Opioid Pain Medication Prescription for Chronic Pain in Primary Care Centers: The Roles of Pain Acceptance, Pain Intensity, Depressive Symptoms, Pain Catastrophizing, Sex, and Age
Abstract
1. Introduction
2. Materials and Methods
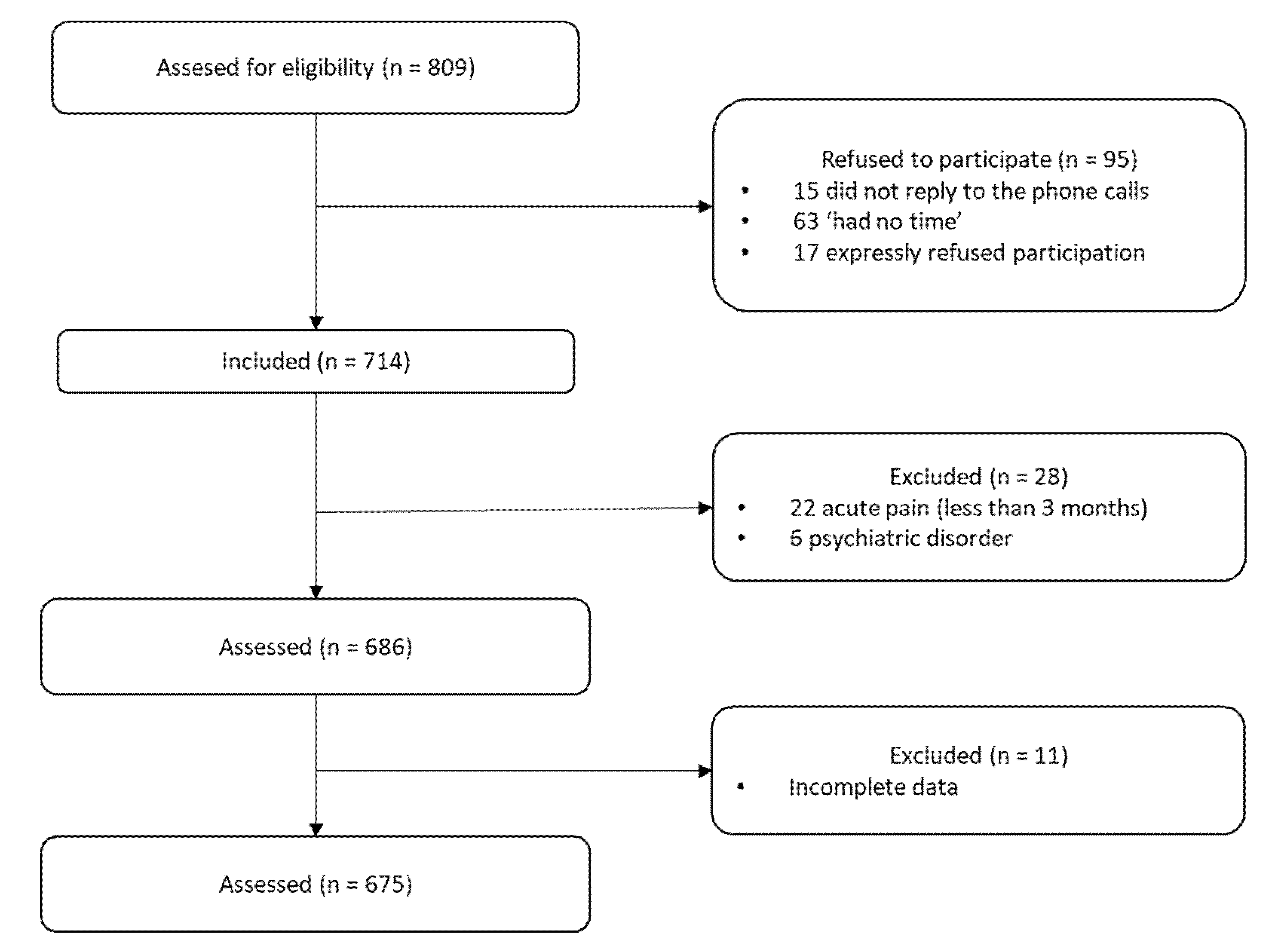
2.1. Participants
2.2. Variables and Instruments
2.2.1. Demographic and Clinical Variables
2.2.2. Pain Intensity
2.2.3. Depressive Symptoms
2.2.4. Pain Catastrophizing
2.2.5. Pain Acceptance
2.3. Statistical Analysis
3. Results
3.1. Frequency Data for the Demographic and Clinical Variables
3.2. Univariate Differences between Individuals Given and Not Given an Opioid Prescription
3.3. Multivariate Associations between the Study Predictors and Differences in Opioid Prescribing
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Use of Opioids in the Management of Chronic Noncancer Pain. Available online: https://www.uptodate.com/contents/use-of-opioids-in-the-management-of-chronic-non-cancer-pain (accessed on 6 July 2020).
