Autism Spectrum Disorders: Prenatal Genetic Testing and Abortion Decision-Making among Taiwanese Mothers of Affected Children
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Data Collection Procedure
2.3. Measures
2.4. Statistical Analysis
3. Results
3.1. Psychosocial Factors Associated with PGT for Detecting ASD Susceptibility Genes and Abortion Decision-Making
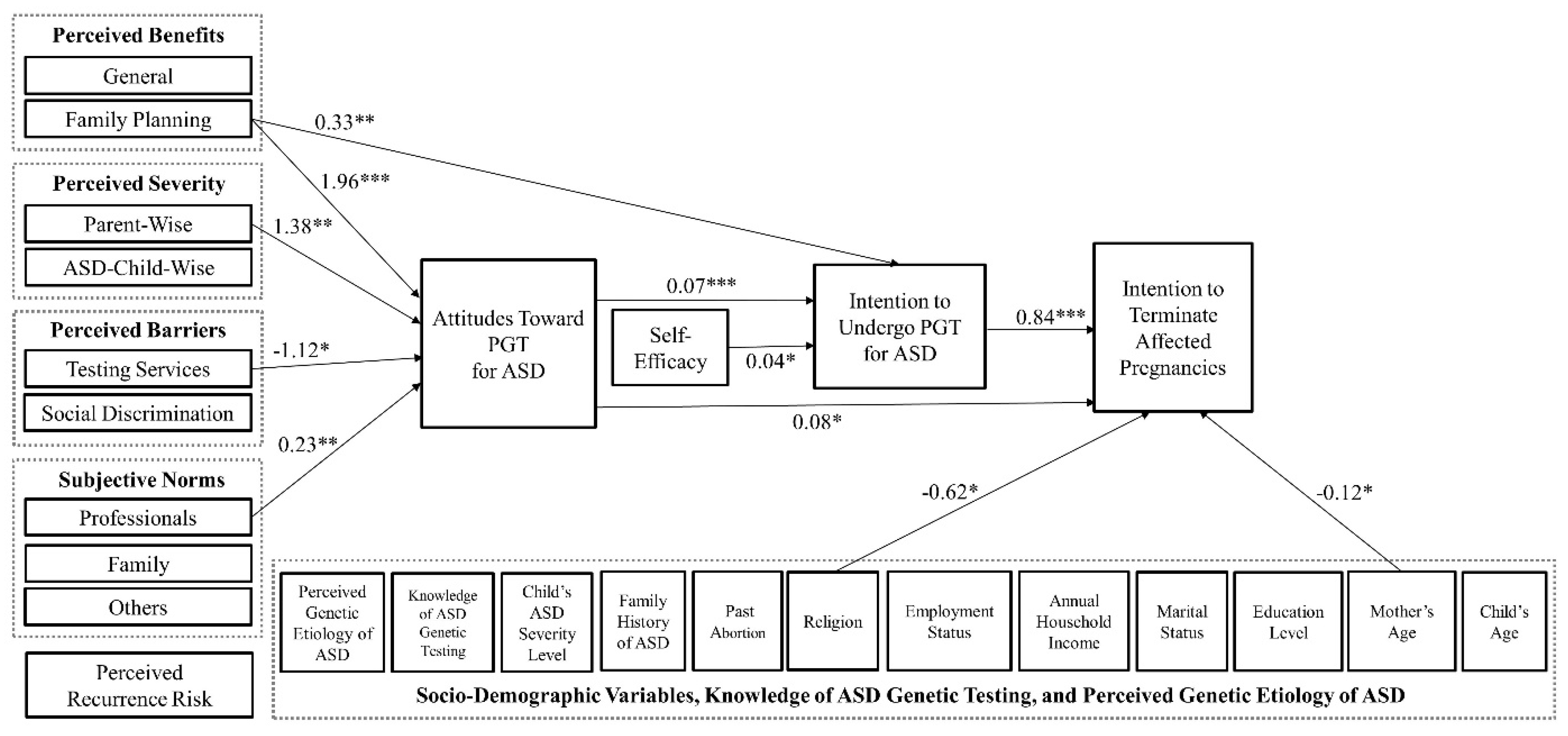
3.2. SEM Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Robert, C.; Pasquier, L.; Cohen, D.; Fradin, M.; Canitano, R.; Damaj, L.; Tordjman, S. Role of genetics in the etiology of Autistic Spectrum Disorder: Towards a hierarchical diagnostic strategy. Int. J. Mol. Sci. 2017, 18, 618. [Google Scholar] [CrossRef] [PubMed]
- Taipei Autism Children Social Welfare Foundation. Prevalence of Autism in Taiwan. Available online: http://www.ican.org.tw/content.asp?id=2 (accessed on 30 October 2019).
