Gingival Crevicular Blood as a Potential Screening Tool: A Cross Sectional Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Governance and Ethics
2.3. Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Müller, H.P.; Behbehani, E. Methods for measuring agreement: Glucose levels in gingival crevice blood. Clin. Oral Investig. 2005, 9, 65–69. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus position statement. Diabetes Care 2005, 29, 537–542. [Google Scholar]
- Ardakani, M.R.; Moeintaghavi, A.; Haerian, A.; Ardakani, M.A.; Hashemzadeh, M. Correlation between levels of sulcular and capillary blood glucose. J. Contemp. Dent. Pract. 2009, 10, 10–17. [Google Scholar] [PubMed]
- Strauss, S.M.; Wheeler, A.J.; Russell, S.L.; Brodsky, A.; Davidson, R.M.; Gluzman, R.; Li, L.; Malo, R.G.; Salis, B.; Schoor, R.; et al. The potential use of gingival crevicular blood for measuring glucose to screen for diabetes: An examination based on characteristics of the blood collection site. J. Periodontol. 2009, 80, 907–914. [Google Scholar] [CrossRef]
- Kempe, K.C.; Budd, D.; Stern, M.; Ellison, J.M.; Saari, L.A.; Adiletto, C.A.; Olin, B.; Price, D.A.; Horwitz, D.L. Palm glucose readings compared with fingertip readings under steady and dynamic glycemic conditions, using the OneTouch Ultra Blood Glucose Monitoring System. Diabetes Technol. Ther. 2005, 7, 916–926. [Google Scholar] [CrossRef] [PubMed]
- Müller, H.P.; Behbehani, E. Screening of elevated glucose levels in gingival crevice blood using a novel, sensitive self-monitoring device. Med. Princ. Pract. 2004, 13, 361–525. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Kohad, R.; Ramreddy, Y.; Shetty, K. Gingival blood glucose estimation with reagent test strips: A method to detect diabetes in a periodontal population. J. Periodontol. 2011, 82, 1548–1555. [Google Scholar] [CrossRef]
- Verma, S.; Bhat, K.M. Diabetes mellitus—A modifier of periodontal disease expression. J. Int. Acad. Periodontol. 2004, 6, 13–20. [Google Scholar]
- Rhodes, C.J. Type 2 diabetes-a matter of beta-cell life and death? Science 2005, 307, 380–384. [Google Scholar] [CrossRef]
- Wadia, R. Screening for diabetes using gingival crevicular blood? Br. Dent. J. 2020, 229, 125. [Google Scholar] [CrossRef] [PubMed]