- Chou, R.; Fanciullo, G.J.; Fine, P.G.; Adler, J.A.; Ballantyne, J.C.; Davies, P.; Donovan, M.I.; Fishbain, D.A.; Foley, K.M.; Fudin, J.; et al. Clinical Guidelines for the Use of Chronic Opioid Therapy in Chronic Noncancer Pain. J. Pain 2009, 10, 113–130. [Google Scholar] [CrossRef] [PubMed]
- Højsted, J.; Sjøgren, P. Addiction to Opioids in Chronic Pain Patients: A Literature Review. Eur. J. Pain 2007, 11, 490–518. [Google Scholar] [CrossRef] [PubMed]
- Kaye, A.D.; Jones, M.R.; Kaye, A.M.; Ripoll, J.G.; Jones, D.E.; Galan, V.; Beakley, B.D.; Calixto, F.; Bolden, J.L.; Urman, R.D.; et al. Prescription Opioid Abuse in Chronic Pain: An Updated Review of Opioid Abuse Predictors and Strategies to Curb Opioid Abuse (Part 2). Pain Physician 2017, 20, S111–S133. [Google Scholar] [PubMed]
- Volkow, N.D.; McLellan, A.T. Opioid Abuse in Chronic Pain—Misconceptions and Mitigation Strategies. N. Engl. J. Med. 2016, 374, 1253–1263. [Google Scholar] [CrossRef]
- Santana Pineda, M.; Jover López-Rodrigalvarez, R.; Rodríguez Sainz, P.; Gómez Cortes, M.; Rodríguez Huertas, F.; Morgado Muñoz, I. Uso y Abuso de Opioides en el Área Norte de La Provincia de Cádiz. Rev. Soc. Esp. Dolor 2016, 23, 127–134. [Google Scholar] [CrossRef]
- Secades Villa, R.; García, E.R.; Barbero, J.V.; Fernández Hermida, J.R.; Seco, G.V.; Garcia, J.M.J. El Consume de Psicofármacos En Pacientes Que Acuden a Atención Primaria En El Principado de Asturias (España). Psicothema 2003, 15, 650–655. [Google Scholar]
- Chenaf, C.; Kaboré, J.L.; Delorme, J.; Pereira, B.; Mulliez, A.; Zenut, M.; Delage, N.; Ardid, D.; Eschalier, A.; Authier, N. Prescription Opioid Analgesic Use in France: Trends and Impact on Morbidity–Mortality. Eur. J. Pain 2019, 23, 124–134. [Google Scholar] [CrossRef]
- Ray, W.A.; Chung, C.P.; Murray, K.T.; Hall, K.; Stein, C.M. Prescription of Long-Acting Opioids and Mortality in Patients with Chronic Noncancer Pain Supplemental Content at Jama.Com Original Investigation. JAMA 2016, 315, 2415–2423. [Google Scholar] [CrossRef]
- Zeng, C.; Dubreuil, M.; Larochelle, M.R.; Lu, N.; Wei, J.; Choi, H.K.; Lei, G.; Zhang, Y. Association of Tramadol with All-Cause Mortality among Patients with Osteoarthritis. JAMA 2019, 321, 969–982. [Google Scholar] [CrossRef]
- Furlan, A.D.; Sandoval, J.A.; Mailis-Gagnon, A.; Tunks, E. Opioids for Chronic Noncancer Pain: A Meta-Analysis of Effectiveness and Side Effects. CMAJ 2006, 174, 1589–1594. [Google Scholar] [CrossRef]
- Kalso, E.; Edwards, J.E.; Moore, R.A.; Mcquay, H.J. Opioids in Chronic Noncancer Pain: Systematic Review of Efficacy and Safety. Pain 2004, 112, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.C.; Solomon, D.H. Towards Defining the Safer Use of Opioids in Rheumatology. Nat. Rev. Rheumatol. 2020, 16, 71–72. [Google Scholar] [CrossRef] [PubMed]
- Bialas, P.; Maier, C.; Klose, P.; Häuser, W. Efficacy and Harms of Long-term Opioid Therapy in Chronic Non-cancer Pain: Systematic Review and Meta-analysis of Open-label Extension Trials with a Study Duration ≥ 26 Weeks. Eur. J. Pain 2020, 24, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Veiga, D.R.; Monteiro-Soares, M.; Mendonça, L.; Sampaio, R.; Castro-Lopes, J.M.; Azevedo, L.F. Effectiveness of Opioids for Chronic Noncancer Pain: A Two-Year Multicenter, Prospective Cohort Study with Propensity Score Matching. J. Pain 2019, 20, 706–715. [Google Scholar] [CrossRef]
- U.S. Opioid Prescribing Rate Maps|Drug Overdose|CDC Injury Center. Available online: https://www.cdc.gov/drugoverdose/maps/rxrate-maps.html (accessed on 24 June 2020).
