Iodine Status of Women and Infants in Russia: A Systematic Review
Abstract
1. Introduction
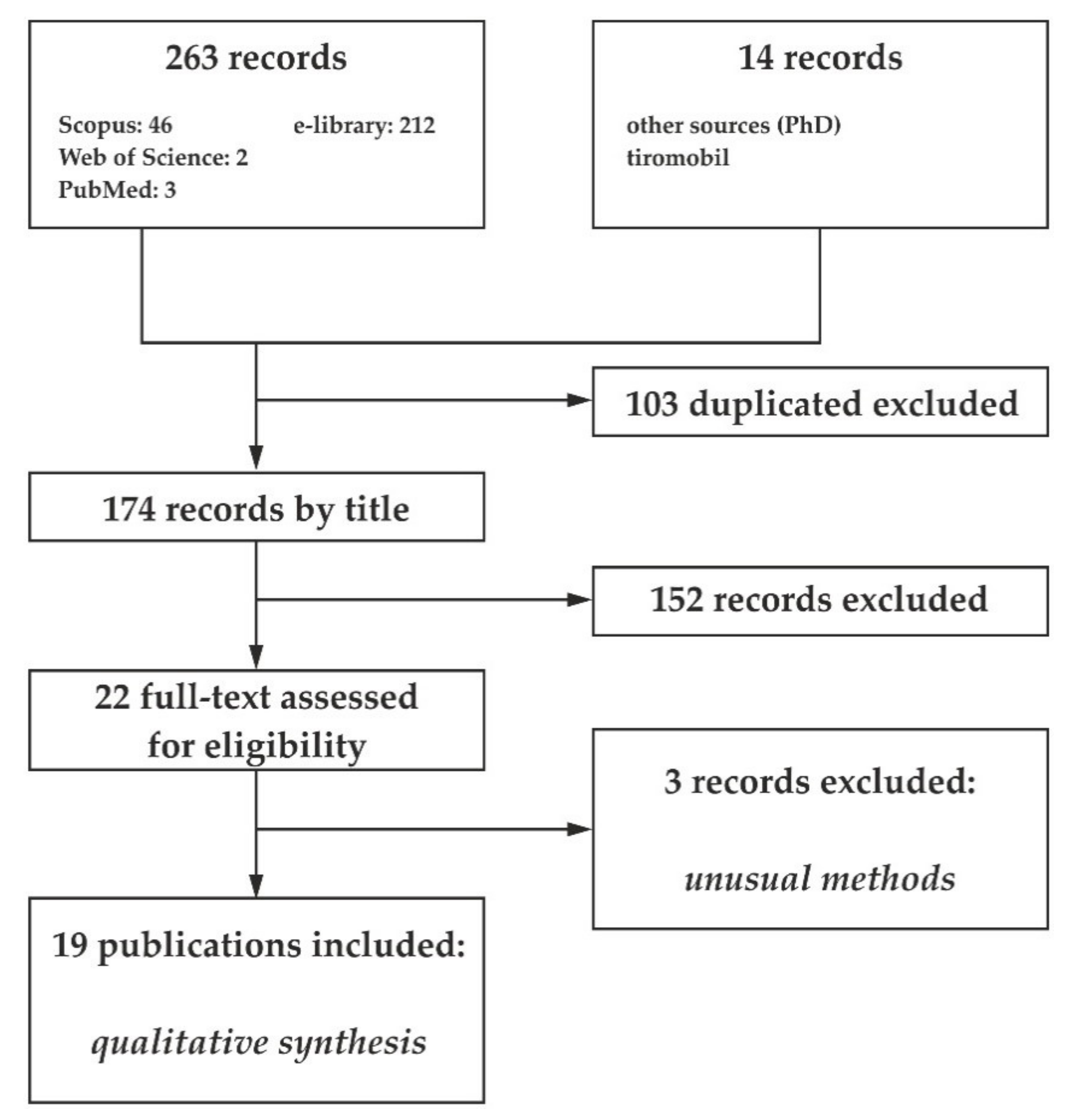
2. Materials and Methods
- Study subjects were residents of the Russian Federation;
- Studies were conducted in Russia from 1991 to 2019;
- Women of reproductive age including non-pregnant, pregnant and lactating women, newborns and infants;
- WHO standardized guidelines for I status are were used; and
- Median UIC data was available.
- Animal studies;
- Insufficient data on sample collection/preparation/analysis;
- Review articles;
- Patients with diagnosed thyroid disease and/or other chronic diseases; and
- Patients treated with radioactive iodine isotopes viz. 123I, 124I, 125I, and 131I.
