Influence of Heterogeneous Karst Microhabitats on the Root Foraging Ability of Chinese Windmill Palm (Trachycarpus fortunei) Seedlings
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Region
2.2. Plant Materials
2.3. Sampling Design
2.4. Measurement of Root Morphological Indicators
2.5. Measurement of Root and Leaf Biomass and Nutritional Components
2.6. Determination of Soil Physical and Chemical Characteristics
2.7. Data Analysis
3. Results and Analysis
3.1. Soil Characteristics
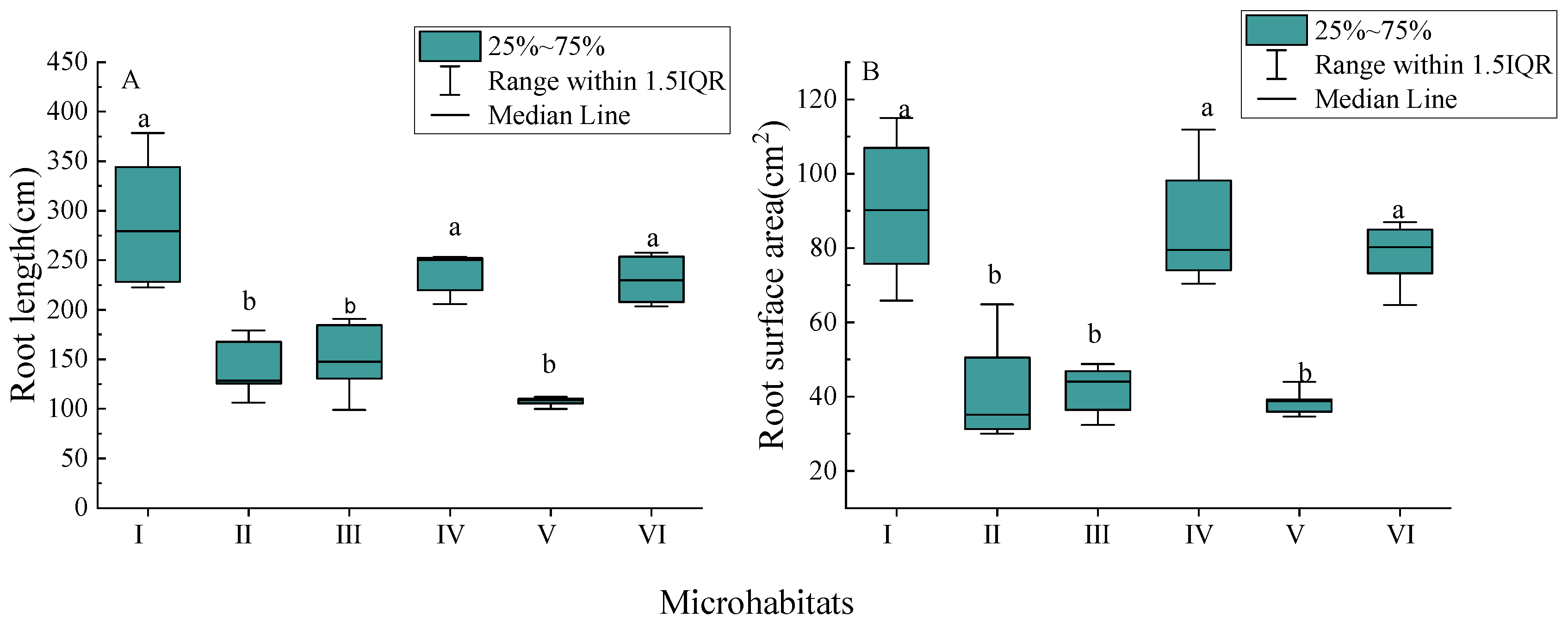
3.2. Morphological Characteristics of New Roots
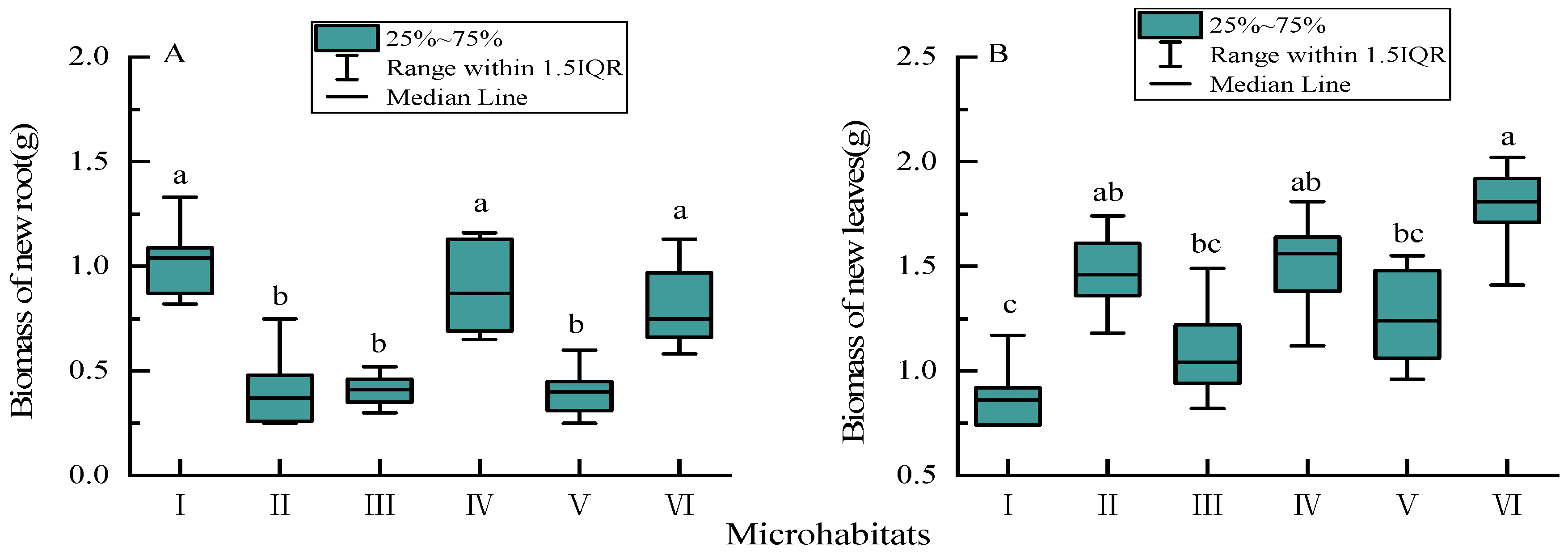
3.3. Biomass of New Annual Roots and Leaves
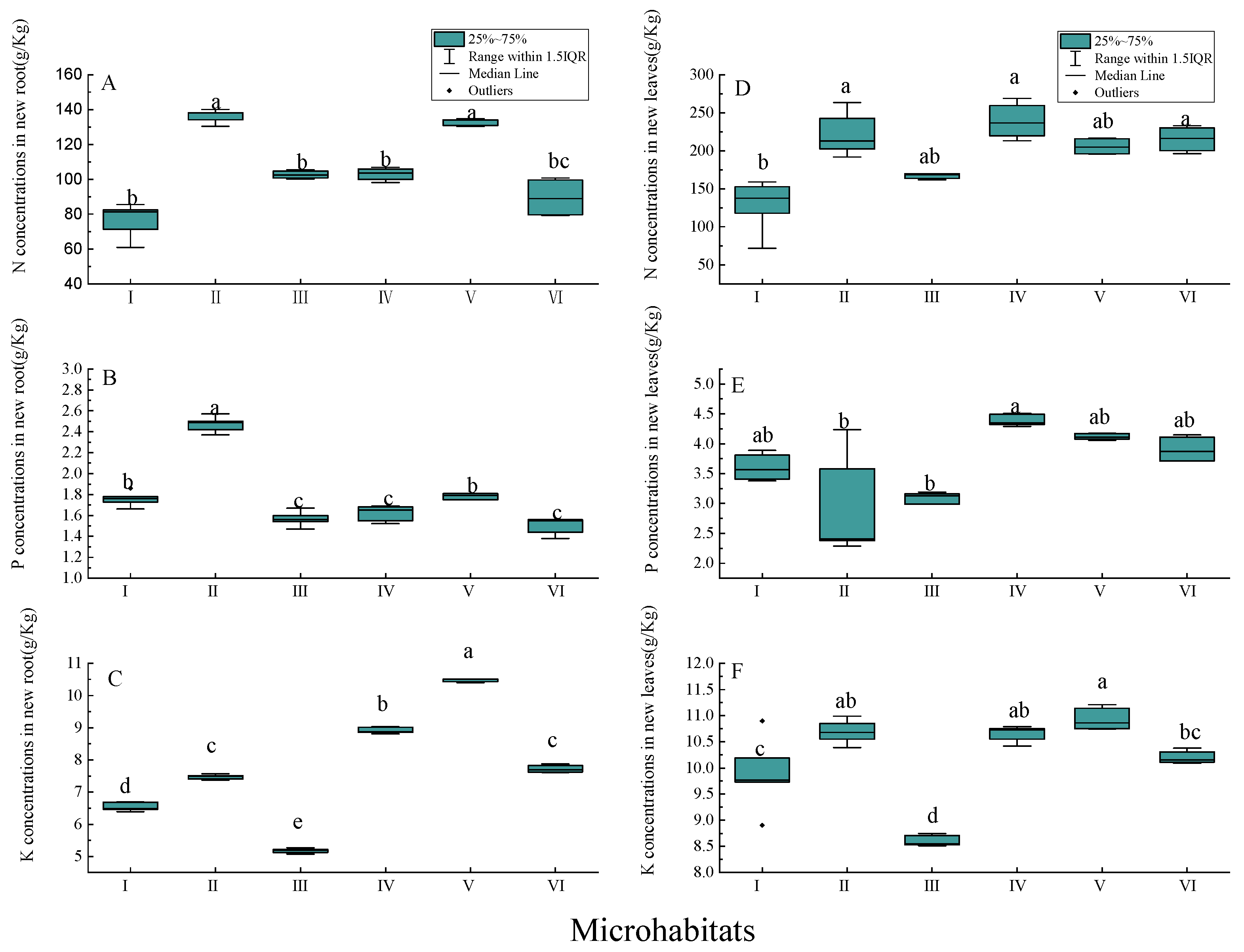
3.4. Nutrient Concentrations of New Annual Roots and Leaves
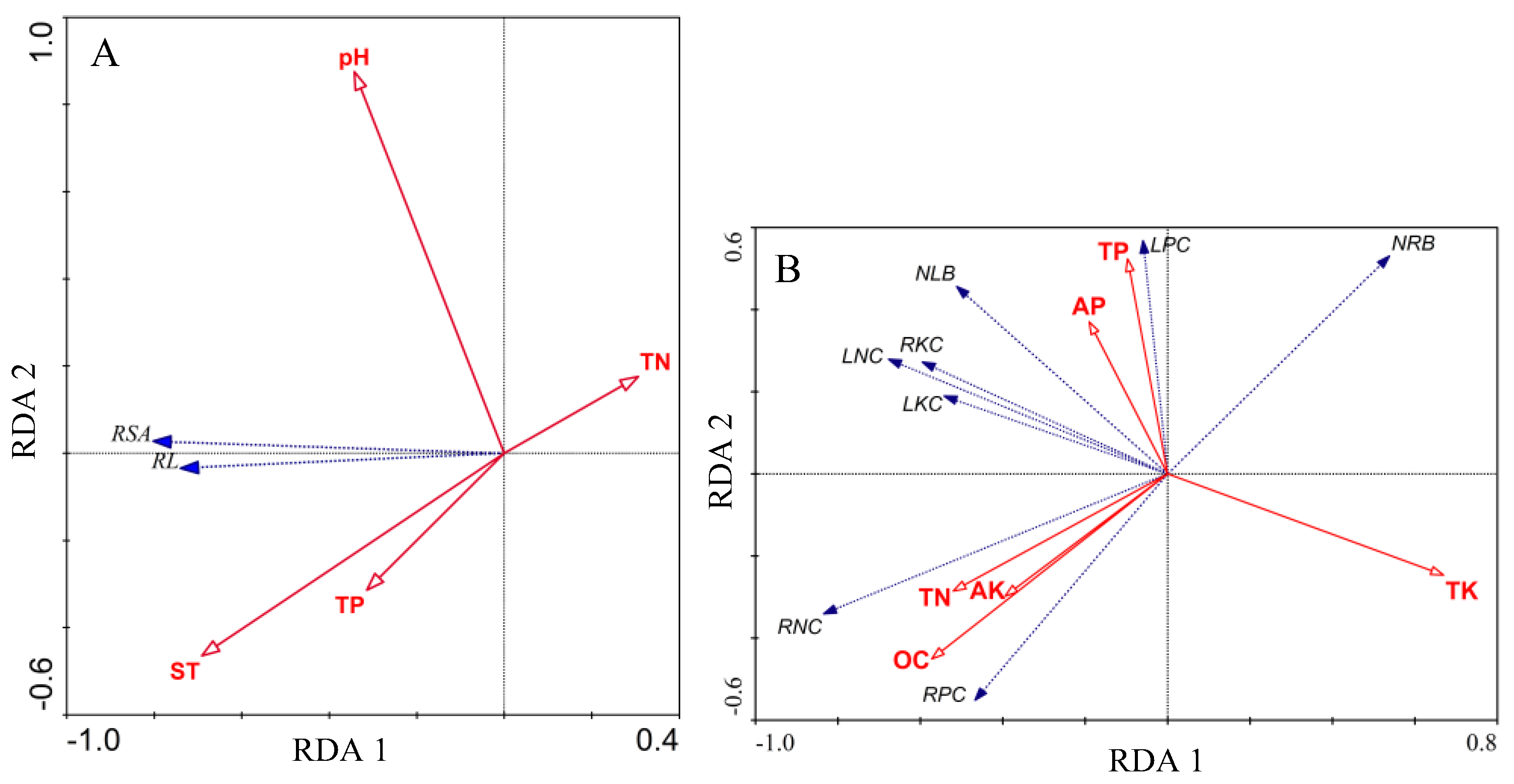
3.5. Redundancy Analysis
4. Discussion
4.1. Root Morphological Characteristics of Palm Seedlings in Different Karst Microhabitats
4.2. Biomass and Nutrient Concentrations of New Annual Roots and Leaves in Different Karst Microhabitats
4.3. Relationship between Root Morphology of Palm Seedlings and Environmental Factors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wang, D.J.; Shen, Y.X.; Li, Y.H.; Huang, J. Rock outcrops redistribute organic carbon and nutrients to nearby soil patches in three karst ecosystems in SW China. PLoS ONE 2016, 11, e0160773. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.A.; Shen, Y.X.; He, B.B.; Zhao, Z.M. Humus soil as a critical driver of flora conversion on karst rock outcrops. Sci. Rep. 2017, 7, 12611. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.; Yu, F. Characteristics of Microclimate Change in Different habitats of karst moderate rocky desertification area. Guizhou Agric. Sci. 2013, 41, 103–108. [Google Scholar]
- Bogunovic, I.; Pereira, P.; Coric, R.; Husnjak, S.; Brevik, E.C. Spatial distribution of soil organic carbon and total nitrogen stocks in a karst polje located in Bosnia and Herzegovina. Environ. Earth Sci. 2018, 77, 612. [Google Scholar] [CrossRef]
