Incidence and Risk Factors for Severe Dehydration in Hospitalized Children in Ujjain, India
Abstract
1. Introduction
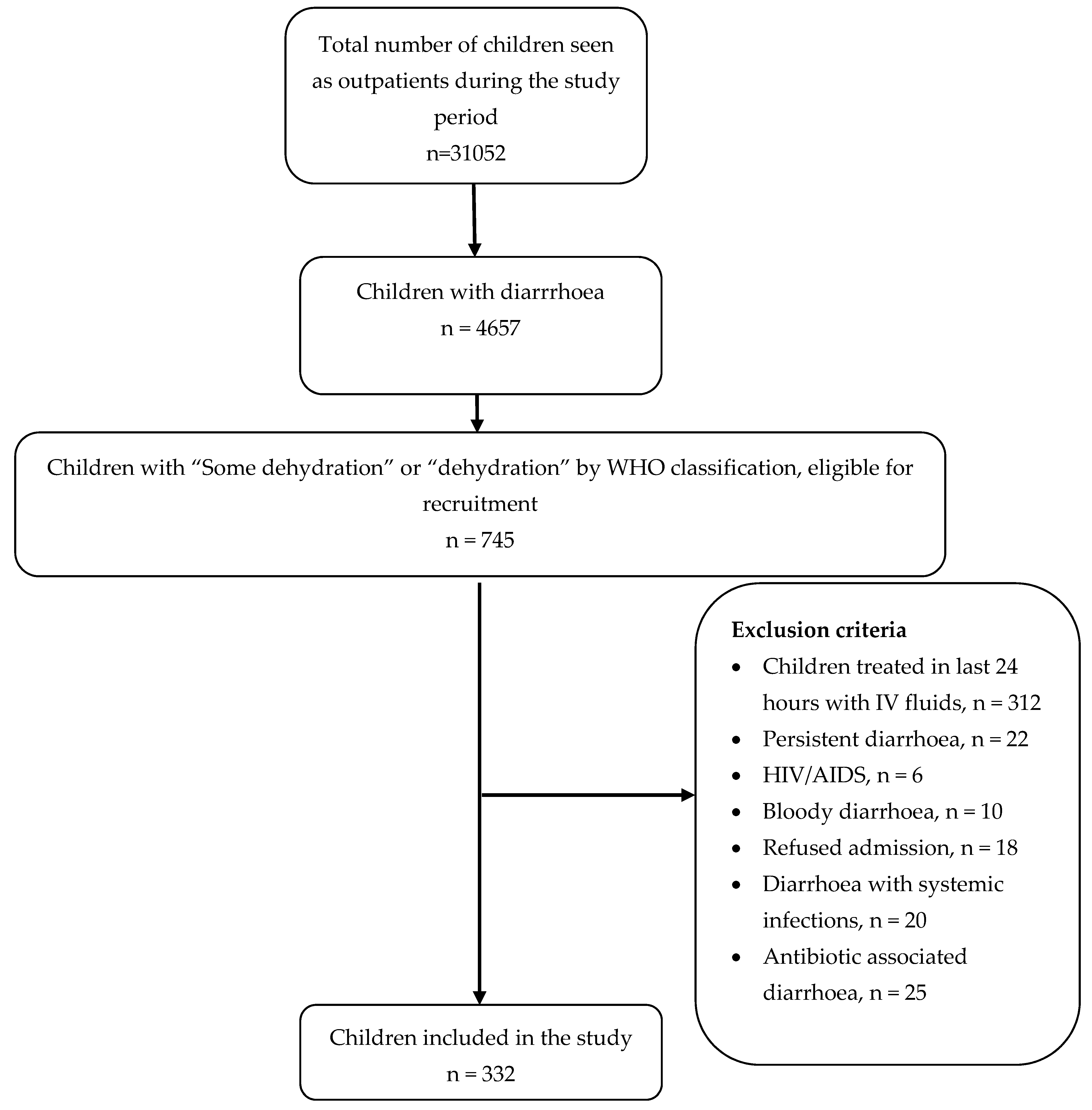
2. Materials and Methods
2.1. Study Setting
2.2. Study Participants
2.3. Definitions
2.4. Data Collection Method
2.5. Clinical Management of Diarrhoea
2.6. Data Management and Statistical Analysis
3. Results
3.1. Multivariate Analysis
3.2. Model Performance
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- UNICEF; WHO. End Preventable Deaths: Global Action Plan for Prevention and Control of Pneumonia and Diarrhoea; World Health Organization: Geneva, Switzerland, 2013; Available online: http://apps.who.int/iris/bitstream/handle/10665/79200/9789241505239_eng.pdf;jsessionid=837AEC1288C76D2368F1E8F8DAB86B55?sequence=1 (accessed on 7 December 2019).