- Tait, R.C.; Chibnall, J.T. Physician Judgments of Chronic Pain Patients. Soc. Sci. Med. 1997, 45, 1199–1205. [Google Scholar] [CrossRef]
- Tait, R.C.; Chibnall, J.T.; Kalauokalani, D. Provider Judgments of Patients in Pain: Seeking Symptom Certainty. Pain Med. 2009, 10, 11–34. [Google Scholar] [CrossRef]
- Serdarevic, M.; Striley, C.W.; Cottler, L.B. Sex Differences in Prescription Opioid Use. Curr. Opin. Psychiatry 2017, 30, 238–246. [Google Scholar] [CrossRef]
- Ickowicz, E. Pharmacological Management of Persistent Pain in Older Persons. J. Am. Geriatr. Soc. 2009, 57, 1331–1346. [Google Scholar] [CrossRef]
- Frenk, S.; Porter, K.; Paulozzi, L. Prescriptyion Opioid Analgesic Use among Adults: United States, 1999–2012; US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Washington, DC, USA, 2015.
- Bedene, A.; Lijfering, W.M.; Niesters, M.; van Velzen, M.; Rosendaal, F.R.; Bouvy, M.L.; Dahan, A.; van Dorp, E.L.A. Opioid Prescription Patterns and Risk Factors Associated with Opioid Use in the Netherlands. JAMA Netw. Open 2019, 2, e1910223. [Google Scholar] [CrossRef]
- Calvo-Falcón, R.; Torres-Morera, L.-M.; Calvo-Falcón, R.; Torres-Morera, L.-M. Tratamiento con Opioides en Dolor Crónico No Oncológico: Recomendaciones para una Prescripción Segura. Rev. Soc. Española Dolor 2017, 24, 313–323. [Google Scholar] [CrossRef]
- Cheung, C.W.; Chan, T.C.; Chen, P.P.; Chu, M.C.; Chui, W.C.; Ho, P.T.; Lam, F.; Law, S.W.; Lee, J.L.; Wong, S.H.; et al. Opioid Therapy for Chronic Noncancer Pain: Guidelines for Hong Kong. Hong Kong Med. J. 2016, 22, 496–505. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fischer, B.; Nakamura, N.; Ialomiteanu, A.; Boak, A.; Rehm, J. Assessing the Prevalence of Nonmedical Prescription Opioid Use in the General Canadian Population: Methodological Issues and Questions. Can. J. Psychiatry 2010, 55, 606–609. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.D.; Lee, J.Y.; Son, J.S.; Byeon, G.J.; Yeo, J.S.; Kim, D.W.; Yoo, S.H.; Hong, J.H.; Park, H.J.; Opioids Research Group in the Korean Pain Society. Guidelines for Prescribing Opioids for Chronic Noncancer Pain in Korea. Korean J. Pain 2017, 30, 18–33. [Google Scholar] [CrossRef] [PubMed]
- Manchikanti, L.; Abdi, S.; Atluri, S.; Balog, C.C.; Benyamin, R.; Boswell, M.V.; Brown, K.R.; Bruel, B.M.; Bryce, D.A.; Burks, P.A.; et al. American Society of Interventional Pain Physicians (ASIPP) Guidelines for Responsible Opioid Prescribing in Chronic Noncancer Pain: Part 2—Guidance. Pain Physician 2012, 15, S67–S116. [Google Scholar]
- Morasco, B.J.; Yarborough, B.J.; Smith, N.X.; Dobscha, S.K.; Deyo, R.A.; Perrin, N.A.; Green, C.A. Higher Prescription Opioid Dose is Associated with Worse Patient-Reported Pain Outcomes and More Health Care Utilization. J. Pain 2017, 18, 437–445. [Google Scholar] [CrossRef]
- Bačkonja, M.M.; Farrar, J.T. Are Pain Ratings Irrelevant? Pain Med. 2015, 16, 1247–1250. [Google Scholar] [CrossRef]
- Morone, N.; Weiner, D. Pain as the Fifth Vital Sign: Exposing the Vital Need for Pain Education. Clin. Ther. 2013, 35, 1728–1732. [Google Scholar] [CrossRef]
- Pasero, C.; Quinlan-Colwell, A.; Rae, D.; Broglio, K.; Drew, D. American Society for Pain Management Nursing Position Statement: Prescribing and Administering Opioid Doses Based Solely on Pain Intensity. Pain Manag. Nurs. 2016, 17, 170–180. [Google Scholar] [CrossRef]
- Adams, M.H.; Dobscha, S.K.; Smith, N.X.; Yarborough, B.J.; Deyo, R.A.; Morasco, B.J. Prevalence and Correlates of Low Pain Interference among Patients with High Pain Intensity who are Prescribed Long-Term Opioid Therapy. J. Pain 2018, 19, 1074–1081. [Google Scholar] [CrossRef]
- Dobscha, S.K.; Morasco, B.J.; Duckart, J.P.; Macey, T.; Deyo, R.A. Correlates of Prescription Opioid Initiation and Long-Term Opioid Use in Veterans with Persistent Pain. Clin. J. Pain 2013, 29, 102. [Google Scholar] [CrossRef]