- Goransson, H.; Edwards, P.J.; Perreijn, K.; Smittenberg, R.H.; Venterink, H.O. Rocks create nitrogen hotspots and N: P heterogeneity by funnelling rain. Biogeochemistry 2014, 121, 329–338. [Google Scholar] [CrossRef]
- Liu, C.C.; Liu, Y.G.; Guo, K.; Fan, D.Y.; Yu, L.F.; Yang, R. Exploitation of patchy soil water resources by the clonal vine Ficus tikoua in karst habitats of southwestern China. Acta Physiol. Plant. 2011, 33, 93–102. [Google Scholar] [CrossRef]
- Wang, D.J.; Shen, Y.X.; Huang, J.; Li, Y.H. Rock outcrops redistribute water to nearby soil patches in karst landscapes. Environ. Sci. Pollut. Res. 2016, 23, 8610–8616. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, B.; Mallik, A.U.; Huang, F.; Xiang, W.; Ding, T.; Wen, S.; Lu, S.; Li, D.; He, Y.; et al. Topographic species-habitat associations of tree species in a heterogeneous tropical karst seasonal rain forest, China. J. Plant Ecol. 2017, 10, 450–460. [Google Scholar] [CrossRef]
- Zhang, Z.; Hu, G.; Zhu, J.; Ni, J. Aggregated spatial distributions of species in a subtropical karst forest, southwestern China. J. Plant Ecol. 2013, 6, 131–140. [Google Scholar] [CrossRef]
- Estrada-Medina, H.; Graham, R.C.; Allen, M.F.; Jiménez-Osornio, J.J.; Robles-Casolco, S. The importance of limestone bedrock and dissolution karst features on tree root distribution in northern Yucatán, México. Plant Soil 2013, 362, 37–50. [Google Scholar] [CrossRef]
- Estrada-Medina, H.; Santiago, L.S.; Graham, R.C.; Allen, M.F.; Jiménez-Osornio, J.J. Source water, phenology and growth of two tropical dry forest tree species growing on shallow karst soils. Trees 2013, 27, 1297–1307. [Google Scholar] [CrossRef]
- Elkington, R.J.; Rebel, K.T.; Heilman, J.L.; Litvak, M.E.; Dekker, S.C.; Moore, G.W. Species-specific water use by woody plants on the Edwards Plateau, Texas. Ecohydrology 2014, 7, 278–290. [Google Scholar] [CrossRef]
