Dissociable Effects of Executive Load on Perceived Exertion and Emotional Valence during Submaximal Cycling
Abstract
1. Introduction
1.1. Effects of Mental Demand on Endurance Performance and Related Variables
1.2. Dissociation between Perceived Exertion and Emotional Dimensions in Dual Cognitive-Physical Tasks
1.3. Rationale and Aims of the Study
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Apparatus and Tasks
2.3.1. Pre-Experimental and Experimental Physical Effort Tasks
2.3.2. Mental Workload Tasks
2.4. Measurements
2.4.1. Rate of Perceived Exertion (RPE)
2.4.2. Self-Assessment Manikin (SAM)
2.4.3. Cognitive Performance on the Mental Load Tasks
2.4.4. Nutritional Assessment
2.5. Procedure
2.5.1. First (Pre-Experimental) Session
2.5.2. Second and Third (Experimental) Sessions
3. Results
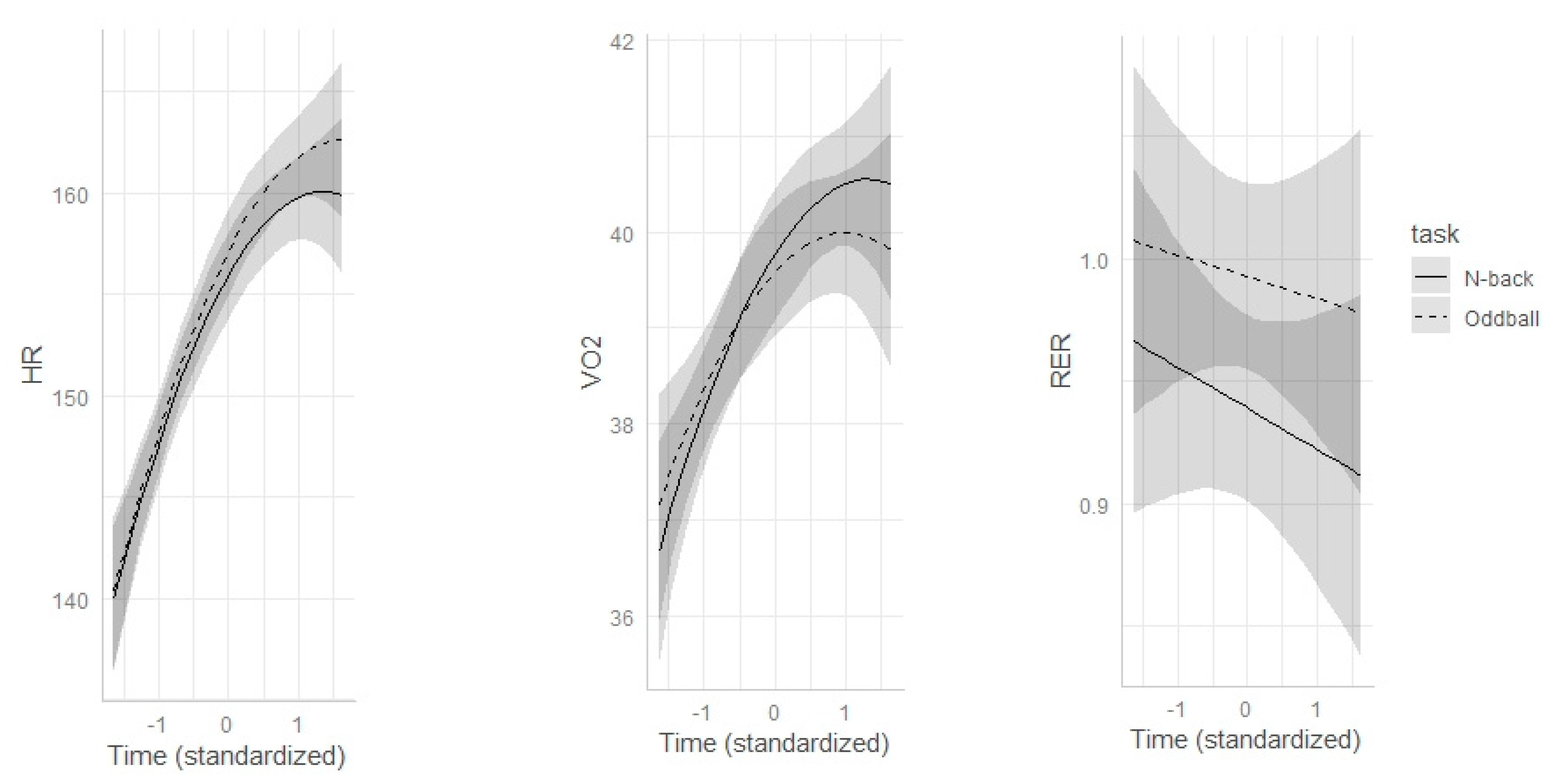
3.1. Indices of Internal Physical Load
3.2. Valence, Arousal, and RPE (Joint Analysis)
3.3. Valence, Arousal, and RPE (Segregated Analyses)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Supplementary Analyses
Appendix A.1. Analysis of Performance in the Cognitive Task
| MSE | F | df | p | η²p | |
|---|---|---|---|---|---|
| Task | 1.191 | 25.146 | 1 | <0.001 | 0.626 |
| Residual | 0.047 | 15 | |||
| Block | 0.006 | 2.213 | 17 | 0.004 | 0.129 |
| Residual | 0.003 | 255 | |||
| Task × Block | 0.006 | 2.461 | 17 | 0.001 | 0.141 |
| Residual | 0.002 | 255 |
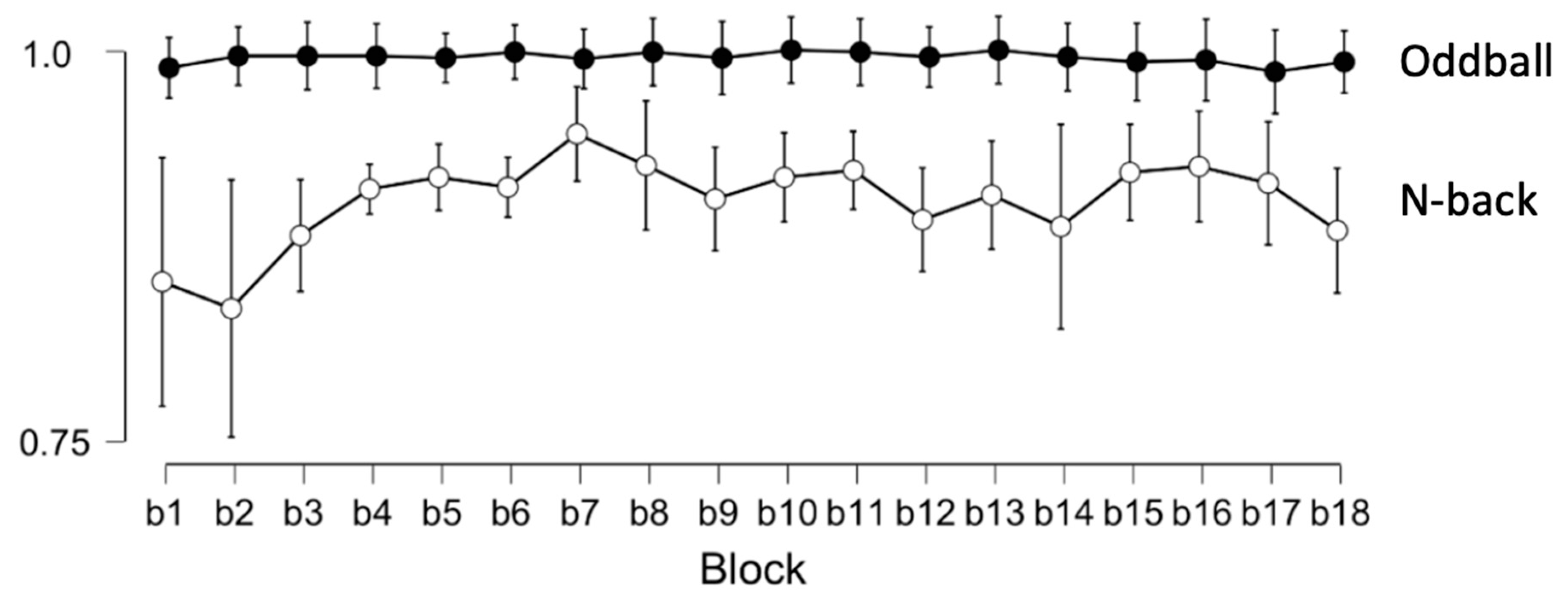
Appendix A.2. Analysis of Internal Physical Load Indices
| Predictors | Estimates | CI | p |
|---|---|---|---|
| Intercept | 153.33 | 148.32–158.33 | <0.001 |
| Time (linear) | 169.06 | 154.66–183.46 | <0.001 |
| Time (quadratic) | −54.49 | −68.92–−40.07 | <0.001 |
| Task | 1.26 | 0.49–2.03 | 0.001 |
| Time (linear)*Task | 20.06 | −0.36–40.49 | 0.054 |
| Time (quadratic)*Task | 3.32 | −17.08–23.71 | 0.750 |
| Random Effects | |||
| σ2 | 26.79 | ||
| τ00participant | 160.66 | ||
| ICC | 0.86 | ||
| Predictors | Estimates | CI | p |
|---|---|---|---|
| Intercept | 39.26 | 37.77–40.76 | <0.001 |
| Time (linear) | 32.60 | 26.37–38.84 | <0.001 |
| Time (quadratic) | −10.71 | −16.96–−4.46 | 0.001 |
| Task | −0.14 | −0.47–0.19 | 0.408 |
| Time (linear)*Task | −9.74 | −18.59–−0.89 | 0.031 |
| Time (quadratic)*Task | 0.60 | −8.23–9.44 | 0.894 |
| Random Effects | |||
| σ2 | 5.03 | ||
| τ00participant | 14.08 | ||
| ICC | 0.74 | ||
| Predictors | Estimates | CI | p |
|---|---|---|---|
| Intercept | 0.94 | 0.90–0.98 | <0.001 |
| Time | −0.02 | −0.05–0.02 | 0.375 |
| Task | 0.05 | 0.00–0.11 | 0.049 |
| Time*Task | 0.01 | −0.05–0.06 | 0.772 |