3. Results and Discussion
3.1. Non-Pregnant Women of Reproductive Age
3.2. Pregnant Women
3.3. Pregnant Women without I Supplementation
- A decrease in median UIC throughout pregnancy reaching a minimum in the III trimester was described in—4 studies (Rostov, Cheboksary, Smolensk) [22];
- a decrease in median UIC in the second trimester with an increase in the third trimester. Median UIC values in the third trimester are almost equal to the values in the first trimester (Anapa, Ivanovo), or even exceed by more than 20% (Nizhnekamsk) [22]; and
3.4. Pregnant Women with I Supplementation
3.5. Nursing Women and Infants
- the other studies indicate that median UICs in newborns exceeded those measured in mothers by 2–3.5 times (e.g., in Amurs where the median UIC values in mother and child are 20 and 75 μg/L [36], respectively);
- of interest are the median UICs in mother-newborn pairs in the Vanino village where the values are 31 and 96 μg/L, respectively. For mothers, this median UIC level is much lower than that recommended and corresponds to a moderate severity of ID, while in newborns the median UIC is close to the recommended value;
- the lowest median UICs were observed in the Jewish Autonomous Region (Birobidzhan); 27 and 25 μg/L, for mother and infants, respectively [36]; and
- the highest median UICs were reported from the Kamchatka territory (Petropavlovsk-Kamchatsky) reaching 155 and 190 μg/L, respectively, for mothers and children [36].
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zimmermann, M.B.; Jooste, P.L.; Pandav, C.S. Iodine-deficiency disorders. Lancet 2008, 372, 1251–1262. [Google Scholar] [CrossRef]
- Bath, S.C. The effect of iodine deficiency during pregnancy on child development. Proc. Nutr. Soc. 2019, 78, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Levie, D.; Korevaar, T.I.M.; Bath, S.C.; Murcia, M.; Dineva, M.; Llop, S.; Espada, M.; E Van Herwaarden, A.; De Rijke, Y.B.; Ibarluzea, J.M.; et al. Association of Maternal Iodine Status with Child IQ: A Meta-Analysis of Individual Participant Data. J. Clin. Endocrinol. Metab. 2019, 104, 5957–5967. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.; Hynes, K.L.; Burgess, J.R. Mild-to-Moderate Gestational Iodine Deficiency Processing Disorder. Nutrients 2019, 11, 1974. [Google Scholar] [CrossRef] [PubMed]
- Nyström, H.F.; Brantsaeter, A.L.; Erlund, I.; Gunnarsdottir, I.; Hulthén, L.; Laurberg, P.; Mattisson, I.; Rasmussen, L.B.; Virtanen, S.; Meltzer, H.M. Iodine status in the Nordic countries–past and present. Food Nutr. Res. 2016, 60, 31969. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination. A Guide for Programme Managers, 2nd ed.; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- World Health Organization. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers, 3rd ed.; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Berg, V.; Nøst, T.H.; Skeie, G.; Thomassen, Y.; Berlinger, B.; Veyhe, A.S.; Jorde, R.; Odland, J.Ø.; Hansen, S. Thyroid homeostasis in mother–child pairs in relation to maternal iodine status: The MISA study. Eur. J. Clin. Nutr. 2017, 71, 1002–1007. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B. Iodine Deficiency. Endocr. Rev. 2009, 30, 376–408. [Google Scholar] [CrossRef] [PubMed]
- Delange, F.; Robertson, A.; McLoughney, E.; Gerasimov, G. Elimination of iodine deficiency disorders (IDD) in Central and Eastern Europe, the Commonwealth of Independent States, and the Baltic States. In Proceedings of the Conference Held in Munich, Germany, 3–6 September 1997; Available online: https://apps.who.int/iris/handle/10665/83307 (accessed on 11 November 2020).