- Ellison, J.M.; Stegmann, J.M.; Colner, S.L.; Michael, R.H.; Sharma, M.K.; Ervin, K.R.; Horwitz, D.L. Rapid changes in postprandial blood glucose produce concentration differences at finger, forearm, and thigh sampling sites. Diabetes Care 2002, 25, 961–964. [Google Scholar] [CrossRef] [PubMed]
- Khader, Y.; Al-Zu’bi, B.; Judeh, A.; Rayyan, M. Screening for type 2 diabetes mellitus using gingival crevicular blood. Int. J. Dent. Hyg. 2006, 4, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Cutler, C.W.; Machen, R.L.; Jotwani, R.; Iacopino, A.M. Heightened gingival inflammation and attachment loss in type 2 diabetics with hyperlipidemia. J. Periodontol. 1999, 70, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Stein, G.M.; Nebbia, A.A. Chairside method of diabetic screening with gingival blood. Oral Surg. Oral Med. Oral Pathol. 1969, 27, 607–612. [Google Scholar] [CrossRef]
- Shetty, N.; Shankarapillai, R.; Mathur, L.K.; Manohar, B.; Mathur, A.; Jain, M. Gingival crevicular blood: As a non-invasive screening tool for diabetes mellitus in dental clinics. J. Indian Soc. Periodontol. 2013, 17, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Ervasti, T.; Knuuttila, M.; Pohjamo, L.; Haukipuro, K. Relation between control of diabetes and gingival bleeding. J. Periodontol. 1985, 56, 154–157. [Google Scholar] [CrossRef]
- Jungheim, K.; Koschinsky, T. Glucose monitoring at the arm: Risky delays of hypoglycemia and hyperglycemia detection. Diabetes Care 2002, 25, 956–960. [Google Scholar] [CrossRef]
- Holt, G.R. Declaration of Helsinki—The World’s Document of Conscience and Responsibility. South. Med. J. 2014, 107, 407. [Google Scholar] [CrossRef]
- Rapone, B.; Ferrara, E.; Corsalini, M.; Converti, I.; Grassi, F.R.; Santacroce, L.; Topi, S.; Gnoni, A.; Scacco, S.; Scarano, A.; et al. The Effect of Gaseous Ozone Therapy in Conjunction with Periodontal Treatment on Glycated Hemoglobin Level in Subjects with Type 2 Diabetes Mellitus: An Unmasked Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 5467. [Google Scholar] [CrossRef]
- Grassi, F.R.; Rapone, B.; Scarano Catanzaro, F.; Corsalini, M.; Kalemaj, Z. Effectiveness of computer-assisted anesthetic delivery system (STA™) in dental implant surgery: A prospective study. Oral Implantol. 2017, 10, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Beikler, T.; Kuczek, A.; Petersilka, G.; Flemmig, T.F. In-dental-office screening for diabetes mellitus using gingival crevicular blood. J. Clin. Periodontol. 2002, 29, 216–218. [Google Scholar] [CrossRef]
