Lithium in Portuguese Bottled Natural Mineral Waters—Potential for Health Benefits?
Abstract
1. Introduction
2. Materials and Methods
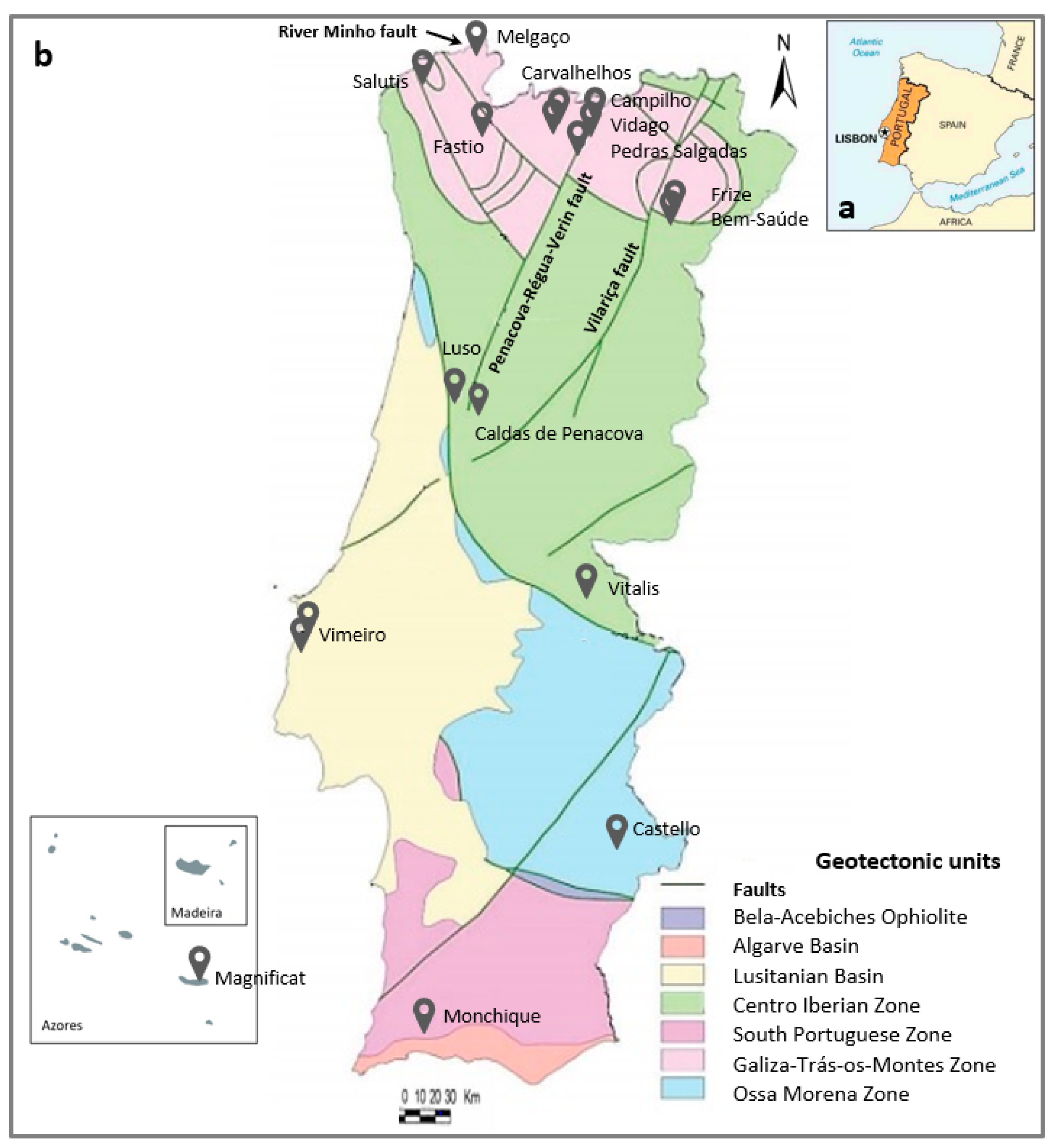
2.1. Sample Collection
2.2. Sample and Data Analysis
3. Results and Discussion
3.1. Bottled Natural Mineral Waters with Low Li Content
3.2. Bottled Natural Mineral Waters with Higher Li Content
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Statistics 2016: Monitoring Health for the SDGs. Chapter 6. pp. 29–42. Available online: https://www.who.int/gho/publications/world_health_statistics/2016/en/ (accessed on 12 May 2020).
