Cardiovascular Health Effects of Shift Work with Long Working Hours and Night Shifts: Study Protocol for a Three-Year Prospective Follow-Up Study on Industrial Workers
Abstract
1. Introduction
Research Objectives
- The main objective is to determine, prospectively over a period of three years, whether several consecutive days/years with long working days included in a shift plan with night work are associated with unhealthy effects on the cardiovascular system.
- The secondary objective is to evaluate the associations between shift work/long working hours, sleep patterns, and CVD-risk outcomes.
- The third objective is to evaluate whether a three-month scheduled plant shutdown affects CVD-risk outcomes in workers.
- The fourth objective is to evaluate if eight weeks of HIIT intervention affect the selected measures of cardiovascular health.
2. Materials and Methods
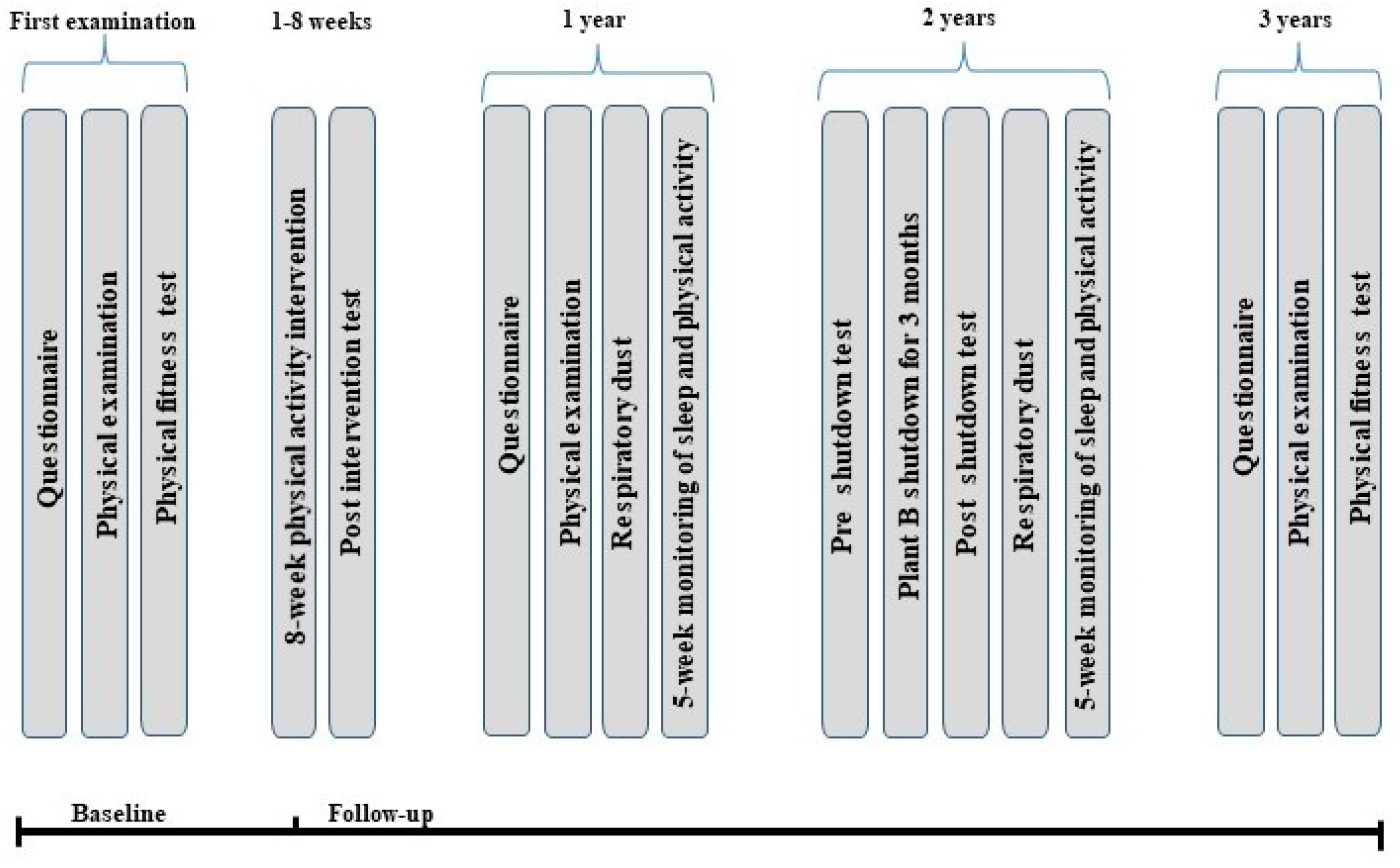
2.1. Setting and Study Population
Shift Work Schedule and Work Content
2.2. Exclusion Criteria for Participation
2.3. Data Collection Period
2.4. Data Collection Procedure at Baseline
2.4.1. Questionnaire
2.4.2. Blood Collection and Plasma Preparation
2.4.3. Quantification of Plasma Cytokines
2.4.4. Blood Pressure and Resting Heart Rate
2.4.5. Central Pulse Pressure and Arterial Stiffness
2.4.6. Carotid Intima-Media Thickness
2.4.7. Aerobic Fitness
2.5. Physical Activity Intervention
2.6. Data Collection Procedure during Follow-Up
2.6.1. Eight-Week Follow-Up
2.6.2. One-Year Follow-Up
2.6.3. Two-Year Follow-Up (Plant B Shutdown)
2.6.4. Three-Year Follow-Up
2.7. Data Management
Data Storage
2.8. Statistical Analyses
2.8.1. Loss to Follow-Up, Compliance, and Missing Data
2.8.2. Study Power
3. Discussion
Strengths and Limitation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body mass index |
| CD40L | CD40 ligand |
| cIMT | Carotis intima media thickness |
| CRP | C-reactive protein |
| CVD | Cardiovascular disease |
| dBP | Diastolic blood pressure |
| EDTA | Ethylene diamine tetra acetic acid |
| HbA1c | Glycosylated hemoglobin |
| HDL | High-density lipoprotein |
| HIIT | High intensity interval training |
| HR | Heart rate |
| HRV | Heart rate variablity |
| IL-6 | Interleukin-6 |
| LDL | Low-density lipoprotein |
| MCP-1 | Monocyte chemoattractant protein-1 |
| sBP | Systolic blood pressure |
| RPM | Revolutions per minute |
| TNF-α | Tumor necrosis factor-alpha |
| VO2max | Maximal oxygen uptake |
| PA | Physical activity |
| PAI | Personalized Activity Intelligence |
| PWV | Pulse wave velocity |
References