- Braden, J.B.; Sullivan, M.D.; Ray, G.T.; Saunders, K.; Merrill, J.; Silverberg, M.J.; Rutter, C.M.; Weisner, C.; Banta-Green, M.C.J.; Campbell, C.; et al. Trends in Long-Term Opioid Therapy for Noncancer Pain among Persons with a History of Depression. Gen. Hosp. Psychiatr. 2009, 31, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Paillard, F.; McKeown, A.; Burke, L.B.; Edwards, R.R.; Katz, N.P.; Papadopoulos, E.J.; Rappaport, B.A.; Slagle, A.; Strain, E.C.; et al. Instruments to Identify Prescription Medication Misuse, Abuse, and Related Events in Clinical Trials: An ACTTION Systematic Review. J. Pain 2015, 16, 389–411. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Thomsen, A.B.; Højsted, J. 10-Year Follow-up of Chronic Non-Malignant Pain Patients: Opioid Use, Health Related Quality of Life and Health Care Utilization. Eur. J. Pain 2006, 10, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Turk, D.C.; Okifuji, A. What Factors Affect Physicians’ Decisions to Prescribe Opioids for Chronic Noncancer Pain Patients? Clin. J. Pain 1997, 13, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.; Cahalan, C.; Mensing, G.; Smith, M.; Haythornthwaite, J. Pain, Catastrophizing, and Depression in the Rheumatic Diseases. Nat. Rev. Rheumatol. 2011, 7, 216–224. [Google Scholar] [CrossRef]
- Sullivan, M.J.L.; Rodgers, W.M.; Kirsch, I. Catastrophizing, Depression and Expectancies for Pain and Emotional Distress. Pain 2001, 91, 147–154. [Google Scholar] [CrossRef]
- Sullivan, M.J.L.; Bishop, S.R.; Pivik, J. The Pain Catastrophizing Scale: Development and Validation. Psychol. Assess. 1995, 7, 524–532. [Google Scholar] [CrossRef]
- Kapoor, S.; Thorn, B.E.; Bandy, O.; Clements, K.L. Pain Referents Used to Respond to the Pain Catastrophizing Scale. Eur. J. Pain 2015, 19, 400–407. [Google Scholar] [CrossRef]
- Martinez-Calderon, J.; Jensen, M.; Morales-Asencio, J.; Luque-Suárez, A. Pain Catastrophizing and Function in Individuals with Chronic Musculoskeletal Pain. Clin. J. Pain 2019, 35, 279–293. [Google Scholar] [CrossRef]
- Kroska, E. A Meta-Analysis of Fear-Avoidance and Pain Intensity: The Paradox of Chronic Pain. Scand. J. Pain 2016, 13, 43–58. [Google Scholar] [CrossRef]
- Ramírez-Maestre, C.; Esteve, R.; López-Martínez, A. Fear-Avoidance, Pain Acceptance and Adjustment to Chronic Pain: A Cross-Sectional Study on a Sample of 686 Patients with Chronic Spinal Pain. Ann. Behav. Med. 2014, 48, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Maestre, C.; Esteve, R.; Ruiz-Párraga, G.; Gómez-Pérez, L.; López-Martínez, A.E. The Key Role of Pain Catastrophizing in the Disability of Patients with Acute Back Pain. Int. J. Behav. Med. 2017, 24, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Mankovsky, T.; Lynch, M.E.; Clark, A.J.; Sawynok, J.; Sullivan, M.J. Pain Catastrophizing Predicts Poor Response to Topical Analgesics in Patients with Neuropathic Pain. Pain Res. Manag. 2012, 17, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Finan, P.H.; Carroll, C.P.; Moscou-Jackson, G.; Martel, M.O.; Campbell, C.M.; Pressman, A.; Smyth, J.M.; Tremblay, J.M.; Lanzkron, S.M.; Haythornthwaite, J.A. Daily Opioid Use Fluctuates as a Function of Pain, Catastrophizing, and Affect in Patients with Sickle Cell Disease: An Electronic Daily Diary Analysis. J. Pain 2018, 19, 46–56. [Google Scholar] [CrossRef] [PubMed]
- McCracken, L.M.; Eccleston, C. Coping or Acceptance: What to Do about Chronic Pain? Pain 2003, 105, 197–204. [Google Scholar] [CrossRef]
- Rhodes, A.; Marks, D.; Block-Lerner, J.; Lomauro, T. Psychological Flexibility, Pain Characteristics and Risk of Opioid Misuse in Noncancerous Chronic Pain Patients. J. Clin. Psychol. Med. Settings 2020. [Google Scholar] [CrossRef]