- Cade, J.F. Lithium salts in the treatment of psychotic excitement. Med. J. Aust. 1949, 2, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Young, W. Review of lithium effects on brain and blood. Cell Trans. 2009, 18, 951–975. [Google Scholar] [CrossRef] [PubMed]
- Oruch, R.; Elderbi, M.A.; Khattab, H.A.; Pryme, I.F.; Lund, A. Lithium: A review of pharmacology, clinical uses, and toxicity. Eur. J. Pharmacol. 2014, 740, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Emsley, J. The Elements, 2nd ed.; Clarendon Press: Oxford, UK, 1991; p. 107. [Google Scholar]
- Schrauzer, G.N. Lithium: Occurrence, dietary intakes, nutritional essentiality. J. Am. Coll. Nutr. 2002, 21, 14–21. [Google Scholar] [CrossRef]
- Cauwenbergh, R.V.; Hendrix, P.; Robberecht, H.; Deelstra, H. Daily dietary lithium intake in Belgium using duplicate portion sampling. Eur. Food Res. Technol. 1991, 208, 153–155. [Google Scholar] [CrossRef]
- Ysart, G.; Miller, P.; Crews, H.; Robb, P.; Baxter, M.; De L’Argy, C.; Lofthouse, S.; Sargent, C.; Harrison, N. Dietary exposure estimates of 30 elements from the UK Total Diet Study. Food Addit. Contam. 1999, 16, 391–403. [Google Scholar] [CrossRef]
- ANSES. Second French Total Diet Study (FTDS2) Report 1. Inorganic Contaminants, Minerals, Persistent Organic Pollutants, Mycotoxins and Phytoestrogens. 2011, p. 60. Available online: https://www.anses.fr/en/system/files/PASER2006sa0361Ra1EN.pdf (accessed on 12 May 2020).
- Marcussen, H.; Jensen, B.H.; Petersen, A.; Holm, P.E. Dietary exposure to essential and potentially toxic elements for the population of Hanoi, Vietnam. Asia Pac. J. Clin. Nutr. 2013, 22, 300–311. [Google Scholar]
- Pearson, A.J.; Ashmore, E. Risk assessment of antimony, barium, beryllium, boron, bromine, lithium, nickel, strontium, thallium and uranium concentrations in the New Zealand diet. Food Addit. Contam. Part A 2020, 37, 451–464. [Google Scholar] [CrossRef]
- González-Weller, D.; Rubio, C.; Gutiérrez, A.J.; González, G.L.; Mesa, J.M.C.; Gironés, C.R.; Ojeda, A.B.; Hardisson, A. Dietary intake of barium, bismuth, chromium, lithium, and strontium in a Spanish population (Canary Islands, Spain). Food Chem. Toxicol. 2013, 62, 856–868. [Google Scholar] [CrossRef]
- Weiner, M.I. Overview of lithium toxicity. In Lithium in Biology and Medicine; Schrauzer, G.N., Klippel, K.F., Eds.; VCH Verlag: Weinheim, Germany, 1991; pp. 83–99. [Google Scholar]
- Goldstein, M.R.; Mascitelli, L. Is violence in part a lithium deficiency? Med. Hypotheses 2016, 89, 40–42. [Google Scholar] [CrossRef]
- Mleczek, M.; Siwulski, M.; Rzymski, P.; Budzyńska, S.; Gąsecka, M.; Kalač, P.; Niedzielski, P. Cultivation of mushrooms for production of food biofortified with lithium. Eur. Food Res. Technol. 2017, 243, 1097–1104. [Google Scholar] [CrossRef]
- Seidel, U.; Jans, K.; Hommen, N.; Ipharraguerre, I.R.; Lüersen, K.; Birringer, M.; Rimbach, G. Lithium Content of 160 Beverages and Its Impact on Lithium Status in Drosophila melanogaster. Foods 2020, 9, 795. [Google Scholar] [CrossRef] [PubMed]
- El-Mallakh, R.S.; Roberts, R.J. Lithiated Lemon-Lime Sodas. Am. J. Psychiatr. 2007, 164, 1662. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N.; Shrestha, K.P. Lithium in drinking water and the incidences of crimes, suicides, and arrests related to drug addictions. Biol. Trace Elem. Res. 1990, 25, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Ohgami, H.; Terao, T.; Shiotsuki, I.; Ishii, N.; Iwata, N. Lithium levels in drinking water and risk of suicide. Br. J. Psychiatr. 2009, 194, 464–465. [Google Scholar] [CrossRef] [PubMed]
