Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic
Abstract
1. Introduction
2. Materials and Methods
2.1. Wastewater Sampling
2.2. Sample Concentration and RNA Extraction
2.3. Process Control Virus
2.4. RT-qPCR Analysis
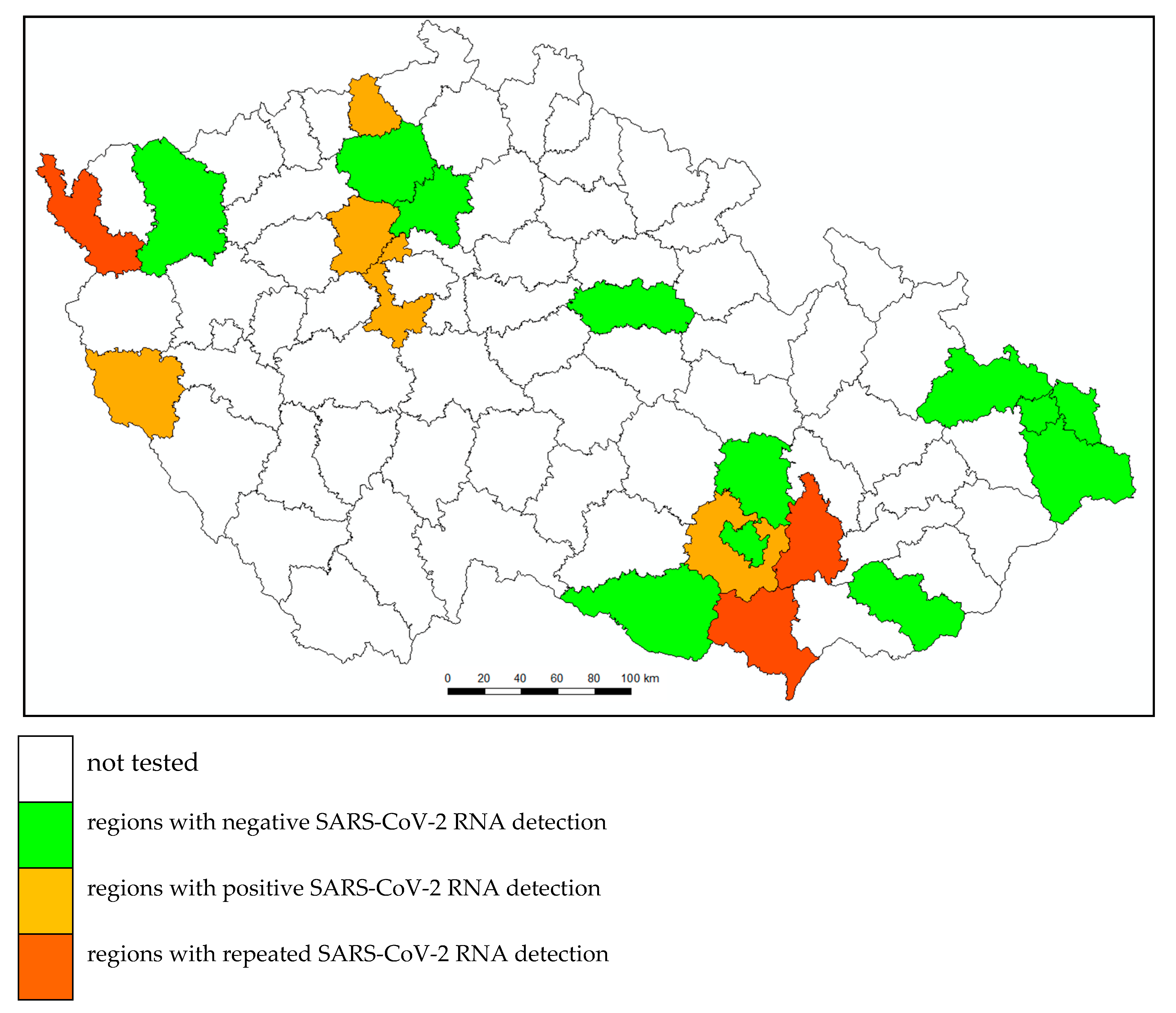
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Drexler, J.F.; Corman, M.V.; Drosten, C. Ecology, evolution and classification of bat coronaviruses in the aftermath of SARS. Antivir. Res. 2014, 101, 45–56. [Google Scholar] [CrossRef]
- Chan, J.F.W.; Lau, S.K.P.; To, K.K.W.; Cheng, V.C.C.; Woo, P.C.Y.; Yuen, K.Y. Middle east respiratory syndrome coronavirus: Another zoonotic betacoronavirus casuing SARS-like disease. Clin. Microbiol. Rev. 2015, 28, 465–522. [Google Scholar] [CrossRef]
- Chan, P.K.; Chan, M.C. Tracing the SARS-coronavirus. J. Thorac. Dis. 2013, 5, S118–S121. [Google Scholar] [CrossRef]
- Ding, Y.; He, L.; Zhang, Q.; Huang, Z.; Che, X.; Hou, J.; Wang, H.; Shen, H.; Qiu, L.; Li, Z.; et al. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: Implications for pathogenesis and virus transmission pathways. J. Pathol. 2004, 203, 622–630. [Google Scholar] [CrossRef]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.K.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, genetic, recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016, 24, 490–502. [Google Scholar] [CrossRef] [PubMed]
- De Wit, E.; Van Doremalen, N.; Falzarano, D.; Munster, V.J. SARS and MERS: Recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016, 14, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Li, F.; Shi, Z.L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019, 17, 181–192. [Google Scholar] [CrossRef]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.A.; Hui, K.P.Y.; Yen, H.-L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe. 2020, 1, e10. [Google Scholar] [CrossRef]
- Corman, V.M.; Albarrak, A.M.; Omrani, A.S.; Albarrak, M.M.; Farah, M.E.; Almasri, M.; Muth, D.; Sieberg, A.; Meyer, B.; Assiri, A.M.; et al. Viral shedding and antibody response in 37 patients with Middle East Respiratory Syndrome coronavirus infection. Clin. Infect. Dis. 2016, 62, 477–483. [Google Scholar] [CrossRef]
- Miri, S.M.; Roozbeh, F.; Omranirad, A.; Alavian, S.M. Panic of Buying Toilet Papers: A Historical Memory or a horrible truth? Systematic review of gastrointestinal manifestations of COVID-19. Hepat. Mon. 2020, 20, e102729. [Google Scholar] [CrossRef]
- Zhang, T.; Cui, X.; Zhao, X.; Wang, J.; Zheng, J.; Zheng, G.; Guo, W.; Cai, C.; He, S.; Xu, Y. Detectable SARS-CoV-2 viral RNA in feces of three children during recovery period of COVID-19 pneumonia. J. Med. Virol. 2020, 92, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Lo, I.L.; Lio, C.F.; Cheong, H.H.; Lei, C.I.; Cheong, T.H.; Zhong, X.; Tian, Y.; Sin, N.N. Evaluation of SARS-CoV-2 RNA shedding in clinical specimens and clinical characteristics of 10 patients with COVID-19 in Macau. Int. J. Biol. Sci. 2020, 16, 1698–1707. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Rong, L.; Nian, W.; He, Y. Review article: Gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment. Pharmacol. Ther. 2020, 51, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Hindson, J. COVID-19, Faecal-oral transmission? Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 259. [Google Scholar] [CrossRef]