- Mccracken, L.M.; Morley, S. The Psychological Flexibility Model: A Basis for Integration and Progress in Psychological Approaches to Chronic Pain Management. J. Pain 2014, 15, 221–232. [Google Scholar] [CrossRef]
- Clementi, M.A.; Kao, G.S.; Monico, E. Pain Acceptance as a Predictor of Medical Utilization and School Absenteeism in Adolescents with Chronic Pain. J. Pediatr. Psychol. 2018, 43, 294–302. [Google Scholar] [CrossRef]
- Elander, J.; Duarte, J.; Maratos, F.A.; Gilbert, P. Predictors of Painkiller Dependence among People with Pain in the General Population. Pain Med. 2014, 15, 613–624. [Google Scholar] [CrossRef]
- Lin, L.A.; Bohnert, A.S.B.; Price, A.M.; Jannausch, M.; Bonar, E.E.; Ilgen, M.A. Pain Acceptance and Opiate Use Disorders in Addiction Treatment Patients with Comorbid Pain. Drug Alcohol. Depend. 2015, 157, 136–142. [Google Scholar] [CrossRef]
- Sinnenberg, L.E.; Wanner, K.J.; Perrone, J.; Barg, F.K.; Rhodes, K.V.; Meisel, Z.F. What Factors Affect Physicians’ Decisions to Prescribe Opioids in Emergency Departments? MDM Policy Pract. 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, S.F.; Costa, M.; Carvalho, H. The Role of Physician Sex on Chronic Low-Back Pain Assessment and Treatment Prescriptions. J. Pain 2013, 14, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.T.; Hochheimer, C.J.; Brooks, E.M.; Sabo, R.T.; Jiang, V.; Day, T.; Rozman, J.S.; Kashiri, P.L.; Krist, A.H. Chronic Opioid Prescribing in Primary Care: Factors and Perspectives. Ann. Fam. Med. 2019, 17, 200–206. [Google Scholar] [CrossRef]
- García Vicente, J.A.; Vedia Urgell, C.; Vallès Fernández, R.; Reina Rodríguez, D.; Rodoreda Noguerola, S.; Samper Bernal, D. Quasi-Experimental Study of an Intervention on the Pharmacological Management of Non-Oncological Chronic Pain in Primary Care. Aten. Primaria 2019, 52, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Esteve, R.; Ramírez-Maestre, C. Pain Fear Avoidance and Pain Acceptance: A Cross-Sectional Study Comparing their Influence on Adjustment to Chronic Pain across Three Samples of Patients. Ann. Behav. Med. 2013, 46, 169–180. [Google Scholar] [CrossRef]
- Austin, P.C.; Steyerberg, E.W. The number of subjects per variable required in linear regression analyses. J. Clin. Epidemiol. 2015, 68, 627–636. [Google Scholar] [CrossRef]
- Jensen, M.; Karoly, P. Self-Report Scales and Procedures for Assessing Pain in Adults. In Handbook of Pain Assessment; Turk, D., Melzack, R., Eds.; Guilford Press: New York, NY, USA, 2001; pp. 15–34. [Google Scholar]
- Zigmond, A.; Snaith, R. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- Ouintana, J.M.; Padierna, A.; Esteban, C.; Arostegui, I.; Bilbao, A.; Ruiz, I. Evaluation of the Psychometric Characteristics of the Spanish Version of the Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 2003, 107, 216–221. [Google Scholar] [CrossRef]
- García-Campayo, J.; Rodero, B.; Del Hoyo, Y.L.; Luciano, J.; Alda, M.; Gili, M. Validation of a Spanish Language Version of the Pain Self-Perception Scale in Patients with Fibromyalgia. BMC Musculoskelet. Disord. 2010, 11, 255. [Google Scholar] [CrossRef]
- Muñoz, M.; Esteve, R. Reports of Memory Functioning by Patients with Chronic Pain. Clin. J. Pain 2005, 21, 287–291. [Google Scholar]
- Mccracken, L.M.; Vowles, K.E.; Eccleston, C. Acceptance of Chronic Pain: Component Analysis and a Revised Assessment Method. Pain 2004, 107, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Bendayan, R.; Esteve, R.; Blanca, M. Empirical Evidence of the Validity of the Spanish Version of the Chronic Pain Acceptance Questionnaire: The Differential Influence of Activity Engagement and Pain Willingness on Adjustment to Chronic Pain. Br. J. Health Psychol. 2012, 17, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates Inc.: Mahwah, NJ, USA, 1988; Volume 13. [Google Scholar]