- Kapusta, N.D.; Mossaheb, N.; Etzersdorfer, E.; Hlavin, G.; Thau, K.; Willeit, M.; Praschak-Rieder, N.; Praschak-Rieder, N.; Sonneck, G.; Leithner-Dziubas, K. Lithium in drinking water and suicide mortality. Br. J. Psychiatr. 2011, 198, 346–350. [Google Scholar] [CrossRef]
- Giotakos, O.; Nisianakis, P.; Tsouvelas, G.; Giakalou, V. Lithium in the public water supply and suicide mortality in Greece. Biol. Trace Elem. Res. Biolog. 2013, 156, 376–379. [Google Scholar] [CrossRef]
- Liaugaudaite, V.; Mickuviene, N.; Raskauskiene, N.; Naginiene, R. Lithium levels in the public drinking water supply and risk of suicide: A pilot study. J. Trace Elem. Med. Biol. 2017, 43, 197–201. [Google Scholar] [CrossRef]
- Kabacs, N.; Memom, A.; Obinwa, T.; Stochl, J.; Perez, J. Lithium in drinking water and suicide rates across the East of England. Br. J. Psychiatr. 2011, 198, 406–407. [Google Scholar] [CrossRef]
- Pompili, M.; Vichi, M.; Dinelli, E.; Pycha, R.; Valera, P.; Albanese, S.; Lima, A.; De Vivo, B.; Cicchella, D.; Fiorillo, A.; et al. Relationships of local lithium concentrations in drinking water to regional suicide rates in Italy. World J. Biol. Psychiatr. 2015, 16, 567–574. [Google Scholar] [CrossRef]
- Knudsen, N.N.; Schullehner, J.; Hansen, B.; Jørgensen, L.F.; Kristiansen, S.M.; Voutchkova, D.D.; Gerds, T.A.; Andersen, P.K.; Bihrmann, K.; Grønbæk, M.; et al. Lithium in Drinking Water and Incidence of Suicide: A Nationwide Individual-Level Cohort Study with 22 Years of Follow-Up. Int. J. Environ. Res. Public Health 2017, 14, 627. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, P.; Zagalo, J.; Madeira, N.; Neves, O. Lithium in Public Drinking Water and Suicide Mortality in Portugal: Initial Approach. Acta Med. Port. 2019, 32, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Marmol, F. Lithium: Bipolar disorder and neurodegenerative diseases. Possible cellular mechanisms of the therapeutic effects of lithium. Prog. NeuroPsychopharmacol. Biol. Psychiatry 2008, 32, 1761–1771. [Google Scholar] [CrossRef]
- Andrade Nunes, M.; Viel, T.A.; Buck, H.S. Microdose lithium treatment stabilized cognitive impairment in patients with Alzheimer’s disease. Curr. Alzheimer Res. 2013, 10, 104–107. [Google Scholar]
- Liu, B.; Wu, Q.; Zhang, S.; Del Rosario, A. Lithium Use and Risk of Fracture: A Systematic Review and Meta-Analysis of Observational Studies. Osteoporos. Int. 2018, 30, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Koh, J.; Kim, S.; Kim, K.E. Effect of Lithium on the Mechanism of Glucose Transport in Skeletal Muscles. J. Nutr. Sci. Vitaminol. 2017, 63, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.; Manne Sjöstrand, M.; Eyal, N. Adding Lithium to Drinking Water for Suicide Prevention—The Ethics. Public Health Ethics 2019, 12, 274–286. [Google Scholar] [CrossRef]
- Reimann, C.; Birke, M. Geochemistry of European Bottled Water; Borntraeger Science Publishers: Stuttgart, Germany, 2010; pp. 46–159. [Google Scholar]
- Schafer, U. Evaluation of beneficial and adverse effects on plants and animals following lithium deficiency and supplementation, and on humans following lithium treatment of mood disorders. Trace Elem. Electrolytes 2012, 29, 91–112. [Google Scholar] [CrossRef]
- STATISTA. Per Capita Consumption of Bottled Water in Europe in 2017, by Country. Available online: https://www.statista.com/statistics/455422/bottled-water-consumption-in-europe-per-capita/ (accessed on 15 May 2020).