- Chen, S. A Study on the Related Factors of Prevalence, Incidence and Comorbidity of Autistic Children. Available online: http://ntcuir.ntcu.edu.tw/handle/987654321/12469 (accessed on 30 October 2019).
- Hsu, S.; Chiang, P.; Lin, L.; Lin, J. Disparity in autism spectrum disorder prevalence among Taiwan National Health Insurance enrollees: Age, gender and urbanization effects. Res. Autism Spectr. Disord. 2012, 6, 836–841. [Google Scholar] [CrossRef]
- Lai, D.; Tseng, Y.; Guo, H. Trends in the prevalence of childhood disability: Analysis of data from the national disability registry of Taiwan, 2000–2011. Res. Dev. Disabil. 2013, 34, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, G.B.; Mendelsohn, N.J. Clinical genetics evaluation in identifying the etiology of autism spectrum disorders: Guideline revisions. Genet. Med. 2013, 15, 399–407. [Google Scholar] [CrossRef]
- Tick, B.; Bolton, P.; Happé, F.; Rutter, M.; Rijsdijk, F. Heritability of autism spectrum disorders: A meta-analysis of twin studies. J. Child Psychol. Psychiatry 2016, 57, 585–595. [Google Scholar] [CrossRef]
- Tordjman, S.; Somogyi, E.; Coulon, N.; Kermarrec, S.; Cohen, D.; Bronsard, G.; Xavier, J. Gene × environment interactions in autism spectrum disorders: Role of epigenetic mechanisms. Front Psychiatry 2014, 5, 53. [Google Scholar] [CrossRef]
- Ko’s Obstetrics & Gynecology. The International Standards for Ctogenomic Arrays Cortium Analysis Application Procedure. Available online: http://www.genephile.com.tw/aCGH/htm/ISCA%20array.htm (accessed on 30 October 2019).
- Neogene Obstetric and Pediatrics Clinic. Array Comparative Genomic Hybridization (aCGH) Introduction. Available online: http://www.neogene.com.tw/ugC_DrNews_Detail.asp?hidDrNewsID=32 (accessed on 30 October 2019).
- National Taiwan University Hospital. Array CGH. Available online: https://www.ntuh.gov.tw/gene/lab/prenatal/Pages/aCGH.aspx (accessed on 30 October 2019).
