Reduced Gray Matter Volume and Risk of Falls in Parkinson’s Disease with Dementia Patients: A Voxel-Based Morphometry Study
Abstract
1. Background
2. Materials and Methods
2.1. Subjects
2.2. Assessment of Clinical Disease Severity
2.3. Neuropsychological Tests (NPT)
2.4. MRI Image Acquisition
2.5. Voxel-Based Morphometry Processing
2.6. Statistical Analysis
2.6.1. Analysis of Demographic Data between Groups
2.6.2. Relationships among Disease Severity, Cognition Function, and Gray Matter Volume
3. Results
3.1. Clinical Characteristics, NPT, and Disease Severity among Groups
3.2. Regional Gray Matter Volume (GMV) Aberrations among Groups
3.2.1. Comparison between all PDD Patients and Normal Controls
3.2.2. Comparison between PDD without Fall and Control Groups, and between PDD with Fall and Control Groups
3.2.3. Comparison between the PDD with Fall and without Fall Groups
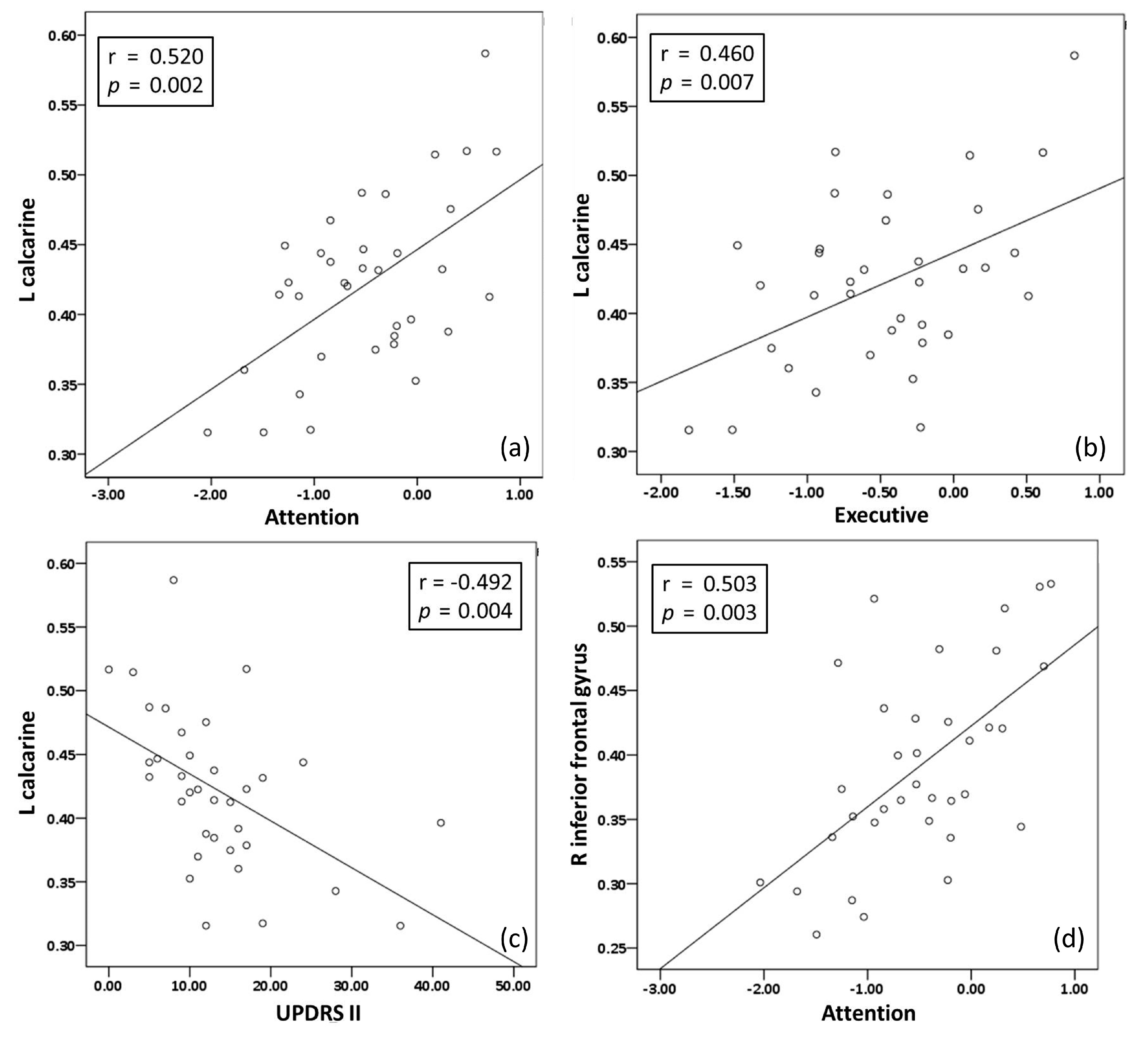
3.3. Relationship among Disease Severity Profiles, NPT, and Extracted Regional GMV from PDD Patients
4. Discussion
4.1. Brief Summary and Potential Implications
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lees, A.J.; Hardy, J.; Revesz, T. Parkinson’s disease. Lancet 2009, 373, 2055–2066. [Google Scholar] [CrossRef]
- Wielinski, C.L.; Erickson-Davis, C.; Wichmann, R.; Walde-Douglas, M.; Parashos, S.A. Falls and injuries resulting from falls among patients with Parkinson’s disease and other parkinsonian syndromes. Mov. Disord. Off. J. Mov. Disord. Soc. 2005, 20, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Vellas, B.J.; Wayne, S.J.; Romero, L.J.; Baumgartner, R.N.; Garry, P.J. Fear of falling and restriction of mobility in elderly fallers. Age Ageing 1997, 26, 189–193. [Google Scholar] [CrossRef] [PubMed]
- DeLong, M.R.; Wichmann, T. Circuits and circuit disorders of the basal ganglia. Arch. Neurol. 2007, 64, 20–24. [Google Scholar] [CrossRef]
- Tang, C.C.; Poston, K.L.; Dhawan, V.; Eidelberg, D. Abnormalities in metabolic network activity precede the onset of motor symptoms in Parkinson’s disease. J. Neurosci. 2010, 30, 1049–1056. [Google Scholar] [CrossRef]
- Albin, R.L.; Koeppe, R.A.; Chervin, R.D.; Consens, F.B.; Wernette, K.; Frey, K.A.; Aldrich, M.S. Decreased striatal dopaminergic innervation in REM sleep behavior disorder. Neurology 2000, 55, 1410–1412. [Google Scholar] [CrossRef]
- Eisensehr, I.; Linke, R.; Noachtar, S.; Schwarz, J.; Gildehaus, F.J.; Tatsch, K. Reduced striatal dopamine transporters in idiopathic rapid eye movement sleep behaviour disorder. Comparison with Parkinson’s disease and controls. Brain 2000, 123, 1155–1160. [Google Scholar] [CrossRef]