- Huhn, A.S.; Tompkins, D.A.; Campbell, C.M.; Dunn, K.E. Individuals with Chronic Pain who Misuse Prescription Opioids Report Sex-Based Differences in Pain and Opioid Withdrawal. Pain Med. 2019, 20, 1942–1947. [Google Scholar] [CrossRef] [PubMed]
- Melchior, M.; Poisbeau, P.; Gaumond, I.; Marchand, S. Insights into the Mechanisms and the Emergence of Sex-Differences in Pain. Neuroscience 2016, 338, 63–80. [Google Scholar] [CrossRef]
- Fillingim, R.B. Sex, Gender, and Pain. In Principles of Gender-Specific Medicine: Gender in the Genomic Era, 3rd ed.; Elsevier: London, UK, 2017; pp. 481–496. [Google Scholar] [CrossRef]
- Edwards, R.R.; Haythornthwaite, J.A.; Sullivan, M.J.; Fillingim, R.B. Catastrophizing as a Mediator of Sex Differences in Pain: Differential Effects for Daily Pain versus Laboratory-Induced Pain. Pain 2004, 111, 335–341. [Google Scholar] [CrossRef]
- Keefe, F.J.; Lefebvre, J.C.; Egert, J.R.; Affeck, G.; Sullivan, M.J.; Caldwell, D.S. The Relationship of Gender to Pain, Pain Behavior, and Disability in Osteoarthritis Patients: The Role of Catastrophizing. Pain 2000, 87, 325–334. [Google Scholar] [CrossRef]
- Sullivan, M.J.L.; Tripp, D.A.; Santor, D. Gender Differences in Pain and Pain Behavior: The Role of Catastrophizing. Cognit. Ther. Res. 2000, 24, 121–134. [Google Scholar] [CrossRef]
- Calcaterra, S.; Glanz, J.; Binswanger, I. National Trends in Pharmaceutical Opioid Related Overdose Deaths Compared to Other Substance Related Overdose Deaths: 1999–2009. Drug Alcohol Depend. 2013, 131, 263–270. [Google Scholar] [CrossRef]
- Unick, G.J.; Rosenblum, D.; Mars, S.; Ciccarone, D. Intertwined Epidemics: National Demographic Trends in Hospitalizations for Heroin- and Opioid-Related Overdoses, 1993–2009. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Coloma-Carmona, A.; Carballo, J.L.; Rodríguez-Marín, J.; Pérez-Carbonell, A. Uso y Dependencia de Fármacos Opioides En Población Española Con Dolor Crónico: Prevalencia y Diferencias de Género. Rev. Clin. Esp. 2017, 217, 315–319. [Google Scholar] [CrossRef]
- Simoni-Wastila, L. The Use of Abusable Prescription Drugs: The Role of Gender. J. Women’s Health Gender Based Med. 2000, 9, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Samulowitz, A.; Gremyr, I.; Eriksson, E.; Hensing, G. “Brave men” and “emotional women”: A theory-guided literature review on gender bias in health care and gendered norms towards patients with chronic pain. Pain Res. Manag. 2018, 2018, 6358624. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.; Blackwell, R.H.; Kuo, P.C.; Gupta, G.N. Rates and Risk Factors for Opioid Dependence and Overdose after Urological Surgery. J. Urol. 2017, 198, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Webster, L.; Webster, R. Predicting Aberrant Behaviors in Opioid-Treated Patients: Preliminary Validation of the Opioid Risk Tool. Pain Med. 2005, 6, 432–442. [Google Scholar] [CrossRef]
- Schieber, L.Z.; Guy, G.P.; Seth, P.; Losby, J.L. Variation in Adult Outpatient Opioid Prescription Dispensing by Age and Sex—United States, 2008–2018. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 298–302. [Google Scholar] [CrossRef]
- McCaffery, M.; Herr, K.; Pasero, C. Assessment Tools. In Pain Assessment and Pharmacologic Management; Pasero, C., McCaffery, M., Eds.; Elsevier/Mosby.: St. Louis, MO, USA, 2011; pp. 33–176. [Google Scholar]
- Krebs, E.E.; Gravely, A.; Nugent, S.; Jensen, A.C.; DeRonne, B.; Goldsmith, E.S.; Kroenke, K.; Bair, M.J.; Noorbaloochi, S. Effect of Opioid vs. Nonopioid Medications on Pain-Related Function in Patients with Chronic Back Pain or Hip or Knee Osteoarthritis Pain the SPACE Randomized Clinical Trial. JAMA 2018, 319, 872–882. [Google Scholar] [CrossRef]