- Melnichenko, G.A.; Troshina, E.A.; Platonova, N.M.; Panfilova, E.A.; Rybakova, A.A.; Abdulkhabirova, F.M.; Bostanova, F.A. Iodine deficiency thyroid disease in the Russian Federation: The current state of the problem. Analytical review of publications and data of official state statistics (Rosstat). Cons. Med. 2019, 21, 14–20. [Google Scholar] [CrossRef]
- Ensminger, M.E.; Ensminger, A.H. Foods & Nutrition Encyclopedia, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2019; p. 1216. [Google Scholar]
- Andersen, S.; Kleinschmidt, K.; Hvingel, B.; Laurberg, P. Thyroid hyperactivity with high thyroglobulin in serum despite sufficient iodine intake in chronic cold adaptation in an Arctic Inuit hunter population. Eur. J. Endocrinol. 2012, 166, 433–440. [Google Scholar] [CrossRef]
- Chashchin, V.P.; Gudkov, A.B.; Chashchin, M.V.; Popova, O.N. Predictive assessment of individual human susceptibility to damaging cold exposure. Ekologiya Cheloveka (Human Ecol.) 2017, 3–13. [Google Scholar] [CrossRef]
- Vlassov, V.; Danishevskiy, K.D. Biomedical journals and databases in Russia and Russian language in the former Soviet Union and beyond. Emerg. Themes Epidemiol. 2008, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Unguryanu, T.; Zhamaliyeva, K.; Grjibovski, A. Brief recommendations on how to write and publish systematic reviews. West Kazakhstan Med. J. 2019, 61, 26–36. [Google Scholar]
- eLIBRARY.RU-Scientific Electronic Library. Available online: https://www.elibrary.ru/defaultx.asp (accessed on 10 November 2020).
- A Troshina, E.; Sekinaeva, A.V.; Abdulhabirova, F.M. Prevention of iodine deficiency in pregnant and lactating women: Epidemiological Studies. Bull. Reprod. Health 2011, 54–60. [Google Scholar] [CrossRef]
- Sekinaev, A.V. Prevention of Iodine Deficiency Disorders in Pregnant Women and Nursing Women Example Regional Research. Ph.D. Thesis, Endocrinological Research Center of Rosmedtechnologies, Moscow, Russia, 2010. (In Russian). [Google Scholar]
- Tironet is All about the Thyroid Gland. Available online: http://thyronet.rusmedserv.com/ (accessed on 11 November 2020).
- Fedak, I.R. Control and Epidemiological Studies of the Effectiveness of Regional Programs for the Prevention of Iodine Deficiency Disorders in the Russian Federation. Ph.D. Thesis, Endocrinological Research center of Rosmedtechnologies, Moscow, Russia, 2008. (In Russian). [Google Scholar]
- Dedov, I.I.; Melnichenko, G.A.; Peterkova, V.A.; Troshina, E.A.; Abdulkhabirova, F.M.; Mazurina, N.V.; Platonova, N.M.; Kavtaradze, S.R.; Polyakova, E.Y.; Kichikova, J.D.; et al. Results of epidemiological studies of iodine deficiency disorders within the “tiromobil” project. Probl. Endocrinol. 2005, 51, 32–36. [Google Scholar]
- Stepanova, E.M. Thyroid Status, Pregnancy, and the Outcome of Giving in Women City Ufa as Area of Iodine Deficiency. Ph.D. Thesis, Kazan State Medical Academy of the Ministry of Health of the Russian Federation, Ufa, Russia, 2004. (In Russian). [Google Scholar]
- Zhivoglazova, L.N.; Kurmacheva, N.A.; Rogozhina, I.E. Efficacy of prophylaxis of micronutrients deficiency in pregnant women and newborns. Saratov J. Med. Sci. Res. 2010, 6, 300–306. [Google Scholar]
- Naumova, Y.V.; Kurmacheva, N.A.; Rogozhina, I.E. Health state of pregnant women and newborns depending on quality of iodine prevention. Saratov J. Med. Sci. Res. 2010, 6, 535–540. [Google Scholar]
- Kurmacheva, O.N. Prevention of iodine deficiency in children of the first year of life (medical-social aspects). Doctoral Thesis, Scientific center for children’s health Russian academy of medical sciences, Saratov, Russia, 2003. (In Russian). [Google Scholar]
- Soboleva, D.E. Iodine and Prevention Iodine Deficiency Disorders in Various Groups Adult Population of ST. Petersburg. Ph.D. Thesis, National Medical Research Center Named after V.A. Almazova, St. Petersburg, Russia, 2017. (In Russian). [Google Scholar]
- Suplotova, L.A.; Makarova, O.B.; Kovalzhina, L.S. Neonatal gipertireotropinemiya – an indicator of the severity of iodine deficiency in the population? Clin. Exp. Thyroid. 2015, 11, 47–53. [Google Scholar] [CrossRef]