- Yeo, C.; Kaushal, S.; Yeo, D. Enteric involvement of coronaviruses: Is faecal-oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol. Hepatol. 2020, 5, 335–337. [Google Scholar] [CrossRef]
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Li, X.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833. [Google Scholar] [CrossRef]
- Heller, L.; Mota, C.R.; Greco, D.B. COVID-19 faecal-oral transmission: Are we asking the right questions? Sci. Total Environ. 2020, 729, 138919–138922. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, Y.; Jiang, N.; Zhou, Q.; Ma, W.L. Persistence of intestinal SARS-CoV-2 infection in patients with COVID-19 leads to re-admission after pneumonia resolved. Int. J. Infect. Dis. 2020, 95, 433–435. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Zhu, B.; Liang, H.; Fang, C.; Gong, Y.; Guo, Q.; Sun, X.; Zhao, D.; Shen, J.; et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020, 26, 502–505. [Google Scholar] [CrossRef]
- Cao, Q.; Chen, Y.C.; Chen, C.L.; Chiu, C.H. SARS-CoV-2 infection in children: Transmission dynamics and clinical characteristics. J. Formos. Med. Assoc. 2020, 119, 670–673. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Sun, J.; Zhu, A.; Li, H.; Zheng, K.; Zhuang, Z.; Chen, Z.; Shi, Y.; Zhang, Z.; Chen, S.; Liu, X.; et al. Isolation of infectious SARS-CoV-2 from urine of a COVID-19 patient. Emerg. Microb. Infect. 2020, 9, 991–993. [Google Scholar] [CrossRef] [PubMed]
- Holshue, M.L.; DeBolt, C.; Lindquist, S.; Lofy, K.H.; Wiesman, J.; Bruce, H.; Spitters, C.; Ericson, K.; Wilkerson, S.; Tural, A.; et al. First case of 2019 novel coronavirus in the United States. N. Engl. J. Med. 2020, 382, 929–936. [Google Scholar] [CrossRef]
- Medema, G.; Heijnen, L.; Elsinga, G.; Italiaander, R.; Brouwer, A. Presence of SARS-Cronavirus-2 in sewage. Environ. Sci. Technol. Lett. 2020. [Google Scholar] [CrossRef]
- Quilliam, R.S.; Weidmann, M.; Moresco, V.; Purshouse, H.; O’Hara, Z.; Oliver, D.M. COVID-19, The environmental implications of shedding SARS-CoV-2 in human faeces. Environ. Int. 2020, 140, 105790. [Google Scholar] [CrossRef]
- Kitajima, M.; Ahmed, W.; Bibby, K.; Carducci, A.; Gerba, C.P.; Hamilton, K.A.; Haramoto, E.; Rose, J.B. SARS-CoV-2 in wastewater: State of the knowledge and research needs. Sci. Total Environ. 2020, 739, 139076. [Google Scholar] [CrossRef]
- Amirian, E.S. Potential fecal transmission of SARS-CoV-2: Current evidence and implications for public health. Int. J. Infect. Dis. 2020, 95, 363–370. [Google Scholar] [CrossRef]