| Random Effects | |||
| σ2 | 0.14 | ||
| τ00code | 0.00 | ||
| ICC | 0.02 | ||
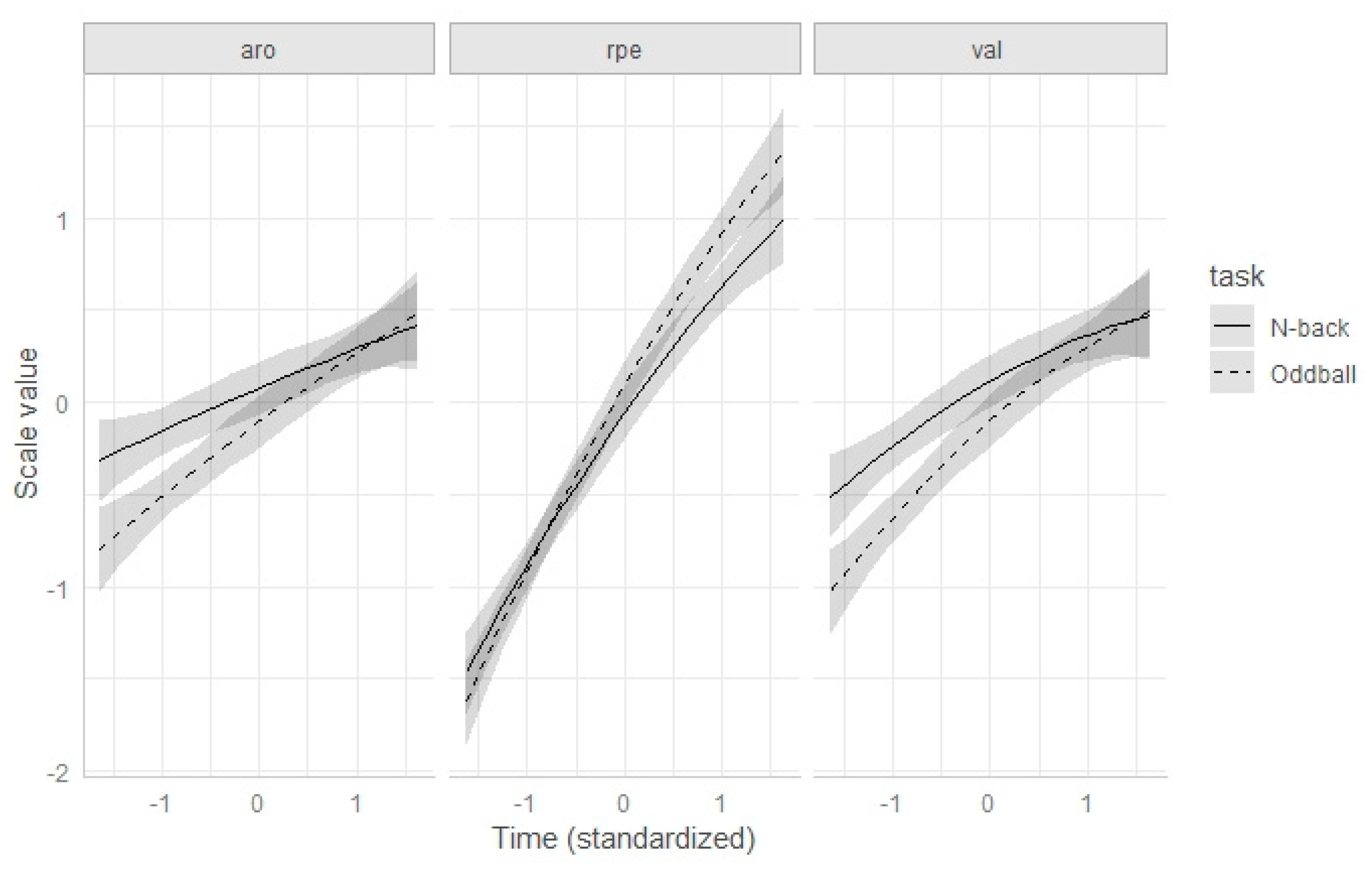
Appendix A.3. Joint Analysis of Arousal, Valence, and RPE without Internal Physical Load Covariates
| Predictors | Estimates | CI | p |
|---|---|---|---|
| Intercept | −0.02 | −0.30–0.26 | 0.881 |
| Time (linear) | 20.29 | 18.30–22.27 | <0.001 |
| Time (quadratic) | −1.80 | −3.79–0.19 | 0.076 |
| Measure (C1) | −0.00 | −0.06–0.05 | 0.866 |
| Measure (C2) | −0.07 | −0.10–−0.04 | <0.001 |
| Task | −0.11 | −0.17–−0.04 | 0.001 |
| Time (linear)*Measure (C1) | 1.84 | 0.13–3.55 | 0.035 |
| Time (quadratic)*Measure (C1) | −0.84 | −2.56–0.87 | 0.336 |
| Time (linear)*Measure (C2) | 7.78 | 6.79–8.77 | <0.001 |
| Time (quadratic)*Measure (C2) | −0.54 | −1.53–0.45 | 0.281 |
| Time (linear)*Task | 7.86 | 5.04–10.68 | <0.001 |
| Time (quadratic)*Task | −0.51 | −3.32–2.31 | 0.724 |
| Measure (C1)*Task | −0.02 | −0.09–0.06 | 0.644 |
| Measure (C2)*Task | 0.11 | 0.07–0.15 | <0.001 |
| Random Effects | |||
| σ2 | 0.51 | ||
| τ00participant | 0.51 | ||
| ICC | 0.50 | ||
References
- Mehta, R.K.; Agnew, M.J. Influence of mental workload on muscle endurance, fatigue, and recovery during intermittent static work. Eur. J. Appl. Physiol. 2012, 112, 2891–2902. [Google Scholar] [CrossRef] [PubMed]
- Vera, J.; Jiménez, R.; García, J.A.; Cárdenas, D. Simultaneous Physical and Mental Effort Alters Visual Function. Optom. Vis. Sci. Off. Publ. Am. Acad. Optom. 2017, 94, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Vera, J.; Perales, J.C.; Jiménez, R.; Cárdenas, D. A test-retest assessment of the effects of mental load on ratings of affect, arousal and perceived exertion during submaximal cycling. J. Sports Sci. 2018, 36, 2521–2530. [Google Scholar] [CrossRef] [PubMed]
- Gabbett, T.J.; Abernethy, B. Dual-task assessment of a sporting skill: Influence of task complexity and relationship with competitive performances. J. Sports Sci. 2012, 30, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, F.; Castillo-Díaz, A.; Madinabeitia, I.; Castillo-Rodríguez, A.; Cárdenas, D. Mental workload impairs the pass precision in soccer players. RPD 2018, 27, 155–164. [Google Scholar]
- Marcora, S.M.; Staiano, W.; Manning, V. Mental fatigue impairs physical performance in humans. J. Appl. Physiol. 2009, 106, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Coutts, A.J.; Merlini, M.; Deprez, D.; Lenoir, M.; Marcora, S.M. Mental fatigue impairs soccer-specific physical and technical performance. Med. Sci. Sports Exerc. 2016, 48, 267–276. [Google Scholar] [CrossRef]