- Sjøgren, P.; Grønbæk, M.; Peuckmann, V.; Ekholm, O. A Population-Based Cohort Study on Chronic Pain: The Role of Opioids. Clin. J. Pain 2010, 26, 763–769. [Google Scholar] [CrossRef]
- Turner, J.; Shortreed, S.; Saunders, K.; LeResche, L. Association of Levels of Opioid Use with Pain and Activity Interference among Patients Initiating Chronic Opioid Therapy: A Longitudinal Study. Pain 2016, 157, 849. [Google Scholar] [CrossRef]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC Guideline for Prescribing Opioids for Chronic Pain-United States, 2016. JAMA 2016, 315, 1624–1645. [Google Scholar] [CrossRef]
- Sullivan, M.D. Depression Effects on Long-Term Prescription Opioid Use, Abuse, and Addiction. Clin J. Pain 2018, 34, 878–884. [Google Scholar] [CrossRef]
- Salazar, A.; Dueñas, M.; Mico, J.A.; Ojeda, B.; Agüera-Ortiz, L.; Cervilla, J.A.; Failde, I. Undiagnosed Mood Disorders and Sleep Disturbances in Primary Care Patients with Chronic Musculoskeletal Pain. Pain Med. 2013, 14, 1416–1425. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sullivan, M.J.L.; Adams, H.; Tripp, D.; Stanish, W.D. Stage of Chronicity and Treatment Response in Patients with Musculoskeletal Injuries and Concurrent Symptoms of Depression. Pain 2008, 135, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Bair, M.; Robinson, R.; Katon, W.; Kroenke, K. Depression and Pain Comorbidity. Arch. Intern. Med. 2003, 12, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Breivik, H.; Collett, B.; Ventafridda, V.; Cohen, R.; Gallacher, D. Survey of Chronic Pain in Europe: Prevalence, Impact on Daily Life, and Treatment. Eur. J. Pain 2006, 10, 287–333. [Google Scholar] [CrossRef] [PubMed]
- Alonso, J.; Angermeyer, M.C.; Bernert, S.; Bruffaerts, R.; Brugha, T.S.; Bryson, H.; Girolamo, G.; Graaf, R.; Demyttenaere, K.; Gasquet, I.; et al. Use of Mental Health Services in Europe: Results from the European Study of the Epidemiology of Mental Disorders (ESEMeD) Project. Acta Psychiatr. Scand. 2004, 109, 47–54. [Google Scholar] [CrossRef]
- Haro, J.M.; Palacín, C.; Vilagut, G.; Martínez, M.; Bernal, M.; Luque, I.; Codony, M.; Doltz, M.; Alonso, J. Grupo ESEMeD-España. Prevalencia de Los Trastornos Mentales y Factores Asociados: Resultados Del Estudio ESEMeD-España. Med. Clin. 2006, 126, 445–451. [Google Scholar] [CrossRef]
- Wasan, A.D.; Michna, E.; Edwards, R.R.; Katz, J.N.; Nedeljkovic, S.S.; Dolman, A.J.; Janfaza, D.; Isaac, Z.; Jamison, R.N. Psychiatric Comorbidity is Associated Prospectively with Diminished Opioid Analgesia and Increased Opioid Misuse in Patients with Chronic Low Back Pain. J. Am. Soc. Anasthesiol. 2015, 123, 861–872. [Google Scholar] [CrossRef]
- Wasan, A.; Davar, G.; Jamison, R. The Association between Negative Affect and Opioid Analgesia in Patients with Discogenic Low Back Pain. Pain 2005, 117, 450–461. [Google Scholar] [CrossRef]
- Feingold, D.; Brill, S.; Goor-Aryeh, I.; Delayahu, Y.; Lev-Ran, S. The Association between Severity of Depression and Prescription Opioid Misuse among Chronic Pain Patients with and without Anxiety: A Cross-Sectional Study. J. Affect. Disord. 2018, 235, 293–302. [Google Scholar] [CrossRef]
- Gilam, G.; Sturgeon, J.; You, D.; Darnall, B.; Mackey, S. Negative Affect as a Predictor of Opioid Prescription Misuse and Abuse in Chronic Pain Patients: A Collaborative Health Outcomes Information Registry Study. J. Pain 2019, 20, S17. [Google Scholar] [CrossRef]
- Davis, M.A.; Lin, L.A.; Liu, H.; Sites, B.D. Prescription Opioid Use among Adults with Mental Health Disorders in the United States. J. Am. Board Fam. Med. 2017, 30, 407–417. [Google Scholar] [CrossRef]
- Seal, K.H.; Shi, Y.; Cohen, G.; Cohen, B.E.; Maguen, S.; Krebs, E.E.; Neylan, T.C. Association of Mental Health Disorders with Prescription Opioids and High-Risk Opioid Use in US Veterans of Iraq and Afghanistan. JAMA 2012, 307, 940–947. [Google Scholar] [CrossRef]