- EFBW. Natural Waters: The Natural Choice for Hydration. Available online: https://www.efbw.org/ (accessed on 15 May 2020).
- Council Directive 2009/54/EC/18-6-2009 on the exploitation and marketing of natural mineral waters. Off. J. EU 2009, L164, 45–58.
- ANZ Guidelines. ANZECC-ARMCANZ-2000-Guidelines-Vol1. Available online: https://www.waterquality.gov.au/media/57 (accessed on 20 May 2020).
- Publications Office of the EU. List of Natural Mineral Waters Recognized by Member States. Available online: https://op.europa.eu/en/publication-detail/-/publication/b9453284-4083-11e3-b4f5-01aa75ed71a1/language-en/format-PDF/source-140676132 (accessed on 15 May 2020).
- Lei 54/2015. Bases do regime jurídico da revelação e do aproveitamento dos recursos geológicos existentes no território nacional, incluindo os localizados no espaço marítimo nacional. DRE 2015, 54, 4296–4308. [Google Scholar]
- Super Bock Group. Águas. Água das Pedras. Ficha Técnica. Available online: https://www.superbockgroup.com/produto/aguas/ (accessed on 15 May 2020).
- Lourenço, C.; Pascoal, R. O Estudo Metagenómico das Águas Minerais Naturais tendo em vista o Reconhecimento Científico das Vocações Terapêuticas. Bol. Minas (Ed. Espec. Term.) 2018–2019, 53, 39–53. [Google Scholar]
- Greenberg, A.; Clesceri, L.S.; Andrew, D.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater, 18th ed.; American Public Health Association, American Water Works association, Water Environment Federations: Washington, DC, USA, 1992; pp. 25–27. [Google Scholar]
- Marques, J.M.; Carreira, P.M.; Neves, O.; Espinha Marques, J.; Teixeira, J. Revision of the hydrogeological conceptual models of two Portuguese thermomineral water systems: Similarities and differences. Sustain. Water Resour. Manag. 2019, 5, 117–133. [Google Scholar] [CrossRef]
- Marques, J.M.; Carreira, P.M.; Aires-Barros, L.A.; Monteiro Santos, F.A.; Antunes da Silva, M.; Represas, P. Assessment of Chaves Low-Temperature CO2-Rich Geothermal System (N-Portugal) Using an Interdisciplinary Geosciences Approach. Geofluids 2019, 2019, 1379093. [Google Scholar] [CrossRef]
- Lourenço, C.; Ribeiro, L.; Cruz, J. Classification of natural mineral and spring bottled waters of Portugal using Principal Component Analysis. J. Geoch. Explor. 2010, 107, 362–372. [Google Scholar] [CrossRef]
- Neves, O.; Machete, I.; Marques, J.M.; da Silva, J.A.L.; Simões do Couto, F. Lítio em águas engarrafadas e de abastecimento público portuguesas. Com. Geol. 2015, 102, 103–106. [Google Scholar]
- Rybakowski, J.; Drogowska, J.; Abramowicz, M.; Chłopocka-Woźniak, M.; Czekalski, S. The effect of long-term lithium treatment on kidney function. Psychiatr. Pol. 2012, 246, 627–636. [Google Scholar]
- Szklarska, D.; Rzymski, P. Is Lithium a Micronutrient? From Biological Activity and Epidemiological Observation to Food Fortification. Biol. Trace Elem. Res. 2019, 189, 18–27. [Google Scholar] [CrossRef]
- Bochud, M.; Staessen, J.A.; Woodiwiss, A.; Norton, G.; Maillard, M.; Burnier, M. Context dependency of serum and urinary lithium: Implications for measurement of proximal sodium reabsorption. Hypertension 2007, 49, e34. [Google Scholar] [CrossRef][Green Version]