- Cárdenas, D.; Perales, J.C.; Chirosa, L.J.; Conde-González, J.; Aguilar-Martínez, D.; Araya, S. The effect of mental workload on the intensity and emotional dynamics of perceived exertion. An. Psicol. 2013, 29, 662–673. [Google Scholar] [CrossRef]
- Muraven, M.; Tice, D.M.; Baumeister, R.F. Self-control as limited resource: Regulatory depletion patterns. J. Pers. Soc. Psychol. 1998, 74, 774–789. [Google Scholar] [CrossRef]
- Giboin, L.S.; Wolff, W. The effect of ego depletion or mental fatigue on subsequent physical endurance performance: A meta-analysis. Perform. Enhanc. Health 2019, 7, 100150. [Google Scholar] [CrossRef]
- Pageaux, B. The psychobiological model of endurance performance: An effort-based decision-making theory to explain self-paced endurance performance. Sports Med. 2014, 44, 1319–1320. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.W.; Self, E.A. The intensity of motivation. Annu. Rev. Psychol. 1989, 40, 109–131. [Google Scholar] [CrossRef] [PubMed]
- E Silva-Júnior, F.L.; Emanuel, P.; Sousa, J.; Silva, M.; Teixeira, S.; Pires, F.O.; Machado, S.; Arias-Carrion, O. Prior Acute Mental Exertion in Exercise and Sport. Clin. Pr. Epidemiol. Ment. Health 2016, 12, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, J.; Marcora, S.M.; De Pauw, K.; Bailey, S.; Meeusen, R.; Roelands, B. The Effects of Mental Fatigue on Physical Performance: A Systematic Review. Sports Med. 2017, 47, 1569–1588. [Google Scholar] [CrossRef]
- Brown, D.M.; Graham, J.D.; Innes, K.I.; Harris, S.; Flemington, A.; Bray, S.R. Effects of Prior Cognitive Exertion on Physical Performance: A Systematic Review and Meta-analysis. Sports Med. 2019, 50, 497–529. [Google Scholar] [CrossRef]
- Pageaux, B.; Lepers, R. The effects of mental fatigue on sport-related performance. Prog. Brain Res. 2018, 240, 291–315. [Google Scholar] [CrossRef]
- Holgado, D.; Troya, E.; Perales, J.C.; Vadillo, M.A.; Sanabria, D. Does mental fatigue impair physical performance? A replication study. Eur. J. Sport Sci. 2020, 1–9. [Google Scholar] [CrossRef]
- Di Domenico, A.; Nussbaum, M.A. Interactive effects of physical and mental workload on subjective workload assessment. Int. J. Ind. Erg. 2008, 38, 977–983. [Google Scholar] [CrossRef]
- Mehta, R.; Agnew, M.J. Subjective Evaluation of Physical and Mental Workload Interactions Across Different Muscle Groups. J. Occup. Environ. Hyg. 2014, 12, 62–68. [Google Scholar] [CrossRef]
- Pageaux, B. Perception of effort in Exercise Science: Definition, measurement and perspectives. Eur. J. Sport Sci. 2016, 16, 885–894. [Google Scholar] [CrossRef]