- Merrill, J.O.; Von Korff, M.; Banta-Green, C.J.; Sullivan, M.D.; Saunders, K.W.; Campbell, C.I.; Weisner, C. Prescribed Opioid Difficulties, Depression and Opioid Dose among Chronic Opioid Therapy Patients. Gen. Hosp. Psychiatry 2012, 34, 581–587. [Google Scholar] [CrossRef]
- McCracken, L.M.; Vowles, K.E. Acceptance of Chronic Pain. Curr. Pain Headache Rep. 2006, 10, 90–94. [Google Scholar] [CrossRef]
| Variables | Mean | SD | Min/Max |
|---|---|---|---|
| Age (years) | 45.4 | 12.9 | 16/79 |
| Time in pain (months) | 49 | 59.3 | 4/240 |
| Pain intensity | 5.3 | 1.6 | 0.5/9.5 |
| N | % | ||
| Sex | |||
| Men | 274 | 41 | |
| Women | 401 | 59 | |
| Marital status | |||
| Single | 125 | 18.5 | |
| Married | 407 | 60.3 | |
| Unmarried couple | 54 | 8.0 | |
| Divorced | 39 | 5.8 | |
| Separated | 24 | 3.6 | |
| Widowed | 26 | 3.9 | |
| Education | |||
| Reading and writing | 70 | 10.4 | |
| Primary school | 231 | 34.2 | |
| High school | 239 | 35.3 | |
| University education | 135 | 20.1 | |
| Work status | |||
| Housekeeping | 112 | 16.6 | |
| Working | 352 | 52.2 | |
| Studying | 17 | 2.5 | |
| Unemployed | 102 | 15.1 | |
| Retired | 92 | 13.6 | |
| Site of pain | |||
| Upper back | 435 | 64.4 | |
| Sacrum | 431 | 63.9 | |
| Neck | 361 | 53.5 | |
| Lower back | 251 | 37.2 |
| Participants Prescribed Opioids (N = 85) | Participants not Prescribed Opioids (N = 590) | p Value | Cohen’s δ | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Age | 49.04 | 11.05 | 44.88 | 13.12 | 0.002 | 0.34 |
| Pain intensity | 6.03 | 1.49 | 5.14 | 1.61 | <0.001 | 0.57 |
| Depressive symptoms | 17.35 | 5.49 | 14.67 | 5.51 | <0.001 | 0.49 |
| Pain acceptance | 65.49 | 7.97 | 69.60 | 9.96 | <0.001 | 0.45 |
| Pain catastrophizing | 23.87 | 10.82 | 23.34 | 8.85 | 0.669 | |
| Beta | Wald | Exp(B) | p | |
|---|---|---|---|---|
| Age | 0.017 | 2.729 | 1.017 | 0.099 |
| Pain intensity | 0.245 | 9.206 | 1.278 | 0.002 |
| Depressive symptoms | 0.061 | 6.318 | 1.063 | 0.012 |
| Pain acceptance | −0.013 | 1.027 | 0.987 | 0.311 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Maestre, C.; Reyes-Pérez, Á.; Esteve, R.; López-Martínez, A.E.; Bernardes, S.; Jensen, M.P. Opioid Pain Medication Prescription for Chronic Pain in Primary Care Centers: The Roles of Pain Acceptance, Pain Intensity, Depressive Symptoms, Pain Catastrophizing, Sex, and Age. Int. J. Environ. Res. Public Health 2020, 17, 6428. https://doi.org/10.3390/ijerph17176428
Ramírez-Maestre C, Reyes-Pérez Á, Esteve R, López-Martínez AE, Bernardes S, Jensen MP. Opioid Pain Medication Prescription for Chronic Pain in Primary Care Centers: The Roles of Pain Acceptance, Pain Intensity, Depressive Symptoms, Pain Catastrophizing, Sex, and Age. International Journal of Environmental Research and Public Health. 2020; 17(17):6428. https://doi.org/10.3390/ijerph17176428
Chicago/Turabian StyleRamírez-Maestre, Carmen, Ángela Reyes-Pérez, Rosa Esteve, Alicia E. López-Martínez, Sonia Bernardes, and Mark P. Jensen. 2020. "Opioid Pain Medication Prescription for Chronic Pain in Primary Care Centers: The Roles of Pain Acceptance, Pain Intensity, Depressive Symptoms, Pain Catastrophizing, Sex, and Age" International Journal of Environmental Research and Public Health 17, no. 17: 6428. https://doi.org/10.3390/ijerph17176428
APA StyleRamírez-Maestre, C., Reyes-Pérez, Á., Esteve, R., López-Martínez, A. E., Bernardes, S., & Jensen, M. P. (2020). Opioid Pain Medication Prescription for Chronic Pain in Primary Care Centers: The Roles of Pain Acceptance, Pain Intensity, Depressive Symptoms, Pain Catastrophizing, Sex, and Age. International Journal of Environmental Research and Public Health, 17(17), 6428. https://doi.org/10.3390/ijerph17176428