- Wong, I.S.; Dawson, D.; Vandongen, H.P. International consensus statements on non-standard working time arrangements and occupational health and safety. Ind. Health 2019, 57, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.M.; Costello, S.; Neophytou, A.M.; Balmes, J.R.; Bradshaw, P.T.; Cullen, M.R.; Eisen, E.A. Night and rotational work exposure within the last 12 months and risk of incident hypertension. Scand. J. Work Environ. Health 2019, 45, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Spurgeon, A.; Harrington, J.M.; Cooper, C.L. Health and safety problems associated with long working hours: A review of the current position. Occup. Environ. Med. 1997, 54, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Schnall, P.L.; Dobson, M.; Landsbergis, P. Globalization, Work, and Cardiovascular Disease. Int. J. Health Serv. Plan. Adm. Eval. 2016, 46, 656–692. [Google Scholar] [CrossRef] [PubMed]
- Russek, H.I.; Zohman, B.L. Relative significance of heredity, diet and occupational stress in coronary heart disease of young adults; based on an analysis of 100 patients between the ages of 25 and 40 years and a similar group of 100 normal control subjects. Am. J. Med. Sci. 1958, 235, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Vyas, M.V.; Garg, A.X.; Iansavichus, A.V.; Costella, J.; Donner, A.; Laugsand, L.E.; Janszky, I.; Mrkobrada, M.; Parraga, G.; Hackam, D.G. Shift work and vascular events: Systematic review and meta-analysis. BMJ 2012, 345, e4800. [Google Scholar] [CrossRef] [PubMed]
- Torquati, L.; Mielke, G.I.; Brown, W.J.; Kolbe-Alexander, T. Shift work and the risk of cardiovascular disease. A systematic review and meta-analysis including dose-response relationship. Scand. J. Work Environ. Health 2018, 44, 229–238. [Google Scholar] [CrossRef]
- Virtanen, M.; Heikkilä, K.; Jokela, M.; Ferrie, J.E.; Batty, G.D.; Vahtera, J.; Kivimäki, M. Long working hours and coronary heart disease: A systematic review and meta-analysis. Am. J. Epidemiol. 2012, 176, 586–596. [Google Scholar] [CrossRef]
- Brotman, D.J.; Golden, S.H.; Wittstein, I.S. The cardiovascular toll of stress. Lancet 2007, 370, 1089–1100. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Herman, J.P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 2009, 10, 397–409. [Google Scholar] [CrossRef]
- Leung, M.; Tranmer, J.; Hung, E.; Korsiak, J.; Day, A.G.; Aronson, K.J. Shift Work, Chronotype, and Melatonin Patterns among Female Hospital Employees on Day and Night Shifts. Cancer Epidemiol. Biomark. Prev. A Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2016, 25, 830–838. [Google Scholar] [CrossRef]
- Ulhôa, M.A.; Marqueze, E.C.; Burgos, L.G.A.; Moreno, C.R.D.C. Shift work and endocrine disorders. Int. J. Endocrinol. 2015, 2015, 826249. [Google Scholar] [CrossRef] [PubMed]
- Aho, V.; Ollila, H.M.; Rantanen, V.; Kronholm, E.; Surakka, I.; van Leeuwen, W.M.; Lehto, M.; Matikainen, S.; Ripatti, S.; Härmä, M.; et al. Partial sleep restriction activates immune response-related gene expression pathways: Experimental and epidemiological studies in humans. PLoS ONE 2013, 8, e77184. [Google Scholar] [CrossRef] [PubMed]