- Liu, L.; Oza, S.; Hogan, D.; Perin, J.; Rudan, I.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of child mortality in 2000-13, with projections to inform post-2015 priorities: An updated systematic analysis. Lancet 2015, 385, 430–440. [Google Scholar] [CrossRef]
- Walker, C.L.; Rudan, I.; Liu, L.; Nair, H.; Theodoratou, E.; Bhutta, Z.A.; O’Brien, K.L.; Campbell, H.; Black, R.E. Global burden of childhood pneumonia and diarrhoea. Lancet 2013, 381, 1405–1416. [Google Scholar] [CrossRef]
- Troeger, C.; Colombara, D.V.; Rao, P.C.; Khalil, I.A.; Brown, A.; Brewer, T.G.; Guerrant, R.L.; Houpt, E.R.; Kotloff, K.L.; Misra, K.; et al. Global disability-adjusted life-year estimates of long-term health burden and undernutrition attributable to diarrhoeal diseases in children younger than 5 years. Lancet Glob. Health 2018, 6, e255–e269. [Google Scholar] [CrossRef]
- Santosham, M.; Chandran, A.; Fitzwater, S.; Fischer-Walker, C.; Baqui, A.H.; Black, R. Progress and barriers for the control of diarrhoeal disease. Lancet 2010, 376, 63–67. [Google Scholar] [CrossRef]
- Ram, U.; Jha, P.; Ram, F.; Kumar, K.; Awasthi, S.; Shet, A.; Pader, J.; Nansukusa, S.; Kumar, R. Neonatal, 1-59 month, and under-5 mortality in 597 indian districts, 2001 to 2012: Estimates from national demographic and mortality surveys. Lancet Glob. Health 2013, 1, e219–e226. [Google Scholar] [CrossRef]
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- National Family Health Survey-4; Ministry of Health and Family Welfare, Government of India: New Delhi, India, 2016; pp. 105–116. Available online: http://rchiips.org/nfhs/NFHS-4Reports/India.pdf (accessed on 7 December 2019).
- Acharya, D.; Singh, J.K.; Adhikari, M.; Gautam, S.; Pandey, P.; Dayal, V. Association of water handling and child feeding practice with childhood diarrhoea in rural community of southern Nepal. J. Infect Public Health 2018, 11, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Bawankule, R.; Singh, A.; Kumar, K.; Pedgaonkar, S. Disposal of children’s stools and its association with childhood diarrhea in india. BMC Public Health 2017, 17, 12. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Singh, K.P.; Jain, A.; Srivastava, S.; Kumar, V.; Singh, M. Aetiology of childhood viral gastroenteritis in lucknow, north india. Indian J. Med. Res. 2015, 141, 469–472. [Google Scholar] [PubMed]
- Lakshminarayanan, S.; Jayalakshmy, R. Diarrheal diseases among children in india: Current scenario and future perspectives. J. Nat. Sci. Biol. Med. 2015, 6, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Sur, D.; Manna, B.; Deb, A.K.; Deen, J.L.; Danovaro-Holliday, M.C.; von Seidlein, L.; Clemens, J.D.; Bhattacharya, S.K. Factors associated with reported diarrhoea episodes and treatment-seeking in an urban slum of kolkata, india. J. Health Popul. Nutr. 2004, 22, 130–138. [Google Scholar] [PubMed]
- Nilima; Kamath, A.; Shetty, K.; Unnikrishnan, B.; Kaushik, S.; Rai, S.N. Prevalence, patterns, and predictors of diarrhea: A spatial-temporal comprehensive evaluation in india. BMC Public Health 2018, 18, 1288. [Google Scholar] [CrossRef] [PubMed]
- The Treatment of Diarrhoea: A Manual for Physicians and Other Senior Health Workers. Geneva: World Health Organization. Available online: https://www.who.int/maternal_child_adolescent/documents/9241593180/en/ (accessed on 7 December 2019).