- Bradley, M.; Lang, P.J. Measuring Emotion: The Self-assesment Manikin and the Semantic Differential. J. Behav. Exp. Psychiatry 1994, 25, 49–59. [Google Scholar] [CrossRef]
- Hartman, M.E.; Ekkekakis, P.; Dicks, N.D.; Pettitt, R.W. Dynamics of pleasure–displeasure at the limit of exercise tolerance: Conceptualizing the sense of exertional physical fatigue as an affective response. J. Exp. Boil. 2018, 222. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, R.E.; Kates, A. Can the Affective Response to Exercise Predict Future Motives and Physical Activity Behavior? A Systematic Review of Published Evidence. Ann. Behav. Med. 2015, 49, 715–731. [Google Scholar] [CrossRef] [PubMed]
- Brand, S.; Gerber, M.; Colledge, F.; Holsboer-Trachsler, E.; Pühse, U.; Ludyga, S. Acute Exercise and Emotion Recognition in Young Adolescents. J. Sport Exerc. Psychol. 2019, 41, 129–136. [Google Scholar] [CrossRef]
- Ekkekakis, P. Pleasure and displeasure from the body: Perspectives from exercise. Cogn. Emot. 2003, 17, 213–239. [Google Scholar] [CrossRef]
- Ekkekakis, P.; Parfitt, G.; Petruzzello, S.J. The Pleasure and Displeasure People Feel When they Exercise at Different Intensities: Decennial update and progress towards a tripartite rationale for exercise intensity prescription. Sports Med. 2011, 41, 641–671. [Google Scholar] [CrossRef]
- Luque-Casado, A.; Zabala, M.; Morales, E.; Mateo-March, M.; Sanabria, D. Cognitive Performance and Heart Rate Variability: The Influence of Fitness Level. PLoS ONE 2013, 8, e56935. [Google Scholar] [CrossRef]
- Filipas, L.; Gallo, G.; Pollastri, L.; La Torre, A. Mental fatigue impairs time trial performance in sub-elite under 23 cyclists. PLoS ONE 2019, 14, e0218405. [Google Scholar] [CrossRef]
- Riebe, D.; Franklin, B.A.; Thompson, P.D.; Garber, C.E.; Whitfield, G.P.; Magal, M.; Pescatello, L.S. Updating ACSM’s Recommendations for Exercise Preparticipation Health Screening. Med. Sci. Sports Exerc. 2015, 47, 2473–2479. [Google Scholar] [CrossRef]
- Green, P.; MacLeod, C.J. SIMR: An R package for power analysis of generalized linear mixed models by simulation. Methods Ecol. Evol. 2016, 7, 493–498. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Casajús, J.A.; Piedrafita, E.; Aragonés, M.T. Criterios De Maximalidad En Pruebas De Esfuerzo Criteria for Maximal Exercise Test. Int. J. Med. Sci. Phys. Act. Sport 2009, 9, 217–231. [Google Scholar]
- Torregrosa-García, A.; Ávila-Gandía, V.; Luque-Rubia, A.J.; Ruiz, M.S.A.; Calderón, M.Q.; López-Román, F.J. Pomegranate Extract Improves Maximal Performance of Trained Cyclists after an Exhausting Endurance Trial: A Randomised Controlled Trial. Nutrients 2019, 11, 721. [Google Scholar] [CrossRef] [PubMed]
