Hypertension Is Associated with Increased Risk of Diabetic Lung
Abstract
1. Introduction
2. Materials and Methods
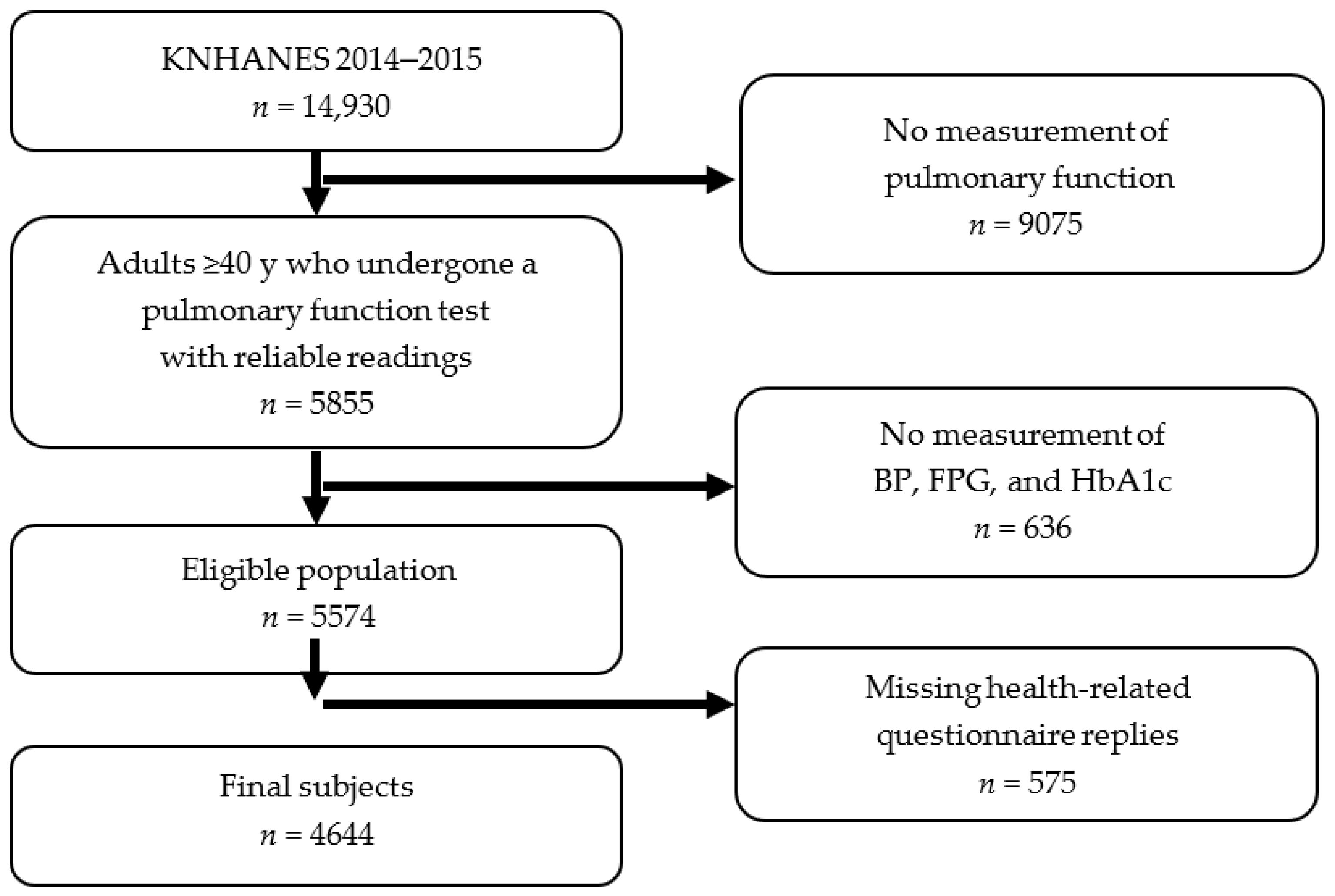
2.1. Study Population and KNHANES
2.2. Pulmonary Function Test
2.3. Diabetes and Impaired Fasting Glucose
2.4. Hypertension and Prehypertension
2.5. Height, Weight, Waist Circumference, and Body Mass Index
2.6. Handgrip, Smoking History, Alcohol Consumption, and Physical Activity
2.7. Statistical Analysis
2.8. Ethical Statement
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Klein, O.L.; Meltzer, D.; Carnethon, M.; Krishnan, J.A. Type II diabetes mellitus is associated with decreased measures of lung function in a clinical setting. Respir. Med. 2011, 105, 1095–1098. [Google Scholar] [CrossRef]
- Van den Borst, B.; Gosker, H.R.; Zeegers, M.P.; Schols, A.M. Pulmonary function in diabetes: A metaanalysis. Chest 2010, 138, 393–406. [Google Scholar] [CrossRef]
- Walter, R.E.; Beiser, A.; Givelber, R.J.; O’Connor, G.T.; Gottlieb, D.J. Association between glycemic state and lung function: The Framingham Heart Study. Am. J. Respir. Crit. Care Med. 2003, 167, 911–916. [Google Scholar] [CrossRef]
- Yeh, H.-C.; Punjabi, N.M.; Wang, N.-Y.; Pankow, J.S.; Duncan, B.B.; Cox, C.E.; Selvin, E.; Brancati, F.L. Cross-sectional and prospective study of lung function in adults with type 2 diabetes: The Atherosclerosis Risk in Communities (ARIC) study. Diabetes Care 2008, 31, 741–746. [Google Scholar] [CrossRef]
- Shah, S.H.; Sonawane, P.; Nahar, P.; Vaidya, S.; Salvi, S. Pulmonary function tests in type 2 diabetes mellitus and their association with glycemic control and duration of the disease. Lung India 2013, 30, 108–112. [Google Scholar] [CrossRef]
- Sandler, M. Is the lung a ‘target organ’ in diabetes mellitus? Arch. Intern. Med. 1990, 150, 1385–1388. [Google Scholar] [CrossRef] [PubMed]
- Rask-Madsen, C.; King, G.L. Vascular complications of diabetes: Mechanisms of injury and protective factors. Cell Metab. 2013, 17, 20–33. [Google Scholar] [CrossRef]
- Kannel, W.B.; Hubert, H.; Lew, E.A. Vital capacity as a predictor of cardiovascular disease: The Framingham study. Am. Heart J. 1983, 105, 311–315. [Google Scholar] [CrossRef]