- Nie, Y.P.; Chen, H.S.; Wang, K.L.; Ding, Y.L. Rooting characteristics of two widely distributed woody plant species growing in different karst habitats of southwest China. Plant Ecol. 2014, 215, 1099–1109. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Z.; Zhang, J.; Song, H.; Liang, Q.; Tao, J.; Liu, J. Do shallow soil, low water availability, or their combination increase the competition between grasses with different root systems in karst soil? Environ. Sci. Pollut. Res. 2017, 24, 10640–10651. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Liang, Y.; Wang, K.; Zhang, W. Responses of fine root functional traits to soil nutrient limitations in a karst ecosystem of Southwest China. Forests 2018, 9, 743. [Google Scholar] [CrossRef]
- Li, H.B.; Wang, X.; Rengel, Z.; Ma, Q.H.; Zhang, F.S.; Shen, J.B. Root over-production in heterogeneous nutrient environment has no negative effects on Zea mays shoot growth in the field. Plant Soil 2016, 409, 405–417. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, X.H.; Zhou, Z.C. The influence of light conditions and interspecific competition on the root foraging traits and seedling growth of two tree species. Plant Biol. 2012, 146, 7–14. [Google Scholar] [CrossRef]
- Gloser, V.; Libera, K.; Orians, C.M. Contrasting below- and aboveground responses of two deciduous trees to patchy nitrate availability. Tree Physiol. 2008, 28, 37–44. [Google Scholar] [CrossRef][Green Version]
- Giehl, R.F.; von Wirén, N. Root Nutrient Foraging. Plant Physiol. 2014, 166, 509–517. [Google Scholar] [CrossRef]
- Li, X.X.; Zeng, R.; Liao, H. Improving crop nutrient efficiency through root architecture modifications. J. Integr. Plant Biol. 2016, 58, 193–202. [Google Scholar] [CrossRef]
- Chen, W.L.; Koide, R.T.; Adams, T.S.; Defotest, J.L.; Cheng, L.; Eissenstat, D.M. Root morphology and mycorrhizal symbioses together shape nutrient foraging strategies of temperate trees. Proc. Natl. Acad. Sci. USA 2016, 113, 8741–8746. [Google Scholar] [CrossRef]
