Dietary Folate Intake and Folic Acid Supplements among Pregnant Women from Southern Italy: Evidence from the “Mamma & Bambino” Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Collection
2.3. Assessment of Dietary Folate Intake and Adherence to Mediterranean Diet
2.4. Use of Folic Acid Supplements
2.5. Neonatal Outcomes
2.6. Statistical Analysis
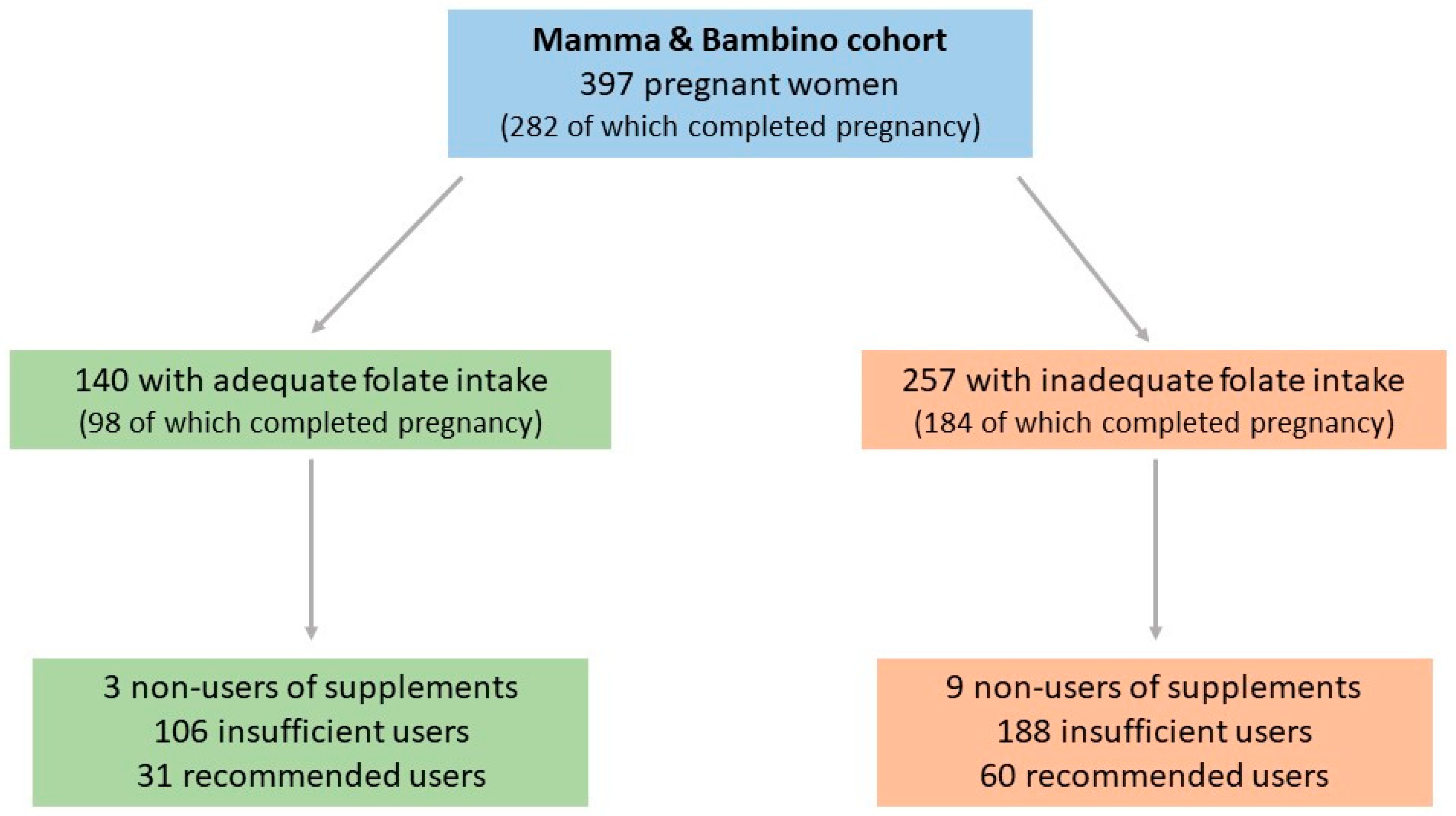
3. Results
3.1. Dietary Folate Intake Among Pregnant Women
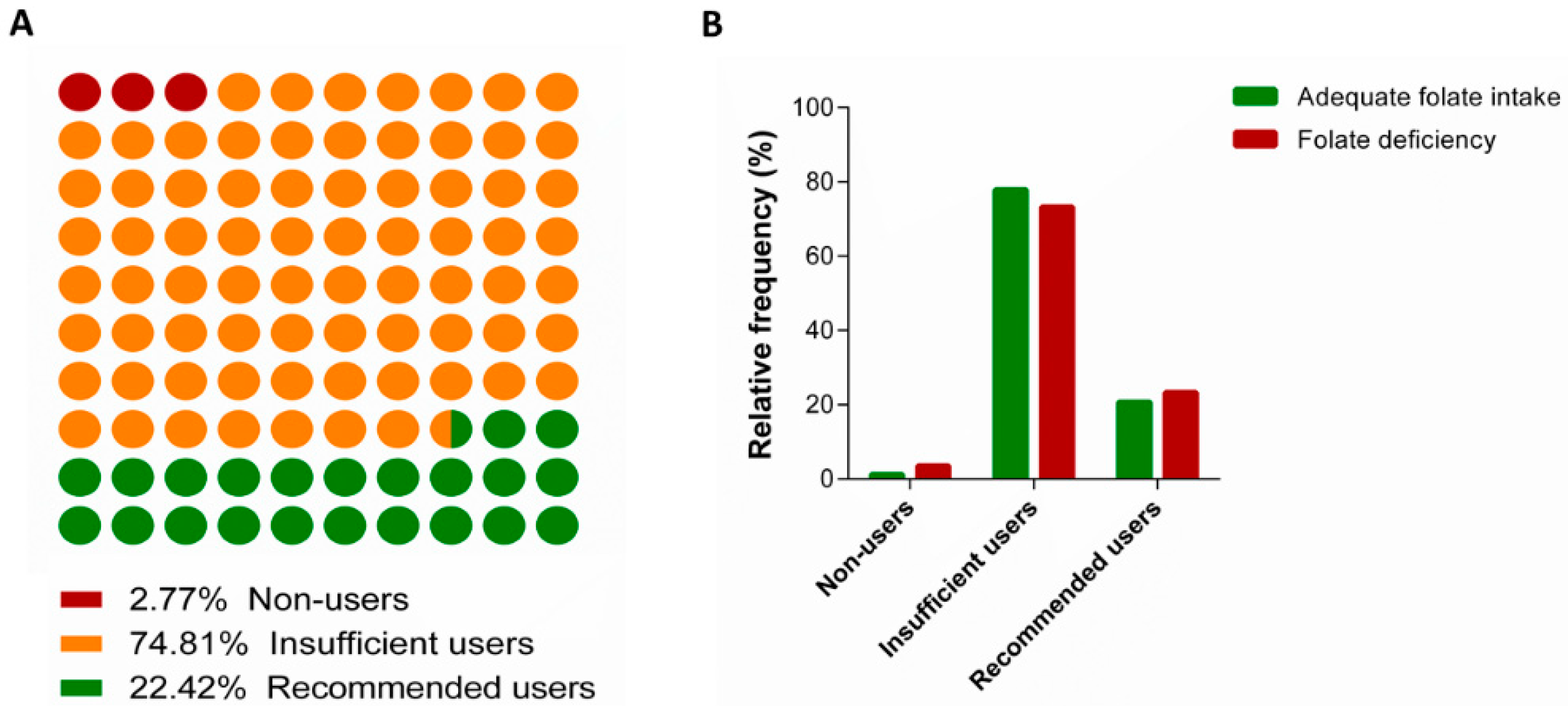
3.2. Use of Folic Acid Supplements
3.3. Determinants of Folic Acid Supplement USE Among Women with Inadequate Folate Intake
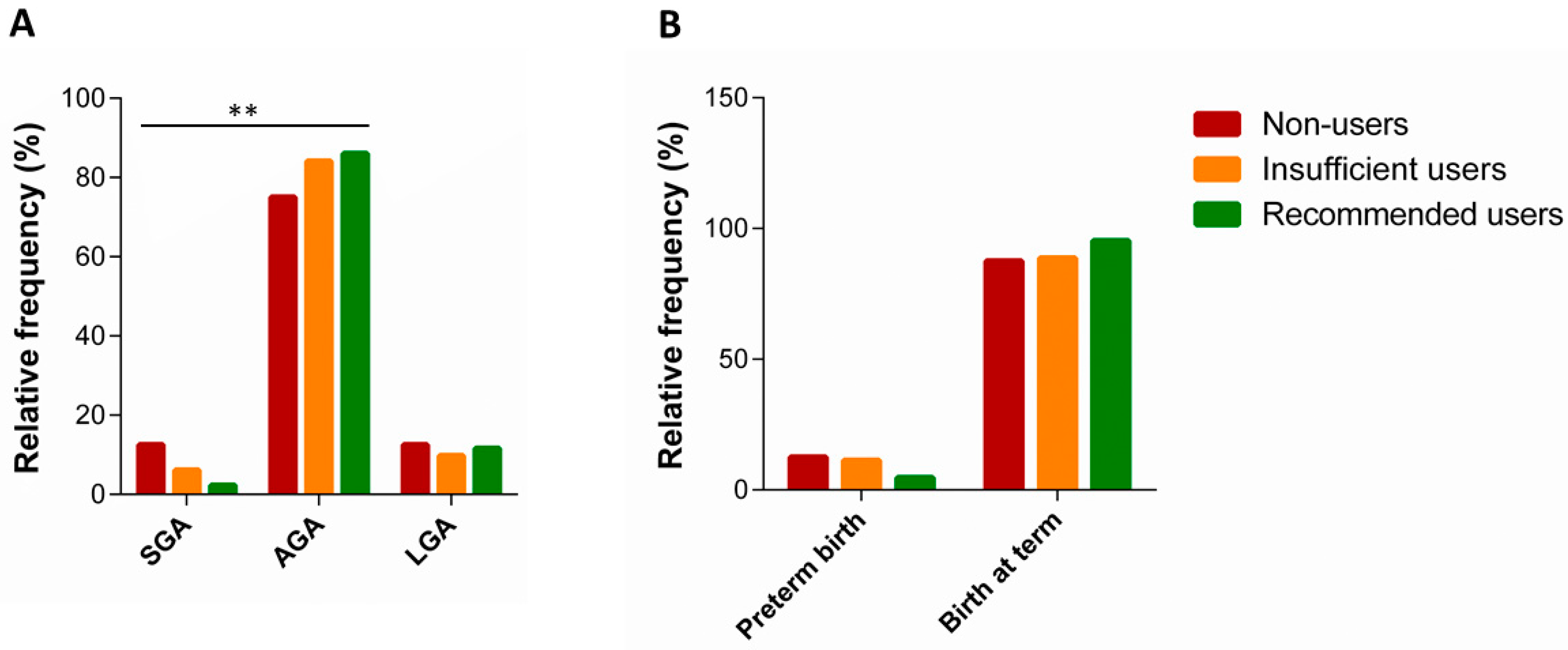
3.4. Use of Supplements and Neonatal Outcomes
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Christian, P. Micronutrients, birth weight, and survival. Annu. Rev. Nutr. 2010, 30, 83–104. [Google Scholar] [CrossRef]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Das, J.K.; Rizvi, A.; Gaffey, M.F.; Walker, N.; Horton, S.; Webb, P.; Lartey, A.; Black, R.E.; Lancet Nutrition Interventions Review Group; et al. Evidence-based interventions for improvement of maternal and child nutrition: What can be done and at what cost? Lancet 2013, 382, 452–477. [Google Scholar] [CrossRef]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; De Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Rush, D. Nutrition and maternal mortality in the developing world. Am. J. Clin. Nutr. 2000, 72, 212S–240S. [Google Scholar] [CrossRef]
- Carducci, B.; Bhutta, Z.A. Care of the growth-restricted newborn. Best Pract. Res. Clin. Obstet. Gynaecol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.A.; Chernausek, S.D.; Hokken-Koelega, A.C.; Czernichow, P.; for the International SGA Advisory Board. International Small for Gestational Age Advisory Board consensus development conference statement: Management of short children born small for gestational age, April 24–October 1, 2001. Pediatrics 2003, 111, 1253–1261. [Google Scholar]
- Kramer, M.S. The epidemiology of low birthweight. Nestle Nutr. Inst. Workshop Ser. 2013, 74, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gabbe, S.G.; Niebyl, J.R.; Simpson, J.L. Obstetrics: Normal and Problem Pregnancies; Churchill Livingstone: New York, NY, USA, 2016. [Google Scholar]
- Suliburska, J.; Kocyłowski, R.; Grzesiak, M.; Gaj, Z.; Chan, B.; von Kaisenberg, C.; Lamers, Y. Evaluation of folate concentration in amniotic fluid and maternal and umbilical cord blood during labor. Arch. Med. Sci. AMS 2019, 15, 1425–1432. [Google Scholar] [CrossRef]
- Castillo-Lancellotti, C.; Tur, J.A.; Uauy, R. Impact of folic acid fortification of flour on neural tube defects: A systematic review. Public Health Nutr. 2013, 16, 901–911. [Google Scholar] [CrossRef]
- Agodi, A.; Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Canto, C.; Marchese, A.E.; Vinciguerra, M. Low fruit consumption and folate deficiency are associated with LINE-1 hypomethylation in women of a cancer-free population. Genes Nutr. 2015, 10, 480. [Google Scholar] [CrossRef]