- Schnabel, E.; Nowak, D.; Brasche, S.; Wichmann, H.-E.; Heinrich, J. Association between lung function, hypertension and blood pressure medication. Respir. Med. 2011, 105, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Vollmer, W.M.; Buist, A.S.; Tsai, R.; Cen, R.; Wu, X.; Chen, P.; Li, Y.; Guo, C.; Mai, J.; et al. Relationship between lung function and blood pressure in Chinese men and women of Beijing and Guangzhou. PRC-USA Cardiovascular and Cardiopulmonary Epidemiology Research Group. Int. J. Epidemiol. 1998, 27, 49–56. [Google Scholar] [CrossRef]
- Lee, H.M.; Zhao, Y.; Liu, M.A.; Yanez, D.; Carnethon, M.; Graham Barr, R.; Wong, N.D. Impact of lung--function measures on cardiovascular disease events in older adults with metabolic syndrome and diabetes. Clin. Cardiol. 2018, 41, 959–965. [Google Scholar] [CrossRef]
- Engström, G.; Hedblad, B.; Valind, S.; Janzon, L. Increased incidence of myocardial infarction and stroke in hypertensive men with reduced lung function. J. Hypertens. 2001, 19, 295–301. [Google Scholar] [CrossRef] [PubMed]
- De Boer, I.H.; Bangalore, S.; Benetos, A.; Davis, A.M.; Michos, E.D.; Muntner, P.; Rossing, P.; Zoungas, S.; Bakris, G. Diabetes and hypertension: A position statement by the American Diabetes Association. Diabetes Care 2017, 40, 1273–1284. [Google Scholar] [CrossRef]
- Kweon, S.; Kim, Y.; Jang, M.-J.; Kim, Y.; Kim, K.; Choi, S.; Chun, C.; Khang, Y.-H.; Oh, K. Data resource profile: The Korea National Health and Nutrition Examination Survey (KNHANES). Int. J. Epidemiol. 2014, 43, 69–77. [Google Scholar] [CrossRef]
- Choi, J.K.; Paek, D.; Lee, J.O. Normal predictive values of spirometry in Korean population. Tuberc. Respir. Dis. 2005, 58, 230–242. [Google Scholar] [CrossRef]
- Quanjer, P.H.; Stanojevic, S.; Cole, T.J.; Baur, X.; Hall, G.L.; Culver, B.H.; Enright, P.L.; Hankinson, J.L.; Ip, M.S.; Zheng, J.; et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: The global lung function 2012 equations. Eur. Respir. J. 2012, 40, 1324–1343. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2009, 32 (Suppl. S1), S62–S67. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jung, A. Optimum cutoff value of urinary cotinine distinguishing South Korean adult smokers from nonsmokers using data from the KNHANES (2008–2010). Nicotine Tob. Res. 2013, 15, 1608–1616. [Google Scholar] [CrossRef]
- World Health Organization. European Action Plan to Reduce the Harmful Use of Alcohol 2012–2020. 2020. Available online: https://www.euro.who.int/__data/assets/pdf_file/0008/178163/E96726.pdf (accessed on 9 September 2020).