- Kivimäki, M.; Jokela, M.; Nyberg, S.T.; Singh-Manoux, A.; Fransson, E.I.; Alfredsson, L.; Bjorner, J.B.; Borritz, M.; Burr, H.; Casini, A.; et al. Long working hours and risk of coronary heart disease and stroke: A systematic review and meta-analysis of published and unpublished data for 603,838 individuals. Lancet 2015, 386, 1739–1746. [Google Scholar] [CrossRef]
- Frost, P.; Kolstad, H.A.; Bonde, J.P. Shift work and the risk of ischemic heart disease—A systematic review of the epidemiologic evidence. Scand. J. Work Environ. Health 2009, 35, 163–179. [Google Scholar] [CrossRef] [PubMed]
- Ishii, N.; Dakeishi, M.; Sasaki, M.; Iwata, T.; Murata, K. Cardiac autonomic imbalance in female nurses with shift work. Auton. Neurosci. Basic Clin. 2005, 122, 94–99. [Google Scholar] [CrossRef]
- Su, T.C.; Lin, L.Y.; Baker, D.; Schnall, P.L.; Chen, M.F.; Hwang, W.C.; Chen, C.F.; Wang, J.D. Elevated blood pressure, decreased heart rate variability and incomplete blood pressure recovery after a 12-h night shift work. J. Occup. Health 2008, 50, 380–386. [Google Scholar] [CrossRef]
- Van Amelsvoort, L.G.; Schouten, E.G.; Maan, A.C.; Swenne, C.A.; Kok, F.J. Changes in frequency of premature complexes and heart rate variability related to shift work. Occup. Environ. Med. 2001, 58, 678–681. [Google Scholar] [CrossRef]
- Härmä, M.; Ropponen, A.; Hakola, T.; Koskinen, A.; Vanttola, P.; Puttonen, S.; Sallinen, M.; Salo, P.; Oksanen, T.; Pentti, J.; et al. Developing register-based measures for assessment of working time patterns for epidemiologic studies. Scand. J. Work Environ. Health 2015, 41, 268–279. [Google Scholar]
- Kecklund, G.; Axelsson, J. Health consequences of shift work and insufficient sleep. BMJ 2016, 355, i5210. [Google Scholar] [CrossRef]
- Grandner, M.A.; Chakravorty, S.; Perlis, M.L.; Oliver, L.; Gurubhagavatula, I. Habitual sleep duration associated with self-reported and objectively determined cardiometabolic risk factors. Sleep Med. 2014, 15, 42–50. [Google Scholar] [CrossRef]
- Sivertsen, B.; Lallukka, T.; Salo, P.; Pallesen, S.; Hysing, M.; Krokstad, S.; Øverland, S. Insomnia as a risk factor for ill health: Results from the large population-based prospective HUNT Study in Norway. J. Sleep Res. 2014, 23, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Van der Hulst, M. Long workhours and health. Scand. J. Work Environ. Health 2003, 29, 171–188. [Google Scholar] [CrossRef] [PubMed]
- Sjogren, B. Occupational exposure to dust: Inflammation and ischaemic heart disease. Occup. Environ. Med. 1997, 54, 466–469. [Google Scholar] [CrossRef] [PubMed]
- Medical, A. Actor Medical 2019. Available online: www.atcormedical.com (accessed on 20 November 2019).