- Ristova, D.; Busch, W. Natural variation of root traits: From development to nutrient uptake. Plant Physiol. 2014, 166, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Nan, H.W.; Liang, J.; Cheng, X.Y.; Zhao, C.Z.; Yin, H.J.; Yin, C.Y.; Liu, Q. Responses of nutrient capture and fine root morphology of subalpine coniferous tree Picea asperata to nutrient heterogeneity and competition. PLoS ONE 2017, 12, e0187496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.Q. Chinese Windmill Palm; Anhui Publishing House of Science and Technology: Hefei, China, 1982; pp. 5–11. [Google Scholar]
- Zhu, S.Q. Ecological Research of Karst Forest; Guizhou Publishing House of Science and Technology: Guiyang, China, 1993; p. 52. [Google Scholar]
- Brissette, J.C.; Chambers, J.L. Leaf water status and root system water flux of shortleaf pine (Pinus echinata Mill.) seedlings in relation to new root growth after transplanting. Tree Physiol. 1992, 11, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Novozamsky, I.; Eck, R.V.; Schouwenburg, J.C.; Walinga, I. Total nitrogen determination in plant material by means of the indophenol-blue method. Neth. J. Agric. Sci. 1974, 22, 3. [Google Scholar]
- Delong, W.A.; Mackay, D.C.; Steppler, H.A. Coordinated soil-plant analysis I. Nutrient Cations. Soil Sci. Soc. Am. J. 1953, 17, 262–266. [Google Scholar] [CrossRef]
- Tang, L.; Wan, K.; Cheng, C.; Li, R.; Wang, D.; Pan, J.; Tao, Y.; Xie, J.; Chen, F. Effect of fertilization patterns on the assemblage of weed communities in an upland winter wheat field. J. Plant Ecol. 2014, 7, 39–50. [Google Scholar] [CrossRef]
- Bogunovic, I.; Pereira, P.; KisicI, I.; Sajko, K.; Sraka, M. Tillage management impacts on soil compaction, erosion and crop yield in Stagnosols (Croatia). Catena 2018, 160, 376–384. [Google Scholar] [CrossRef]
- Wei, H.; Lei, L.; Zhang, S. Adsorption of tetrabromobisphenol A on soils: Contribution of soil components and influence of soil properties. Colloid Surf. A 2013, 428, 60–64. [Google Scholar]
- Jing, Z.; Cheng, J.; Su, J.; Bai, Y.; Jin, J. Changes in plant community composition and soil properties under 3-decade grazing exclusion in semiarid grassland. Ecol. Eng. 2014, 64, 171–178. [Google Scholar] [CrossRef]