- Steiner, M.J.; DeWalt, D.A.; Byerley, J.S. Is this child dehydrated? JAMA 2004, 291, 2746–2754. [Google Scholar] [CrossRef] [PubMed]
- Park, K. Park’s Textbook of Preventive and Social Medicine, 23th ed.; Banarsidas Bhanot Publication: Jabalpur, India, 2015. [Google Scholar]
- WHO. Global Strategy on Infant and Young Child Feeding Geneva, 2002. Available online: https://apps.who.int/iris/bitstream/handle/10665/42590/9241562218.pdf?sequence=1 (accessed on 7 December 2019).
- Dalwai, S.; Choudhury, P.; Bavdekar, S.B.; Dalal, R.; Kapil, U.; Dubey, A.P.; Ugra, D.; Agnani, M.; Sachdev, H.P.S. Consensus statement of the indian academy of pediatrics on integrated management of severe acute malnutrition. Indian Pediatr. 2013, 50, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Statistics and Programme Implementation (MSPI). Concepts and Definitions Used in NSS; Ministry of Statistics and Programme Implementation: New Delhi, India, 2001.
- Dekate, P.; Jayashree, M.; Singhi, S.C. Management of acute diarrhea in emergency room. Indian J. Pediatr. 2013, 80, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Steyerberg, E.W.; Vickers, A.J.; Cook, N.R.; Gerds, T.; Gonen, M.; Obuchowski, N.; Pencina, M.J.; Kattan, M.W. Assessing the performance of prediction models: A framework for traditional and novel measures. Epidemiology 2010, 21, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Alnawajha, S.K.; Bakry, G.A.; Aljeesh, Y.I. Predictors of acute diarrhoea among hospitalized children in gaza governorates: A case-control study. J. Health Popul. Nutr. 2015, 33, 1–8. [Google Scholar]
- Ganguly, E.; Sharma, P.K.; Bunker, C.H. Prevalence and risk factors of diarrhea morbidity among under-five children in india: A systematic review and meta-analysis. Indian J. Child Health 2015, 2, 152–160. [Google Scholar]
- Siziya, S.; Muula, A.S.; Rudatsikira, E. Correlates of diarrhoea among children below the age of 5 years in sudan. Afr. Health Sci. 2013, 13, 376–383. [Google Scholar] [CrossRef]
- Das, J.K.; Bhutta, Z.A. 3.6 reducing the burden of acute and prolonged childhood diarrhea. World Rev. Nutr. Diet 2015, 113, 168–172. [Google Scholar]
- Dos Santos, S.; Ouedraogo Fde, C.; Soura, A.B. Water-related factors and childhood diarrhoea in african informal settlements. A cross-sectional study in ouagadougou (burkina faso). J. Water Health 2015, 13, 562–574. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sinmegn Mihrete, T.; Asres Alemie, G.; Shimeka Teferra, A. Determinants of childhood diarrhea among underfive children in benishangul gumuz regional state, north west ethiopia. BMC Pediatr. 2014, 14, 102. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Das, J.K.; Walker, N.; Rizvi, A.; Campbell, H.; Rudan, I.; Black, R.E.; Lancet Diarrhoea and Pneumonia Interventions Study Group. Interventions to address deaths from childhood pneumonia and diarrhoea equitably: What works and at what cost? Lancet 2013, 381, 1417–1429. [Google Scholar] [CrossRef]
- Das, J.; Das, S.K.; Ahmed, S.; Ferdous, F.; Farzana, F.D.; Sarker, M.H.; Ahmed, A.M.; Chisti, M.J.; Malek, M.A.; Rahman, A.; et al. Determinants of percent expenditure of household income due to childhood diarrhoea in rural bangladesh. Epidemiol. Infect. 2015, 143, 2700–2706. [Google Scholar] [CrossRef]
