Effects of Eccentric Single-Leg Decline Squat Exercise on the Morphological and Structural Properties of the Vastus Lateralis and Patellar Tendon
Abstract
1. Introduction
2. Materials and Methods
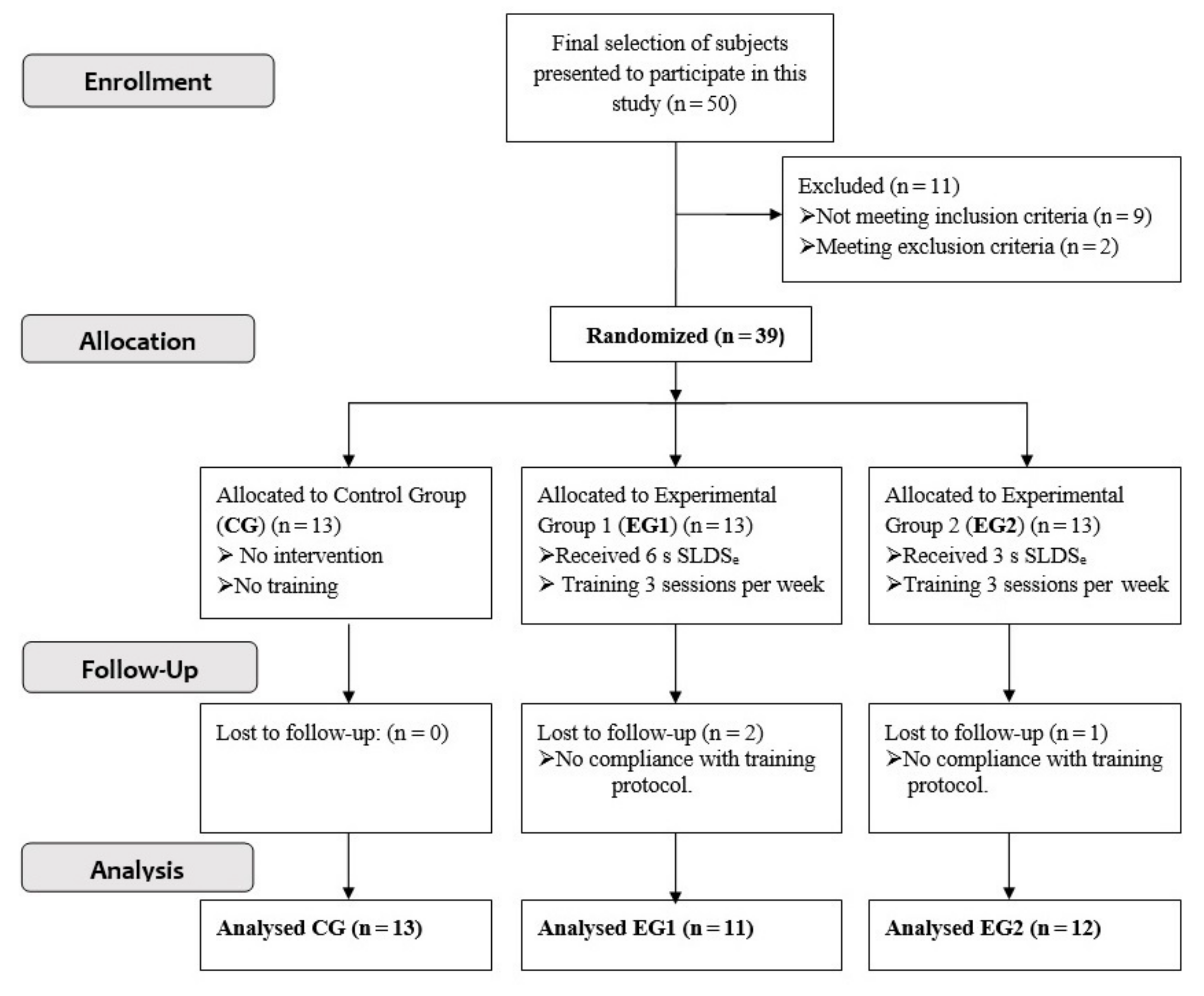
2.1. Subjects and Inclusion Procedure
2.2. Design and Procedure
2.3. Outcome Measures
2.4. Statistical Analysis
3. Results
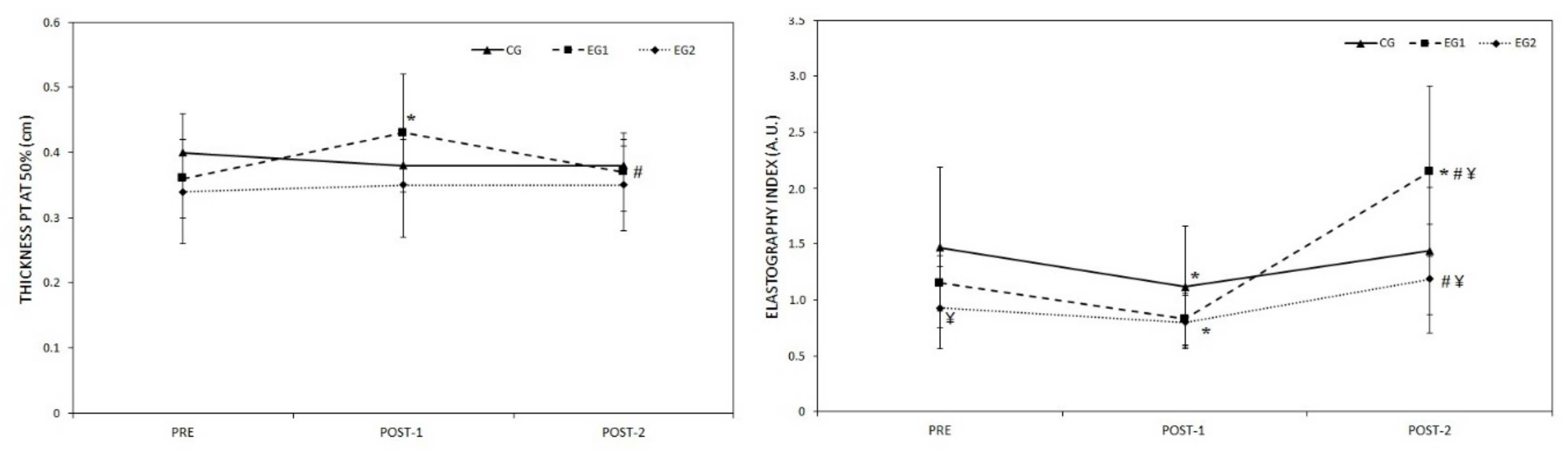
3.1. Morphological and Elastic Properties of PT
3.2. Vastus Lateralis Structure
3.3. Composition of the Thigh
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fanchini, M.; Steendahl, I.B.; Impellizzeri, F.M.; Pruna, R.; Dupont, G.; Coutts, A.J.; Meyer, T.; McCall, A. Exercise-Based Strategies to Prevent Muscle Injury in Elite Footballers: A Systematic Review and Best Evidence Synthesis. Sports Med. 2020, 50, 1653–1666. [Google Scholar] [CrossRef] [PubMed]
- Mendonca Lde, M.; Ocarino, J.M.; Bittencourt, N.F.; Fernandes, L.M.; Verhagen, E.; Fonseca, S.T. The Accuracy of the VISA-P Questionnaire, Single-Leg Decline Squat, and Tendon Pain History to Identify Patellar Tendon Abnormalities in Adult Athletes. J. Orthop. Sports Phys. Ther. 2016, 46, 673–680. [Google Scholar] [CrossRef]
- Hernández, J.L.; Monteagudo, P.; Sabido, R. Comparison of six weeks eccentric overload training between bilateral and unilateral squat in basketball players. Eur. J. Hum. Mov. 2018, 40, 111–121. [Google Scholar]
- Penailillo, L.; Blazevich, A.; Numazawa, H.; Nosaka, K. Metabolic and muscle damage profiles of concentric versus repeated eccentric cycling. Med. Sci. Sports Exerc. 2013, 45, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, S.D.; Mills, M.D.; Sterczala, A.J.; Mala, J.; Comstock, B.A.; Szivak, T.K.; DuPont, W.H.; Looney, D.P.; McDermott, D.M.; Hooper, D.R.; et al. The relationship between muscle action and repetition maximum on the squat and bench press in men and women. J. Strength Cond. Res. 2014, 28, 2437–2442. [Google Scholar] [CrossRef]
- Hody, S.; Croisier, J.L.; Bury, T.; Rogister, B.; Leprince, P. Eccentric Muscle Contractions: Risks and Benefits. Front. Physiol. 2019, 10, 536. [Google Scholar] [CrossRef]
- Camargo, P.R.; Zanca, G.G.; Okino, P.S.; Russo, T.L.; Michener, L.A. Scapular kinematics and muscle performance in a single case of Parsonage-Turner. Man. Ther. 2014, 19, 77–81. [Google Scholar] [CrossRef]