- Gil-Antuñano, N.P.; Bonafonte, L.F.; Marqueta, P.M.; González, B.M.; Villegas García, J.A. Consenso sobre bebidas para el deportista. composición y pautas de reposición de líquidos-Documento de consenso de la federación Española de medicina del deporte. Arch. Med. Del. Deport 2008, 25, 245–258. [Google Scholar]
- Luque-Casado, A.; Perales, J.C.; Cárdenas, D.; Sanabria, D.; Vélez, D.C. Heart rate variability and cognitive processing: The autonomic response to task demands. Biol. Psychol. 2016, 113, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Owen, A.M.; McMillan, K.M.; Laird, A.R.; Bullmore, E.; Bullmore, E.T. N-back working memory paradigm: A meta-analysis of normative functional neuroimaging studies. Hum. Brain Mapp. 2005, 25, 46–59. [Google Scholar] [CrossRef]
- Eston, R.G. Use of Ratings of Perceived Exertion in Sports. Int. J. Sports Physiol. Perform. 2012, 7, 175–182. [Google Scholar] [CrossRef]
- Chen, M.; Fan, X.; Moe, S.T. Criterion-related validity of the Borg ratings of perceived exertion scale in healthy individuals: A meta-analysis. J. Sports Sci. 2002, 20, 873–899. [Google Scholar] [CrossRef]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. International Affective Picture System (IAPS): Affective Ratings of Pictures and Instruction Manual. Technical Report A-8; University of Florida: Gainesville, FL, USA, 2008. [Google Scholar]
- Stanislaw, H.; Todorov, N. Calculation of signal detection theory measures. Behav. Res. Methods Instrum. Comput. 1999, 31, 137–149. [Google Scholar] [CrossRef]
- Castell, G.S.; Serra-Majem, L.; Ribas-Barba, L. What and how much do we eat? 24-hour dietary recall method. Nutr. Hosp. 2015, 31 (Suppl. S3), 46–48. [Google Scholar] [CrossRef]
- Oliveira, B.R.R.; Slama, F.A.; Deslandes, A.; Furtado, E.S.; Santos, T.M. Continuous and High-Intensity Interval Training: Which Promotes Higher Pleasure? PLoS ONE 2013, 8, e79965. [Google Scholar] [CrossRef] [PubMed]
- Crewe, H.; Tucker, R.; Noakes, T.D. The rate of increase in rating of perceived exertion predicts the duration of exercise to fatigue at a fixed power output in different environmental conditions. Graefe’s Arch. Clin. Exp. Ophthalmol. 2008, 103, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Ekkekakis, P.; Hall, E.E.; Petruzzello, S.J. Variation and homogeneity in affective responses to physical activity of varying intensities: An alternative perspective on dose–response based on evolutionary considerations. J. Sports Sci. 2005, 23, 477–500. [Google Scholar] [CrossRef] [PubMed]