- Seidel, U.; Baumhof, E.; Hägele, F.A.; Bosy-Westphal, A.; Birringer, M.; Rimbach, G. Lithium-Rich Mineral Water is a Highly Bioavailable Lithium Source for Human Consumption. Mol. Nutr. Food Res. 2019, 63, e1900039. [Google Scholar] [CrossRef]
- Uwai, Y.; Arima, R.; Takatsu, C.; Furuta, R.; Kawasaki, T.; Nabekura, T. Sodium-Phosphate Cotransporter Mediates Reabsorption of Lithium in Rat Kidney. Pharmacol. Res. 2014, 87, 94–98. [Google Scholar] [CrossRef]
- Kovacsics, C.E.; Gottesman, I.I.; Gould, T.D. Lithium’s Antisuicidal Efficacy: Elucidation of Neurobiological Targets Using Endophenotype Strategies. Annu. Rev. Pharmacol. Toxicol. 2009, 49, 175–198. [Google Scholar] [CrossRef] [PubMed]
- Carreira, P.M.; Marques, J.M.; Carvalho, M.R.; Nunes, D.; Antunes da Silva, M. Carbon isotopes and geochemical processes in CO2-rich cold mineral water, N-Portugal. Environ. Earth Sci. 2014, 71, 2941–2953. [Google Scholar] [CrossRef]
- Lourenço, M.C. Modelação Estatística das Águas Gasocarbónicas de Vidago e Pedras Salgadas. Master’s Thesis, Instituto Superior Técnico, Lisbon, Portugal, April 2000. [Google Scholar]
- APIAM. Águas Minerais Naturais e Águas de Nascente—Livro Branco. Available online: http://www.apiam.pt/publicacoes/Livro-Branco-APIAM/-/47/23/175 (accessed on 18 May 2020).
- Calado, C.; Almeida, C. Geoquímica do Flúor em Águas Minerais da Zona Centro Ibérica. Memórias 1993, 3, 319–323. [Google Scholar]
- Council Directive 2003/40/EC/16-5-2003/ establishing the list, concentration limits and labelling requirements for the constituents of natural mineral waters and the conditions for using ozone-enriched air for the treatment of natural mineral waters and spring waters. Off. J EU 2003, L126, 34–39.
- Council Directive 98/83/EC/3-11-1998/on the quality of water intended for human consumption. Off. J EU 1998, L330, 32–54.
- Campbell, M.; Adams, P.B.; Small, A.M.; Kafantaris, V.; Silva, R.R.; Shell, J.; Perry, R.; Overall, J.E. Lithium in hospitalized aggressive children with conduct disorder: A double-blind and placebo-controlled study. J. Am. Acad. Child Adolesc. Psychiatr. 1995, 34, 445–453. [Google Scholar] [CrossRef]
- Cipriani, A.; Pretty, H.; Hawton, K.; Geddes, J. Lithium in the prevention of suicidal behavior and all-cause mortality in patients with mood disorders: A systematic review of randomized trials. Am. J. Psychiatr. 2005, 162, 1805–1819. [Google Scholar] [CrossRef]
- Jones, R.; Arlidge, J.; Gillham, R.; Reagu, S.; van den Bree, M.; Taylor, P. Efficacy of mood stabilisers in the treatment of impulsive or repetitive aggression: Systematic review and meta-analysis. Br. J. Psychiatr. 2011, 198, 93–98. [Google Scholar] [CrossRef]
- Rodriguez, C.J.; Bibbins-Domingo, K.; Jin, Z.; Daviglus, M.L.; Goff, D.C., Jr.; Jacobs, D.R. Association of sodium and potassium intake with left ventricular mass: Coronary artery risk development in young adults. Hypertension 2011, 58, 410–416. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration. Sodium in Your Diet—Use the Nutrition Facts Label and Reduce Your Intake. Available online: https://www.fda.gov/food/nutrition-education-resources-materials/sodium-your-diet (accessed on 18 September 2020).