- Prakash, K.; Swaroop, H.M. A simplified approach of determining the specific gravity of soil solids. Geotech. Geol. Eng. 2012, 30, 1063–1067. [Google Scholar] [CrossRef]
- Xu, M.X.; Zhao, Y.G.; Liu, G.B.; Argent, R.M. Soil quality indices and their application in the hilly loess plateau region of China. Aust. J. Soil Res. 2006, 44, 245–254. [Google Scholar] [CrossRef]
- Karthikeyan, A.S.; Jain, A.; Nagarajan, V.K.; Sinilal, B.; Sahi, S.V.; Raghothama, K.G. Arabidopsis thaliana mutant lpsi reveals impairment in the root responses to local phosphate availability. Plant Physiol. Biochem. 2014, 77, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.E.; Hocking, P.J.; Simpson, R.J.; George, T.S. Plant mechanisms to optimise access to soil phosphorus. Crop Pasture Sci. 2009, 60, 124–143. [Google Scholar] [CrossRef]
- Liao, H.K.; Long, J.; Li, J.; Yang, J.J.; Feng, Y.Q. Distribution characteristics of soil carbon and nitrogen under different vegetation types in Micro-habitats of karst dry-hot valley region of South Western China. Soils 2012, 44, 421–428. [Google Scholar]
- Du, X.L.; Wang, S.J. Micro-habitat characteristics in the karst desertification area: A case study of the wangjiazhai catchment in Guizhou province. Earth Environ. 2010, 38, 255–261. [Google Scholar]
- Da Silva, A.; Bruno, I.P.; Franzini, V.I.; Marcante, N.C.; Benitiz, L.; Muraoka, T. Phosphorus uptake efficiency, root morphology and architecture in Brazilian wheat cultivars. J. Radioanal. Nucl. Chem. 2016, 307, 1055–1063. [Google Scholar] [CrossRef]
- Matthes-Sears, U.; Larson, D.W. Rooting characteristics of trees in rock: A study of Thuja occidentalis on cliff faces. Int. J. Plant Sci. 1995, 156, 679–686. [Google Scholar] [CrossRef]
- Martínková, J.; Klimeš, A.; Klimešová, J. No evidence for nutrient foraging in root-sprouting clonal plants. Basic Appl. Ecol. 2018, 28, 27–36. [Google Scholar] [CrossRef]
- Singh, P.; Singh, B. Biomass and nitrogen dynamics of fine roots of poplar under differential N and P levels in an agroforestry system in Punjab. Trop. Ecol. 2016, 57, 143–152. [Google Scholar]