- Agodi, A.; Barchitta, M.; Valenti, G.; Marzagalli, R.; Frontini, V.; Marchese, A.E. Increase in the prevalence of the MTHFR 677 TT polymorphism in women born since 1959: Potential implications for folate requirements. Eur. J. Clin. Nutr. 2011, 65, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.N.; Copp, A.J.; Shawe, J. Knowledge and periconceptional use of folic acid for the prevention of neural tube defects in ethnic communities in the United Kingdom: Systematic review and meta-analysis. Birth Defects Res. Part A Clin. Mol. Teratol. 2013, 97, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Badiga, S.; Johanning, G.L.; Macaluso, M.; Azuero, A.; Chambers, M.M.; Siddiqui, N.R.; Piyathilake, C.J. A lower degree of PBMC L1 methylation in women with lower folate status may explain the MTHFR C677T polymorphism associated higher risk of CIN in the US post folic acid fortification era. PLoS ONE 2014, 9, e110093. [Google Scholar] [CrossRef]
- Hertrampf, E.; Cortés, F. National food-fortification program with folic acid in Chile. Food Nutr. Bull. 2008, 29, S231–S237. [Google Scholar] [CrossRef] [PubMed]
- McStay, C.L.; Prescott, S.L.; Bower, C.; Palmer, D.J. Maternal Folic Acid Supplementation during Pregnancy and Childhood Allergic Disease Outcomes: A Question of Timing? Nutrients 2017, 9, 123. [Google Scholar] [CrossRef]
- Caut, C.; Leach, M.; Steel, A. Dietary guideline adherence during preconception and pregnancy: A systematic review. Matern. Child Nutr. 2019, e12916. [Google Scholar] [CrossRef] [PubMed]
- Popa, A.D.; Niţă, O.; Graur Arhire, L.I.; Popescu, R.M.; Botnariu, G.E.; Mihalache, L.; Graur, M. Nutritional knowledge as a determinant of vitamin and mineral supplementation during pregnancy. BMC Public Health 2013, 13, 1105. [Google Scholar] [CrossRef]
- Nasr Hage, C.; Jalloul, M.; Sabbah, M.; Adib, S.M. Awareness and intake of folic acid for the prevention of neural tube defects among Lebanese women of childbearing age. Matern. Child Health J. 2012, 16, 258–265. [Google Scholar] [CrossRef]
- Roth, C.; Bjørke-Monsen, A.L.; Reichborn-Kjennerud, T.; Nilsen, R.M.; Smith, G.D.; Stoltenberg, C.; Surén, P.; Susser, E.; Ueland, P.M.; Vollset, S.E.; et al. Use of folic acid supplements in early pregnancy in relation to maternal plasma levels in week 18 of pregnancy. Mol. Nutr. Food Res. 2013, 57, 653–660. [Google Scholar] [CrossRef]
- Tamim, H.; Harrison, G.; Atoui, M.; Mumtaz, G.; El-Kak, F.; Seoud, M.; Yunis, K.; National Collaborative Perinatal Neonatal Network (NCPNN). Preconceptional folic acid supplement use in Lebanon. Public Health Nutr. 2009, 12, 687–692. [Google Scholar] [CrossRef]
- Murray, C.J.; Lopez, A.D. Regional patterns of disability-free life expectancy and disability-adjusted life expectancy: Global Burden of Disease Study. Lancet 1997, 349, 1347–1352. [Google Scholar] [CrossRef]