- Zhao, S.; Wang, C.; Huang, T.; Chen, Y.; Hsiao, C.; Tseng, C.; Xu, L.; Chen, L. A qualitative study exploring the attitudes toward prenatal genetic testing for autism spectrum disorders among parents of affected children in Taiwan. Res. Autism Spectr. Disord. 2018, 48, 36–43. [Google Scholar] [CrossRef]
- Klugman, S.; Suskin, B.; Spencer, B.L.; Dar, P.; Bajaj, K.; Powers, J.; Reichling, J.; Wasserman, D.; Dolan, S.M.; Merkatz, I.R. Clinical utility of chromosomal microarray analysis in prenatal diagnosis: Report of first 6 months in clinical practice. J. Matern. Fetal Neonatal Med. 2014, 27, 1333–1338. [Google Scholar] [CrossRef]
- Armengol, L.; Nevado, J.; Serra-Juhe, C.; Plaja, A.; Mediano, C.; Garcia-Santiago, F.A.; Garcia-Aragones, M.; Villa, O.; Mansilla, E.; Preciado, C.; et al. Clinical utility of chromosomal microarray analysis in invasive prenatal diagnosis. Hum. Genet. 2012, 131, 513–523. [Google Scholar] [CrossRef]
- Hillman, S.C.; McMullan, D.J.; Hall, G.; Togneri, F.S.; James, N.; Maher, E.J.; Meller, C.H.; Williams, D.; Wapner, R.J.; Maher, E.R.; et al. Use of prenatal chromosomal microarray: Prospective cohort study and systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2013, 41, 610–620. [Google Scholar] [CrossRef]
- Miller, D.T.; Adam, M.P.; Aradhya, S.; Biesecker, L.G.; Brothman, A.R.; Carter, N.P.; Church, D.M.; Crolla, J.A.; Eichler, E.E.; Epstein, C.J.; et al. Consensus Statement: Chromosomal Microarray Is a First-Tier Clinical Diagnostic Test for Individuals with Developmental Disabilities or Congenital Anomalies. Am. J. Hum. Genet. 2010, 86, 749–764. [Google Scholar] [CrossRef] [PubMed]
- National Health Insurance Administration Ministry of Health and Welfare. 2017–2018 National Health Insurance Annual Report; National Health Insurance Administration Ministy of Health and Welfare: Taipei, Taiwan, 2017. [Google Scholar]
- Chen, L.; Xu, L.; Dhar, S.; Li, M.; Talwar, D.; Jung, E. Autism spectrum disorders: A qualitative study of attitudes toward prenatal genetic testing and termination decisions of affected pregnancies. Clin. Genet. 2015, 88, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhao, M.; Zhou, Q.; Xu, L. Chinese Americans’ views of prenatal genetic testing in the genomic era: A qualitative study. Clin. Genet. 2012, 82, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Masi, A.; DeMayo, M.M.; Glozier, N.; Guastella, A.J. An overview of Autism Spectrum Disorder, heterogeneity and treatment options. Neurosci. Bull. 2017, 33, 183–193. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, Y.; Xiu, X.; Choy, K.; Pursley, A.; Cheung, S. Genetic diagnosis of autism spectrum disorders: The opportunity and challenge in the genomics era. Crit. Rev. Clin. Lab. Sci. 2014, 51, 249–262. [Google Scholar] [CrossRef][Green Version]
- Rossi, M.; El-Khechen, D.; Black, M.H.; Hagman, K.D.F.; Tang, S.; Powis, Z. Outcomes of Diagnostic Exome Sequencing in Patients With Diagnosed or Suspected Autism Spectrum Disorders. Pediatr. Neurol. 2017, 70, 34–43. [Google Scholar] [CrossRef]
- Ho, K.S.; Wassman, E.R.; Baxter, A.L.; Hensel, C.H.; Martin, M.M.; Prasad, A.; Twede, H.; Vanzo, R.J.; Butler, M.G. Chromosomal Microarray Analysis of Consecutive Individuals with Autism Spectrum Disorders Using an Ultra-High Resolution Chromosomal Microarray Optimized for Neurodevelopmental Disorders. Int. J. Mol. Sci. 2016, 17, 2070. [Google Scholar] [CrossRef]
- Creswell, J.W.; Creswell, D.J. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches; SAGE Publications, Inc.: Los Angeles, CA, USA, 2018. [Google Scholar]