- Ahmed, W.; Angel, N.; Edson, J.; Bibby, K.; Bivins, A.; O’Brien, J.W.; Choi, P.M.; Kitajima, M.; Simpson, S.L.; Li, J.; et al. First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: A proof of concept for the wastewater surveillance of COVID-19 in the community. Sci. Total Environ. 2020, 728, 138764. [Google Scholar] [CrossRef]
- Hata, A.; Honda, R. Potential sensitivity of wastewater monitoring for SARS-CoV-2: Comparison with Norovirus cases. Environ. Sci. Technol. 2020, 54, 6451–6452. [Google Scholar] [CrossRef]
- La Rosa, G.; Iaconelli, M.; Mancini, P.; Bonanno Ferraro, G.; Veneri, C.; Bonadonna, L.; Lucentini, L.; Suffredini, E. First detection of SARS-COV-2 in untreated wastewaters in Italy. Sci. Total Environ. 2020, 736, 139652. [Google Scholar] [CrossRef]
- Lodder, W.; Husman, A.M.D. SARS-CoV-2 in wastewater: Potential health risk, but also source data. Lancet Gastroenterol. 2020, 5, 533–534. [Google Scholar] [CrossRef]
- Randazzo, W.; Truchado, P.; Cuevas-Ferrando, E.; Simón, P.; Allende, A.; Sánchez, G. SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res. 2020, 181, 115942. [Google Scholar] [CrossRef] [PubMed]
- Sodre, F.F.; Brandao, C.C.S.; Vizzotto, C.S.; Maldaner, A.O. Wastewater-based epidemiology as a strategy for community monitoring, mapping of hotspots and early warning systems of Covid-19. Quim. Nova 2020, 43, 515–519. [Google Scholar] [CrossRef]
- Mlejnková, H.; Očenášková, V.; Sovová, K.; Vašíčková, P.; Juranová, E. Koronavirus SARS-CoV-2 v povrchových a odpadních vodách (Coronavirus SARS-CoV-2 in surface and wastewater). VTEI 2020, 2, 28–32. [Google Scholar] [CrossRef]
- La Rosa, G.; Mancini, P.; Bonanno Ferraro, G.; Veneri, C.; Iaconelli, M.; Bonadonna, L.; Lucentini, L.; Suffredini, E. SARS-CoV-2 has been circulating in northern Italy since December 2019: Evidence from environmental monitoring. medRxiv 2020. [Google Scholar] [CrossRef]
- Očenášková, V. Komunální odpadní voda jako diagnostické médium (Municipal wastewater as a diagnostic medium). VTEI 2018, 60, 28–30. [Google Scholar]
- Gracia-Lor, E.; Castiglioni, S.; Bade, R.; Been, F.; Castrignanò, E.; Covaci, A.; González-Mariño, I.; Hapeshi, E.; Kasprzyk-Hordern, B.; Kinyua, J.; et al. Measuring biomarkers in wastewater as a new source of epidemiological information: Current state and future perspectives. Environ. Int. 2017, 99, 131–150. [Google Scholar] [CrossRef]
- Cacace, D.; Fatta-Kassinos, D.; Manaia, C.M.; Cytryn, E.; Kreuzinger, N.; Rizzo, L.; Karaolia, P.; Schwartz, T.; Alexander, J.; Merlin, C.; et al. Antibiotic resistance genes in treated wastewater and in the receiving water bodies: A pan-European survey of urban settings. Water Res. 2019, 162, 320–330. [Google Scholar] [CrossRef]
- Daughton, C.G. Wasterwater surveillance for population—Wide Covid-19: The present and future. Sci. Total Environ. 2020, 736, 139631. [Google Scholar] [CrossRef]
- Mao, K.; Zhang, K.; Du, W.; Ali, W.; Feng, X.; Zhang, H. The potential of wastewater-based epidemiology as surveillance and early warning of infectious disease outbreaks. Curr. Opin. Environ. Sci. Health 2020, 17, 1–7. [Google Scholar] [CrossRef]