- Lin, Y.; Comita, L.S.; Johnson, D.J.; Chen, M.; WU, S. Biotic vs. abiotic drivers of seedling persistence in a tropical karst forest. J. Veg. Sci. 2017, 28, 206–217. [Google Scholar] [CrossRef]
- Wang, P.; Yang, Y.; Mou, P.; Zhao, Q.Z.; Li, Y.B. Local root growth and death are mediated by contrasts in nutrient availability and root quantity between soil patches. Proc. R. Soc. B 2018, 285, 20180699. [Google Scholar] [CrossRef] [PubMed]
- Kramer-Walter, K.R.; Laughlin, D.C. Root nutrient concentration and biomass allocation are more plastic than morphological traits in response to nutrient limitation. Plant Soil 2017, 416, 539–550. [Google Scholar] [CrossRef]
- Long, P.; Wei, X.L.; Peng, L.S. Effects of plantations of different site conditions on the growth and yield of palm. J. Fujian Agric. Univ. 2019, 48, 182–187. [Google Scholar]
- Zhao, W.; Dong, H.; Zahoor, R.; Zhou, Z.; Snider, J.L.; Chen, Y.; Siddique, K.H.M.; Wang, Y. Ameliorative effects of potassium on drought-induced decreases in fiber length of cotton (Gossypium hirsutum L.) are associated with osmolyte dynamics during fiber development. Crop J. 2019, 7, 619–634. [Google Scholar] [CrossRef]

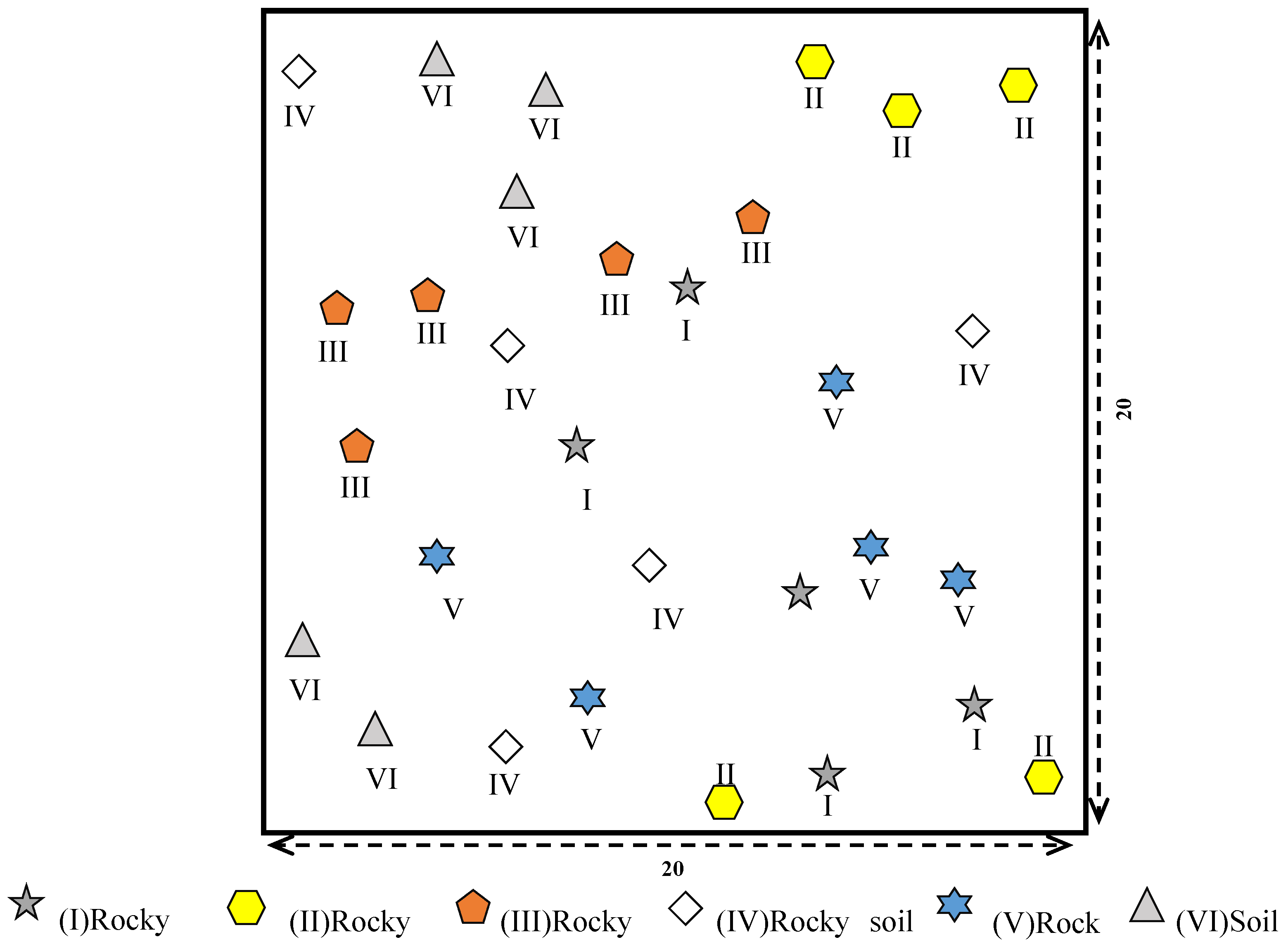
| Microhabitat | Rocky Trough (I) | Rocky Surface (II) | Rocky Gully (III) | Rocky Soil Surface (IV) | Rocky Pit (V) | Soil Surface (VI) |
|---|---|---|---|---|---|---|
| Soil thickness (cm) | 54.33 ± 6.03 a | 19.67 ± 2.08 c | 41.00 ± 2.65 b | 39.67 ± 3.06 b | 38.00 ± 2.00 b | 44.33 ± 4.51 ab |
| Volumetric weight (g/cm3) | 1.56 ± 0.13 a | 1.36 ± 0.43 b | 1.63 ± 0.04 a | 1.54 ± 0.09 a | 1.44 ± 0.09 b | 1.58 ± 0.11 a |
| Specific gravity (g/cm3) | 2.53 ± 0.05 a | 2.35 ± 0.02 b | 2.51 ± 0.02 a | 2.45 ± 0.04 a | 2.51 ± 0.06 a | 2.55 ± 0.02 a |
| Porosity (%) | 37.19 ± 1.15 bcd | 45.47 ± 8.21 a | 34.92 ± 1.37 d | 36.77 ± 0.97 cd | 42.16 ± 1.30 ab | 38.47 ± 1.72 bc |
| Water content (%) | 39.11 ± 1.99 bc | 37.62 ± 9.06 bcd | 41.33 ± 2.19 a | 36.51 ± 1.04 cd | 32.08 ± 6.52 d | 40.82 ± 1.76 ab |