- Ross, R. The pathogenesis of atherosclerosis: A perspective for the 1990s. Nature 1993, 362, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Touboul, P.J.; Hennerici, M.G.; Meairs, S.; Adams, H.; Amarenco, P.; Bornstein, N.; Csiba, L.; Desvarieux, M.; Ebrahim, S.; Hernandez, R.H.; et al. Mannheim carotid intima-media thickness and plaque consensus (2004-2006-2011). An Update on Behalf of the Advisory Board of the 3rd, 4th and 5th Watching the Risk Symposia, at the 13th, 15th and 20th European Stroke Conferences, Mannheim, Germany, 2004, Brussels, Belgium, 2006, and Hamburg, Germany, 2011. Cerebrovasc. Dis. 2012, 34, 290–296. [Google Scholar]
- Gerrit, L.; Gert-Jan, R.; van den Oord, S.C.; Dedic, A.; Dharampal, A.S.; Nieman, K.; de Feyter, P.J.; Sijbrands, E.J.; van der Steen, A.F.; Schinkel, A.F. Carotid plaque burden as a measure of subclinical coronary artery disease in patients with heterozygous familial hypercholesterolemia. Am. J. Cardiol. 2013, 111, 1305–1310. [Google Scholar]
- Tjønna, A.E.; Leinan, I.M.; Bartnes, A.T.; Jenssen, B.M.; Gibala, M.J.; Winett, R.A.; Wisløff, U. Low- and high-volume of intensive endurance training significantly improves maximal oxygen uptake after 10-weeks of training in healthy men. PLoS ONE 2013, 8, e65382. [Google Scholar] [CrossRef]
- Borg, G. Perceived exertion as an indicator of somatic stress. Scand. J. Rehabilit. Med. 1970, 2, 92–98. [Google Scholar]
- Nes, B.M.; Gutvik, C.R.; Lavie, C.J.; Nauman, J.; Wisloff, U. Personalized Activity Intelligence (PAI) for Prevention of Cardiovascular Disease and Promotion of Physical Activity. Am. J. Med. 2017, 130, 328–336. [Google Scholar] [CrossRef]
- Kieffer, S.K.; Croci, I.; Wisloff, U.; Nauman, J. Temporal Changes in a Novel Metric of Physical Activity Tracking (Personal Activity Intelligence) and Mortality: The HUNT Study, Norway. Prog. Cardiovasc. Dis. 2019, 62, 186–192. [Google Scholar] [CrossRef]
- Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur. Heart J. 1996, 17, 354–381. [Google Scholar]
- Carney, C.E.; Buysse, D.J.; Ancoli-Israel, S.; Edinger, J.D.; Krystal, A.D.; Lichstein, K.L.; Morin, C.M. The consensus sleep diary: Standardizing prospective sleep self-monitoring. Sleep 2012, 35, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Wisland, D.T.; Granhaug, K.; Pleym, J.R.; Andersen, N.; Støa, S.; Hjortland, H.A. Remote monitoring of vital signs using a CMOS UWB radar transceiver. In 2016 14th IEEE International New Circuits and Systems Conference (NEWCAS); IEEE: Piscataway, NJ, USA, 2016; pp. 1–4. [Google Scholar]
- Krzywinski, M.; Altman, N. Comparing samples—part II. Nat. Methods 2014, 11, 355. [Google Scholar] [CrossRef]
- Skogstad, M.; Lunde, L.K.; Ulvestad, B.; Aass, H.; Clemm, T.; Mamen, A.; Skare, Ø. The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study. Int. J. Environ. Res. Public Health 2018, 15, 264. [Google Scholar] [CrossRef] [PubMed]
- Jankowiak, S.; Backé, E.; Liebers, F.; Schulz, A.; Hegewald, J.; Garthus-Niegel, S.; Nübling, M.; Blankenberg, S.; Pfeiffer, N.; Lackner, K.J.; et al. Current and cumulative night shift work and subclinical atherosclerosis: Results of the Gutenberg Health Study. Int. Arch. Occup. Environ. Health 2016, 89, 1169–1182. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Shiu, L.J.; Li, Y.L.; Tung, K.Y.; Chan, K.Y.; Yeh, C.J.; Chen, S.C.; Wong, R.H. Shift work and arteriosclerosis risk in professional bus drivers. Ann. Epidemiol. 2010, 20, 60–66. [Google Scholar] [CrossRef]
