Impact of Matrix Metalloproteinases 11 Gene Variants on Urothelial Cell Carcinoma Development and Clinical Characteristics
Abstract
1. Introduction
2. Materials and Methods
2.1. Subject Selection
2.2. Sample Preparation and DNA Extraction
2.3. Selection of MMP11 SNPs
2.4. MMP11 SNPs Genotyping
2.5. Statistical Analysis
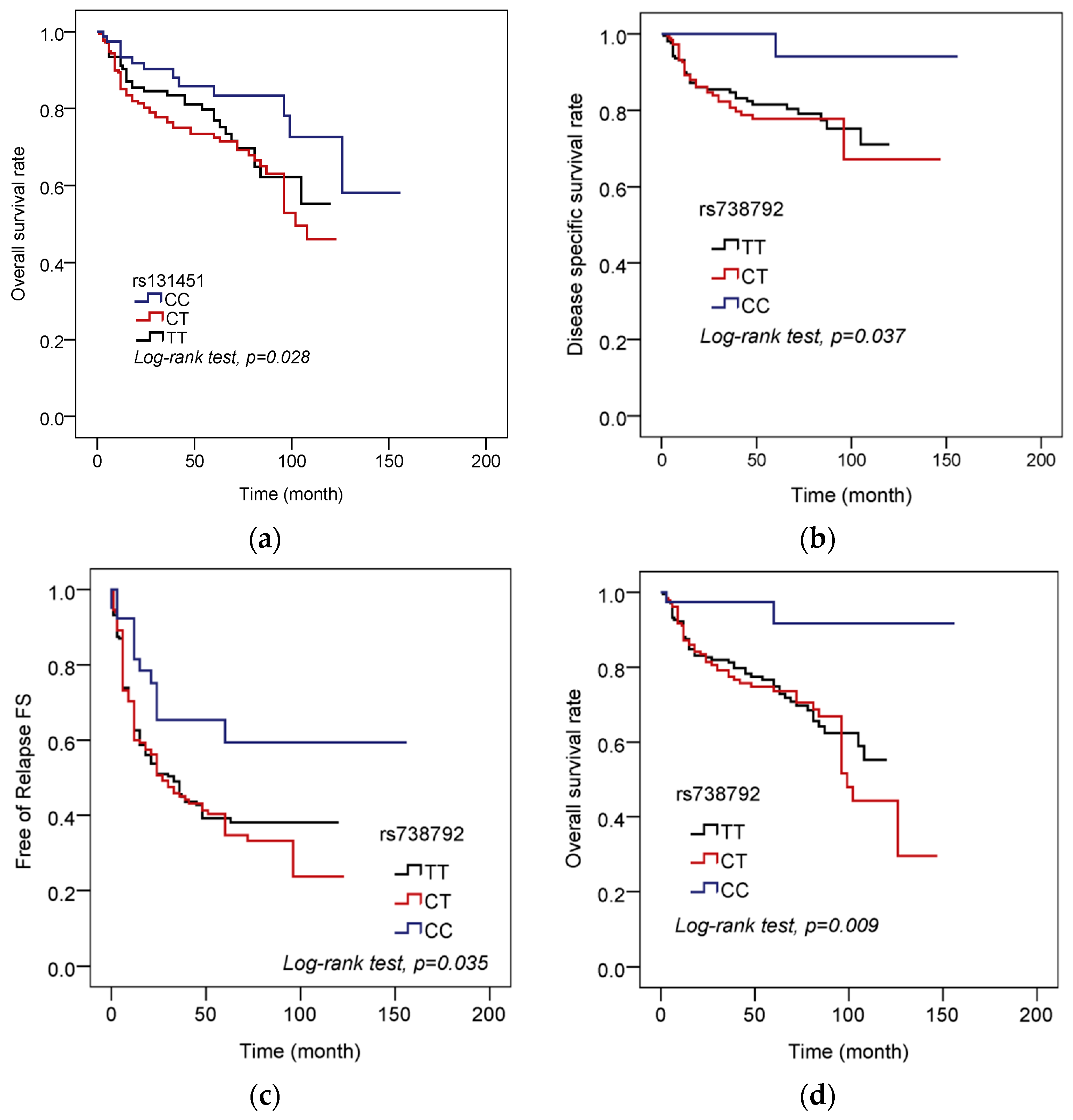
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar]
- Hung, C.F.; Yang, C.K.; Ou, Y.C. Urologic cancer in Taiwan. Jpn. J. Clin. Oncol. 2016, 46, 605–609. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence. Bladder cancer: Diagnosis and management of bladder cancer: (c) nice (2015) bladder cancer: Diagnosis and management of bladder cancer. BJU Int. 2017, 120, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Cohen, S.M.; Arnold, L.L.; Pennington, K.L.; Kato, H.; Naiki, T.; Naiki-Ito, A.; Yamashita, Y.; Takahashi, S. Cotinine, a major nicotine metabolite, induces cell proliferation on urothelium In Vitro and In Vivo. Toxicology 2020, 429, 152325. [Google Scholar] [CrossRef]
- Cumberbatch, M.G.K.; Jubber, I.; Black, P.C.; Esperto, F.; Figueroa, J.D.; Kamat, A.M.; Kiemeney, L.; Lotan, Y.; Pang, K.; Silverman, D.T.; et al. Epidemiology of bladder cancer: A systematic review and contemporary update of risk factors in 2018. Eur. Urol. 2018, 74, 784–795. [Google Scholar]
- Czerniak, B.; Dinney, C.; McConkey, D. Origins of bladder cancer. Annu. Rev. Pathol. 2016, 11, 149–174. [Google Scholar] [CrossRef]
- Miyazaki, J.; Nishiyama, H. Epidemiology of urothelial carcinoma. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2017, 24, 730–734. [Google Scholar] [CrossRef]
- Richters, A.; Aben, K.K.H.; Kiemeney, L. The global burden of urinary bladder cancer: An update. World J. Urol. 2019. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, S.; Guo, J.; Zhou, L.; You, L.; Zhang, T.; Zhao, Y. Insights into the distinct roles of MMP11 in tumor biology and future therapeutics (review). Int. J. Oncol. 2016, 48, 1783–1793. [Google Scholar] [CrossRef]
- Pei, D.; Weiss, S.J. Furin-dependent intracellular activation of the human stromelysin-3 zymogen. Nature 1995, 375, 244–247. [Google Scholar] [CrossRef]