| Potential of hydrogen | 7.11 ± 0.08 abc | 7.15 ± 0.11 ab | 7.00 ± 0.06 bc | 7.28 ± 0.16 a | 6.96 ± 0.06 c | 6.94 ± 0.06 c |
| Organic carbon (g/kg) | 7.19 ± 0.34 c | 13.86 ± 0.15 a | 5.58 ± 0.55 e | 6.91 ± 0.19 d | 8.58 ± 1.14 b | 6.43 ± 0.74 d |
| Total nitrogen (N%) | 0.2 ± 0 b | 0.34 ± 0.04 a | 0.14 ± 0.02 c | 0.18 ± 0.04 b | 0.2 ± 0.02 b | 0.18 ± 0.03 b |
| Hydrolyzed nitrogen (mg/kg) | 112.74 ± 2.15 d | 185.5 ± 73.99 a | 87.41 ± 7.28 e | 125.91 ± 6.25 c | 131.99 ± 10.34 b | 116.18 ± 15.87 d |
| Total phosphorus (g/kg) | 0.13 ± 0.02 c | 0.22 ± 0.03 b | 0.11 ± 0.01 d | 0.13 ± 0 | 0.11 ± 0.01 d | 0.46 ± 0.05 a |
| Available phosphorus (mg/kg) | 0.62 ± 0.14 d | 2.5 ± 0.1 b | 0.4 ± 0.12 e | 0.21 ± 0.07 f | 0.82 ± 0.16 c | 5.24 ± 0.07 a |
| Total potassium (g/kg) | 8.99 ± 0.91 a | 8.17 ± 0.04 ab | 7.96 ± 0.64 b | 6.85 ± 0.12 c | 6.45 ± 0.11 d | 8.66 ± 2.21 ab |
| Available potassium (mg/kg) | 103.47 ± 1.01 d | 138.86 ± 1.16 b | 89.94 ± 2.51 e | 52.45 ± 0.63 f | 160.44 ± 1.62 a | 135.49 ± 2.87 c |
| Item | Axis 1 | Axis 2 | Axis 3 | Axis 4 | Total Variance |
|---|---|---|---|---|---|
| Eigenvalues | 0.599 | 0.001 | 0.370 | 0.030 | 1 |
| Species–environment correlations | 0.788 | 0.169 | 0 | 0 | |
| CV of species data (%) | 59.9 | 60.0 | 97.0 | 100 | |
| CV of species–environment relationship (%) | 99.8 | 100 | 0 | 0 | |
| Sum of all eigenvalues | 1 | ||||
| Variance explained by all variables | 0.817 | ||||
| Variance explained by selected variables | 0.6534 | ||||
| Forward selection of variables | RDA 1 | RDA 2 | F value | p value | Extra fit |
| Soil thickness (cm) | −0.5449 | −0.0787 | 11.27 | 0.0014 ** | 0.287 |
| Potential of hydrogen | −0.2701 | 0.1480 | 7.41 | 0.0094 ** | 0.1535 |
| Total phosphorus (g/kg) | −0.2478 | −0.0532 | 9.98 | 0.0025 ** | 0.1552 |
| Total nitrogen (N%) | 0.2411 | 0.0297 | 4.64 | 0.0352 * | 0.0577 |
| Item | Axis 1 | Axis 2 | Axis 3 | Axis 4 | Total Variance |
|---|---|---|---|---|---|
| Eigenvalues | 0.393 | 0.155 | 0.077 | 0.042 | 1 |
| Species–environment correlations | 0.906 | 0.808 | 0.762 | 0.823 | |
| CV of species data (%) | 39.3 | 54.7 | 62.5 | 66.7 | |