- Myasishcheva, A.B. Health Conditions of Children under Conditions Combined Exposure to Natural Iodine Deficiency and Anthropogenic Load. Ph.D. Thesis, Astrakhan State Medical Academy Health Agency and Social Development, Astrakhan, Russia, 2010. (In Russian). [Google Scholar]
- Rymar, O.D.; Mustafina, S.V.; I Simonova, G.; Denisova, D.V.; Nikitin, Y.P. Epidemiological evaluation of iodine deficiencyand thyroid disorders in the megalopolis of western Siberia in 1995–2010. Clin. Exp. Thyroid. 2012, 8, 50–54. [Google Scholar] [CrossRef]
- Maksimova, L.L. Structure and Function of the Thyroid Gland Pregnant Women and Newborns, Living in the Conditions of Easy Iodine Deficiency. Ph.D. Thesis, Siberian State Medical University Agency for Health and Social Development, Tomsk, Russia, 2007. (In Russian). [Google Scholar]
- Savchenkov, M.F. Iodine deficiency in population and its medical consequences (according to the research data conducted in Irkutsk city and Irkutsk region). Sib. Med. J. 2009, 1, 65–67. [Google Scholar]
- Savvateeva, V.G.; Okhremchuk, L.V.; Muratova, N.M.; Bogdanova, L.P.; Alsaeva, D.D. Value of iodine deficiency in pregnant women and nursing women. Sib. Med. J. 2002, 30, 66–67. [Google Scholar]
- Nikitina, I.L.; Baranova, T.I. Prevention of iodine-deficiency disorders in the population of Transbaikal region. OSU Bull. 2011, 91–94. [Google Scholar]
- Sirotina, Z.V.; Senkevich, O.A. Microelement imbalance in the formation of the pathology of small newborns in the Far East. Far East. Med. J. 2010, 4, 47–50. [Google Scholar]
- Senkevich, O.A.; Kovalskiy, J.G.; Sirotina, Z.V. Iodine deficit and goiter microcells in hair of women of reproductive age. Far East. Med. J. 2008, 3, 58–62. [Google Scholar]
- World Health Organization. Urinary Iodine Concentrations for Determining Iodine Status in Populations; World Health Organization: Geneva, Switzerland, 2013; Volume 13, pp. 1–5. [Google Scholar]
- Haap, M.; Roth, H.J.; Huber, T.; Dittmann, H.; Wahl, R. Urinary iodine: Comparison of a simple method for its determination in microplates with measurement by inductively-coupled plasma mass spectrometry. Sci. Rep. 2017, 7, 39835. [Google Scholar] [CrossRef]
- Rodriguez, P.A.; Pardue, H.L. Kinetics of the iodide-catalyzed reaction between cerium(IV) and arsenic(III) in sulfuric acid medium. Anal. Chem. 1969, 41, 1369–1376. [Google Scholar] [CrossRef]
- Jooste, P.L.; Strydom, E. Methods for determination of iodine in urine and salt. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 77–88. [Google Scholar] [CrossRef]
- Newsletter “Rybak Kamchatki”. Available online: http://rybak-kamchatky.ru/news/1744-v-rossii-zafiksirovan-rost-potreblenija-ryby-i-rybnyh-produktov.html (accessed on 11 November 2020).
- Samuelsson, G. Salt jodization: Effectively combating iodine deficiency. Food Nutr. Res. 2003, 47, 161. [Google Scholar] [CrossRef][Green Version]
- Manousou, S.; Dahl, L.; Thuesen, B.H.; Hulthén, L.; Nyström, H.F. Iodine deficiency and nutrition in Scandinavia. Minerva Med. 2016, 108, 147–158. [Google Scholar]
- Bromage, S.; Ganmaa, D.; Rich-Edwards, J.W.; Rosner, B.; Bater, J.; Fawzi, W.W. Projected effectiveness of mandatory industrial fortification of wheat flour, milk, and edible oil with multiple micronutrients among Mongolian adults. PLoS ONE 2018, 13, e0201230. [Google Scholar] [CrossRef]
- Deng, J.; Xu, W.-M.; Zhu, X.-X.; Jin, Q.; Huang, Y.; Liu, H.; Jin, X.-Y. [The iodine status in Hangzhou, Zhejiang province 2010]. Zhonghua Liu Xing Bing Xue Za Zhi = Zhonghua Liuxingbingxue Zazhi 2011, 32, 1009–1013. [Google Scholar] [PubMed]
- Mokhort, T.V.; Kolomiets, N.D.; Petrenko, S.; Fedorenko, E.V.; Mokhort, A. Dynamic monitoring of iodine sufficiency in Belarus: Results and problems. Probl. Endocrinol. 2018, 64, 170–179. [Google Scholar] [CrossRef]
- Phedorenko, E.; Kolomiets, N.; Mokhort, T.; Volchenko, A.; Mokhort, E.; Petrenko, S.; Sychik, S. Risk communication as a component that provides stability of strategy aimed at eliminating diseases caused by iodine deficiency in Belarus. Health Risk Anal. 2019, 58–67. [Google Scholar] [CrossRef][Green Version]