- Veeranki, S.P.; Gebretsadik, T.; Mitchel, E.F.; Tylavsky, F.A.; Hartert, T.V.; Cooper, W.O.; Dupont, W.D.; Dorris, S.L.; Hartman, T.J.; Carroll, K.N. Maternal Folic Acid Supplementation During Pregnancy and Early Childhood Asthma. Epidemiology (Camb. Mass.) 2015, 26, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Whitrow, M.J.; Moore, V.M.; Rumbold, A.R.; Davies, M.J. Effect of supplemental folic acid in pregnancy on childhood asthma: A prospective birth cohort study. Am. J. Epidemiol. 2009, 170, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Håberg, S.E.; London, S.J.; Stigum, H.; Nafstad, P.; Nystad, W. Folic acid supplements in pregnancy and early childhood respiratory health. Arch. Dis. Child. 2009, 94, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Picciano, M.F. Folate and human reproduction. Am. J. Clin. Nutr. 2006, 83, 993–1016. [Google Scholar] [CrossRef]
- Baker, B.C.; Mackie, F.L.; Lean, S.C.; Greenwood, S.L.; Heazell, A.E.P.; Forbes, K.; Jones, R.L. Placental dysfunction is associated with altered microRNA expression in pregnant women with low folate status. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Bergen, N.E.; Jaddoe, V.W.V.; Timmermans, S.; Hofman, A.; Lindemans, J.; Russcher, H.; Raat, H.; Steegers-Theunissen, R.P.M.; Steegers, E.A.P. Homocysteine and folate concentrations in early pregnancy and the risk of adverse pregnancy outcomes: The Generation R Study. BJOG Int. J. Obstet. Gynaecol. 2012, 119, 739–751. [Google Scholar] [CrossRef]
- Bukowski, R.; Malone, F.D.; Porter, F.T.; Nyberg, D.A.; Comstock, C.H.; Hankins, G.D.; Eddleman, K.; Gross, S.J.; Dugoff, L.; Craigo, S.D.; et al. Preconceptional folate supplementation and the risk of spontaneous preterm birth: A cohort study. PLoS Med. 2009, 6, e1000061. [Google Scholar] [CrossRef]
- Catov, J.M.; Bodnar, L.M.; Olsen, J.; Olsen, S.; Nohr, E.A. Periconceptional multivitamin use and risk of preterm or small-for-gestational-age births in the Danish National Birth Cohort. Am. J. Clin. Nutr. 2011, 94, 906–912. [Google Scholar] [CrossRef]
- Chen, S.; Zhu, R.; Zhu, H.; Yang, H.; Gong, F.; Wang, L.; Jiang, Y.; Lian, B.Q.; Yan, C.; Li, J.; et al. The prevalence and risk factors of preterm small-for-gestational-age infants: A population-based retrospective cohort study in rural Chinese population. BMC Pregnancy Childbirth 2017, 17, 237. [Google Scholar] [CrossRef]
- Dwarkanath, P.; Barzilay, J.R.; Thomas, T.; Thomas, A.; Bhat, S.; Kurpad, A.V. High folate and low vitamin B-12 intakes during pregnancy are associated with small-for-gestational age infants in South Indian women: A prospective observational cohort study. Am. J. Clin. Nutr. 2013, 98, 1450–1458. [Google Scholar] [CrossRef] [PubMed]
- Furness, D.L.; Yasin, N.; Dekker, G.A.; Thompson, S.D.; Roberts, C.T. Maternal red blood cell folate concentration at 10-12 weeks gestation and pregnancy outcome. J. Matern.-Fetal Neonatal Med. 2012, 25, 1423–1427. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Tamura, T.; Cliver, S.P.; Cutter, G.R.; Hoffman, H.J.; Copper, R.L. Serum folate and fetal growth retardation: A matter of compliance? Obstet. Gynecol. 1992, 79, 719–722. [Google Scholar] [PubMed]
- Hodgetts, V.A.; Morris, R.K.; Francis, A.; Gardosi, J.; Ismail, K.M. Effectiveness of folic acid supplementation in pregnancy on reducing the risk of small-for-gestational age neonates: A population study, systematic review and meta-analysis. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 478–490. [Google Scholar] [CrossRef]