- Janz, N.K.; Becker, M.H. The Health Belief Model: A decade later. Health Educ. Q. 1984, 11, 1–47. [Google Scholar] [CrossRef]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Decis. Process. 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Bandura, A. Social Foundations of Thought and Action: A Social Cognitive Theory; Prentice-Hall, Inc.: Englewood Cliffs, NJ, USA, 1986. [Google Scholar]
- Gooding, H.C.; Organista, K.; Burack, J.; Biesecker, B.B. Genetic susceptibility testing from a stress and coping perspective. Soc. Sci. Med. 2006, 62, 1880–1890. [Google Scholar] [CrossRef]
- Levine, F.R.; Coxworth, J.E.; Stevenson, D.A.; Tuohy, T.; Burt, R.W.; Kinney, A.Y. Parental attitudes, beliefs, and perceptions about genetic testing for FAP and colorectal cancer surveillance in minors. J. Genet. Couns. 2010, 19, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.M.; Shoda, Y.; Hurley, K. Applying cognitive-social theory to health-protective behavior: Breast self-examination in cancer screening. Psychol. Bull. 1996, 119, 70–94. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.; Chao, Y.; Yuh, Y. Informed choice of pregnant women in prenatal screening tests for Down’s syndrome. J. Med. Ethics 2006, 32, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Huang, T.; Ye, J.; Zhao, S.; Chen, L. Perceived recurrence risk of having another affected child: A survey on parents of children with autism spectrum disorders in Taiwan. Patient Educ. Couns. 2018, 101, 926–931. [Google Scholar] [CrossRef]
- Leung, T.N.; Chau, M.; Chang, J.; Leung, T.Y.; Fung, T.Y.; Tze, T.K. Attitudes towards termination of pregnancy among Hong Kong Chinese women attending prenatal diagnosis counselling clinic. Prenat. Diagn. 2004, 24, 546–551. [Google Scholar] [CrossRef]
- Chen, L.; Ye, J.; Talwar, D.; Zhao, S.; Wang, C.; Huang, T.; Barry, A.E. Perceived causes of autism spectrum disorders among Taiwanese parents of affected children: A qualitative study. Glob. J. Anthropol. Res. 2014, 1, 12–19. [Google Scholar] [CrossRef][Green Version]
- Lin, J.; Sung, C.; Lin, L.; Hsu, S.; Chien, W.; Su, S.; Weu, J. Ten-year trend analysis of autism severity: A nationwide population-based register study. Res. Autism Spectr. Disord. 2011, 5, 570–574. [Google Scholar] [CrossRef]
- Chen, L.; Xu, L.; Huang, T.; Dhar, S.U. Autism genetic testing: A qualitative study of awareness, attitudes, and experiences among parents of children with autism spectrum disorders. Genet. Med. 2013, 15, 274–281. [Google Scholar] [CrossRef]
- Chen, L.; Li, C.; Wang, C.; Amuta, A.; Li, M.; Huang, T.; Jung, E. Autism spectrum disorders: Perceptions of genetic etiology and recurrence risk among Taiwanese parents of affected children. Clin. Genet. 2015, 88, 129–134. [Google Scholar] [CrossRef]
- Doukas, D.J.; Localio, A.R.; Li, Y. Attitudes and beliefs concerning prostate cancer genetic screening. Clin. Genet. 2004, 66, 445–451. [Google Scholar] [CrossRef]
- Li, M.; Amuta, A.; Xu, L.; Dhar, S.U.; Talwar, D.; Jung, E.; Chen, L. Autism genetic testing information needs among parents of affected children: A qualitative study. Patient Educ. Couns. 2016, 99, 1011–1016. [Google Scholar] [CrossRef]
- Mercer, L.; Creighton, S.; Holden, J.A.; Lewis, M.S. Parental perspectives on the causes of an Autism Spectrum Disorder in their children. J. Genet. Couns. 2006, 15, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Sagi, M.; Shiloh, S.; Cohen, T. Application of the Health Belief Model in a study on parents’ intentions to utilize prenatal diagnosis of cleft lip and/or palate. Am. J. Med. Genet. 1992, 44, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Selkirk, C.G.; Veach, P.M.; Lian, F.; Schimmenti, L.; LeRoy, B.S. Parents’ perceptions of autism spectrum disorder etiology and recurrence risk and effects of their perceptions on family planning: Recommendations for genetic counselors. J. Genet. Couns. 2009, 18, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Yanushka Bunn, J.; Bosompra, K.; Ashikaga, T.; Flynn, B.S.; Worden, J.K. Factors influencing intention to obtain a genetic test for colon cancer risk: A population-based study. Prev. Med. 2002, 34, 567–577. [Google Scholar] [CrossRef]