- Agunova, L.V. Analysis of meat production of functional use for correction of iodine deficiency. East.-Eur. J. Enterp. Technol. 2015, 2, 9–14. [Google Scholar] [CrossRef][Green Version]
- Aburto, N.; Abudou, M.; Candeias, V.; Tiaxiang, W. Effect and Safety of Salt Iodization to Prevent Iodine Deficiency Disorders: A Systematic Review with Meta-Analyses; WHO eLibrary of Evidence for Nutrition Actions (eLENA); World Health Organization: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Panth, P.; Guerin, G.; DiMarco, N.M. A Review of Iodine Status of Women of Reproductive Age in the USA. Biol. Trace Elem. Res. 2019, 188, 208–220. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, W.; Du, C.; Fan, L.; Wang, W.; Gao, M.; Zhang, Y.; Cui, T.; Hao, Y.; Pearce, E.N.; et al. Iodine Nutrition and Thyroid Function in Pregnant Women Exposed to Different Iodine Sources. Biol. Trace Elem. Res. 2018, 190, 52–59. [Google Scholar] [CrossRef]
- Abel, M.H.; Caspersen, I.H.; Sengpiel, V.; Jacobsson, B.; Meltzer, H.M.; Magnus, P.; Alexander, J.; Brantsæter, A. Insufficient maternal iodine intake is associated with subfecundity, reduced foetal growth, and adverse pregnancy outcomes in the Norwegian Mother, Father and Child Cohort Study. BMC Med. 2020, 18, 1–17. [Google Scholar] [CrossRef]
- Hynes, K.L.; Otahal, P.; Burgess, J.; Oddy, W.H.; Hay, I. Reduced Educational Outcomes Persist into Adolescence Following Mild Iodine Deficiency in Utero, Despite Adequacy in Childhood: 15-Year Follow-Up of the Gestational Iodine Cohort Investigating Auditory Processing Speed and Working Memory. Nutrients 2017, 9, 1354. [Google Scholar] [CrossRef]
- Ahi, S.; Dehdar, M.R.; Hatami, N. Vitamin D deficiency in non-autoimmune hypothyroidism: A case-control study. BMC Endocr. Disord. 2020, 20, 1–6. [Google Scholar] [CrossRef]
- Lunze, K.; Yurasova, E.; Idrisov, B.; Gnatienko, N.; Migliorini, L. Food security and nutrition in the Russian Federation – a health policy analysis. Glob. Health Action 2015, 8, 27537. [Google Scholar] [CrossRef]
- AMAP Assessment 2015: Human Health in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2015.
- Prado, E.L.; Dewey, K.G. Nutrition and brain development in early life. Nutr. Rev. 2014, 72, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B. The Influence of Iron Status on Iodine Utilization and Thyroid Function. Annu. Rev. Nutr. 2006, 26, 367–389. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Luzuriaga, M.J.; Larson, L.M.; Mannar, V.; Martorell, R. Impact of Double-Fortified Salt with Iron and Iodine on Hemoglobin, Anemia, and Iron Deficiency Anemia: A Systematic Review and Meta-Analysis. Adv. Nutr. 2018, 9, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Berg, V.; Nøst, T.H.; Pettersen, R.D.; Hansen, S.; Veyhe, A.-S.; Jorde, R.; Odland, J. Øyvind; Sandanger, T.M. Persistent Organic Pollutants and the Association with Maternal and Infant Thyroid Homeostasis: A Multipollutant Assessment. Environ. Health Perspect. 2017, 125, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Dietz, R.; Letcher, R.J.; Desforges, J.-P.; Eulaers, I.; Sonne, C.; Wilson, S.; Andersen-Ranberg, E.; Basu, N.; Barst, B.D.; Bustnes, J.O.; et al. Current state of knowledge on biological effects from contaminants on arctic wildlife and fish. Sci. Total Environ. 2019, 696, 133792. [Google Scholar] [CrossRef]
- Cetin, I.; Bühling, K.; Demir, C.; Kortam, A.; Prescott, S.L.; Yamashiro, Y.; Yarmolinskaya, M.; Koletzko, B. Impact of Micronutrient Status during Pregnancy on Early Nutrition Programming. Ann. Nutr. Metab. 2019, 74, 269–278. [Google Scholar] [CrossRef]
- Resolution of the Chief State Sanitary Doctor of the Russian Federation of July 23, 2008 N 45 “Sanitary and epidemiological requirements for catering of students in education institutions, institutions of primary and secondary professional education”. Available online: http://www.consultant.ru/document/cons_doc_LAW_79255/92d969e26a4326c5d02fa79b8f9cf4994ee5633b/ (accessed on 11 November 2020).