- Kim, M.W.; Ahn, K.H.; Ryu, K.J.; Hong, S.C.; Lee, J.S.; Nava-Ocampo, A.A.; Oh, M.J.; Kim, H.J. Preventive effects of folic acid supplementation on adverse maternal and fetal outcomes. PLoS ONE 2014, 9, e97273. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Li, Z.; Ye, R.; Liu, J.; Ren, A. Impact of Periconceptional Folic Acid Supplementation on Low Birth Weight and Small-for-Gestational-Age Infants in China: A Large Prospective Cohort Study. J. Pediatrics 2017, 187, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Navarrete-Muñoz, E.M.; Giménez Monzó, D.; García de La Hera, M.; Climent, M.D.; Rebagliato, M.; Murcia, M.; Iñiguez, C.; Ballester, F.; Ramón, R.; Vioque, J. Folic acid intake from diet and supplements in a population of pregnant women in Valencia, Spain. Med. Clin. 2010, 135, 637–643. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Stratakis, N.; Roumeliotaki, T.; Sarri, K.; Merlo, D.F.; Kogevinas, M.; Chatzi, L. The effect of high doses of folic acid and iron supplementation in early-to-mid pregnancy on prematurity and fetal growth retardation: The mother-child cohort study in Crete, Greece (Rhea study). Eur. J. Nutr. 2013, 52, 327–336. [Google Scholar] [CrossRef]
- Pastor-Valero, M.; Navarrete-Muñoz, E.M.; Rebagliato, M.; Iñiguez, C.; Murcia, M.; Marco, A.; Ballester, F.; Vioque, J. Periconceptional folic acid supplementation and anthropometric measures at birth in a cohort of pregnant women in Valencia, Spain. Br. J. Nutr. 2011, 105, 1352–1360. [Google Scholar] [CrossRef]
- Rolschau, J.; Kristoffersen, K.; Ulrich, M.; Grinsted, P.; Schaumburg, E.; Foged, N. The influence of folic acid supplement on the outcome of pregnancies in the county of Funen in Denmark. Part, I. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 87, 105–110, discussion 103–104. [Google Scholar] [CrossRef]
- Ronnenberg, A.G.; Goldman, M.B.; Chen, D.; Aitken, I.W.; Willett, W.C.; Selhub, J.; Xu, X. Preconception homocysteine and B vitamin status and birth outcomes in Chinese women. Am. J. Clin. Nutr. 2002, 76, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, S.; Jaddoe, V.W.; Hofman, A.; Steegers-Theunissen, R.P.; Steegers, E.A. Periconception folic acid supplementation, fetal growth and the risks of low birth weight and preterm birth: The Generation R Study. Br. J. Nutr. 2009, 102, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ge, X.; Zhu, B.; Xuan, Y.; Huang, K.; Rutayisire, E.; Mao, L.; Huang, S.; Yan, S.; Tao, F. Maternal Continuing Folic Acid Supplementation after the First Trimester of Pregnancy Increased the Risk of Large-for-Gestational-Age Birth: A Population-Based Birth Cohort Study. Nutrients 2016, 8, 493. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.Q.; Xu, Y.Q.; Su, P.Y.; Cao, H.; Pan, W.J.; Tao, F.B. Relationship between folic acid supplements during peri-conceptional period and the adverse pregnancy outcomes: A cohort study. Zhonghua Liu Xing Bing Xue Za Zhi 2013, 34, 1–4. [Google Scholar] [PubMed]
- Barchitta, M.; Maugeri, A.; La Rosa, M.C.; Magnano San Lio, R.; Favara, G.; Panella, M.; Cianci, A.; Agodi, A. Single Nucleotide Polymorphisms in Vitamin D Receptor Gene Affect Birth Weight and the Risk of Preterm Birth: Results From the "Mamma & Bambino" Cohort and A Meta-Analysis. Nutrients 2018, 10, 1172. [Google Scholar] [CrossRef]