- Li, W.M.; Wei, Y.C.; Huang, C.N.; Ke, H.L.; Li, C.C.; Yeh, H.C.; Chang, L.L.; Huang, C.H.; Li, C.F.; Wu, W.J. Matrix metalloproteinase-11 as a marker of metastasis and predictor of poor survival in urothelial carcinomas. J. Surg. Oncol. 2016, 113, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Matziari, M.; Dive, V.; Yiotakis, A. Matrix metalloproteinase 11 (MMP11; stromelysin-3) and synthetic inhibitors. Med. Res. Rev. 2007, 27, 528–552. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Shi, Y.B. Matrix metalloproteinase stromelysin-3 in development and pathogenesis. Histol. Histopathol. 2005, 20, 177–185. [Google Scholar]
- Soni, S.; Mathur, M.; Shukla, N.K.; Deo, S.V.; Ralhan, R. Stromelysin-3 expression is an early event in human oral tumorigenesis. Int. J. Cancer 2003, 107, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Arora, S.; Kaur, J.; Sharma, C.; Mathur, M.; Bahadur, S.; Shukla, N.K.; Deo, S.V.; Ralhan, R. Stromelysin 3, ets-1, and vascular endothelial growth factor expression in oral precancerous and cancerous lesions: Correlation with microvessel density, progression, and prognosis. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2005, 11, 2272–2284. [Google Scholar] [CrossRef] [PubMed]
- Denys, H.; De Wever, O.; Nusgens, B.; Kong, Y.; Sciot, R.; Le, A.T.; Van Dam, K.; Jadidizadeh, A.; Tejpar, S.; Mareel, M.; et al. Invasion and mmp expression profile in desmoid tumours. Br. J. Cancer 2004, 90, 1443–1449. [Google Scholar] [CrossRef] [PubMed]
- Kettunen, E.; Anttila, S.; Seppanen, J.K.; Karjalainen, A.; Edgren, H.; Lindstrom, I.; Salovaara, R.; Nissen, A.M.; Salo, J.; Mattson, K.; et al. Differentially expressed genes in nonsmall cell lung cancer: Expression profiling of cancer-related genes in squamous cell lung cancer. Cancer Genet. Cytogenet. 2004, 149, 98–106. [Google Scholar] [CrossRef]
- Hourihan, R.N.; O’Sullivan, G.C.; Morgan, J.G. Transcriptional gene expression profiles of oesophageal adenocarcinoma and normal oesophageal tissues. Anticancer. Res. 2003, 23, 161–165. [Google Scholar]
- von Marschall, Z.; Riecken, E.O.; Rosewicz, S. Stromelysin 3 is overexpressed in human pancreatic carcinoma and regulated by retinoic acid in pancreatic carcinoma cell lines. Gut 1998, 43, 692–698. [Google Scholar] [CrossRef]
- Perret, A.G.; Duthel, R.; Fotso, M.J.; Brunon, J.; Mosnier, J.F. Stromelysin-3 is expressed by aggressive meningiomas. Cancer 2002, 94, 765–772. [Google Scholar] [CrossRef]
- Mueller, J.; Brebeck, B.; Schmalfeldt, B.; Kuhn, W.; Graeff, H.; Hofler, H. Stromelysin-3 expression in invasive ovarian carcinomas and tumours of low malignant potential. Virchows Archiv. 2000, 437, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Wlodarczyk, J.; Stolte, M.; Mueller, J. E-cadherin, beta-catenin and stromelysin-3 expression in de novo carcinoma of the colorectum. Polish J. Pathol. Off. J. Polish Soc. Pathol. 2001, 52, 119–124. [Google Scholar]