- El-Azeem, A.A.; Hamdy, G.; Amin, M.; Rashad, A. Pulmonary function changes in diabetic lung. Egypt. J. Chest Dis. Tuberc. 2013, 62, 513–517. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kim, N.K.; Yang, J.Y.; Noh, J.H.; Lee, S.-S.; Ko, K.S.; Rhee, B.D.; Kim, D.-J. Low pulmonary function in individuals with impaired fasting glucose: The 2007–2009 Korea national health and nutrition examination survey. PLoS ONE 2013, 8, e76244. [Google Scholar] [CrossRef] [PubMed]
- Kopf, S.; Groener, J.B.; Kender, Z.; Fleming, T.; Brune, M.; Riedinger, C.; Volk, N.; Herpel, E.; Pesta, D.; Szendrödi, J.; et al. Breathlessness and restrictive lung disease: An important diabetes-related feature in patients with type 2 diabetes. Respiration 2018, 96, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.V.; Shaw, L.C.; Grant, M.B. Inflammation in the pathogenesis of microvascular complications in diabetes. Front. Endocrinol. 2012, 3, 170. [Google Scholar] [CrossRef] [PubMed]
- Fisman, E.Z.; Tenenbaum, A. Cardiovascular Diabetology: Clinical, Metabolic and Inflammatory Facets; Karger Medical and Scientific Publishers: Basel, Switzerland, 2008; Volume 45, p. 44. [Google Scholar]
- Lago, R.M.; Singh, P.P.; Nesto, R.W. Diabetes and hypertension. Nat. Clin. Pr. Endocrinol. Metab. 2007, 3, 667. [Google Scholar] [CrossRef] [PubMed]
- Petrie, J.R.; Guzik, T.J.; Touyz, R.M. Diabetes, hypertension, and cardiovascular disease: Clinical insights and vascular mechanisms. Can. J. Cardiol. 2018, 34, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Savoia, C.; Sada, L.; Zezza, L.; Pucci, L.; Lauri, F.M.; Befani, A.; Alonzo, A.; Volpe, M. Vascular inflammation and endothelial dysfunction in experimental hypertension. Int. J. Hypertens. 2011, 2011, 281240. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Oliveira, A., Jr.; Nogueira, A.I.; Pereira, R.M.; Boas, W.W.; Dos Santos, R.A.; e Silva, A.C. The renin–angiotensin system and diabetes: An update. Vasc. Health Risk Manag. 2008, 4, 787–803. [Google Scholar]
- Manrique, C.; Lastra, G.; Gardner, M.; Sowers, J.R. The renin angiotensin aldosterone system in hypertension: Roles of insulin resistance and oxidative stress. Med. Clin. N. Am. 2009, 93, 569–582. [Google Scholar] [CrossRef]
- De Miguel, C.; Rudemiller, N.P.; Abais, J.M.; Mattson, D.L. Inflammation and hypertension: New understandings and potential therapeutic targets. Curr. Hypertens. Rep. 2015, 17, 507. [Google Scholar] [CrossRef]
- Rodríguez-Iturbe, B.; Pons, H.; Quiroz, Y.; Johnson, R.J. The immunological basis of hypertension. Am. J. Hypertens. 2014, 27, 1327–1337. [Google Scholar] [CrossRef]
- McMaster, W.G.; Kirabo, A.; Madhur, M.S.; Harrison, D.G. Inflammation, immunity, and hypertensive end-organ damage. Circ. Res. 2015, 116, 1022–1033. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Mainous, A.G., 3rd; Buchanan, T.A. Pearson WS. C-reactive protein and glycemic control in adults with diabetes. Diabetes Care 2003, 26, 1535–1539. [Google Scholar] [CrossRef]