- Li, Y. AI Medical Future: Taiwan’s First Intelligent, Key Medical Report; Hao Ren Publishing: Taipei, Taiwan, 2018. [Google Scholar]
- Chien, S.; Su, P.; Chen, S. Development of genetic counseling services in Taiwan. J. Genet. Couns. 2013, 22, 839–843. [Google Scholar] [CrossRef]
- Hu, L.T.; Bentler, P.M. Cutoff criteria for fit indexes in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. A Multidiscip. J. 1999, 6, 1–55. [Google Scholar] [CrossRef]
- Wiesner, M.; Schanding, G.T. Exploratory structural equation modeling, bifactor models, and standard confirmatory factor analysis models: Application to the BASC-2 behavioral and emotional screening system teacher form. J. Sch. Psychol. 2013, 51, 751–763. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics Using SPSS; SAGE Publications Ltd.: Thousand Oaks, CA, USA, 2009. [Google Scholar]
- Tsai, G.J.; Cameron, C.A.; Czerwinski, J.L.; Mendez-Figueroa, H.; Peterson, S.K.; Noblin, S.J. Attitudes towards prenatal genetic counseling, prenatal genetic testing, and termination of pregnancy among Southeast and East Asian women in the United States. J. Genet. Couns. 2017, 26, 1041–1058. [Google Scholar] [CrossRef]
- Ministry of Health and Welfare. Genetic Health Act. Available online: https://law.moj.gov.tw/ENG/LawClass/LawAll.aspx?pcode=L0070001 (accessed on 10 January 2020).
- Learman, L.A.; Kuppermann, M.; Gates, E.; Nease, R.F.; Gildengorin, V.; Washington, A.E. Social and familial context of prenatal genetic testing decisions: Are there racial/ethnic differences? Am. J. Med. Genet. 2003, 119C, 19–26. [Google Scholar] [CrossRef]
- Novelli, A.; Grati, F.R.; Ballarati, L.; Bernardini, L.; Bizzoco, D.; Camurri, L.; Casalone, R.; Cardarelli, L.; Cavalli, P.; Ciccone, R.; et al. Microarray application in prenatal diagnosis: A position statement from the cytogenetics working group of the Italian Society of Human Genetics (SIGU), November 2011. Ultrasound Obstet. Gynecol. 2012, 39, 384–388. [Google Scholar] [CrossRef] [PubMed]
| Variable | M (SD)/% |
|---|---|
| Age | 38.93 (4.88) |
| Education level | |
| Below college | 67.9% |
| College or above | 32.1% |
| Marital status | |
| Married | 90.1% |
| Others | 9.9% |
| Annual household income | |
| Less than NT$600,000 (~US$19,745) | 35.3% |
| NT$600,000–1,200,000 (~US$19,745–39,489) | 41.0% |
| Above NT$1,200,000 (~US$39,489) | 23.7% |
| Employment status | |
| Employed | 55.1% |
| Not employed | 44.9% |
| Religion | |
| Folk religion | 27.6% |
| Buddhism | 27.6% |
| Christianity | 10.0% |
| No religion | 22.4% |
| Multiple religions | 12.4% |
| Previous abortion experience | |
| Yes | 42.9% |
| No | 57.1% |
| Family history of ASD | |
| Yes | 35.7% |
| No | 64.3% |
| Severity level of the child(ren) with ASD a | 1.58 (0.76) |
| Current age of child(ren) with ASD | 9.61 (2.13) |
| Knowledge of PGT for detecting ASD susceptibility genes b | 0.33 (0.22) |
| Perceived genetic etiology of ASD | |
| Yes | 43.4% |
| No | 56.6% |
| Table. Cont. | Definition | No. of Items | Example Question | M a/% | SD a | Min a | Max a | TheoreticalRange b | Reliability: Cronbach’s α c | Validity: CFA d |
|---|---|---|---|---|---|---|---|---|---|---|
| Perceived severity of ASD | α Overall = 0.89 | χ2 = 51.85, df = 19, p < 0.001, RMSEA = 0.072, CFI = 0.976, SRMR = 0.032 | ||||||||