- Maugeri, A.; Barchitta, M.; Agrifoglio, O.; Favara, G.; La Mastra, C.; La Rosa, M.C.; Magnano San Lio, R.; Panella, M.; Cianci, A.; Agodi, A. The impact of social determinants and lifestyles on dietary patterns during pregnancy: Evidence from the “Mamma & Bambino” study. Annali di Igiene Medicina Preventiva e di Comunita 2019, 31, 81–89. [Google Scholar] [PubMed]
- Eveleth, P.B.; Andres, R.; Chumlea, W.C.; Eiben, O.; Ge, K.; Harris, T.; Heymsfield, S.B.; Launer, L.J.; Rosenberg, I.H.; Solomons, N.W.; et al. Uses and interpretation of anthropometry in the elderly for the assessment of physical status. Report to the Nutrition Unit of the World Health Organization: The Expert Subcommittee on the Use and Interpretation of Anthropometry in the Elderly. J. Nutr. Health Aging 1998, 2, 5–17. [Google Scholar]
- Barchitta, M.; Maugeri, A.; Quattrocchi, A.; Agrifoglio, O.; Scalisi, A.; Agodi, A. The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy. Nutrients 2018, 10, 469. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; Quattrocchi, A.; Barone, G.; Mazzoleni, P.; Catalfo, A.; De Guidi, G.; Iemmolo, M.G.; Crimi, N.; Agodi, A. Mediterranean Diet and Particulate Matter Exposure Are Associated With LINE-1 Methylation: Results From a Cross-Sectional Study in Women. Front. Genet. 2018, 9, 514. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; Magnano San Lio, R.; Favara, G.; La Rosa, M.C.; La Mastra, C.; Quattrocchi, A.; Agodi, A. Dietary Patterns are Associated with Leukocyte LINE-1 Methylation in Women: A Cross-Sectional Study in Southern Italy. Nutrients 2019, 11, 1843. [Google Scholar] [CrossRef]
- nstitute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. The National Academies Collection: Reports funded by National Institutes of Health. In Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press (US) National Academy of Sciences: Washington, DC, USA, 1998. [Google Scholar]
- Couto, E.; Boffetta, P.; Lagiou, P.; Ferrari, P.; Buckland, G.; Overvad, K.; Dahm, C.C.; Tjønneland, A.; Olsen, A.; Clavel-Chapelon, F.; et al. Mediterranean dietary pattern and cancer risk in the EPIC cohort. Br. J. Cancer 2011, 104, 1493–1499. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Barchitta, M.; Quattrocchi, A.; Adornetto, V.; Marchese, A.E.; Agodi, A. Tumor necrosis factor-alpha -308 G>A polymorphism, adherence to Mediterranean diet, and risk of overweight/obesity in young women. BioMed. Res. Int. 2014, 2014, 742620. [Google Scholar] [CrossRef] [PubMed]
- Network Italiano Promozione Acido Folico per la Prevenzione Primaria diDifetti Congeniti. RACCOMANDAZIONE PER LA RIDUZIONE DEL RISCHIO DI DIFETTI CONGENITI. Available online: http://old.iss.it/binary/acid4/cont/raccomandazione.pdf (accessed on 1 December 2019).
- Bertino, E.; Spada, E.; Occhi, L.; Coscia, A.; Giuliani, F.; Gagliardi, L.; Gilli, G.; Bona, G.; Fabris, C.; De Curtis, M.; et al. Neonatal anthropometric charts: The Italian neonatal study compared with other European studies. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 353–361. [Google Scholar] [CrossRef] [PubMed]