- Sommer, U.; Hummel, T.; Cormann, K.; Mueller, A.; Frasnelli, J.; Kropp, J.; Reichmann, H. Detection of presymptomatic Parkinson’s disease: Combining smell tests, transcranial sonography, and SPECT. Mov. Disord. Off. J. Mov. Disord. Soc. 2004, 19, 1196–1202. [Google Scholar] [CrossRef]
- Lee, C.C.; Yo, C.H.; Lee, M.G.; Tsai, K.C.; Lee, S.H.; Chen, Y.S.; Lee, W.C.; Hsu, T.C.; Lee, S.H.; Chang, S.S. Adult sepsis—A nationwide study of trends and outcomes in a population of 23 million people. J. Infect. 2017, 75, 409–419. [Google Scholar] [CrossRef]
- Chiang, P.L.; Chen, H.L.; Lu, C.H.; Chen, P.C.; Chen, M.H.; Yang, I.H.; Tsai, N.W.; Lin, W.C. White matter damage and systemic inflammation in Parkinson’s disease. BMC Neurosci. 2017, 18, 48. [Google Scholar] [CrossRef]
- Antonini, A.; DeNotaris, R. PET and SPECT functional imaging in Parkinson’s disease. Sleep Med 2004, 5, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, T.P.; Malykhin, N.; Martin, W.R.; Hanstock, C.C.; Emery, D.J.; Fisher, N.J.; Camicioli, R.M. Age and dementia-associated atrophy predominates in the hippocampal head and amygdala in Parkinson’s disease. Neurobiol. Aging 2008, 29, 1027–1039. [Google Scholar] [CrossRef] [PubMed]
- Camicioli, R.; Moore, M.M.; Kinney, A.; Corbridge, E.; Glassberg, K.; Kaye, J.A. Parkinson’s disease is associated with hippocampal atrophy. Mov. Disord. Off. J. Mov. Disord. Soc. 2003, 18, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Junque, C.; Ramirez-Ruiz, B.; Tolosa, E.; Summerfield, C.; Marti, M.J.; Pastor, P.; Gomez-Anson, B.; Mercader, J.M. Amygdalar and hippocampal MRI volumetric reductions in Parkinson’s disease with dementia. Mov. Disord. Off. J. Mov. Disord. Soc. 2005, 20, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Laakso, M.P.; Partanen, K.; Riekkinen, P.; Lehtovirta, M.; Helkala, E.L.; Hallikainen, M.; Hanninen, T.; Vainio, P.; Soininen, H. Hippocampal volumes in Alzheimer’s disease, Parkinson’s disease with and without dementia, and in vascular dementia: An MRI study. Neurology 1996, 46, 678–681. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Yang, J.; Chen, K.; Luo, C.; Burgunder, J.; Gong, Q.; Shang, H. Resting-state fMRI reveals potential neural correlates of impaired cognition in Huntington’s disease. Parkinsonism Relat. Disord. 2016, 27, 41–46. [Google Scholar] [CrossRef]
- Guo, W.; Song, Y.; Liu, F.; Zhang, Z.; Zhang, J.; Yu, M.; Liu, J.; Xiao, C.; Liu, G.; Zhao, J. Dissociation of functional and anatomical brain abnormalities in unaffected siblings of schizophrenia patients. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2015, 126, 927–932. [Google Scholar] [CrossRef]
- O’connell, B.; Myers, H. The sensitivity and specificity of the Morse Fall Scale in an acute care setting. J. Clin. Nurs. 2002, 11, 134–136. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1998, 50, 318. [Google Scholar] [CrossRef]