- Kou, Y.B.; Zhang, S.Y.; Zhao, B.L.; Ding, R.; Liu, H.; Li, S. Knockdown of mmp11 inhibits proliferation and invasion of gastric cancer cells. Int. J. Immunopathol. Pharmacol. 2013, 26, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Kader, A.K.; Shao, L.; Dinney, C.P.; Schabath, M.B.; Wang, Y.; Liu, J.; Gu, J.; Grossman, H.B.; Wu, X. Matrix metalloproteinase polymorphisms and bladder cancer risk. Cancer Res. 2006, 66, 11644–11648. [Google Scholar] [CrossRef] [PubMed]
- Kader, A.K.; Liu, J.; Shao, L.; Dinney, C.P.; Lin, J.; Wang, Y.; Gu, J.; Grossman, H.B.; Wu, X. Matrix metalloproteinase polymorphisms are associated with bladder cancer invasiveness. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2007, 13, 2614–2620. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.H.; Chang, W.S.; Tsai, C.W.; Hu, P.S.; Wu, H.C.; Hsu, S.W.; Chen, G.L.; Yueh, T.C.; Shen, T.C.; Hsia, T.C.; et al. Association of matrix metalloproteinase-7 genotypes with the risk of bladder cancer. In Vivo 2018, 32, 1045–1050. [Google Scholar] [CrossRef]
- Mao, F.; Niu, X.B.; Gu, S.; Ji, L.; Wei, B.J.; Wang, H.B. The association between matrix metalloproteinase-7 genetic variant and bladder cancer risk in a Chinese han population. Clin. Exp. Med. 2019, 19, 565–570. [Google Scholar] [CrossRef]
- Meng, J.; Wang, S.; Shen, X.; Bai, Z.; Niu, Q.; Ma, D.; Xu, Y.; Liang, C. Polymorphism of mmp-9 gene is not associated with the risk of urinary cancers: Evidence from an updated meta-analysis. Pathol. Res. Pract. 2018, 214, 1966–1973. [Google Scholar] [CrossRef]
- Pence, S.; Ozbek, E.; Ozan Tiryakioglu, N.; Ersoy Tunali, N.; Pence, H.H.; Tunali, H. Rs3918242 variant genotype frequency and increased timp-2 and mmp-9 expression are positively correlated with cancer invasion in urinary bladder cancer. Cell. Mol. Biol. 2017, 63, 46–52. [Google Scholar] [CrossRef]
- Wieczorek, E.; Reszka, E.; Jablonowski, Z.; Jablonska, E.; Krol, M.B.; Grzegorczyk, A.; Gromadzinska, J.; Sosnowski, M.; Wasowicz, W. Genetic polymorphisms in matrix metalloproteinases (mmps) and tissue inhibitors of mps (timps), and bladder cancer susceptibility. BJU Int. 2013, 112, 1207–1214. [Google Scholar] [CrossRef]