- Draft Federal Law “On the Prevention of Iodine Deficiency Disorders” of March 27, 2019. Available online: https://www.endocrincentr.ru/sites/default/files/all/prikaz_doc/01.04.19/iod_rek.pdf (accessed on 1 October 2020).
| Area of Residence | Data | Subjects | Median UIC (μg/L) | Analytical Method | References | |
|---|---|---|---|---|---|---|
| Year of Publication | Year of Sample Collection | |||||
| Moscow region | ||||||
| * | 2011 | 75 pregnant women on I trimester (aged 18–42 years) (using nutritional supplements) | 128 | Cerium-arsenite reaction | [19] | |
| * | 2010 | Initially 75 pregnant women | 128 | Cerium-arsenite reaction | [20] | |
| Group 1: 59 pregnant women receiving IS a 200 μg/day as KI | 124 | |||||
| Group 2: 16 pregnant women receiving IS a 300 μg/day as KI | 196 | |||||
| Nursing mothers from Group 1 | 118 | |||||
| Nursing mothers from Group 2 | 82 | |||||
| 16 infants of mothers from Group 1 | 180 | |||||
| 7 infants of mothers from Group 2 | 200 | |||||
| Ivanovo region | ||||||
| Ivanovo city | 2005 | 84 pregnant women | 93 | [21] | ||
| Ivanovo city | 2008 | 2001–2003 | 84 pregnant women aged 26 ± 4 years | Colorimetric | [22] | |
| 14 I trimester | 116 | |||||
| 25 II trimester | 82 | |||||
| 45 III trimester | 94 | |||||
| Smolensk region | ||||||
| Smolensk city | 2008 | 2004 | 150 pregnant women aged 25 ± 5 years | Colorimetric | [22] | |
| 50 I trimester | 93 | |||||
| 50 II trimester | 86 | |||||
| 50 III trimester | 51 | |||||
| * | 2011 | 119 pregnant women on I trimester (aged 18–42 years) | 63 | Cerium-arsenite reaction | [19] | |
| (taken nutritional supplements) | ||||||
| * | 2010 | Initially 119 pregnant women | 63 | Cerium-arsenite reaction | [20] | |
| Group 1: 50 pregnant women receiving IS a 200 μg/day as KI | 84 | |||||
| Group 2: 69 pregnant women receiving IS a 300 μg/day as KI | 121 | |||||
| Nursing mothers from Group 1 | 41 | |||||
| Nursing mothers from Group 2 | 70 | |||||
| 22 infants of mothers from Group 1 | 174 | |||||
| 19 infants of mothers from Group 2 | 136 | |||||
| Tatarstan republic | ||||||
| 2008 | 2003 | 180 pregnant women (aged 25 ± 5 years) | Colorimetric | [22] | ||
| Al’met’evsk | 92 pregnant women: | 112 | ||||
| (54.9 °N, 52.17 °E) | 13 I trimester | 102 | ||||
| 26 II trimester | 129 | |||||
| 53 III trimester | 110 | |||||
| Nizhnekamsk | 88 pregnant women: | 164 | ||||
| (55.63 °N, 51.82 °E) | 21 I trimester | 155 | ||||
| 36 II trimester | 148 | |||||
| 31 III trimester | 192 | |||||
| * | 2005 | 182 pregnant women | 150 | [21] | ||
| Kirov region | ||||||
| Kirov city | 2005 | 2004 | 92 pregnant women (aged 26 ± 4 years) | Colorimetric | [22,23] | |
| 25 I trimester | 69 | |||||
| 32 II trimester | 87 | |||||
| 35 III trimester | 73 | |||||
| * | 2004 | 92 pregnant women | 73 | [21] | ||
| Nizhny Novgorod region | ||||||
| * | 2011 | 220 pregnant women on I trimester (aged 18–42 years) | 141 | Cerium-arsenite reaction | [19] | |
| * | 2010 | Initially 220 pregnant women | 141 | Cerium-arsenite reaction | [20] | |
| Group 1: 111 pregnant women receiving IS a 200 μg/day as KI | 97 | |||||
| Group 2: 109 pregnant women receiving IS a 300 μg/day as KI | 260 | |||||
| Nursing mothers from Group 1 | 77 | |||||