- Brick, N.E.; Campbell, M.; Metcalfe, R.S.; Mair, J.L.; MacIntyre, T. Altering Pace Control and Pace Regulation. Med. Sci. Sports Exerc. 2016, 48, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Lucía, A.; Juan, A.F.S.; Montilla, M.; Canete, S.; Santalla, A.; Earnest, C.P.; Perez, M. In Professional Road Cyclists, Low Pedaling Cadences Are Less Efficient. Med. Sci. Sports Exerc. 2004, 36, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Vercruyssen, F.; Suriano, R.; Bishop, D.J.; Hausswirth, C.; Brisswalter, J. Cadence selection affects metabolic responses during cycling and subsequent running time to fatigue. Br. J. Sports Med. 2005, 39, 267–272. [Google Scholar] [CrossRef]
- Ludyga, S.; Hottenrott, K.; Gronwald, T. Effects of different training loads and environmental conditions on EEG activity. Dtsch. Z. Sportmed. 2015, 66, 113–120. [Google Scholar] [CrossRef]
- Boksem, M.A.; Meijman, T.F.; Lorist, M.M. Effects of mental fatigue on attention: An ERP study. Cogn. Brain Res. 2005, 25, 107–116. [Google Scholar] [CrossRef]
- Siegenthaler, E.; Costela, F.M.; McCamy, M.B.; Di Stasi, L.L.; Otero-Millan, J.; Sonderegger, A.; Groner, R.; Macknik, S.L.; Martinez-Conde, S. Task difficulty in mental arithmetic affects microsaccadic rates and magnitudes. Eur. J. Neurosci. 2013, 39, 287–294. [Google Scholar] [CrossRef]
- Nibbeling, N.; Oudejans, R.R.D.; Daanen, H.A. Effects of anxiety, a cognitive secondary task, and expertise on gaze behavior and performance in a far aiming task. Psychol. Sport Exerc. 2012, 13, 427–435. [Google Scholar] [CrossRef]
- Williams, A.M.; Vickers, J.; Rodrigues, S.T. The Effects of Anxiety on Visual Search, Movement Kinematics, and Performance in Table Tennis: A Test of Eysenck and Calvo’s Processing Efficiency Theory. J. Sport Exerc. Psychol. 2002, 24, 438–455. [Google Scholar] [CrossRef]
- Yang, Q.; Pourtois, G. Conflict-driven adaptive control is enhanced by integral negative emotion on a short time scale. Cogn. Emot. 2018, 32, 1637–1653. [Google Scholar] [CrossRef] [PubMed]
- Dignath, D.; Wirth, R.; Kühnhausen, J.; Gawrilow, C.; Kunde, W.; Kiesel, A. Motivation drives conflict adaptation. Motiv. Sci. 2020, 6, 84–89. [Google Scholar] [CrossRef]
- Kanske, P. On the Influence of Emotion on Conflict Processing. Front. Integr. Neurosci. 2012, 6, 42. [Google Scholar] [CrossRef]
- Ekkekakis, P.; Brand, R. Affective responses to and automatic affective valuations of physical activity: Fifty years of progress on the seminal question in exercise psychology. Psychol. Sport Exerc. 2019, 42, 130–137. [Google Scholar] [CrossRef]