- Santos, A.; Martins, M.J.; Severo, M.; Guimarães, J.; Azevedo, I. Ingestão de água mineral natural gasocarbónica hipersalina e pressão arterial. Rev. Port. Cardiol. 2010, 29, 159–172. [Google Scholar]
- Schorr, U.; Distler, A.; Sharma, A.M. Effect of sodium chloride and sodium bicarbonate-rich mineral water on blood pressure and metabolic parameters in elderly normotensive individuals: A randomized double-blind crossover trial. J. Hypertens. 1996, 14, 131–135. [Google Scholar] [PubMed]

| Mineral Water Brand | Type | pH | EC | DR | F− | Cl− | HCO3− | NO3− | SO42− | Na+ | K+ | Ca2+ | Mg2+ | Mn2+ | Li+ | Rb+ | Cs+ | Water Type | Place of Exploitation | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| μS/cm | mg/L | µg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | µg/L | µg/L | µg/L | µg/L | ||||||
| 1 | Salutis | S | 5.2 | 49 | 32 | 16 | 7.8 | 1.2 | 3.8 | 1.8 | 4.3 | 0.7 | 0.8 | 0.57 | 10 | 1 | 2.4 | 0.04 | Na-Cl | Ferreira—Paredes de Coura |
| 2 | Fastio | S | 6.0 | 35 | 141 | 19 | 3.8 | 8.5 | 2.3 | 0.8 | 4.3 | 0.6 | 1.2 | 0.47 | 1 | <1 | 1.7 | 0.11 | Na-HCO3 | Chamoim—Terras de Bouro |
| 3 | Monchique | S | 9.4 | 419 | 107 | 1058 | 31.7 | 126.9 | nd | 47.8 | 76.9 | 2.0 | 1.3 | 0.05 | <1 | 1 | 5.1 | 0.03 | Na-HCO3 | Caldas de Monchique—Monchique |
| 4 | Vimeiro Lisa | S | 7.1 | 83 | 75 | <10 | 8.4 | 21.4 | 0.4 | 3.6 | 9.2 | 0.3 | 5.6 | 1.46 | <1 | 1 | 0.3 | 0.02 | Na-HCO3 | Maceira—Torres |
| 5 | Vitalis | S | 5.7 | 50 | 71 | 28 | 6.3 | 3.7 | 2.2 | 2.7 | 5.2 | 2.1 | 0.9 | 0.55 | 8 | 1 | 14.5 | 0.34 | Na-Cl | Castelo de Vide |
| 6 | Caldas de Penacova | S | 5.5 | 48 | 41 | <10 | 7.8 | 4.3 | 1.8 | 1.3 | 6.1 | 0.3 | 0.7 | 1.10 | 4 | 2 | 0.9 | 0.19 | Na-Cl | Penacova |
| 7 | Magnificat | NC | 5.0 | 166 | 234 | 533 | 18.0 | 81.0 | 13.6 | 4.2 | 21.9 | 9.7 | 8.4 | 5.04 | 142 | 3 | 33.9 | 0.15 | Na-HCO3 | Serra do Trigo—Açores |
| 8 | Castello | AC | 5.4 | 793 | 472 | 123 | 46.2 | 361.7 | 18.3 | 18.0 | 34.3 | 0.9 | 94.8 | 26.20 | <1 | 7 | 0.4 | 0.04 | Ca-HCO3 | Pisões -Moura |
| 9 | Luso | S | 5.6 | 58 | 56 | 31 | 7.3 | 12.2 | 1.5 | 1.4 | 7.4 | 0.8 | 0.8 | 1.81 | 3 | 7 | 3.5 | 0.39 | Na-Cl | Luso—Mealhada |
| 10 | Vimeiro | AC | 5.7 | 1050 | 2291 | 221 | 176.3 | 425.8 | 7.8 | 79.4 | 144.0 | 3.7 | 112 | 27.1 | <1 | 11 | 3.4 | 0.19 | Na/Ca-HCO3 | Maceira—Torres |