- Magen, E.; Mishal, J.; Paskin, J.; Glick, Z.; Yosefy, C.; Kidon, M.; Schlesinger, M. Resistant arterial hypertension is associated with higher blood levels of complement C3 and C--reactive protein. J. Clin. Hypertens. 2008, 10, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Dinh, Q.N.; Drummond, G.R.; Sobey, C.G.; Chrissobolis, S. Roles of inflammation, oxidative stress, and vascular dysfunction in hypertension. Biomed. Res. Int. 2014, 2014, 406960. [Google Scholar] [CrossRef] [PubMed]
- Margretardottir, O.B.; Thorleifsson, S.J.; Gudmundsson, G.; Olafsson, I.; Benediktsdottir, B.; Janson, C.; Buist, A.S.; Gislason, T. Hypertension, systemic inflammation and body weight in relation to lung function impairment—an epidemiological study. COPD 2009, 6, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S.; Vaz Fragoso, C.A.; Van Ness, P.H.; Fried, L.P.; Tinetti, M.E. Association between combined interleukin--6 and C--reactive protein levels and pulmonary function in older women: Results from the Women’s Health and Aging Studies I and II. J. Am. Geriatr. Soc. 2011, 59, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Andersson, T.; Alfredsson, L.; Källberg, H.; Zdravkovic, S.; Ahlbom, A. Calculating measures of biological interaction. Eur. J. Epidemiol. 2005, 20, 575–579. [Google Scholar] [CrossRef]
| Characteristics Mean ± STD or n (%) | Normal FPG; A (n = 1803) | IFG; B (n = 2175) | DM; C (n = 666) | p-Value * (Post Hoc) |
|---|---|---|---|---|
| Sex | ||||
| Male | 681 (32.1) | 1060 (50.0) | 379 (17.9) | <0.001 (A<B<C) |
| Female | 1122 (44.5) | 1115 (44.1) | 287 (11.4) | <0.001B>C) |
| Age | 54.01 ± 10.21 | 58.74 ± 10.25 | 61.71 ± 9.75 | <0.001 (A<B<C) |
| FPG (mg/dL) | 90.00 ± 5.66 | 100.95 ± 9.30 | 145.49 ± 40.75 | <0.001 |
| HbA1c (%) | 5.35 ± 0.21 | 5.80 ± 0.30 | 7.32 ± 1.16 | <0.001 |
| BMI (kg/m2) | 23.26 ± 2.78 | 24.56 ± 3.06 | 25.15 ± 3.19 | <0.001 |
| Waist circumference (cm) | 79.98 ± 8.42 | 84.56 ± 8.71 | 87.48 ± 8.61 | <0.001 (A<B<C) |
| Lifetime smoking status | ||||
| Nonsmoker | 1169 (42.7) | 1233 (45.1) | 334 (12.2) | <0.001 (A>B>C) |
| Former smoker | 374 (33.3) | 547 (48.7) | 202 (18.0) | <0.001 (A<B<C) |
| Current smoker | 260 (32.7) | 395 (49.7) | 140 (17.6) | <0.001 (A<B=C) |
| Regular physical activity | ||||
| Yes | 907 (50.3) | 1030 (47.4) | 309 (46.4)) | 0.098 |
| No | 896 (40.4) | 1145 (45.9) | 357 (13.8) | |
| Heavy drinking | 310 (17.2) | 436 (20.0) | 146 (21.5) | 0.022 (A<B=C) |
| Handgrip strength | 30.90 ± 9.35 | 31.81 ± 9.57 | 31.75 ± 9.34 | 0.007 (A<B=C) |
| Blood pressure | ||||
| Systolic BP (mmHg) | 117.23 ±16.30 | 122.16 ± 16.71 | 125.21 ± 15.19 | <0.001 (A<B<C) |
| Diastolic BP (mmHg) | 75.84 ± 9.91 | 76.65 ± 9.99 | 75.42 ± 10.37 | 0.005 (A=C<B) |
| Blood pressure status | ||||
| Normal BP | 937 (52.0) | 883 (40.6) | 213 (32.0) | <0.001 (A>B>C) |
| Prehypertension | 604 (33.5) | 874 (40.2) | 312 (46.8) | <0.001 (A<B<C) |
| Hypertension | 262 (14.5) | 418 (19.2) | 141 (21.2) | <0.001 (A<B=C) |
| Pulmonary function pattern | ||||
| Restrictive (FVCp < 80%) | 113 (6.3) | 266 (12.3) | 150 (22.1) | <0.001 (A<B<C) |