| Nursing mothers from Group 2 | 107 | |||||
| 99 infants of mothers from Group 1 | 110 | |||||
| 93 infants of mothers from Group 2 | 150 | |||||
| Chuvash republic | ||||||
| Cheboksary | 2007 | 98 pregnant women | 83 | [21] | ||
| (56.12 °N, 47.23 °E) | ||||||
| Novocheboksarsk | ||||||
| (56.12 °N, 47.49 °E) | ||||||
| Cheboksary | 2005 | 2003 | 96 pregnant women (aged 26 ± 6 years) | 83 | Colorimetric | [22,23] |
| 62 pregnant women | 83 | |||||
| 12 I trimester | 180 | |||||
| 23 II trimester | 94 | |||||
| 27 III trimester | 78 | |||||
| Novocheboksarsk | 34 pregnant women | 77 | ||||
| 13 I trimester | 76 | |||||
| 10 II trimester | 84 | |||||
| 11 III trimester | 68 | |||||
| Bashkortostan republic | ||||||
| Ufa city | 2004 | 2000–2003 | Non-pregnant | 127 | Cerium-arsenite reaction b | [24] |
| Pregnant women receiving IS a | ||||||
| I trimester | 143 | |||||
| II trimester | 120 | |||||
| Saratov region | ||||||
| Engel’s | 2010 | Group 1: 62 pregnant women | 115 | Cerium-arsenite reaction | [25] | |
| (51.46 °N, 46.12 °E) | Group 2: 54 pregnant women receiving IS a | 177 | ||||
| Engel’s | 2010 | Group 1: a random of 106 pregnant women | 116 | Cerium-arsenite reaction | [26] | |
| Group 2: 90 pregnant women receiving IS a | 164 | |||||
| * | 2003 | 123 pregnant women did not receive IS a | 33 | Cerium-arsenite reaction c | [27] | |
| 120 pregnant women who received IS a | 134 | |||||
| St. Petersburg | ||||||
| St. Petersburg city | 2017 | 2013-2015 | 184 pregnant women (aged 18-45 years) | 112 | Cerium-arsenite reaction d | [28] |
| Tyumen region | ||||||
| * | 2015 | 1999 | Pregnant women | 93 | Cerium-arsenite reaction | [29] |
| 2009 | Pregnant women | 124 | ||||
| Kabardino-Balkar republic | ||||||
| Nalchik city | 2007 | Pregnant women | 67 | [21] | ||
| Krasnodar territory | ||||||
| Slavyansk-on-Kuban’ | 2005 | 2003 | 120 pregnant women (aged 26 ± 6 years) | Colorimetric | [22,23] | |
| (45.26 °N, 38.12 °E) | 17 I trimester | 98 | ||||
| Anapa | 46 II trimester | 91 | ||||
| (44.89 °N, 37.32 °E) | 57 III trimester | 95 | ||||
| sta. Kanevskaya | ||||||
| (46.05 °N, 38.95 °E) | ||||||
| * | 2005 | 121 pregnant women | 90 | [21] | ||
| Astrakhan region | ||||||
| Astrakhan city | 2010 | 2006-2009 | 68 pregnant women with IS a | 176 | Cerium-arsenite reaction e | [30] |
| infants | 95 | |||||
| I trimester | 63 | |||||
| II trimester | 50 | |||||
| III trimester | 27 | |||||
| 67 pregnant women in the III trimester without IS a | 49 | |||||
| infants | 39 | |||||
| Adygea republic | ||||||
| Majkop | 2005 | 60 pregnant women | 84 | [21] | ||
| (44.36 °N, 40.60 °E) | ||||||
| Majkop | 2005 | 2005 | 60 pregnant women (aged 26 ± 5 years | 84 | Colorimetric | [22] |
| 32 pregnant women with IS a | 115 | |||||
| 28 pregnant women without IS a | 73 | |||||
| Rostov region | ||||||
| * | 2005 | 303 pregnant women | 95 | [21] | ||
| * | 2005 | 2004 | 299 pregnant women (aged 25 ± 5 years) | 95 | Colorimetric | [22] |
| 55 I trimester | 104 | |||||
| 107 II trimester | 99 | |||||
| 137 III trimester | 95 | |||||
| Novosibirsk region | ||||||
| Novosibirsk city | 1994–1995 | 200 men and women (aged 25–34 years) | 47 | Cerium-arsenite reaction | [31] | |
| Tomsk region | ||||||