- Hoppeler, H. Moderate Load Eccentric Exercise; A Distinct Novel Training Modality. Front. Physiol. 2016, 7, 483. [Google Scholar] [CrossRef]
- Benjafield, A.J.; Killingback, A.; Robertson, C.J.; Adds, P.J. An investigation into the architecture of the vastus medialis oblique muscle in athletic and sedentary individuals: An in vivo ultrasound study. Clin. Anat. 2015, 28, 262–268. [Google Scholar] [CrossRef]
- Engelina, S.; Antonios, T.; Robertson, C.J.; Killingback, A.; Adds, P.J. Ultrasound investigation of vastus medialis oblique muscle architecture: An in vivo study. Clin. Anat. 2014, 27, 1076–1084. [Google Scholar] [CrossRef]
- Jan, M.H.; Lin, D.H.; Lin, J.J.; Lin, C.H.; Cheng, C.K.; Lin, Y.F. Differences in sonographic characteristics of the vastus medialis obliquus between patients with patellofemoral pain syndrome and healthy adults. Am. J. Sports Med. 2009, 37, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Franchi, M.V.; Reeves, N.D.; Narici, M.V. Skeletal muscle remodeling in response to eccentric vs. concentric loading: Morphological, molecular, and metabolic adaptations. Front. Physiol. 2017, 8. [Google Scholar] [CrossRef]
- LaStayo, P.; Marcus, R.; Dibble, L.; Frajacomo, F.; Lindstedt, S. Eccentric exercise in rehabilitation: Safety, feasibility, and application. J. Appl. Physiol. 2014, 116, 1426–1434. [Google Scholar] [CrossRef] [PubMed]
- Nunez, F.J.; Santalla, A.; Carrasquila, I.; Asian, J.A.; Reina, J.I.; Suarez-Arrones, L.J. The effects of unilateral and bilateral eccentric overload training on hypertrophy, muscle power and COD performance, and its determinants, in team sport players. PLoS ONE 2018, 13, e0193841. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Fernandez, D.; Abalo-Núñez, R.; Mateos-Padorno, C.; Martínez-Patiño, M.J. Effects of eccentric exercise on the quadriceps architecture. Sci. Sports 2020. [Google Scholar] [CrossRef]
- Timmins, R.G.; Ruddy, J.D.; Presland, J.; Maniar, N.; Shield, A.J.; Williams, M.D.; Opar, D.A. Architectural Changes of the Biceps Femoris Long Head after Concentric or Eccentric Training. Med. Sci. Sports Exerc. 2016, 48, 499–508. [Google Scholar] [CrossRef]