| 11 | Carvalhelhos | S | 7.0 | 248 | 453 | 963 | 2.6 | 141.5 | 0.2 | 6.9 | 55.0 | 1.4 | 5.6 | 0.70 | 1 | 173 | 16.3 | 30.90 | Na-HCO3 | Carvalhelhos -Boticas |
| 12 | Carvalhelhos | AC | 5.3 | 189 | 208 | 498 | 2.7 | 124.4 | 1.1 | 7.6 | 52.3 | 1.4 | 5.9 | 0.62 | <1 | 177 | 17.9 | 32.60 | Na-HCO3 | Carvalhelhos -Boticas |
| 13 | Melgaço | NC | 5.7 | 840 | 439 | 657 | 10.6 | 691.7 | 1.2 | 7.5 | 87.5 | 3.6 | 145 | 3.01 | 275 | 600 | 19.5 | 6.73 | Ca-HCO3 | Quinta do Peso—Melgaço |
| 14 | Campilho | AC | 5.9 | 1892 | 1289 | 4131 | 15.6 | 1288.3 | 0.9 | 8.7 | 428.0 | 27.1 | 37.4 | 10.06 | 1 | 1590 | 215.0 | 222.00 | Na-HCO3 | Vidago—Chaves |
| 15 | Frize | NC | 6.5 | 2300 | 2336 | 1440 | 100.2 | 1941.0 | 1.6 | nd | 630.0 | 41.1 | 75.6 | 25.05 | 53 | 1760 | 335.0 | 331.00 | Na-HCO3 | Sampaio—Vila Flor |
| 16 | Pedras Salgadas | NC | 6.1 | 2660 | 1825 | 1265 | 22.9 | 1897.1 | 0.2 | 7.5 | 594.0 | 34.4 | 95.6 | 24.70 | 213 | 1800 | 238.0 | 49.00 | Na-HCO3 | Pedras Salgadas—Vila Pouca de Aguiar |
| 17 | Bem-Saúde | NC | 6.0 | 2310 | 1600 | 2100 | 90.0 | 1596.0 | 20.3 | 7.2 | 510.0 | 46.0 | 84.0 | 21.00 | 100 | 2000 | na | na | Na-HCO3 | Sampaio -Vila Flor |
| 18 | Vidago | NC | 6.0 | 1554 | 1797 | 25 | 25.5 | 1869.0 | 0.8 | 7.7 | 624.0 | 53.8 | 73.7 | 14.40 | 20 | 2210 | 415.0 | 253.00 | Na-HCO3 | Vidago—Chaves |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neves, M.O.; Marques, J.; Eggenkamp, H.G.M. Lithium in Portuguese Bottled Natural Mineral Waters—Potential for Health Benefits? Int. J. Environ. Res. Public Health 2020, 17, 8369. https://doi.org/10.3390/ijerph17228369
Neves MO, Marques J, Eggenkamp HGM. Lithium in Portuguese Bottled Natural Mineral Waters—Potential for Health Benefits? International Journal of Environmental Research and Public Health. 2020; 17(22):8369. https://doi.org/10.3390/ijerph17228369
Chicago/Turabian StyleNeves, Maria Orquídia, José Marques, and Hans G.M. Eggenkamp. 2020. "Lithium in Portuguese Bottled Natural Mineral Waters—Potential for Health Benefits?" International Journal of Environmental Research and Public Health 17, no. 22: 8369. https://doi.org/10.3390/ijerph17228369
APA StyleNeves, M. O., Marques, J., & Eggenkamp, H. G. M. (2020). Lithium in Portuguese Bottled Natural Mineral Waters—Potential for Health Benefits? International Journal of Environmental Research and Public Health, 17(22), 8369. https://doi.org/10.3390/ijerph17228369







