2001, 108, 40–43. [Google Scholar] [CrossRef]
- Cruickshanks, K.J.; Nondahl, D.M.; Tweed, T.S.; Wiley, T.L.; Klein, B.E.; Klein, R.; Chappell, R.; Dalton, D.S.; Nash, S.D. Education, occupation, noise exposure history and the 10-yr cumulative incidence of hearing impairment in older adults. Hear. Res. 2010, 264, 3–9. [Google Scholar] [CrossRef]
- Ishii, E.K.; Talbott, E.O. Race/ethnicity differences in the prevalence of noise-induced hearing loss in a group of metal fabricating workers. J. Occup. Environ. Med. 1998, 40, 661–666. [Google Scholar] [CrossRef]
| First Author and Year | Country | n | Mean Age/Age Range | Gender | Smoking Information | Diagnostic Criteria of NIHL | Adjusting or Matching Variables | Quality Assessment |
|---|---|---|---|---|---|---|---|---|
| Cohort study | ||||||||
| Dement, 2018 [11] | USA | 4275 | 59.2 | Both | Never smoked, Past smoker, Current smoker, Smoking | Index weighted average threshold >25 dB at 1, 2, 3, and 4 kHz | Age, race, sex, organic solvent exposure, duration of trade work, loud or very loud noise exposure, hypertension | 8 |
| Pettersson, 2014 [30] | Sweden | 184 | NA | Male | Smoker, Non-smoker | >30 dB at 0.5 kHz or >25 dB at 1–2 kHz and >25 dB for at least one of the frequencies of 3, 4, 6 kHz | VWF in the right hand, age, exposure | 8 |
| Li, 2008 [47] | China | 408 | 26.5 | Both | Smoking number per day: 0, −10, 10–20, >20 | Hearing any tone >25 dB | Cumulative noise exposure (CNE) | 6 |
| Burr, 2005 [40] | Denmark | 1237 | 18–59 | Both | Never, past, currently <15 g/day, currently ≥15 g/day | Question: ‘Do you have reduced hearing to such an extent that you feel it is difficult to follow a conversation between several people without using a hearing aid?’ | Gender, age and smoking stratified by occupational noise exposure | 7 |
| Case-control study | ||||||||
| Jiao, 2017 [19] | China | Case 286/control 286 | 40.2 | Both | Smoker, Non-smoker | Average hearing threshold ≥40 dB at 3, 4, 6 kHz | Gender, age, job category and time of exposure to noise | 7 |
| Jeffree, 2016 [28] | Malaysia | Case 49/control 98 | 41.3 | Male | Smoking in pack-years: 0, 1–10, 11–20, >20 | Average audibility threshold ≥25 dB at 0.5, 1, 2, 3 kHz | Daily noise dose, duration of services, HPD used frequency, perception about HPD | 8 |
| Cross-sectional study | ||||||||
| Wang,2017 [10] | China | 11,196 | 67.1 | Both | Smoking in pack-years: 0, 0–25, >25 | Average audibility threshold ≥25 dB at 0.5, 1, 2, 4 kHz in both ears | Age, sex, race, shift work, occupational noise exposure, drinking status, hypertension, ototoxicity medicine, chronic diseases (diabetes mellitus, coronary heart disease, myocardial infarction and stroke) | 10 |
| Sari, 2017 [12] | Indonesia | 122 | 18–40 | Male | Smoker, Non-smoker | Average hearing threshold >25 dB at 0.5, 1, 2, 4 kHz | NA | 6 |
| Sriopas, 2017 [13] | Thailand | 180 | 20–50 | NA | Pack-years smoking: <10, ≥10 | Average threshold >25 dB at 3, 4, 6, 8 kHz | Noise exposure level, employment duration, age, factory group, job position, and education level/noise exposure level, and education level | 8 |
| Win, 2015 [29] | Brunei | 543 | 35.6 | Both | Smoker, Non-smoker | Hearing loss of >25 dB at 4 kHz | NA | 7 |
| Sun, 2014 [45] | China | 471 | 39.8 | Male | Smoker, Non-smoker | Hearing any tone at 0.5, 1, 2 KHz >25 dB or average hearing threshold ≥40 dB at 3, 4, 6 kHz | Age, alcohol | 9 |
| Ferrite, 2013 [31] | Brazil | 364 | 33.9 | Female | Never smoked, Past smoker, Current smoker | Average threshold >25 dB at 0.5, 1, 2, 3, 4 kHz in the worse ear | Age, job type, solvent exposure and high blood pressure | 10 |
| Tao, 2013 [32] | China | 517 | 37.9 | Male | Smoker, Non-smoker | Hearing threshold >40 dB at 4 kHz in the worse ear | Age, CNE | 8 |
| Shen, 2013 [46] | China | 495 | 40.6 | Male | Smoker, Non-smoker | Average hearing threshold >40 dB at 3, 4, 6 kHz | Age, alcohol | 8 |
| Nasir, 2012 [33] | Malaysia | 358 | 31.9 | Both | Smoker, Non-smoker | Average hearing threshold ≥25 dB at 0.5, 1, 2, 3 kHz | Age, job type, Service duration, exposure duration, exposure to explosion, exposure to vibration | 9 |
| Shrestha, 2011 [7] | Nepal | 110 | 29.8 | Both | Smoker, Non-smoker | Average hearing loss >25 dB at 1, 2, 3 KHz | NA | 6 |