- Hakkinen, K.; Komi, P.V. Electromyographic changes during strength training and detraining. Med. Sci. Sports Exerc. 1983, 15, 455–460. [Google Scholar] [CrossRef]
- Hernandez-Sanchez, S.; Hidalgo, M.D.; Gomez, A. Cross-cultural adaptation of VISA-P score for patellar tendinopathy in Spanish population. J. Orthop. Sports Phys. Ther. 2011, 41, 581–591. [Google Scholar] [CrossRef]
- Cadore, E.L.; Gonzalez-Izal, M.; Pallares, J.G.; Rodriguez-Falces, J.; Hakkinen, K.; Kraemer, W.J.; Pinto, R.S.; Izquierdo, M. Muscle conduction velocity, strength, neural activity, and morphological changes after eccentric and concentric training. Scand. J. Med. Sci. Sports 2014, 24, e343–e352. [Google Scholar] [CrossRef]
- Brzycki, M. Strength Testing-Predicting a One-Rep Max from Reps-to-Fatigue. JOPERD 1993, 64, 88–90. [Google Scholar] [CrossRef]
- Purdam, C.R.; Jonsson, P.; Alfredson, H.; Lorentzon, R.; Cook, J.L.; Khan, K.M. A pilot study of the eccentric decline squat in the management of painful chronic patellar tendinopathy. Br. J. Sports Med. 2004, 38, 395–397. [Google Scholar] [CrossRef] [PubMed]
- Fukutani, A.; Kurihara, T. Comparison of the muscle fascicle length between resistance-trained and untrained individuals: Cross-sectional observation. Springerplus 2015, 4, 341. [Google Scholar] [CrossRef] [PubMed]
- Kastelic, J.; Galeski, A.; Baer, E. The multicomposite structure of tendon. Connect. Tissue Res. 1978, 6, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, K.; Bachinger, H.P.; Mizuno, K.; Boudko, S.; Engel, J.; Berisio, R.; Vitagliano, L. Re: Microfibrillar structure of type I collagen in situ. Acta Cryst. D Biol. Cryst. 2009, 65, 1007–1008. [Google Scholar] [CrossRef] [PubMed]
- Andrikoula, S.; Tokis, A.; Vasiliadis, H.S.; Georgoulis, A. The extensor mechanism of the knee joint: An anatomical study. Knee Surg. Sports Traumatol. Arthrosc. 2006, 14, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Klauser, A.S.; Miyamoto, H.; Tamegger, M.; Faschingbauer, R.; Moriggl, B.; Klima, G.; Feuchtner, G.M.; Kastlunger, M.; Jaschke, W.R. Achilles tendon assessed with sonoelastography: Histologic agreement. Radiology 2013, 267, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Sanchez, A.; Abian, P.; Jimenez, F.; Abian-Vicen, J. Myotendinous asymmetries derived from the prolonged practice of badminton in professional players. PLoS ONE 2019, 14, e0222190. [Google Scholar] [CrossRef]