- Fredrickson, B.L.; Kahneman, D. Duration Neglect in Retrospective Evaluations of Affective Episodes. J. Pers. Soc. Psychol. 1993, 65, 45–55. [Google Scholar] [CrossRef]
- Pashler, H.; Johnston, J.C. Attentional limitations in dual-task performance. In Attention; Pashler, H., Ed.; Taylor and Francis: Oxfordshore, UK, 1998; pp. 155–189. [Google Scholar]
- Renfree, A.; West, J.; Corbett, M.; Rhoden, C.; Gibson, A.S.C. Complex Interplay Between Determinants of Pacing and Performance During 20-km Cycle Time Trials. Int. J. Sports Physiol. Perform. 2012, 7, 121–129. [Google Scholar] [CrossRef]
- Audiffren, M.; André, N. The exercise–Cognition relationship: A virtuous circle. J. Sport Health Sci. 2019, 8, 339–347. [Google Scholar] [CrossRef]
- Holroyd, C.B.; Coles, M.G.H. The neural basis of human error processing: Reinforcement learning, dopamine, and the error-related negativity. Psychol. Rev. 2002, 109, 679–709. [Google Scholar] [CrossRef]
- Umemoto, A.; Inzlicht, M.; Holroyd, C.B. Electrophysiological indices of anterior cingulate cortex function reveal changing levels of cognitive effort and reward valuation that sustain task performance. Neuropsychologia 2019, 123, 67–76. [Google Scholar] [CrossRef]
- Dreisbach, G.; Fischer, R. Conflicts as aversive signals. Brain Cogn. 2012, 78, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Schouppe, N.; Braem, S.; De Houwer, J.; Silvetti, M.; Verguts, T.; Ridderinkhof, K.R.; Notebaert, W. No pain, no gain: The affective valence of congruency conditions changes following a successful response. Cogn. Affect. Behav. Neurosci. 2014, 15, 251–261. [Google Scholar] [CrossRef] [PubMed]

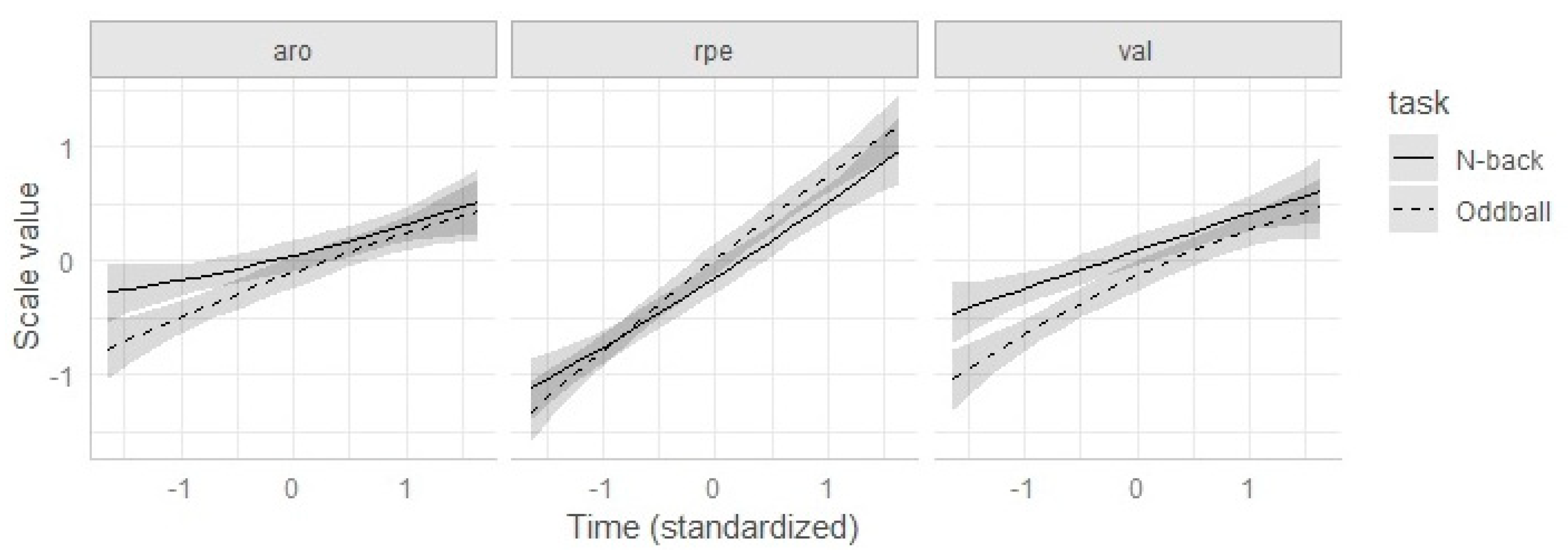
| Model | Effects | AIC | df | c2 | p |
|---|---|---|---|---|---|
| Saturated | Task, Measure, Time in task (and interactions) * | 4845.5 | 47 | ||
| Model 1 ** | Saturated minus Task*Measure × Time in task | 4843.0 | 43 | 5.48 | 0.241 (M1 > Saturated) |
| Model 2 | Model 1 minus Task*Measure | 4869.6 | 41 | 30.56 | <0.001 (M1 > M2) |
| Predictors | Estimates | CI | p |
|---|---|---|---|
| Intercept | −2.92 | −3.94–−1.89 | <0.001 |
| Time (linear) | −41.60 | −68.88–−14.33 | 0.003 |
| Time (quadratic) | −45.20 | −68.66–−21.74 | <0.001 |
| Measure (C1) | −0.98 | −1.53–−0.43 | <0.001 |
| Measure (C2) | −0.93 | −1.25–−0.62 | <0.001 |
| Task | −0.13 | −0.19–−0.07 | <0.001 |
| Time (linear)*Measure (C1) | −53.54 | −84.59–−22.49 | 0.001 |
| Time (quadratic)*Measure (C1) | −40.45 | −67.45–−13.46 | 0.003 |
| Time (linear)*Measure (C2) | 2.14 | −15.78–20.06 | 0.815 |
| Time (quadratic)*Measure (C2) | 27.33 | 11.75–42.90 | 0.001 |
| Time (linear)*Task | 6.28 | 3.48–9.09 | <0.001 |