| Mohammadi, 2010 [34] | Iran | 622 | 42.1 | Male | Smoker, Non-smoker; Smoking in pack-years: 0, <20, ≥20 | Average audibility threshold ≥25 dB at 0.5, 1, 2, 3 kHz | Age, duration of exposure | 9 |
| Attarchi, 2010 [35] | Iran | 478 | 33.5 | Male | Smoker, Non-smoker; Smoking in pack-years: 0, ≤8, >8 | Hearing threshold differences ≥30 dB between 4 KHz and 1 KHz in both ears | Age, duration of exposure | 8 |
| Chang, 2009 [36] | China | 75 | 42.4 | Male | Smoker, Non-smoker | Average hearing loss >25 dB at 0.5, 1, and 2 kHz | Exposure status, age, tea or coffee, physical activity, BMI | 8 |
| Pouryaghoub, 2007 [37] | Iran | 412 | 42.1 | Male | Smoking in pack-years: 0, ≤10, >10 | Hearing threshold >25 dB at 4 KHz in the better ear | Age, duration of exposure | 8 |
| Rachiotis, 2006 [38] | Greece | 145 | 40.3 | Both | Smoker, Non-smoker | Average threshold ≥25 dB at 4 KHz | Sex, age, occupational exposure to waste, duration of employment | 8 |
| Dement, 2005 [39] | USA | 2469 | 56.6 | Both | Smoker, Non-smoker | Index weighted average threshold >25 dB at 1, 2, 3, and 4 kHz | Age, race, and gender | 9 |
| Nomura, 2005 [41] | Japan | 163 | 21–66 | Male | Never smoked, Past smoker, Current smoker | Hearing loss >40 dB at 4 kHz | NA | 7 |
| Fortunato, 2004 [42] | Italy | 94 | 43 | Male | Smokers in cigarettes/day: ≤10, >10 | Hearing any tone >25 dB | PON2 (S/C) and SOD2 IVS3-23 T/Gmand IVS3-60 T/G polymorphisms, age | 8 |
| Palmer, 2004 [20] | Britain | 2232 | 16–64 | Both | Never smoked, Past smoker, Current smoker | Question: ‘‘How well can you hear a person who is talking to you when he is sitting on your right [left] side in a quiet room?’’. | age, sex, and self report of frequent | 8 |
| Mizoue, 2003 [43] | Japan | 1386 | NA | Male | cigarettes/day: 0, 1–14, 15–24, ≥25 | Hearing threshold >25 dB at 1 KHz and threshold > 40 dB at 4 KHz | Age | 8 |
| Barone, 1987 [44] | USA | 1210 | 35.4 | Male | Never smoked, Past smoker, Current smoker | Average hearing loss >25 dB at 1,2,3 KHz with a 5:1 weighting of the better to poorer ear | Age, years of present job | 9 |
| Subgroup | Number of Studies | Pooled OR | 95% CI | P Value for Q Test | I2 (%) |
|---|---|---|---|---|---|
| Study design | |||||
| Cohort | 4 | 1.19 | 1.10–1.28 | 0.504 | 0 |
| Case-control | 3 | 2.25 | 1.59–3.19 | 0.418 | 0 |
| Cross-sectional | 22 | 2.21 | 1.74–2.81 | <0.001 | 89 |
| Gender | |||||
| Both | 13 | 1.50 | 1.28–1.76 | <0.001 | 68 |
| Male | 14 | 3.05 | 1.90–4.89 | <0.001 | 92 |
| Female | 1 | 1.52 | 1.03–2.27 | - | - |
| Mean age | |||||
| <40 | 11 | 2.18 | 1.51–3.14 | <0.001 | 86 |
| ≥40 | 16 | 2.03 | 1.59–2.61 | <0.001 | 89 |
| Race | |||||
| Mongoloid | 16 | 1.88 | 1.50–2.36 | <0.001 | 76 |
| Caucasian | 12 | 2.41 | 1.70–3.42 | <0.001 | 93 |
| others | 1 | 1.52 | 1.03–2.27 | - | - |
| Quality of studies | |||||
| High quality | 24 | 2.14 | 1.73–2.64 | <0.001 | 88 |
| Moderate quality | 5 | 1.91 | 1.05–3.45 | <0.001 | 82 |
| Number of adjusting variables | |||||
| 0 | 4 | 1.58 | 0.86–2.90 | <0.001 | 79 |
| ≥1 | 25 | 2.18 | 1.77–2.69 | <0.001 | 88 |
| Publication year | |||||
| <2010 | 12 | 2.27 | 1.53–3.34 | <0.001 | 87 |
| ≥2010 | 17 | 1.90 | 1.53–2.36 | <0.001 | 87 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Rong, X.; Wang, Z.; Lin, A. Association between Smoking and Noise-Induced Hearing Loss: A Meta-Analysis of Observational Studies. Int. J. Environ. Res. Public Health 2020, 17, 1201. https://doi.org/10.3390/ijerph17041201
Li X, Rong X, Wang Z, Lin A. Association between Smoking and Noise-Induced Hearing Loss: A Meta-Analysis of Observational Studies. International Journal of Environmental Research and Public Health. 2020; 17(4):1201. https://doi.org/10.3390/ijerph17041201
Chicago/Turabian StyleLi, Xiaowen, Xing Rong, Zhi Wang, and Aihua Lin. 2020. "Association between Smoking and Noise-Induced Hearing Loss: A Meta-Analysis of Observational Studies" International Journal of Environmental Research and Public Health 17, no. 4: 1201. https://doi.org/10.3390/ijerph17041201
APA StyleLi, X., Rong, X., Wang, Z., & Lin, A. (2020). Association between Smoking and Noise-Induced Hearing Loss: A Meta-Analysis of Observational Studies. International Journal of Environmental Research and Public Health, 17(4), 1201. https://doi.org/10.3390/ijerph17041201