- Puttonen, S.; Kivimäki, M.; Elovainio, M.; Pulkki-Råback, L.; Hintsanen, M.; Vahtera, J.; Telama, R.; Juonala, M.; Viikari, J.S.; Raitakari, O.T.; et al. Shift work in young adults and carotid artery intima-media thickness: The Cardiovascular Risk in Young Finns study. Atherosclerosis 2009, 205, 608–613. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in atherosclerosis. Nature 2002, 420, 868–874. [Google Scholar] [CrossRef]
- Fleg, J.L.; Stone, G.W.; Fayad, Z.A.; Granada, J.F.; Hatsukami, T.S.; Kolodgie, F.D.; Ohayon, J.; Pettigrew, R.; Sabatine, M.S.; Tearney, G.J.; et al. Detection of high-risk atherosclerotic plaque: Report of the NHLBI Working Group on current status and future directions. JACC Cardiovasc. Imaging 2012, 5, 941–955. [Google Scholar] [CrossRef]
- Tuttolomondo, A.; Di Raimondo, D.; Pecoraro, R.; Arnao, V.; Pinto, A.; Licata, G. Atherosclerosis as an inflammatory disease. Curr. Pharm. Des. 2012, 18, 4266–4288. [Google Scholar] [CrossRef]
- Stoner, L.; Lucero, A.A.; Palmer, B.R.; Jones, L.M.; Young, J.M.; Faulkner, J. Inflammatory biomarkers for predicting cardiovascular disease. Clin. Biochem. 2013, 46, 1353–1371. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Cushman, M.; Stampfer, M.J.; Tracy, R.P.; Hennekens, C.H. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N. Engl. J. Med. 1997, 336, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Herder, M.; Johnsen, S.H.; Arntzen, K.A.; Mathiesen, E.B. Risk factors for progression of carotid intima-media thickness and total plaque area: A 13-year follow-up study: The Tromso Study. Stroke 2012, 43, 1818–1823. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.W.; Markus, H.S.; Bots, M.L.; Rosvall, M.; Sitzer, M. Prediction of clinical cardiovascular events with carotid intima-media thickness: A systematic review and meta-analysis. Circulation 2007, 115, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Fujishiro, K.; Roux, A.V.D.; Landsbergis, P.; Baron, S.; Barr, R.G.; Kaufman, J.D.; Polak, J.F.; Stukovsky, K.H. Associations of occupation, job control and job demands with intima-media thickness: The Multi-Ethnic Study of Atherosclerosis (MESA). Occup. Environ. Med. 2011, 68, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Charles, L.E.; Fekedulegn, D.; Burchfiel, C.M.; Fujishiro, K.; Landsbergis, P.; Roux, A.V.D.; MacDonald, L.; Foy, C.G.; Andrew, M.E.; Stukovsky, K.H.; et al. Associations of work hours with carotid intima-media thickness and ankle-brachial index: The Multi-Ethnic Study of Atherosclerosis (MESA). Occup. Environ. Med. 2012, 69, 713–720. [Google Scholar] [CrossRef]
- Williams, B.; Lacy, P.S.; Thom, S.M.; Cruickshank, K.; Stanton, A.; Collier, D.; Hughes, A.D.; Thurston, H.; O’Rourke, M. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: Principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation 2006, 113, 1213–1225. [Google Scholar] [CrossRef]
- Roman, M.J.; Devereux, R.B.; Kizer, J.R.; Okin, P.M.; Lee, E.T.; Wang, W.; Umans, J.G.; Calhoun, D.; Howard, B.V. High central pulse pressure is independently associated with adverse cardiovascular outcome the strong heart study. J. Am. Coll. Cardiol. 2009, 54, 1730–1734. [Google Scholar] [CrossRef]
- McEniery, C.M.; Cockcroft, J.R.; Roman, M.J.; Franklin, S.S.; Wilkinson, I.B. Central blood pressure: Current evidence and clinical importance. Eur. Heart J. 2014, 35, 1719–1725. [Google Scholar] [CrossRef]
- Nelson, M.R.; Stepanek, J.; Cevette, M.; Covalciuc, M.; Hurst, R.T.; Tajik, A.J. Noninvasive measurement of central vascular pressures with arterial tonometry: Clinical revival of the pulse pressure waveform? Mayo Clin. Proc. 2010, 85, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Lear, S.A.; Hu, W.; Rangarajan, S.; Gasevic, D.; Leong, D.; Iqbal, R.; Casanova, A.; Swaminathan, S.; Anjana, R.M.; Kumar, R.; et al. The effect of physical activity on mortality and cardiovascular disease in 130 000 people from 17 high-income, middle-income, and low-income countries: The PURE study. Lancet 2017, 390, 2643–2654. [Google Scholar] [CrossRef]