- Srivastava, P.; Kapoor, R.; Mittal, R.D. Association of single nucleotide polymorphisms in promoter of matrix metalloproteinase-2, 8 genes with bladder cancer risk in northern India. Urol. Oncol. 2013, 31, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Gangwar, R.; Kapoor, R.; Mittal, R.D. Bladder cancer risk associated with genotypic polymorphism of the matrix metalloproteinase-1 and 7 in north Indian population. Dis. Markers 2010, 29, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Mandhani, A.; Kapoor, R.; Mittal, R.D. Role of mmp-3 and mmp-9 and their haplotypes in risk of bladder cancer in north indian cohort. Ann. Surg. Oncol. 2010, 17, 3068–3075. [Google Scholar] [CrossRef] [PubMed]
- Tasci, A.I.; Tugcu, V.; Ozbek, E.; Ozbay, B.; Simsek, A.; Koksal, V. A single-nucleotide polymorphism in the matrix metalloproteinase-1 promoter enhances bladder cancer susceptibility. BJU Int. 2008, 101, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Montironi, R.; Davidson, D.D.; Lopez-Beltran, A. Staging and reporting of urothelial carcinoma of the urinary bladder. Mod. Pathol. 2009, 22 (Suppl. 2), S70. [Google Scholar] [CrossRef] [PubMed]
- Osunkoya, A.O.; Grignon, D.J. Practical issues and pitfalls in staging tumors of the genitourinary tract. Semin. Diagn. Pathol. 2012, 29, 154–166. [Google Scholar] [CrossRef] [PubMed]
- International HapMap, C. The international hapmap project. Nature 2003, 426, 789–796. [Google Scholar] [CrossRef]
- Wang, B.; Hsu, C.J.; Lee, H.L.; Chou, C.H.; Su, C.M.; Yang, S.F.; Tang, C.H. Impact of matrix metalloproteinase-11 gene polymorphisms upon the development and progression of hepatocellular carcinoma. Int. J. Med. Sci. 2018, 15, 653–658. [Google Scholar] [CrossRef]
- Ng, S.C.; Wang, P.H.; Lee, Y.C.; Lee, C.Y.; Yang, S.F.; Shen, H.P.; Hsiao, Y.H. Impact of matrix metalloproteinase-11 gene polymorphisms on development and clinicopathologcial variables of uterine cervical cancer in taiwanese women. Int. J. Med. Sci. 2019, 16, 774–782. [Google Scholar] [CrossRef]
- Lin, C.W.; Yang, S.F.; Chuang, C.Y.; Lin, H.P.; Hsin, C.H. Association of matrix metalloproteinase-11 polymorphisms with susceptibility and clinicopathologic characteristics for oral squamous cell carcinoma. Head Neck 2015, 37, 1425–1431. [Google Scholar] [CrossRef]