- Mahmood, D.A.; Feachem, R.G.; Huttly, S.R. Infant feeding and risk of severe diarrhoea in basrah city, iraq: A case-control study. Bull. World Health Organ. 1989, 67, 701–706. [Google Scholar]
- Imdad, A.; Mayo-Wilson, E.; Herzer, K.; Bhutta, Z.A. Vitamin a supplementation for preventing morbidity and mortality in children from six months to five years of age. Cochrane Database Syst. Rev. 2017, 3, CD008524. [Google Scholar] [CrossRef]
- Marufu, T.; Siziya, S.; Tshimanga, M.; Murugasampillay, S.; Mason, E.; Manyame, B. Factors associated with measles complications in gweru, zimbabwe. East Afr. Med. J. 2001, 78, 135–138. [Google Scholar] [CrossRef]
- Pathak, D.; Pathak, A.; Marrone, G.; Diwan, V.; Lundborg, C.S. Adherence to treatment guidelines for acute diarrhoea in children up to 12 years in ujjain, india--a cross-sectional prescription analysis. BMC Infect. Dis. 2011, 11, 32. [Google Scholar] [CrossRef]
- Knee, J.; Sumner, T.; Adriano, Z.; Berendes, D.; de Bruijn, E.; Schmidt, W.P.; Nala, R.; Cumming, O.; Brown, J. Risk factors for childhood enteric infection in urban maputo, mozambique: A cross-sectional study. PLoS Negl. Trop. Dis. 2018, 12, e0006956. [Google Scholar] [CrossRef]
- Kabhele, S.; New-Aaron, M.; Kibusi, S.M.; Gesase, A.P. Prevalence and factors associated with diarrhoea among children between 6 and 59 months of age in mwanza city tanzania. J. Trop. Pediatr. 2018, 64, 523–530. [Google Scholar] [CrossRef]
- Tickell, K.D.; Atlas, H.E.; Walson, J.L. Environmental enteric dysfunction: A review of potential mechanisms, consequences and management strategies. BMC Med. 2019, 17, 181. [Google Scholar] [CrossRef] [PubMed]
- Ejemot-Nwadiaro, R.I.; Ehiri, J.E.; Arikpo, D.; Meremikwu, M.M.; Critchley, J.A. Hand washing promotion for preventing diarrhoea. Cochrane Database Syst. Rev. 2015, 9, CD004265. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.A.; Ensink, J.H.J.; Ramos, M.; Benelli, P.; Holdsworth, E.; Dreibelbis, R.; Cumming, O. Does targeting children with hygiene promotion messages work? The effect of handwashing promotion targeted at children, on diarrhoea, soil-transmitted helminth infections and behaviour change, in low- and middle-income countries. Trop. Med. Int. Health 2017, 22, 526–538. [Google Scholar] [CrossRef] [PubMed]
| Variable | Total (n = 332) (%) | Variable | Total (n = 332) (%) |
|---|---|---|---|
| Eye-ball appearance a | Abnormal respiratory pattern b | ||
| Sunken | 270 (81) | Yes | 89 (27) |
| Normal | 62 (19) | No | 243 (73) |
| Ability to drink a | Poor oral intake | ||
| Normal | 9 (3) | Yes | 291 (88) |
| Eagerly | 300 (90) | No | 41 (12) |
| Poorly | 23 (7) | Normal | 13 (4) |
| Irritability/Restlessness a | Sunken anterior fontanelle c | ||
| Yes | 64 (19) | Yes | 37 (11) |
| No | 268 (80) | No | 295 (89) |
| Lethargic/Unconscious a | Fever | ||
| Yes | 179 (54) | Yes | 289 (87) |
| No | 153 (46) | No | 43 (13) |
| Skin Pinch a | Vomiting | ||
| Normal | 9 (3) | Yes | 235 (71) |
| Slow | 300 (90) | No | 97 (29) |
| Very Slow | 23 (7) | Tenesmus | |
| Capillary refill time b | Yes | 95 (29) | |
| Delayed/prolonged | 298 (90) | No | 237 (71) |
| Normal | 34 (10) | ||
| Urine output b | |||