- McLean, E.; de Benoist, B.; Allen, L.H. Review of the magnitude of folate and vitamin B12 deficiencies worldwide. Food Nutr. Bull. 2008, 29, S38–S51. [Google Scholar] [CrossRef] [PubMed]
- Metz, J. Haematological implications of folate food fortification. S. Afr. Med. J. 2013, 103, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Agodi, A.; Barchitta, M.; Quattrocchi, A.; Marchese, A.E.; Boffetta, P. Folate deficiency is not associated with increased mitochondrial genomic instability: Results from dietary intake and lymphocytic mtDNA 4977-bp deletion in healthy young women in Italy. Mutagenesis 2014, 29, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Canto, C.; La Rosa, N.; Cantarella, M.A.; Spampinato, G.; Scalisi, A.; Agodi, A. LINE-1 hypermethylation in white blood cell DNA is associated with high-grade cervical intraepithelial neoplasia. BMC Cancer 2017, 17, 601. [Google Scholar] [CrossRef] [PubMed]
- Lamers, Y.; MacFarlane, A.J.; O’Connor, D.L.; Fontaine-Bisson, B. Periconceptional intake of folic acid among low-risk women in Canada: Summary of a workshop aiming to align prenatal folic acid supplement composition with current expert guidelines. Am. J. Clin. Nutr. 2018, 108, 1357–1368. [Google Scholar] [CrossRef]
- Palaniappan, U.; Jacobs Starkey, L.; O’Loughlin, J.; Gray-Donald, K. Fruit and vegetable consumption is lower and saturated fat intake is higher among Canadians reporting smoking. J. Nutr. 2001, 131, 1952–1958. [Google Scholar] [CrossRef]
- Dyer, A.R.; Elliott, P.; Stamler, J.; Chan, Q.; Ueshima, H.; Zhou, B.F.; Group, I.R. Dietary intake in male and female smokers, ex-smokers, and never smokers: The INTERMAP study. J. Hum. Hypertens. 2003, 17, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Birkett, N.J. Intake of fruits and vegetables in smokers. Public Health Nutr. 1999, 2, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Dallongeville, J.; Marécaux, N.; Fruchart, J.C.; Amouyel, P. Cigarette smoking is associated with unhealthy patterns of nutrient intake: A meta-analysis. J. Nutr. 1998, 128, 1450–1457. [Google Scholar] [CrossRef] [PubMed]
- De Santis, M.; Quattrocchi, T.; Mappa, I.; Spagnuolo, T.; Licameli, A.; Chiaradia, G.; De Luca, C. Folic acid use in planned pregnancy: An Italian survey. Matern. Child Health J. 2013, 17, 661–666. [Google Scholar] [CrossRef]
- EUROCAT Central Registry. Special Report: Prevention of Neural Tube Defects by Periconceptional Folic Acid Supplementation in Europe; University of Ulster: Newtownabbey, Ireland, 2009. [Google Scholar]
- De Walle, H.E.; De Jong-van den Berg, L.T. Insufficient folic acid intake in the Netherlands: What about the future? Teratology 2002, 66, 40–43. [Google Scholar] [CrossRef]
- Vitale, K.; Mujkić, A.; Todorović, G.; Tulchinsky, T. Is level of knowledge, attitude and use of folic acid among pregnant women in Croatia a call for public health action? Period. Biol. 2009, 111, 329–335. [Google Scholar]
- McIntire, D.D.; Bloom, S.L.; Casey, B.M.; Leveno, K.J. Birth weight in relation to morbidity and mortality among newborn infants. N. Engl. J. Med. 1999, 340, 1234–1238. [Google Scholar] [CrossRef]