- Orive, G.; Lertxundi, U.; Barcelo, D. Early SARS-CoV-2 outbreak detection by sewage-based epidemiology. Sci. Total Environ. 2020, 732, 139298. [Google Scholar] [CrossRef] [PubMed]
- Barcelo, D. An environmental and health perspective for COVID-19 outbreak: Meteorology and air quality influence, sewage epidemiology indicator, hospitals disinfection, drug therapies and recommendations. J. Environ. Chem. Eng. 2020, 8, 104006. [Google Scholar] [CrossRef] [PubMed]
- Hart, O.E.; Halden, R.U. Computational analysis of SARS-CoV-2/COVID-19 surveillance by wastewater-based epidemiology locally and globally: Feasibility, economy, opportunities and challenges. Sci. Total Environ. 2020, 730, 138875. [Google Scholar] [CrossRef] [PubMed]
- Sims, N.; Kasprzyk-Hordern, B. Future perspectives of wastewater-based epidemiology: Monitoring infectious disease spread and resistance to the community level. Environ. Int. 2020, 139, 105689. [Google Scholar] [CrossRef]
- Calgua, B.; Rodriguez-Manzano, J.; Hundesa, A.; Suñen, E.; Calvo, M.; Bofill-Mas, S.; Girones, R. New methods for the concentration of viruses from urban sewage using quantitative PCR. J. Virol. Methods 2013, 187, 215–221. [Google Scholar] [CrossRef]
- ISO 15216-2: 2019 Microbiology of the Food Chain—Horizontal Method for Determination of Hepatitis A Virus and Norovirus Using Real-Time RT-PCR—Part 2: Method for Qualitative Detection; Organization for Standardization: Geneva, Switzerland, 2019.
- Vemulapalli, R.; Gulani, J.; Santrich, C. A real-time TaqMan RT-PCR assay with an internal amplification control for rapid detection of transmissible gastroenteritis virus in swine fecal samples. J. Virol. Methods 2009, 162, 231–235. [Google Scholar] [CrossRef]
- Vasickova, P.; Kralik, P.; Slana, I.; Pavlik, I. Optimisation of a triplex real time RT-PCR for detection of hepatitis E virus RNA and validation on biological samples. J. Virol. Methods 2012, 180, 38–42. [Google Scholar] [CrossRef]
- Mikel, P.; Vasickova, P.; Tesarik, R.; Malenovska, H.; Kulich, P.; Vesely, T.; Kralik, P. Preparation of MS2 phage-like particles and their use as potential process control viruses for detection and quantification of enteric RNA viruses in different matrices. Front. Microbiol. 2016, 7, 1911. [Google Scholar] [CrossRef]
- Ye, Y.; Ellenberg, R.M.; Graham, K.E.; Wigginton, K.R. Survivability, partitioning, and recovery of enveloped viruses in untreated municipal wastewater. Environ. Sci. Technol. 2016, 50, 5077–5085. [Google Scholar] [CrossRef]
- Casanova, L.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Survival of surrogate coronaviruses in water. Water Res. 2009, 43, 1893–1898. [Google Scholar] [CrossRef]
| WWTP Capacity According to the Population Served (Number of Inhabitants) | Number of WWTPs Involved in the Study |
|---|---|
| <2000 | 2 |
| 2000–10,000 | 7 |
| 10,000–100,000 | 21 |
| >100,000 | 3 |
| WWTP Code | Size of WWTP | Week 17 | Week 18 | Week 19 | Week 20 | Week 21 | Week 22 | Week 23 | Week 24 | Week 25 |
|---|---|---|---|---|---|---|---|---|---|---|