| * | 2007 | 2003-2005 | 238 pregnant women (aged years 27) of whom received IS a | Potentiometric f | [32] | |
| I trimester | 65 | |||||
| II trimester | 96 | |||||
| III trimester | 70 | |||||
| 30 pregnant women of the control group III trimester without IS a | 60 | |||||
| Irkutsk region | ||||||
| Irkutsk city | 2009 | Pregnant women | 48 | [33] | ||
| Lactating women | 75 | |||||
| Lactating women with thyroid goiter | 50 | |||||
| * | 2002 | 150 pregnant women | 60 | Cerium-arsenite reaction | [34] | |
| Zabaykalskyterritory | ||||||
| * | 2011 | Pregnant women | 128 | [35] | ||
| Khabarovsk territory | ||||||
| Khabarovsk city | 2010 | Mother (25)- | 55 | Cerium-arsenite reaction g | [36] | |
| (48.48 °N, 135.07 °E) | infant (25) pairs | 69 | ||||
| Komsomol’sk-on- | Mother (30)– | 26 | ||||
| Amur | infant (31) pairs | 64 | ||||
| (50.55 °N, 137 °E) | Mother (19)- | 20 | ||||
| Amursk | infant (17) pairs | 75 | ||||
| (50.22 °N, 136.9 °E) | Mother (21)- | 75 | ||||
| Nikolaevsk-on-Amur | infant (21) pairs | 86 | ||||
| (53.15 °N, 140.73 °E) | Mother (19)- | 31 | ||||
| s. Vanino | infant pairs (20) | 96 | ||||
| (49.08 °N, 140.27 °E) | ||||||
| Khabarovsk city | 2008 | 30 healthy women in the early postpartum period | 55 | Cerium-arsenite reaction g | [37] | |
| and 22 non-pregnant women | 69 | |||||
| Jewish Autonomous region | ||||||
| Birobidzhan city | 2010 | Mother (20)- | 27 | Cerium-arsenite reaction g | [36] | |
| infant (22) pairs | 25 | |||||
| Sakha (Yakutiya) republic | ||||||
| Neryungri | 2010 | Mother (23)- | 69 | Cerium-arsenite reaction g | [36] | |
| (56.66 °N, 124.73 °E) | infant (23) pairs | 67 | ||||
| Sakhalin region | ||||||
| Oha | 2010 | Mother (26)- | 49 | Cerium-arsenite reaction g | [36] | |
| (53.58 °N, 142.93 °E) | infant (24) pairs | 61 | ||||
| Kamchatka territory | ||||||
| Petropavlovsk-Kamchatsky city | 2010 | Mother (20)– | 155 | Cerium-arsenite reaction g | [36] | |
| infant (23) pairs | 190 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korobitsyna, R.; Aksenov, A.; Sorokina, T.; Trofimova, A.; Sobolev, N.; Grjibovski, A.M.; Chashchin, V.; Thomassen, Y. Iodine Status of Women and Infants in Russia: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 8346. https://doi.org/10.3390/ijerph17228346
Korobitsyna R, Aksenov A, Sorokina T, Trofimova A, Sobolev N, Grjibovski AM, Chashchin V, Thomassen Y. Iodine Status of Women and Infants in Russia: A Systematic Review. International Journal of Environmental Research and Public Health. 2020; 17(22):8346. https://doi.org/10.3390/ijerph17228346
Chicago/Turabian StyleKorobitsyna, Rimma, Andrey Aksenov, Tatiana Sorokina, Anna Trofimova, Nikita Sobolev, Andrej M Grjibovski, Valery Chashchin, and Yngvar Thomassen. 2020. "Iodine Status of Women and Infants in Russia: A Systematic Review" International Journal of Environmental Research and Public Health 17, no. 22: 8346. https://doi.org/10.3390/ijerph17228346
APA StyleKorobitsyna, R., Aksenov, A., Sorokina, T., Trofimova, A., Sobolev, N., Grjibovski, A. M., Chashchin, V., & Thomassen, Y. (2020). Iodine Status of Women and Infants in Russia: A Systematic Review. International Journal of Environmental Research and Public Health, 17(22), 8346. https://doi.org/10.3390/ijerph17228346