| Parent-wise perceived severity | Beliefs regarding the seriousness of ASD and its negative impacts on parents’ lives | 4 | Treatment and education regarding ASD cause extra financial burden on the caregivers [on a 4-point scale ranging from strongly disagree to strongly agree] | 3.38 | 0.54 | 1.25 | 4.00 | 1–4 | α = 0.88 | |
| ASD-child-wise perceived severity | Beliefs regarding the seriousness of ASD and its negative impacts on children with ASD | 4 | ASD affects social life of the children with ASD [on a 4-point scale ranging from strongly disagree to strongly agree] | 3.28 | 0.51 | 1.50 | 4.00 | 1–4 | α = 0.81 | |
| Perceived benefits of PGT for detecting ASD susceptibility genes within a fetus | α Overall = 0.93 | χ2 = 121.49, df = 39, p < 0.001, RMSEA = 0.080, CFI = 0.971, SRMR = 0.037 | ||||||||
| General benefits | Beliefs regarding the helpfulness of PGT for detecting ASD susceptibility genes overall | 6 | ASD genetic testing is helpful in early treatment and utilization of relevant resources [on a 4-point scale ranging from strongly disagree to strongly agree] | 3.19 | 0.46 | 2.00 | 4.00 | 1–4 | α = 0.91 | |
| Family planning-related benefits | Beliefs regarding the helpfulness of PGT for detecting ASD susceptibility genes in family planning | 5 | ASD genetic testing might be helpful in family planning for parents of children with ASD [on a 4-point scale ranging from strongly disagree to strongly agree] | 2.96 | 0.56 | 1.00 | 4.00 | 1–4 | α = 0.91 | |
| Perceived barriers to undergoing PGT for detecting ASD susceptibility genes within a fetus | α Overall = 0.83 | χ2 = 63.14, df = 24, p < 0.001, RMSEA = 0.071, CFI = 0.965, SRMR = 0.043 | ||||||||
| Testing-related barriers | Beliefs regarding testing related obstacles in undergoing PGT for detecting ASD susceptibility genes | 5 | The process of undergoing ASD genetic testing is uncomfortable [on a 4-point scale ranging from strongly disagree to strongly agree] | 2.58 | 0.47 | 1.00 | 4.00 | 1–4 | α = 0.70 | |
| Social discrimination barriers | Beliefs regarding the prejudice and discrimination related obstacles in undergoing PGT for detecting ASD susceptibility genes | 4 | The testing results might lead to discrimination against people with ASD [on a 4-point scale ranging from strongly disagree to strongly agree] | 2.66 | 0.60 | 1.00 | 4.00 | 1–4 | α = 0.88 | |
| Subjective norms related to PGT for detecting ASD susceptibility genes within a fetus | α Overall = 0.91 | χ2 = 161.07, df = 42, p < 0.001, RMSEA = 0.093, CFI = 0.960, SRMR = 0.057 | ||||||||
| Professionals | Views and influence of physicians, other non-physician health professionals (e.g., nurses, social workers, occupational/physical/speech therapists, and psychologists), and school teachers on the uptake decision-making of PGT for detecting ASD susceptibility genes | 3 | If you were pregnant, physicians would recommend PGT for detecting ASD susceptibility genes within your baby [on a 4-point scale ranging from very unlikely to very likely] | 9.23 | 2.87 | 1.67 | 16.00 | 1–16 | α = 0.82 | |
| Family members | Views and influence of spouse, spouse’s biological family, participants’ own biological family, and their children without ASD on the uptake decision-making of PGT for detecting ASD susceptibility genes | 4 | If you were pregnant, your spouse would suggest you undergo PGT for detecting ASD susceptibility genes within your baby [on a 4-point scale ranging from very unlikely to very likely] | 6.31 | 2.69 | 1.00 | 16.00 | 1–16 | α = 0.92 | |