| B1 | L | −28.06% | −23.52% | −33.95% | −39.59% | −33.97% | −31.27% | −32.76% | ||
| B2 | M | −26.25% | +(36.53) * 25.50% | +(36.36) * 25.28% | −35.51% | −42.95% | +(38.77) 36.75% | −39.79% | −39.48% | −43.51% |
| B3 | M | −43.22% | −46.72% | |||||||
| B4 | M | −46.92% | −33.07% | |||||||
| B5 | S | +(39.62) 27.75% | +(34.40) * 24.17% | −33.10% | −42.23% | −31.58% | ||||
| C1 | S | −31.29% | −24.16% | |||||||
| C2-A | M | −41.38% | −43.15% | |||||||
| C2-B | −47.95% | −40.27% | ||||||||
| C2-E | −46.90% | −23.06% | ||||||||
| C2-F | −30.12% | −47.29% | ||||||||
| D1 | S | −32.20% | −26.92% | |||||||
| D2 | M | −29.49% | +(38.84) * 23.27% | −29.82% | ||||||
| F | M | −28.20% | −37.01% | |||||||
| H1 | S | −44.76% | +(37.19) 37.09% | −39.44% | −46.59% | −31.95% | −26.63% | −26.36% | ||
| I | M | −48.57% | −42.40% | −28.96% | ||||||
| K | M | −45.84% | −46.50% | |||||||
| K1 | M | −30.91% | −39.92% | −27.36% | −34.46% | −44.94% | −23.71% | −46.84% | ||
| L1 | M | −26.63% | −31.48% | −22.90% | −31.30% | |||||
| M1 | M | +(36.59) 42.86% | −46.01% | +(39.35) 33.53% | −31.82% | |||||
| M2 | M | −41.92% | ||||||||
| N1 | XS | −37.20% | −24.92% | |||||||
| O | M | −36.78% | −25.42% | |||||||
| OV | L | −29.64% | −26.10% | |||||||
| P1 | XS | −35.13% | +(34.65) 44.05% | −39.52% | ||||||
| P2-A | L | −30.25% | −34.68% | |||||||
| P2-B | −44.59% | |||||||||
| R1 | S | −29.08% | −26.92% | −22.49% | ||||||
| S | S | +(38.55) 33.40% | −46.83% | −43.27% | −25.95% | −44.14% | ||||
| T | M | −23.45% | +(34.97) 36.93% | −30.50% | −33.10% | −39.95% | ||||
| T1 | M | −24.85% | −40.09% | −44.82% | ||||||
| U | M | −46.58% | −46.69% | |||||||
| U1 | M | −47.82% | −46.59% | |||||||
| UB | M | −35.75% | −36.88% | |||||||
| V | M | −34.75% | ||||||||
| V1 | M | +(39.86) 46.68% | −45.51% | |||||||
| Z | S | −28.77% | −25.07% | −28.91% | ||||||
| Z1 | M | −40.34% | −44.84% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mlejnkova, H.; Sovova, K.; Vasickova, P.; Ocenaskova, V.; Jasikova, L.; Juranova, E. Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic. Int. J. Environ. Res. Public Health 2020, 17, 5508. https://doi.org/10.3390/ijerph17155508
Mlejnkova H, Sovova K, Vasickova P, Ocenaskova V, Jasikova L, Juranova E. Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic. International Journal of Environmental Research and Public Health. 2020; 17(15):5508. https://doi.org/10.3390/ijerph17155508
Chicago/Turabian StyleMlejnkova, Hana, Katerina Sovova, Petra Vasickova, Vera Ocenaskova, Lucie Jasikova, and Eva Juranova. 2020. "Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic" International Journal of Environmental Research and Public Health 17, no. 15: 5508. https://doi.org/10.3390/ijerph17155508
APA StyleMlejnkova, H., Sovova, K., Vasickova, P., Ocenaskova, V., Jasikova, L., & Juranova, E. (2020). Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic. International Journal of Environmental Research and Public Health, 17(15), 5508. https://doi.org/10.3390/ijerph17155508