- Csapo, R.; Alegre, L.M.; Baron, R. Time kinetics of acute changes in muscle architecture in response to resistance exercise. J. Sci. Med. Sport 2011, 14, 270–274. [Google Scholar] [CrossRef]
- Draganidis, D.; Jamurtas, A.Z.; Stampoulis, T.; Laschou, V.C.; Deli, C.K.; Georgakouli, K.; Papanikolaou, K.; Chatzinikolaou, A.; Michalopoulou, M.; Papadopoulos, C.; et al. Disparate Habitual Physical Activity and Dietary Intake Profiles of Elderly Men with Low and Elevated Systemic Inflammation. Nutrients 2018, 10, 566. [Google Scholar] [CrossRef]
- Alegre, L.M.; Aguado, X.; Rojas-Martin, D.; Martin-Garcia, M.; Ara, I.; Csapo, R. Load-controlled moderate and high-intensity resistance training programs provoke similar strength gains in young women. Muscle Nerve 2015, 51, 92–101. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Arampatzis, A.; Peper, A.; Bierbaum, S.; Albracht, K. Plasticity of human Achilles tendon mechanical and morphological properties in response to cyclic strain. J. Biomech. 2010, 43, 3073–3079. [Google Scholar] [CrossRef] [PubMed]
- Farup, J.; Rahbek, S.K.; Vendelbo, M.H.; Matzon, A.; Hindhede, J.; Bejder, A.; Ringgard, S.; Vissing, K. Whey protein hydrolysate augments tendon and muscle hypertrophy independent of resistance exercise contraction mode. Scand. J. Med. Sci. Sports 2014, 24, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Malliaras, P.; Kamal, B.; Nowell, A.; Farley, T.; Dhamu, H.; Simpson, V.; Morrissey, D.; Langberg, H.; Maffulli, N.; Reeves, N.D. Patellar tendon adaptation in relation to load-intensity and contraction type. J. Biomech. 2013, 46, 1893–1899. [Google Scholar] [CrossRef] [PubMed]
- Heinemeier, K.M.; Kjaer, M. In vivo investigation of tendon responses to mechanical loading. J. Musculoskelet. Neuronal Interact 2011, 11, 115–123. [Google Scholar] [PubMed]
- Young, M.A.; Cook, J.L.; Purdam, C.R.; Kiss, Z.S.; Alfredson, H. Eccentric decline squat protocol offers superior results at 12 months compared with traditional eccentric protocol for patellar tendinopathy in volleyball players. Br. J. Sports Med. 2005, 39, 102–105. [Google Scholar] [CrossRef]
- Zwerver, J.; Bredeweg, S.W.; Hof, A.L. Biomechanical analysis of the single-leg decline squat. Br. J. Sports Med. 2007, 41, 264–268. [Google Scholar] [CrossRef]