- Sarlati, F.; Pakmehr, E.; Khoshru, K.; Akhondi, N. Gingival crevicular blood for assessment of blood glucose levels. J. Periodontol. Implant. Dent. 2010, 2, 17–24. [Google Scholar]
- Patil, D.J.; Kamalakkannan, D. Diagnostic efficacy of gingival crevicular blood for assessment of blood glucose levels in dental office: A cross sectional study. Oral Hyg. Health 2014, 2, 1–4. [Google Scholar]
- Sibyl, S.; Bennadi, D.; Kshetrimayum, N.; Manjunath, M. Correlations between gingival crevicular blood glucose and capillary blood glucose: A preliminary report. J. Lab. Phys. 2017, 9, 260–263. [Google Scholar] [CrossRef]
- Kandwal, A.; Batra, M. Gingival crevicular blood as a screening tool for diabetic patient: A randomized clinical trial. Ann. Dent. Spec. 2014, 2, 6–8. [Google Scholar]
- Rapone, B.; Corsalini, M.; Converti, I.; Loverro, M.T.; Gnoni, A.; Trerotoli, P.; Ferrara, E. Does Periodontal Inflammation Affect Type 1 Diabetes in Childhood and Adolescence? A Meta-Analysis. Front. Endocrinol. 2020, 11, 27. [Google Scholar] [CrossRef]
- Corsalini, M.; Di Venere, D.; Rapone, B.; Stefanachi, G.; Laforgia, A.; Pettini, F. Evidence of signs and symptoms of Craniomandibular Disorders in Fibromyalgia patients. Open Dent. J. 2017, 11, 91–98. [Google Scholar] [CrossRef]
- Di Venere, D.; Nardi, G.M.; Lacarbonara, V.; Laforgia, A.; Stefanachi, G.; Corsalini, M.; Grassi, F.R.; Rapone, B.; Pettini, F. Early mandibular canine-lateral incisor transposition: Case Report. Oral Implantol. 2017, 10, 181–189. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Kawabata, Y.; Kambe, S.; Wårdell, K.; Nystrom, F.H.; Naitoh, K.; Yoshida, H. Non-invasive monitoring of gingival crevicular fluid for estimation of blood glucose level. Med. Biol. Eng. Comput. 2004, 42, 322–327. [Google Scholar] [CrossRef]
- Di Venere, D.; Corsalini, M.; Nardi, G.M.; Laforgia, A.; Grassi, F.R.; Rapone, B.; Pettini, F. Obstructive site localization in patients with Obstructive Sleep Apnea Syndrome: A comparison between otolaryngologic data and cephalometric values. Oral Implantol. 2017, 10, 295–310. [Google Scholar] [CrossRef]
- Parihar, S.; Tripathi, R.; Parihar, A.V.; Samadi, F.M.; Chandra, A.; Bhavsar, N. Estimation of gingival crevicular blood glucose level for the screening of diabetes mellitus: A simple yet reliable method. J. Oral Biol. Craniofac. Res. 2016, 6, 198–203. [Google Scholar] [CrossRef]