- Mustafić, H.; Jabre, P.; Caussin, C.; Murad, M.H.; Escolano, S.; Tafflet, M.; Périer, M.C.; Marijon, E.; Vernerey, D.; Empana, J.P.; et al. Main air pollutants and myocardial infarction: A systematic review and meta-analysis. JAMA 2012, 307, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Byrkjeland, R.; Stensæth, K.H.; Anderssen, S.; Njerve, I.U.; Arnesen, H.; Seljeflot, I.; Solheim, S. Effects of exercise training on carotid intima-media thickness in patients with type 2 diabetes and coronary artery disease. Influence of carotid plaques. Cardiovasc. Diabetol. 2016, 15, 13. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Morris, C.J.; Caputo, R.; Wang, W.; Garaulet, M.; Scheer, F.A. Sex differences in the circadian misalignment effects on energy regulation. Proc. Natl. Acad. Sci. USA 2019, 116, 23806–23812. [Google Scholar] [CrossRef]
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weekday | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| Plant A | E | E | E | N | N | D | D | E | E | N | N | D | D | D | D | D | N | N | N | ||||||||||||||||
| Plant B | D | D | D | D | N | N | N | N | E | E | N | N | N | D | D | D | E | E | |||||||||||||||||
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weekday | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| Before | N | N | N | D | D | D | D | D | D | D | N | N | N | N | N | N | N | ||||||||||||||||||
| After | D | D | D | D | N | N | N | N | E | E | N | N | N | D | D | D | E | E | |||||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lunde, L.-K.; Skare, Ø.; Mamen, A.; Sirnes, P.A.; Aass, H.C.D.; Øvstebø, R.; Goffeng, E.; Matre, D.; Nielsen, P.; Heglum, H.S.A.; et al. Cardiovascular Health Effects of Shift Work with Long Working Hours and Night Shifts: Study Protocol for a Three-Year Prospective Follow-Up Study on Industrial Workers. Int. J. Environ. Res. Public Health 2020, 17, 589. https://doi.org/10.3390/ijerph17020589
Lunde L-K, Skare Ø, Mamen A, Sirnes PA, Aass HCD, Øvstebø R, Goffeng E, Matre D, Nielsen P, Heglum HSA, et al. Cardiovascular Health Effects of Shift Work with Long Working Hours and Night Shifts: Study Protocol for a Three-Year Prospective Follow-Up Study on Industrial Workers. International Journal of Environmental Research and Public Health. 2020; 17(2):589. https://doi.org/10.3390/ijerph17020589
Chicago/Turabian StyleLunde, Lars-Kristian, Øivind Skare, Asgeir Mamen, Per Anton Sirnes, Hans C. D. Aass, Reidun Øvstebø, Elisabeth Goffeng, Dagfinn Matre, Pia Nielsen, Hanne Siri Amdahl Heglum, and et al. 2020. "Cardiovascular Health Effects of Shift Work with Long Working Hours and Night Shifts: Study Protocol for a Three-Year Prospective Follow-Up Study on Industrial Workers" International Journal of Environmental Research and Public Health 17, no. 2: 589. https://doi.org/10.3390/ijerph17020589
APA StyleLunde, L.-K., Skare, Ø., Mamen, A., Sirnes, P. A., Aass, H. C. D., Øvstebø, R., Goffeng, E., Matre, D., Nielsen, P., Heglum, H. S. A., Hammer, S. E., & Skogstad, M. (2020). Cardiovascular Health Effects of Shift Work with Long Working Hours and Night Shifts: Study Protocol for a Three-Year Prospective Follow-Up Study on Industrial Workers. International Journal of Environmental Research and Public Health, 17(2), 589. https://doi.org/10.3390/ijerph17020589