- Porta, F.; Damjanov, N.; Galluccio, F.; Iagnocco, A.; Matucci-Cerinic, M. Ultrasound elastography is a reproducible and feasible tool for the evaluation of the patellar tendon in healthy subjects. Int. J. Rheum. Dis. 2014, 17, 762–766. [Google Scholar] [CrossRef]
- Kubo, K.; Ikebukuro, T.; Yata, H.; Tsunoda, N.; Kanehisa, H. Time course of changes in muscle and tendon properties during strength training and detraining. J. Strength Cond. Res. 2010, 24, 322–331. [Google Scholar] [CrossRef]
- Alegre, L.M.; Hasler, M.; Wenger, S.; Nachbauer, W.; Csapo, R. Does knee joint cooling change in vivo patellar tendon mechanical properties? Eur. J. Appl. Physiol. 2016, 116, 1921–1929. [Google Scholar] [CrossRef]
- Pruyn, E.C.; Watsford, M.L.; Murphy, A.J. Validity and reliability of three methods of stiffness assessment. J. Sport Health Sci. 2016, 5, 476–483. [Google Scholar] [CrossRef]
- McHugh, M.P.; Connolly, D.A.; Eston, R.G.; Kremenic, I.J.; Nicholas, S.J.; Gleim, G.W. The role of passive muscle stiffness in symptoms of exercise-induced muscle damage. Am. J. Sports Med. 1999, 27, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Blazevich, A.J.; Cannavan, D.; Coleman, D.R.; Horne, S. Influence of concentric and eccentric resistance training on architectural adaptation in human quadriceps muscles. J. Appl. Physiol. 2007, 103, 1565–1575. [Google Scholar] [CrossRef] [PubMed]
- Baroni, B.M.; Geremia, J.M.; Rodrigues, R.; De Azevedo Franke, R.; Karamanidis, K.; Vaz, M.A. Muscle architecture adaptations to knee extensor eccentric training: Rectus femoris vs. vastus lateralis. Muscle Nerve 2013, 48, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Sharifnezhad, A.; Marzilger, R.; Arampatzis, A. Effects of load magnitude, muscle length and velocity during eccentric chronic loading on the longitudinal growth of the vastus lateralis muscle. J. Exp. Biol. 2014, 217, 2726–2733. [Google Scholar] [CrossRef] [PubMed]
- Hornberger, T.A.; Chien, S. Mechanical stimuli and nutrients regulate rapamycin-sensitive signaling through distinct mechanisms in skeletal muscle. J. Cell Biochem. 2006, 97, 1207–1216. [Google Scholar] [CrossRef]