| Decreased | 301 (91) | ||
| Normal | 31(9) |
| Variable | Severe Dehydration | ||||||
|---|---|---|---|---|---|---|---|
| Total (%) a (n = 332) | Yes (%) b (n = 232) | No (%) b (n = 100) | OR | 95% CI | p Value | ||
| Lower | Upper | ||||||
| Gender | |||||||
| Boys | 179 (54) | 123 (69) | 56 (31) | R | |||
| Girls | 153 (46) | 109 (71) | 44 (29) | 1.03 | 0.90 | 1.19 | 0.616 |
| Age | |||||||
| 1 to 24 months | 246 (74) | 177 (72) | 69 (28) | R | |||
| >2yr to 12yr | 86 (26) | 55 (64) | 31 (36) | 0.88 | 0.74 | 1.06 | 0.792 |
| Resident | |||||||
| Urban | 243 (73) | 160 (66) | 83 (34) | R | |||
| Rural | 89 (27) | 72 (81) | 17 (19) | 1.22 | 1.07 | 1.40 | 0.003 |
| Prematurity | |||||||
| Yes | 321 (97) | 224 (70) | 97 (30) | R | |||
| No | 11 (3) | 8 (73) | 3 (27) | 1.04 | 0.72 | 1.50 | 0.826 |
| Term low birth weight | |||||||
| Yes | 45 (14) | 28 (62) | 17 (38) | R | |||
| No | 287 (86) | 204 (71) | 83 (29) | 1.14 | 0.89 | 1.45 | 0.276 |
| Mother’s education | |||||||
| Literate | 252 (76) | 167 (66) | 85 (34) | R | |||
| Illiterate | 80 (24) | 65 (81) | 15 (19) | 1.22 | 1.06 | 1.40 | 0.004 |
| Mother’s occupation | |||||||
| Housewife | 281 (85) | 190 (68) | 91 (32) | R | |||
| Working | 51 (15) | 42 (82) | 9 (18) | 1.21 | 1.04 | 1.41 | 0.010 |
| Variable | Severe Dehydration | ||||||
|---|---|---|---|---|---|---|---|
| Total (%)a (n = 332) | Yes (%)b (n = 232) | No (%)b (n = 100) | OR | 95%CI | p Value | ||
| Lower | Upper | ||||||
| Exclusive breastfeeding (first 6 months) | |||||||
| Yes | 228 (69) | 143 (63) | 85 (37) | R | |||
| No | 104 (31) | 89 (86) | 15 (14) | 1.36 | 1.20 | 1.54 | <0.001 |
| Bottle feeding (at present) | |||||||
| No | 253 (76) | 169 (67) | 84 (33) | R | |||
| Yes | 79 (24) | 63 (80) | 16 (20) | 1.91 | 1.03 | 1.37 | 0.014 |
| Vitamin A supplementation (past 1 month) | |||||||
| Yes | 280 (84) | 195 (70) | 85 (30) | R | |||
| No | 52 (16) | 37 (71) | 15 (29) | 1.02 | 0.84 | 1.23 | 0.824 |
| History of measles (past 1 month) | |||||||
| No | 299 (90) | 203 (68) | 96 (32) | R | |||
| Yes | 33 (10) | 29 (88) | 4 (12) | 1.29 | 1.11 | 1.50 | 0.001 |
| History of diarrhoea (past 1 month) | |||||||
| No | 140 (42) | 88 (63) | 52 (37) | R | |||
| Yes | 19 2(58) | 144 (75) | 48 (25) | 1.19 | 1.02 | 1.38 | 0.022 |
| SAM (diagnosed on admission) | |||||||
| No | 221 (67) | 144 (65) | 77 (35) | R | |||
| Yes | 111 (33) | 88 (79) | 23 (21) | 1.21 | 1.06 | 1.39 | 0.005 |
| Vaccination on schedule | |||||||
| Yes | 316 (95) | 218 (69) | 98 (31) | R | |||
| No | 16 (5) | 14 (88) | 2 (12) | 1.26 | 1.03 | 1.54 | 0.019 |
| Antibiotic received for present episode | |||||||
| No | 209 (67) | 123 (59) | 86 (41) | R | |||
| Yes | 123 (37) | 109 (89) | 14 (11) | 1.50 | 1.32 | 1.71 | <0.001 |
| ORS received for present episode | |||||||
| Yes | 111 (33) | 98 (88) | 13 (12) | R | |||
| No | 221 (67) | 134 (61) | 87 (39) | 1.47 | 1.29 | 1.66 | <0.001 |
| Oral Zinc received for present episode | |||||||
| Yes | 64 (19) | 21 (33) | 43 (67) | R | |||
| No | 268 (81) | 211 (79) | 57 (21) | 2.23 | 1.68 | 3.42 | <0.001 |
| Home treatment given for present episode | |||||||
| Yes | 67 (20) | 58 (87) | 9 (13) | R | |||
| No | 265 (80) | 174 (66) | 91 (34) | 1.31 | 1.15 | 1.49 | <0.001 |