- Pallotto, E.K.; Kilbride, H.W. Perinatal outcome and later implications of intrauterine growth restriction. Clin. Obstet. Gynecol. 2006, 49, 257–269. [Google Scholar] [CrossRef]
- Hack, M.; Taylor, H.G.; Drotar, D.; Schluchter, M.; Cartar, L.; Andreias, L.; Wilson-Costello, D.; Klein, N. Chronic conditions, functional limitations, and special health care needs of school-aged children born with extremely low-birth-weight in the 1990s. JAMA 2005, 294, 318–325. [Google Scholar] [CrossRef]
- Hille, E.T.; den Ouden, A.L.; Saigal, S.; Wolke, D.; Lambert, M.; Whitaker, A.; Pinto-Martin, J.A.; Hoult, L.; Meyer, R.; Feldman, J.F.; et al. Behavioural problems in children who weigh 1000 g or less at birth in four countries. Lancet 2001, 357, 1641–1643. [Google Scholar] [CrossRef]
- Nafee, T.M.; Farrell, W.E.; Carroll, W.D.; Fryer, A.A.; Ismail, K.M. Epigenetic control of fetal gene expression. BJOG Int. J. Obstet. Gynaecol. 2008, 115, 158–168. [Google Scholar] [CrossRef]
- Schlotz, W.; Phillips, D.I. Fetal origins of mental health: Evidence and mechanisms. Brain Behav. Immun. 2009, 23, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Tosh, D.N.; Fu, Q.; Callaway, C.W.; McKnight, R.A.; McMillen, I.C.; Ross, M.G.; Lane, R.H.; Desai, M. Epigenetics of programmed obesity: Alteration in IUGR rat hepatic IGF1 mRNA expression and histone structure in rapid vs. delayed postnatal catch-up growth. Am. J. Physiol.-Gastrointest. Liver Physiol. 2010, 299, G1023–G1029. [Google Scholar] [CrossRef] [PubMed]
- Waterland, R.A. Is epigenetics an important link between early life events and adult disease? Horm. Res. 2009, 71 (Suppl. 1), 13–16. [Google Scholar] [CrossRef] [PubMed]
- Cabo, R.; Hernes, S.; Slettan, A.; Haugen, M.; Ye, S.; Blomhoff, R.; Mansoor, M.A. Effect of genetic polymorphisms involved in folate metabolism on the concentration of serum folate and plasma total homocysteine (p-tHcy) in healthy subjects after short-term folic acid supplementation: A randomized, double blind, crossover study. Genes Nutr. 2015, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Agodi, A.; Quattrocchi, A.; Maugeri, A.; Barchitta, M. The link between MTHFR C677T polymorphism, folate metabolism and global DNA methylation: A literature review. In Methylenetetrahydrofolate Reductase (MTHFR) in Health and Disease; Evans, R., Ed.; Nova Science Publishers: New York, NY, USA, 2015; pp. 71–82. [Google Scholar]
| Characteristics a | Inadequate Folate Intake (n = 257) | Adequate Folate Intake (n = 140) | p-Value b |
|---|---|---|---|
| Age, years | 37.0 (5.0) | 37.5 (4.0) | 0.375 |
| Educational level | |||
| Low | 19.3% | 18.7% | 0.214 |
| Medium | 51.4% | 43.6% | |
| High | 29.3% | 37.7% | |
| Employed | 56.4% | 58.6% | 0.679 |
| Living in couple | 92.3% | 93.8% | 0.973 |
| Having children | 68.5% | 68.6% | 0.985 |
| Food intolerance (% yes) | 16.7% | 10.0% | 0.150 |
| Dietary restriction (% yes) | 19.8% | 8.6% | 0.003 |
| Smoking status | |||
| Non-smoker | 55.0% | 57.6% | 0.028 |
| Former smoker | 15.7% | 23.5% | |
| Smoker | 29.3% | 18.8% | |
| Pregestational BMI, Kg/m2 | 23.2 (4.9) | 22.1 (5.4) | 0.029 |
| Pregestational BMI categories | |||