- Di Venere, D.; Pettini, F.; Nardi, G.M.; Laforgia, A.; Stefanachi, G.; Notaro, V.; Rapone, B.; Grassi, F.R.; Corsalini, M. Correlation between parodontal indexes and orthodontic retainers: Prospective study in a group of 16 patients. Oral Implantol. 2017, 10, 78–86. [Google Scholar] [CrossRef]
- Mealey, B.L.; Oates, T.W. American Academy of Periodontology. Diabetes mellitus and periodontal diseases. J. Periodontol. 2006, 77, 1289–1303. [Google Scholar] [CrossRef]
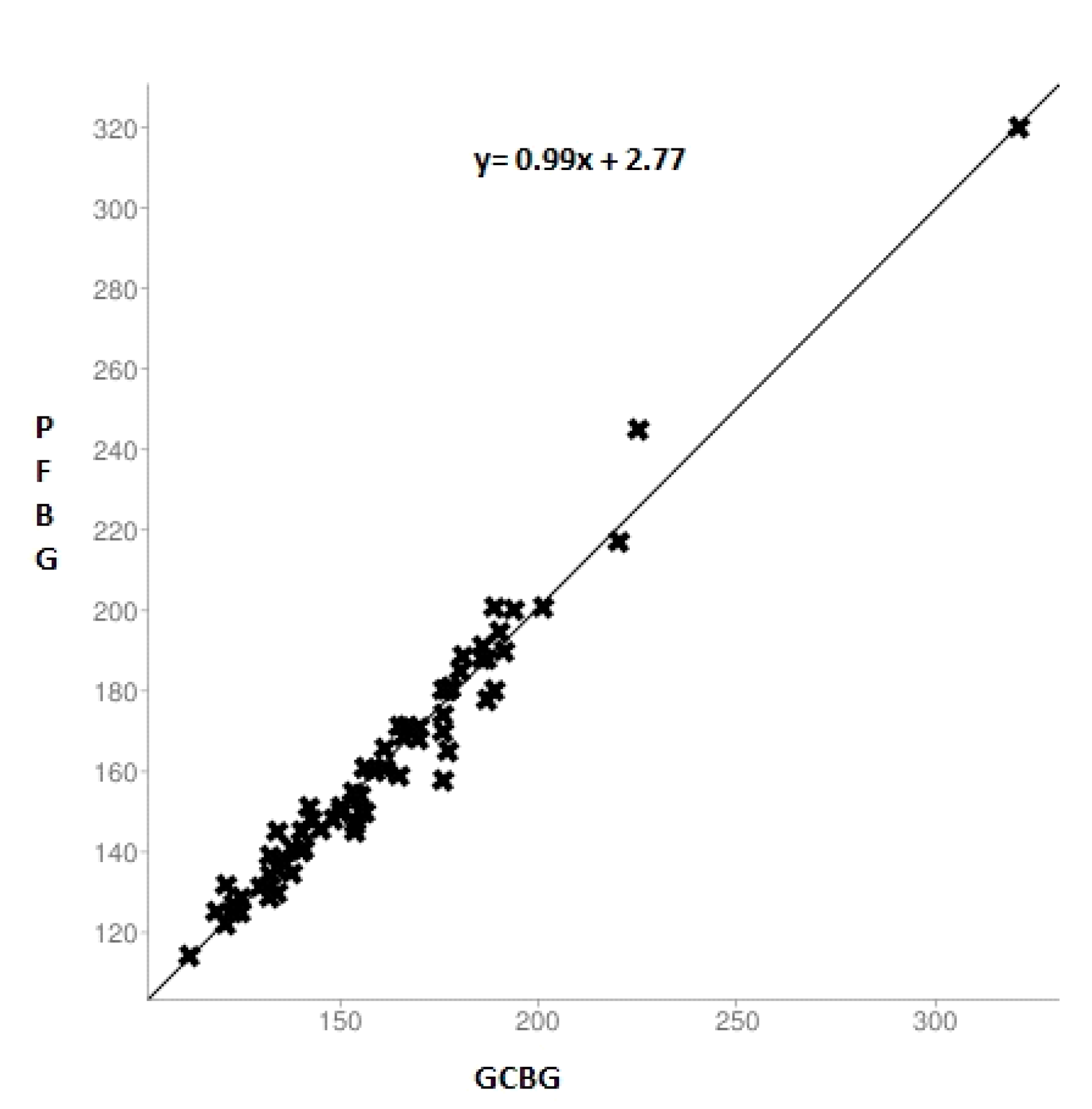
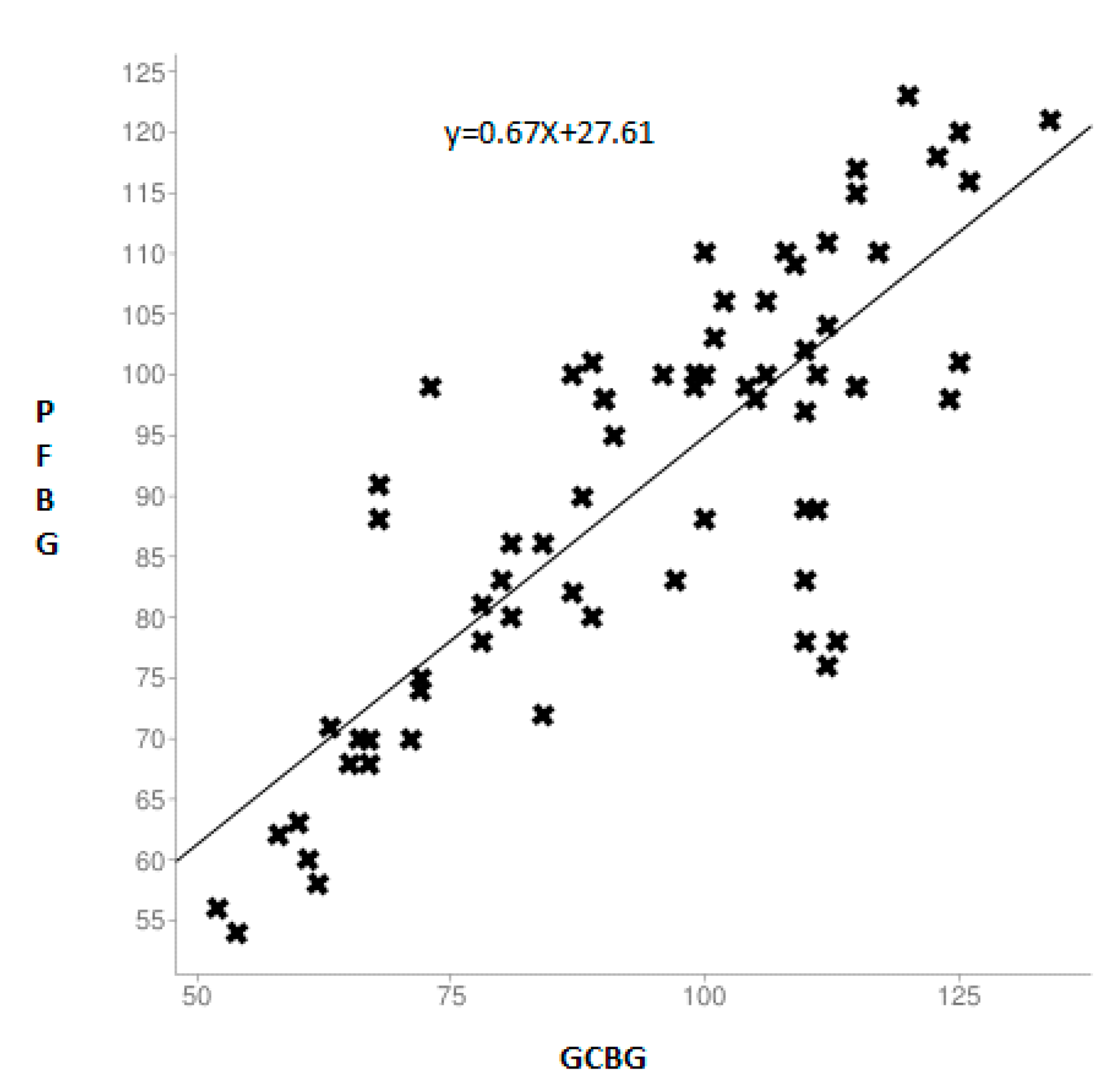
| Groups | No | Statistical Data | GCBG (mg/dL) | PFBG (mg/dL) | P-Value * | T-Value * |
|---|---|---|---|---|---|---|
| Diabetic | 70 | Mean ± SD | 160.42 ± 31.31 | 161.64 ± 31.56 | 0.9834 | 0.185 |
| Non-diabetic | 70 | Mean ± SD | 93.93 ± 20.93 | 90.88 ± 19.38 | 0.8153 | 1.127 |
| Group | * Correlation (r) |
|---|---|
| Diabetic | 0.9834 |
| Non-diabetic | 0.8153 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rapone, B.; Ferrara, E.; Santacroce, L.; Topi, S.; Converti, I.; Gnoni, A.; Scarano, A.; Scacco, S. Gingival Crevicular Blood as a Potential Screening Tool: A Cross Sectional Comparative Study. Int. J. Environ. Res. Public Health 2020, 17, 7356. https://doi.org/10.3390/ijerph17207356
Rapone B, Ferrara E, Santacroce L, Topi S, Converti I, Gnoni A, Scarano A, Scacco S. Gingival Crevicular Blood as a Potential Screening Tool: A Cross Sectional Comparative Study. International Journal of Environmental Research and Public Health. 2020; 17(20):7356. https://doi.org/10.3390/ijerph17207356
Chicago/Turabian StyleRapone, Biagio, Elisabetta Ferrara, Luigi Santacroce, Skender Topi, Ilaria Converti, Antonio Gnoni, Antonio Scarano, and Salvatore Scacco. 2020. "Gingival Crevicular Blood as a Potential Screening Tool: A Cross Sectional Comparative Study" International Journal of Environmental Research and Public Health 17, no. 20: 7356. https://doi.org/10.3390/ijerph17207356
APA StyleRapone, B., Ferrara, E., Santacroce, L., Topi, S., Converti, I., Gnoni, A., Scarano, A., & Scacco, S. (2020). Gingival Crevicular Blood as a Potential Screening Tool: A Cross Sectional Comparative Study. International Journal of Environmental Research and Public Health, 17(20), 7356. https://doi.org/10.3390/ijerph17207356