| Time (quadratic)*Task | −2.30 | −5.08–0.47 | 0.104 |
| Measure (C1)*Task | −0.04 | −0.11–0.03 | 0.316 |
| Measure (C2)*Task | 0.11 | 0.07–0.15 | <0.001 |
| Random Effects | |||
| σ2 | 0.47 | ||
| τ00participant | 0.48 | ||
| ICC | 0.51 | ||
| Predictors | Arousal | Valence | RPE | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimates | CI | p | Estimates | CI | p | Estimates | CI | p | |
| Intercept | −1.88 | −3.38–−0.37 | 0.015 | −2.00 | −3.50–−0.50 | 0.009 | −4.80 | −6.43–−3.17 | <0.001 |
| Time (linear) | 14.22 | −9.73–38.17 | 0.245 | −61.22 | −85.30–−37.14 | <0.001 | −25.72 | −52.64–1.20 | 0.061 |
| Time (quadratic) | −15.05 | −35.66–5.56 | 0.152 | −52.93 | −73.65–−32.21 | <0.001 | −10.64 | −33.81–12.53 | 0.368 |
| Task | −0.22 | −0.31–−0.13 | <0.001 | −0.27 | −0.36–−0.18 | <0.001 | 0.10 | −0.00–0.20 | 0.054 |
| Time (linear)*Task | 6.12 | 3.65–8.58 | <0.001 | 1.87 | −0.61–4.35 | 0.140 | 2.83 | 0.05–5.61 | 0.046 |
| Time (quadratic)*Task | −1.07 | −3.51–1.37 | 0.391 | −1.98 | −4.44–0.47 | 0.113 | −0.84 | −3.59–1.91 | 0.548 |
| Random Effects | |||||||||
| σ2 | 0.36 | 0.36 | 0.46 | ||||||
| τ00participant | 0.63 | 0.57 | 0.46 | ||||||
| ICC | 0.64 | 0.61 | 0.50 | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ávila-Gandía, V.; Alarcón, F.; Perales, J.C.; López-Román, F.J.; Luque-Rubia, A.J.; Cárdenas, D. Dissociable Effects of Executive Load on Perceived Exertion and Emotional Valence during Submaximal Cycling. Int. J. Environ. Res. Public Health 2020, 17, 5576. https://doi.org/10.3390/ijerph17155576
Ávila-Gandía V, Alarcón F, Perales JC, López-Román FJ, Luque-Rubia AJ, Cárdenas D. Dissociable Effects of Executive Load on Perceived Exertion and Emotional Valence during Submaximal Cycling. International Journal of Environmental Research and Public Health. 2020; 17(15):5576. https://doi.org/10.3390/ijerph17155576
Chicago/Turabian StyleÁvila-Gandía, Vicente, Francisco Alarcón, José C. Perales, F. Javier López-Román, Antonio J. Luque-Rubia, and David Cárdenas. 2020. "Dissociable Effects of Executive Load on Perceived Exertion and Emotional Valence during Submaximal Cycling" International Journal of Environmental Research and Public Health 17, no. 15: 5576. https://doi.org/10.3390/ijerph17155576
APA StyleÁvila-Gandía, V., Alarcón, F., Perales, J. C., López-Román, F. J., Luque-Rubia, A. J., & Cárdenas, D. (2020). Dissociable Effects of Executive Load on Perceived Exertion and Emotional Valence during Submaximal Cycling. International Journal of Environmental Research and Public Health, 17(15), 5576. https://doi.org/10.3390/ijerph17155576