| Variables | Severe Dehydration | ||||||
|---|---|---|---|---|---|---|---|
| Total (%) a (n = 332) | Yes (%) b (n = 232) | No (%) b (n = 100) | OR | 95% CI | p Value | ||
| Lower | Upper | ||||||
| Type of family | |||||||
| Nuclear | 147 (44) | 106 (72) | 41 (28) | R | |||
| Joint | 185 (56) | 126 (68) | 59 (32) | 0.94 | 0.82 | 1.08 | 0.427 |
| Type of home | |||||||
| Pacca | 235 (71) | 157 (67) | 78 (33) | R | |||
| Kutcha | 97 (29) | 75 (77) | 22 (23) | 1.15 | 1.00 | 1.33 | 0.042 |
| Over-crowding | |||||||
| No | 50 (15) | 27 (54) | 23 (46) | R | |||
| Yes | 282 (85) | 205 (73) | 77 (27) | 1.34 | 1.30 | 1.75 | 0.028 |
| Toilet present | |||||||
| Yes | 285 (86) | 193 (68) | 92 (32) | R | |||
| No | 46 (14) | 38 (82) | 8 (18) | 1.21 | 1.04 | 1.42 | 0.012 |
| Use of toilet | |||||||
| Yes | 283 (85) | 191 (67) | 92 (33) | R | |||
| No | 49 (15) | 41 (83) | 8 (17) | 1.23 | 1.06 | 1.43 | 0.004 |
| Open air defecation | |||||||
| No | 284 (86) | 192 (68) | 92 (32) | R | |||
| Yes | 47 (14) | 39 (83) | 8 (17) | 1.22 | 1.05 | 1.42 | 0.008 |
| Hand washing after going to toilet | |||||||
| Yes | 298 (90) | 202 (68) | 86 (32) | R | |||
| No | 34 (10) | 30 (88) | 4 (12) | 1.30 | 1.12 | 1.50 | <0.001 |
| Finger nail trimmed | |||||||
| Yes | 277 (83) | 188 (68) | 89 (32) | R | |||
| No | 55 (17) | 44 (80) | 11 (20) | 1.71 | 1.00 | 1.37 | 0.038 |
| Eating open/stale food | |||||||
| No | 161 (49) | 114 (71) | 47 (29) | R | |||
| Yes | 171 (51) | 118 (69) | 53 (31) | 0.97 | 0.84 | 1.12 | 0.720 |
| Variable | Adjusted OR | 95% CI | p Value | |
|---|---|---|---|---|
| Lower | Upper | |||
| Age in months * (continuous variable) | 1.26 | 0.65 | 2.46 | 0.483 |
| Sex * (boys vs. girls) | 0.99 | 0.98 | 1.01 | 0.980 |
| Lack of exclusive breastfeeding in first 6 months of life | 5.67 | 2.51 | 12.78 | <0.001 |
| SAM (yes vs. no) | 2.05 | 1.10 | 5.32 | 0.027 |
| History of not receiving ORS before hospitalization | 1.34 | 1.01 | 1.78 | 0.038 |
| History of not receiving oral zinc | 2.66 | 1.68 | 4.21 | <0.001 |
| Living in overcrowded house (yes vs. no) | 5.52 | 2.19 | 13.93 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, A.; Mathur, A.; Stålsby Lundborg, C.; Pathak, A. Incidence and Risk Factors for Severe Dehydration in Hospitalized Children in Ujjain, India. Int. J. Environ. Res. Public Health 2020, 17, 616. https://doi.org/10.3390/ijerph17020616
Sharma A, Mathur A, Stålsby Lundborg C, Pathak A. Incidence and Risk Factors for Severe Dehydration in Hospitalized Children in Ujjain, India. International Journal of Environmental Research and Public Health. 2020; 17(2):616. https://doi.org/10.3390/ijerph17020616
Chicago/Turabian StyleSharma, Abhishek, Aditya Mathur, Cecilia Stålsby Lundborg, and Ashish Pathak. 2020. "Incidence and Risk Factors for Severe Dehydration in Hospitalized Children in Ujjain, India" International Journal of Environmental Research and Public Health 17, no. 2: 616. https://doi.org/10.3390/ijerph17020616
APA StyleSharma, A., Mathur, A., Stålsby Lundborg, C., & Pathak, A. (2020). Incidence and Risk Factors for Severe Dehydration in Hospitalized Children in Ujjain, India. International Journal of Environmental Research and Public Health, 17(2), 616. https://doi.org/10.3390/ijerph17020616