- Chen, G.L.; Wang, S.C.; Huang, W.C.; Chang, W.S.; Tsai, C.W.; Li, H.T.; Shen, T.C.; Hsia, T.C.; Bau, D.T. The association of MMP11 promoter polymorphisms with susceptibility to lung cancer in Taiwan. Anticancer Res. 2019, 39, 5375–5380. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, X.; Liu, T.; Meng, X.; Yin, X.; Fang, C.; Huang, D.; Cao, Y.; Weng, H.; Zeng, X.; et al. Identification of biomarkers correlated with the tnm staging and overall survival of patients with bladder cancer. Front. Physiol. 2017, 8, 947. [Google Scholar] [CrossRef] [PubMed]
| Variable | Controls (N = 650) n (%) | Patients (N = 431)n (%) | p Value |
|---|---|---|---|
| Age (yrs) | <0.001 | ||
| ≤65 | 423 (65.1%) | 166 (38.5%) | |
| >65 | 227 (34.9%) | 265 (61.5%) | |
| Mean ± S.D. | 64.1 ± 3.1 | 68.6 ± 11.8 | <0.001 |
| Gender | 0.991 | ||
| Female | 240 (36.9%) | 159 (36.9%) | |
| Male | 410 (63.1%) | 272 (63.1%) | |
| Tobacco consumption | 0.442 | ||
| No | 438 (67.4%) | 300 (69.6%) | |
| Yes | 212 (32.6%) | 131 (30.4%) | |
| Stage | |||
| Non muscle invasive tumor (pTa–pT1) | 235 (54.5%) | ||
| Muscle invasive tumor (pT2–pT4) | 196 (45.5%) | ||
| Tumor T status | |||
| Ta | 90 (20.9%) | ||
| T1-T4 | 341 (79.1%) | ||
| Lymph node status | |||
| N0 | 380 (88.2%) | ||
| N1 + N2 | 51 (11.8%) | ||
| Metastasis | |||
| M0 | 417 (96.8%) | ||
| M1 | 14 (3.2%) | ||
| Histopathologic grading | |||
| Low grade | 53 (12.3%) | ||
| High grade | 378 (87.7%) |
| Variable | Controls (N = 650) n (%) | Patients (N = 431) n (%) | OR (95% CI) | AOR (95% CI) |
|---|---|---|---|---|
| rs738791 | ||||
| CC | 309 (47.5%) | 181 (42%) | 1.000 (reference) | 1.000 (reference) |
| CT | 270 (41.5%) | 203 (47.1%) | 1.284 (0.991–1.662) | 1.230 (0.939–1.610) |
| TT | 71 (10.9%) | 47 (10.9%) | 1.130 (0.749–1.706) | 1.192 (0.775–1.832) |
| CT + TT | 341 (52.5%) | 250 (58%) | 1.252 (0.979–1.600) | 1.222 (0.946–1.579) |
| rs2267029 | ||||
| GG | 365 (56.2%) | 230 (53.4%) | 1.000 (reference) | 1.000 (reference) |
| AG | 239 (36.8%) | 172 (39.9%) | 1.142 (0.884–1.476) | 1.126 (0.862–1.47) |
| AA | 46 (7.1%) | 29 (6.7%) | 1.000 (0.611–1.638) | 1.009 (0.605–1.684) |
| AG + AA | 285 (43.8%) | 201 (46.6%) | 1.119 (0.876–1.429) | 1.107 (0.858–1.428) |
| rs738792 | ||||
| TT | 341 (52.5%) | 207 (48%) | 1.000 (reference) | 1.000 (reference) |
| CT | 253 (38.9%) | 185 (42.9%) | 1.205 (0.932–1.557) | 1.169 (0.895–1.527) |
| CC | 56 (8.6%) | 39 (9%) | 1.147 (0.736–1.788) | 1.117 (0.704–1.773) |
| CT + CC | 309 (47.5%) | 224 (52%) | 1.194 (0.936–1.524) | 1.160 (0.900–1.495) |
| rs28382575 | ||||
| TT | 631 (97.1%) | 405 (94%) | 1.000 (reference) | 1.000 (reference) |
| CT | 19 (2.9%) | 26 (6%) | 2.132 (1.165–3.903) | 2.045 (1.088–3.843) |