- Walker, P.M.; Brunotte, F.; Rouhier-Marcer, I.; Cottin, Y.; Casillas, J.M.; Gras, P.; Didier, J.P. Nuclear magnetic resonance evidence of different muscular adaptations after resistance training. Arch. Phys. Med. Rehabil. 1998, 79, 1391–1398. [Google Scholar] [CrossRef]
- Perez-Gomez, J.; Vicente-Rodriguez, G.; Ara Royo, I.; Martinez-Redondo, D.; Puzo Foncillas, J.; Moreno, L.A.; Diez-Sanchez, C.; Casajus, J.A. Effect of endurance and resistance training on regional fat mass and lipid profile. Nutr. Hosp. 2013, 28, 340–346. [Google Scholar] [CrossRef]

| CG (n = 13) | EG1 (n = 11) | EG2 (n = 12) | |
|---|---|---|---|
| Age (years) | 20.77 ± 1.88 | 21.55 ± 2.46 | 21.08 ± 1.24 |
| Weight (kg) | 69.84 ± 10.89 | 71.85 ± 11.82 | 71.27 ± 8.28 |
| Height (cm) | 1.75 ± 0.06 | 1.76 ± 0.07 | 1.74 ± 0.07 |
| Fat percentage (%) | 18.99 ± 7.02 | 18.91 ± 05.04 | 19.54 ± 4.81 |
| PRE | POST-1 | POST-2 | p-Value | Timeline Effect | Time × Group Interaction | |||
|---|---|---|---|---|---|---|---|---|
| F | p-Value | F | p-Value | |||||
| Pennation angle VL distal (°) | ||||||||
| CG | 15.92 ± 2.69 | 16.15 ± 3.29 | 17.15 ± 2.19 | 0.437 | 3.94 | 0.032 | 3.83 | 0.034 |
| EG1 | 17.38 ± 2.39 | 19.63 ± 2.88 *¥ | 16.63± 1.77 # | 0.025 | ||||
| EG2 | 15.50 ± 3.24 | 17.10 ± 3.96 | 17.40 ± 2.98 | 0.108 | ||||
| Thickness VL distal (cm) | ||||||||
| CG | 1.53 ± 0.32 | 1.39 ± 0.33 | 1.48 ± 0.27 | 0.267 | 1.26 | 0.299 | 5.01 | 0.014 |
| EG1 | 1.61 ± 0.45 | 1.79 ± 0.41 ¥ | 1.68 ± 0.40 | 0.244 | ||||
| EG2 | 1.42 ± 0.24 | 1.63 ± 0.26 * | 1.55 ± 0.36 | 0.088 | ||||
| Fascicle length VL distal (cm) | ||||||||
| CG | 5.65 ± 1.14 | 5.45 ± 0.99 | 5.30 ± 0.63 | 0.716 | 0.47 | 0.628 | 0.06 | 0.993 |
| EG1 | 5.15 ± 2.47 | 5.10 ± 0.56 | 4.89 ± 0.85 | 0.860 | ||||
| EG2 | 5.51 ± 1.19 | 5.57 ± 1.35 | 5.39 ± 0.90 | 0.877 | ||||
| Pennation angle VL 50% (°) | ||||||||
| CG | 15.46 ± 3.89 | 15.62 ± 3.69 | 15.46 ± 3.13 | 0.954 | 2.53 | 0.099 | 0.60 | 0.662 |
| EG1 | 14.75 ± 2.76 | 16.38 ± 2.62 | 16.63 ± 2.07 | 0.168 | ||||
| EG2 | 14.20 ± 2.97 | 15.40 ± 2.46 | 15.10 ± 2.51 | 0.297 | ||||
| Thickness VL 50% (cm) | ||||||||
| CG | 2.09 ± 0.39 | 2.08 ± 0.33 | 2.07 ± 0.33 | 0.961 | 4.34 | 0.023 | 2.72 | 0.083 |
| EG1 | 1.90 ± 0.38 | 2.13 ± 0.36 | 2.08 ± 1.82 | 0.164 | ||||
| EG2 | 2.02 ± 0.71 | 2.25 ± 0.30 * | 2.08 ± 0.32 # | 0.016 | ||||
| Fascicle length VL 50% (cm) | ||||||||