| Other people | Views and influence of friends, neighbors, other parents of children without ASD, and general public on the uptake decision-making of PGT for detecting ASD susceptibility genes | 4 | If you were pregnant, your friends would suggest you undergo PGT for detecting ASD susceptibility genes within your baby [on a 4-point scale ranging from very unlikely to very likely] | 4.33 | 2.03 | 1.00 | 12.00 | 1–16 | α = 0.89 | |
| Attitudes toward PGT for detecting ASD susceptibility genes within the fetus | Beliefs and values about PGT for detecting ASD susceptibility genes | 4 | All pregnant women should undergo PGT for detecting ASD susceptibility genes within their babies [4-point scales ranging from strongly disagree to strongly agree and from very unimportant to very important] | 9.01 | 3.41 | 2.25 | 16.00 | 1–16 | α = 0.83 | χ2 = 4.70, df = 2, p = 0.095, RMSEA = 0.067, CFI = 0.994, SRMR = 0.015 |
| Self-efficacy in undergoing PGT for detecting ASD susceptibility genes within the fetus | Confidence in going through PGT for detecting ASD susceptibility genes | 4 | If you were pregnant, considering the factor of time, on a scale of 0 to 10, how confident are you in undergoing PGT for detecting ASD susceptibility genes within your baby? [11-point scale ranging from 0 to 10] | 6.13 | 3.11 | 0 | 10.00 | 0–10 | α = 0.93 | χ2 = 9.57, df = 3, p = 0.023, RMSEA = 0.082, CFI = 0.995, SRMR = 0.012 |
| Perceived recurrence risk of having another child with ASDe | Beliefs regarding the chance of having another child with ASD | 1 | Suppose you plan to have another child; the chance of having another child with ASD is ______ % [0-100% (0% = child will not have ASD; 100% = child will definitely have ASD)] | 36.53 | 27.68 | 0 | 100.00 | 0–100 | ||
| Intention to undergo PGT for detecting ASD susceptibility genes within the fetus e | Likelihood of undertaking PGT for detecting ASD susceptibility genes in the future | 1 | If you were pregnant, how likely would you to undergo PGT for detecting ASD susceptibility genes within your baby? [4-point scale ranging from very unlikely to very likely] | 2.89 | 0.83 | 1.00 | 4.00 | 1–4 | ||
| Intention to terminate ASD-affected pregnanciese | Likelihood in decision regarding continuation or termination of ASD-affected pregnancies in the future | 1 | If PGT results indicate that you might have a child with ASD, what would be your choice? [give birth to the child or do not keep the child (abortion)] | Give birth to the child: 46.9% Do not keep the child (abortion): 53.1% | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.-J.; Zhao, S.; Huang, T.-Y.; Kwok, O.-M.; Chen, L.-S. Autism Spectrum Disorders: Prenatal Genetic Testing and Abortion Decision-Making among Taiwanese Mothers of Affected Children. Int. J. Environ. Res. Public Health 2020, 17, 476. https://doi.org/10.3390/ijerph17020476
Chen W-J, Zhao S, Huang T-Y, Kwok O-M, Chen L-S. Autism Spectrum Disorders: Prenatal Genetic Testing and Abortion Decision-Making among Taiwanese Mothers of Affected Children. International Journal of Environmental Research and Public Health. 2020; 17(2):476. https://doi.org/10.3390/ijerph17020476
Chicago/Turabian StyleChen, Wei-Ju, Shixi Zhao, Tse-Yang Huang, Oi-Man Kwok, and Lei-Shih Chen. 2020. "Autism Spectrum Disorders: Prenatal Genetic Testing and Abortion Decision-Making among Taiwanese Mothers of Affected Children" International Journal of Environmental Research and Public Health 17, no. 2: 476. https://doi.org/10.3390/ijerph17020476
APA StyleChen, W.-J., Zhao, S., Huang, T.-Y., Kwok, O.-M., & Chen, L.-S. (2020). Autism Spectrum Disorders: Prenatal Genetic Testing and Abortion Decision-Making among Taiwanese Mothers of Affected Children. International Journal of Environmental Research and Public Health, 17(2), 476. https://doi.org/10.3390/ijerph17020476