| CC | 0 (0%) | 0 (0%) | NA | NA |
| CT + CC | 19 (2.9%) | 26 (6%) | 2.132 (1.165–3.903) | 2.045 (1.088–3.843) |
| rs131451 | ||||
| TT | 233 (35.8%) | 139 (32.3%) | 1.000 (reference) | 1.000 (reference) |
| CT | 301 (46.3%) | 215 (49.9%) | 1.197 (0.911–1.574) | 1.115 (0.839–1.482) |
| CC | 116 (17.8%) | 77 (17.9%) | 1.113 (0.779–1.589) | 1.136 (0.784–1.645) |
| CT + CC | 417 (64.2%) | 292 (67.7%) | 1.174 (0.907–1.519) | 1.121 (0.857–1.465) |
| MMP11 (rs131451) | ||||
|---|---|---|---|---|
| Variable | CT + TT (%) (n = 354) | CC (%) (n = 77) | OR (95% CI) | p Value |
| Stage | ||||
| Non muscle invasive tumor (pTa–pT1) | 189 (53.4%) | 46 (59.7%) | 1.000 (reference) | |
| Muscle invasive tumor (pT2–pT4) | 165 (46.6%) | 31 (40.3%) | 0.772 (0.468–1.274) | 0.311 |
| Tumor T status | ||||
| Ta | 69 (19.5%) | 21 (27.3%) | 1.000 (reference) | |
| T1-T4 | 285 (80.5%) | 56 (72.7%) | 0.646 (0.366–1.137) | 0.130 |
| Lymph node status | ||||
| N0 | 314 (88.7%) | 66 (85.7%) | 1.000 (reference) | |
| N1 + N2 | 40 (11.3%) | 11 (14.3%) | 1.308 (0.638–2.683) | 0.463 |
| Metastasis | ||||
| M0 | 344 (97.2%) | 73 (94.8%) | 1.000 (reference) | |
| M1 | 10 (2.8%) | 4 (5.2%) | 1.885 (0.575–6.176) | 0.295 |
| Histopathologic grading | ||||
| Low grade | 42 (11.9%) | 11 (14.3%) | 1.000 (reference) | |
| High grade | 312 (88.1%) | 66 (85.7%) | 0.808 (0.395–1.651) | 0.558 |
| MMP11 (rs131451) | ||||
|---|---|---|---|---|
| Variable | CT + TT (%) (n = 134) | CC (%) (n = 32) | OR (95% CI) | p Value |
| Stage | ||||
| Non muscle invasive tumor (pTa–pT1) | 69 (51.5%) | 19 (59.4%) | 1.000 (reference) | |
| Muscle invasive tumor (pT2–pT4) | 65 (48.5%) | 13 (40.6%) | 0.726 (0.332–1.589) | 0.423 |
| Tumor T status | ||||
| Ta | 22 (16.4%) | 11 (34.4%) | 1.000 (reference) | |
| T1-T4 | 112 (83.6%) | 21 (65.6%) | 0.375 (0.159–0.887) | 0.026 |
| Lymph node status | ||||
| N0 | 112 (83.6%) | 27 (84.4%) | 1.000 (reference) | |
| N1 + N2 | 22 (16.4%) | 5 (15.6%) | 0.943 (0.327–2.716) | 0.913 |
| Metastasis | ||||
| M0 | 130 (97%) | 29 (90.6%) | 1.000 (reference) | |
| M1 | 4 (3%) | 3 (9.4%) | 3.362 (0.714–15.842) | 0.125 |
| Histopathologic grading | ||||
| Low grade | 17 (12.7%) | 6 (18.8%) | 1.000 (reference) | |
| High grade | 117 (87.3%) | 26 (81.3%) | 0.630 (0.226–1.752) | 0.375 |
| Variable | N | Number of Death | HR (95% CI) | AHR (95% CI) |
|---|---|---|---|---|
| Stage | ||||
| Non muscle invasive tumor (pTa–pT1) | 235 | 39 | 1.000 (reference) | 1.000 (reference) |