| CG | 8.05 ± 1.74 | 7.99 ± 1.72 | 7.98 ± 1.82 | 0.997 | 0.78 | 0.470 | 0.35 | 0.843 |
| EG1 | 7.04 ± 3.56 | 8.01 ± 1.85 | 7.62 ± 1.01 | 0.353 | ||||
| EG2 | 7.84 ± 03.40 | 8.33 ± 1.79 | 7.92 ± 1.26 | 0.455 | ||||
| PRE | POST-1 | POST-2 | p-Value | Timeline Effect | Time × Group Interaction | |||
|---|---|---|---|---|---|---|---|---|
| F | p-Value | F | p-Value | |||||
| Lean mass thigh (kg) | ||||||||
| CG | 7.01 ± 1.16 | 7.03 ± 1.17 | 7.16 ± 1.21 | 0.164 | 10.75 | p < 0.001 | 2.89 | 0.029 |
| EG1 | 7.43 ± 1.19 | 7.73 ± 1.21 * | 7.51 ± 1.20 # | 0.005 | ||||
| EG2 | 7.19 ± 0.77 | 7.55 ± 0.88 * | 7.36 ± 0.74 | 0.001 | ||||
| Fat percentage thigh (%) | ||||||||
| CG | 20.36 ± 8.44 | 20.36 ± 8.06 | 20.57 ± 7.95 | 0.822 | 17.90 | p < 0.001 | 2.88 | 0.030 |
| EG1 | 19.30 ± 5.80 | 18.45 ± 6.21 * | 19.42± 6.10 # | p < 0.001 | ||||
| EG2 | 19.89 ± 5.55 | 18.88 ± 4.78 * | 19.83 ± 4.80 # | p < 0.001 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abián, P.; Martínez, F.; Jiménez, F.; Abián-Vicén, J. Effects of Eccentric Single-Leg Decline Squat Exercise on the Morphological and Structural Properties of the Vastus Lateralis and Patellar Tendon. Int. J. Environ. Res. Public Health 2020, 17, 9410. https://doi.org/10.3390/ijerph17249410
Abián P, Martínez F, Jiménez F, Abián-Vicén J. Effects of Eccentric Single-Leg Decline Squat Exercise on the Morphological and Structural Properties of the Vastus Lateralis and Patellar Tendon. International Journal of Environmental Research and Public Health. 2020; 17(24):9410. https://doi.org/10.3390/ijerph17249410
Chicago/Turabian StyleAbián, Pablo, Fernando Martínez, Fernando Jiménez, and Javier Abián-Vicén. 2020. "Effects of Eccentric Single-Leg Decline Squat Exercise on the Morphological and Structural Properties of the Vastus Lateralis and Patellar Tendon" International Journal of Environmental Research and Public Health 17, no. 24: 9410. https://doi.org/10.3390/ijerph17249410
APA StyleAbián, P., Martínez, F., Jiménez, F., & Abián-Vicén, J. (2020). Effects of Eccentric Single-Leg Decline Squat Exercise on the Morphological and Structural Properties of the Vastus Lateralis and Patellar Tendon. International Journal of Environmental Research and Public Health, 17(24), 9410. https://doi.org/10.3390/ijerph17249410