| Obstructive (FEV1/FVC < 0.7) | 196 (10.9) | 356 (16.5) | 142 (20.9) | <0.001 (A<B<C) |
| Crude Model | Adjusted * | |||
|---|---|---|---|---|
| β | p-Value | β | p-Value | |
| FVCp (%) | ||||
| Normal FPG | 1 (Ref) | 1 (Ref) | ||
| IFG | −3.020 | <0.001 | −1.496 | <0.001 |
| DM | −6.342 | <0.001 | −3.639 | <0.001 |
| Normal BP | 1 (Ref) | 1 (Ref) | ||
| Prehypertension | −1.515 | <0.001 | −0.320 | 0.395 |
| Hypertension | −2.832 | <0.001 | −1.528 | 0.001 |
| FEV1p (%) | ||||
| Normal FPG | 1 (Ref) | 1 (Ref) | ||
| IFG | −2.086 | <0.001 | −1.628 | <0.001 |
| DM | −4.605 | <0.001 | −3.422 | <0.001 |
| Normal BP | 1 (Ref) | 1 (Ref) | ||
| Prehypertension | −0.445 | 0.311 | −0.204 | 0.647 |
| Hypertension | −0.678 | 0.225 | −0.621 | 0.274 |
| FEV1/FVC | ||||
| Normal FPG | 1 (Ref) | 1 (Ref) | ||
| IFG | −0.016 | <0.001 | 0.001 | 0.683 |
| DM | −0.024 | <0.001 | 0.004 | 0.171 |
| Normal BP | 1 (Ref) | 1 (Ref) | ||
| Prehypertension | −0.013 | <0.001 | 0.002 | 0.435 |
| Hypertension | −0.010 | 0.001 | 0.007 | 0.008 |
| Crude | Adjusted * | |
|---|---|---|
| Subgroups | ||
| Normal BP–Normal FPG (n = 1541) | 1 (Ref) | 1 (Ref) |
| Normal BP–IFG (n = 1757) | 2.21 (1.70–2.87) | 1.49 (1.13–1.97) |
| Normal BP–DM (n = 525) | 4.33 (3.20–5.87) | 2.30 (1.66–3.18) |
| HTN–Normal FPG (n = 262) | 1.94 (1.24–3.06) | 1.45 (0.91–2.33) |
| HTN–IFG (n = 418) | 3.23 (2.31–4.54) | 1.93 (1.34–2.77) |
| HTN–DM (n = 141) | 6.47 (4.22–9.93) | 3.30 (2.09–5.22) |
| Age | 1.02 (1.00–1.03) | |
| Sex | ||
| Male | 1 (Ref) | |
| Female | 0.21 (0.14–0.31) | |
| BMI | 1.11 (1.05–1.18) | |
| Waist circumference | 1.02 (1.00–1.05) | |
| Smoking history | ||
| Nonsmoker | 1 (Ref) | |
| Former smoker | 0.83 (0.61–1.13) | |
| Current smoker | 0.99 (0.71–1.38) | |
| Aerobic physical activity engagement | 0.77 (0.64–0.94) | |
| Drinking history | ||
| Not a heavy drinker | 1 (Ref) | |
| Heavy drinker | 0.66 (0.73–1.23) | |
| Handgrip strength | 0.92 (0.91–0.94) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Kwon, D.; Lee, Y.; Jung, I.; Hyun, D.; Lee, H.; Ahn, Y.-S. Hypertension Is Associated with Increased Risk of Diabetic Lung. Int. J. Environ. Res. Public Health 2020, 17, 7513. https://doi.org/10.3390/ijerph17207513
Lee J, Kwon D, Lee Y, Jung I, Hyun D, Lee H, Ahn Y-S. Hypertension Is Associated with Increased Risk of Diabetic Lung. International Journal of Environmental Research and Public Health. 2020; 17(20):7513. https://doi.org/10.3390/ijerph17207513
Chicago/Turabian StyleLee, Jihyun, Donghwan Kwon, Youngjang Lee, Inchan Jung, Daesung Hyun, Hunju Lee, and Yeon-Soon Ahn. 2020. "Hypertension Is Associated with Increased Risk of Diabetic Lung" International Journal of Environmental Research and Public Health 17, no. 20: 7513. https://doi.org/10.3390/ijerph17207513
APA StyleLee, J., Kwon, D., Lee, Y., Jung, I., Hyun, D., Lee, H., & Ahn, Y.-S. (2020). Hypertension Is Associated with Increased Risk of Diabetic Lung. International Journal of Environmental Research and Public Health, 17(20), 7513. https://doi.org/10.3390/ijerph17207513