| Muscle invasive tumor (pT2–pT4) | 195 | 72 | 3.297 (2.222–4.893) | 3.375 (2.272–5.015) |
| Tumor T status | ||||
| Ta | 90 | 14 | 1.000 (reference) | 1.000 (reference) |
| T1-T4 | 340 | 97 | 2.681 (1.524–4.716) | 2.661 (1.51–4.69) |
| Lymph node status | ||||
| N0 | 379 | 85 | 1.000 (reference) | 1.000 (reference) |
| N1 + N2 | 51 | 26 | 4.408 (2.803–6.934) | 4.898 (3.105–7.726) |
| Metastasis | ||||
| M0 | 416 | 103 | 1.000 (reference) | 1.000 (reference) |
| M1 | 14 | 8 | 6.561 (3.138–13.717) | 8.566 (3.985–18.41) |
| Histopathologic grading | ||||
| Low grade | 53 | 6 | 1.000 (reference) | 1.000 (reference) |
| High grade | 377 | 105 | 3.569 (1.56–8.164) | 3.431 (1.497–7.863) |
| rs131451 | ||||
| CC | 77 | 13 | 1.000 (reference) | 1.000 (reference) |
| CT + TT | 353 | 98 | 2.089 (1.145–3.81) | 2.05 (1.12–3.754) |
| rs738792 | ||||
| CC | 39 | 2 | 1.000 (reference) | 1.000 (reference) |
| CT + TT | 391 | 109 | 6.468 (1.593–26.258) | 6.562 (1.612–26.704) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.-C.; Hsieh, M.-J.; Wang, S.-S.; Hung, S.-C.; Lin, C.-Y.; Kuo, C.-W.; Yang, S.-F.; Chou, Y.-E. Impact of Matrix Metalloproteinases 11 Gene Variants on Urothelial Cell Carcinoma Development and Clinical Characteristics. Int. J. Environ. Res. Public Health 2020, 17, 475. https://doi.org/10.3390/ijerph17020475
Li C-C, Hsieh M-J, Wang S-S, Hung S-C, Lin C-Y, Kuo C-W, Yang S-F, Chou Y-E. Impact of Matrix Metalloproteinases 11 Gene Variants on Urothelial Cell Carcinoma Development and Clinical Characteristics. International Journal of Environmental Research and Public Health. 2020; 17(2):475. https://doi.org/10.3390/ijerph17020475
Chicago/Turabian StyleLi, Chien-Chang, Ming-Ju Hsieh, Shian-Shiang Wang, Sheng-Chun Hung, Chia-Yen Lin, Chi-Wen Kuo, Shun-Fa Yang, and Ying-Erh Chou. 2020. "Impact of Matrix Metalloproteinases 11 Gene Variants on Urothelial Cell Carcinoma Development and Clinical Characteristics" International Journal of Environmental Research and Public Health 17, no. 2: 475. https://doi.org/10.3390/ijerph17020475
APA StyleLi, C.-C., Hsieh, M.-J., Wang, S.-S., Hung, S.-C., Lin, C.-Y., Kuo, C.-W., Yang, S.-F., & Chou, Y.-E. (2020). Impact of Matrix Metalloproteinases 11 Gene Variants on Urothelial Cell Carcinoma Development and Clinical Characteristics. International Journal of Environmental Research and Public Health, 17(2), 475. https://doi.org/10.3390/ijerph17020475