| CV of species–environment relation (%) | 58.1 | 81 | 92.4 | 98.7 | |
| Sum of all eigenvalues | 1 | ||||
| Variance explained by all variables | 0.69 | ||||
| Variance explained by selected variables | 0.676 | ||||
| Forward selection of variables | RDA 1 | RDA 2 | F value | p value | Extra fit |
| Organic carbon (g/kg) | −0.5188 | −0.3644 | 8.91 | 0.0001 ** | 0.1963 |
| Total nitrogen (N%) | −0.4716 | −0.2308 | 3.97 | 0.0042 ** | 0.0653 |
| Total phosphorus (g/kg) | −0.0882 | 0.4232 | 6.44 | 0.0001 ** | 0.1181 |
| Available phosphorus (mg/kg) | −0.1725 | 0.2992 | 3.21 | 0.0156 * | 0.0453 |
| Total potassium (g/kg) | 0.6069 | −0.1993 | 7.4 | 0.0001 ** | 0.2089 |
| Available potassium (mg/kg) | −0.3577 | −0.241 | 2.71 | 0.0318 * | 0.0417 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Wei, X.; Zhou, Z.; Shao, C.; Su, S. Influence of Heterogeneous Karst Microhabitats on the Root Foraging Ability of Chinese Windmill Palm (Trachycarpus fortunei) Seedlings. Int. J. Environ. Res. Public Health 2020, 17, 434. https://doi.org/10.3390/ijerph17020434
Liu Y, Wei X, Zhou Z, Shao C, Su S. Influence of Heterogeneous Karst Microhabitats on the Root Foraging Ability of Chinese Windmill Palm (Trachycarpus fortunei) Seedlings. International Journal of Environmental Research and Public Health. 2020; 17(2):434. https://doi.org/10.3390/ijerph17020434
Chicago/Turabian StyleLiu, Yingying, Xiaoli Wei, Zijing Zhou, Changchang Shao, and Shicheng Su. 2020. "Influence of Heterogeneous Karst Microhabitats on the Root Foraging Ability of Chinese Windmill Palm (Trachycarpus fortunei) Seedlings" International Journal of Environmental Research and Public Health 17, no. 2: 434. https://doi.org/10.3390/ijerph17020434
APA StyleLiu, Y., Wei, X., Zhou, Z., Shao, C., & Su, S. (2020). Influence of Heterogeneous Karst Microhabitats on the Root Foraging Ability of Chinese Windmill Palm (Trachycarpus fortunei) Seedlings. International Journal of Environmental Research and Public Health, 17(2), 434. https://doi.org/10.3390/ijerph17020434