| Underweight | 5.5% | 8.6% | 0.353 |
| Normal weight | 63.7% | 66.4% | |
| Overweight | 20.3% | 13.6% | |
| Obese | 10.2% | 11.4% | |
| MDS | 4 (2) | 5 (2) | <0.001 |
| Adherence to MD | |||
| Low | 45.1% | 20.0% | <0.001 |
| Medium | 52.9% | 63.6% | |
| High | 1.9% | 16.4% | |
| Preterm birth c | 8.4% | 7.2% | 0.722 |
| Birthweight for gestational age | |||
| SGA | 13.7% | 4.8% | <0.001 |
| AGA | 67.4%% | 88.4% | |
| LGA | 18.9% | 6.9% | |
| Characteristics a | Non-Users (n = 9) | Insufficient Users (n = 188) | Recommended Users (n = 60) | p-Value b |
|---|---|---|---|---|
| Age, years | 38.0 (8.0) | 37.0 (4.0) | 37.0 (4.0)) | 0.884 |
| Educational level | ||||
| Low | 44.4% | 22.9% | 1.7% | <0.001 |
| Medium | 55.6% | 44.1% | 40.0% | |
| High | 0% | 33.0% | 58.3% | |
| Employed | 55.6% | 52.7% | 68.3% | 0.103 |
| Living in couple | 90.1% | 91.2% | 94.2 | 0.878 |
| Having children | 66.7% | 70.2% | 63.3% | 0.603 |
| Food intolerance (% yes) | 0% | 17.0% | 18.3% | 0.264 |
| Dietary restriction (% yes) | 33.3% | 18.6% | 21.7% | 0.514 |
| Smoking status | ||||
| Non-smoker | 55.6% | 55.1% | 66.1% | 0.078 |
| Former smoker | 33.3% | 21.9% | 27.1% | |
| Smoker | 11.1% | 23.0% | 6.8% | |
| Pregestational BMI, Kg/m2 | 22.7 (3.2) | 23.6 (5.3) | 22.2 (3.9) | 0.256 |
| Pregestational BMI categories | ||||
| Underweight | 0% | 5.9% | 5.0% | 0.575 |
| Normal weight | 66.7% | 60.4% | 73.3% | |
| Overweight | 33.3% | 20.9% | 16.7% | |
| Obese | 0% | 12.3% | 5.0% | |
| MDS | 3 (3) | 4 (2) | 4 (2) | 0.047 |
| Adherence to MD | ||||
| Low | 66.7% | 46.8% | 36.7% | 0.424 |
| Medium | 33.3% | 51.1% | 61.7% | |
| High | 0% | 2.1% | 1.7% | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barchitta, M.; Maugeri, A.; Magnano San Lio, R.; Favara, G.; La Mastra, C.; La Rosa, M.C.; Agodi, A. Dietary Folate Intake and Folic Acid Supplements among Pregnant Women from Southern Italy: Evidence from the “Mamma & Bambino” Cohort. Int. J. Environ. Res. Public Health 2020, 17, 638. https://doi.org/10.3390/ijerph17020638
Barchitta M, Maugeri A, Magnano San Lio R, Favara G, La Mastra C, La Rosa MC, Agodi A. Dietary Folate Intake and Folic Acid Supplements among Pregnant Women from Southern Italy: Evidence from the “Mamma & Bambino” Cohort. International Journal of Environmental Research and Public Health. 2020; 17(2):638. https://doi.org/10.3390/ijerph17020638
Chicago/Turabian StyleBarchitta, Martina, Andrea Maugeri, Roberta Magnano San Lio, Giuliana Favara, Claudia La Mastra, Maria Clara La Rosa, and Antonella Agodi. 2020. "Dietary Folate Intake and Folic Acid Supplements among Pregnant Women from Southern Italy: Evidence from the “Mamma & Bambino” Cohort" International Journal of Environmental Research and Public Health 17, no. 2: 638. https://doi.org/10.3390/ijerph17020638
APA StyleBarchitta, M., Maugeri, A., Magnano San Lio, R., Favara, G., La Mastra, C., La Rosa, M. C., & Agodi, A. (2020). Dietary Folate Intake and Folic Acid Supplements among Pregnant Women from Southern Italy: Evidence from the “Mamma & Bambino” Cohort. International Journal of Environmental Research and Public Health, 17(2), 638. https://doi.org/10.3390/ijerph17020638