- Martínez-Martín, P.; Gil-Nagel, A.; Gracia, L.M.; Gómez, J.B.; Martínez-Sarriés, J.; Bermejo, F. Unified Parkinson’s disease rating scale characteristics and structure. Mov. Disord. 1994, 9, 76–83. [Google Scholar] [CrossRef]
- Chen, H.-Y.; Hua, M.-S.; Zhu, J.-J.; Chen, Y.-H. Selection of Factor-Based WAIS-Ⅲ Tetrads in the Taiwan Standardization Sample: A Guide to Clinical Practice. Chin. J. Psychol. 2008, 50, 91–109. [Google Scholar]
- Teng, E.L.; Hasegawa, K.; Homma, A.; Imai, Y.; Larson, E.; Graves, A.; Sugimoto, K.; Yamaguchi, T.; Sasaki, H.; Chiu, D. The Cognitive Abilities Screening Instrument (CASI): A practical test for cross-cultural epidemiological studies of dementia. Int. Psychogeriatr. 1994, 6, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Bennett, D.A.; Schneider, J.A.; Buchman, A.S.; Mendes de Leon, C.; Bienias, J.L.; Wilson, R.S. The Rush Memory and Aging Project: Study design and baseline characteristics of the study cohort. Neuroepidemiology 2005, 25, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Barnes, L.L.; Bennett, D.A. Assessment of lifetime participation in cognitively stimulating activities. J. Clin. Exp. Neuropsychol. 2003, 25, 634–642. [Google Scholar] [CrossRef]
- Poewe, W.; Gauthier, S.; Aarsland, D.; Leverenz, J.B.; Barone, P.; Weintraub, D.; Tolosa, E.; Dubois, B. Diagnosis and management of Parkinson’s disease dementia. Int. J. Clin. Pract. 2008, 62, 1581–1587. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, H.-L.; Chen, P.-C.; Lu, C.-H.; Hsu, N.-W.; Chou, K.-H.; Lin, C.-P.; Wu, R.-W.; Li, S.-H.; Cheng, Y.-F.; Lin, W.-C. Structural and cognitive deficits in chronic carbon monoxide intoxication: A voxel-based morphometry study. BMC Neurol. 2013, 13, 129. [Google Scholar] [CrossRef]
- Mahieux, F.; Fenelon, G.; Flahault, A.; Manifacier, M.J.; Michelet, D.; Boller, F. Neuropsychological prediction of dementia in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1998, 64, 178–183. [Google Scholar] [CrossRef]
- Troster, A.I.; Woods, S.P.; Morgan, E.E. Assessing cognitive change in Parkinson’s disease: Development of practice effect-corrected reliable change indices. Arch. Clin. Neuropsychol. 2007, 22, 711–718. [Google Scholar] [CrossRef]
- Liepelt-Scarfone, I.; Graber, S.; Fruhmann Berger, M.; Feseker, A.; Baysal, G.; Csoti, I.; Godau, J.; Gaenslen, A.; Huber, H.; Srulijes, K.; et al. Cognitive profiles in Parkinson’s disease and their relation to dementia: A data-driven approach. Int. J. Alzheimers Dis. 2012, 2012, 910757. [Google Scholar] [CrossRef]
- Meireles, J.; Massano, J. Cognitive impairment and dementia in Parkinson’s disease: Clinical features, diagnosis, and management. Front. Neurol. 2012, 3, 88. [Google Scholar] [CrossRef]
- Camicioli, R.; Majumdar, S.R. Relationship between mild cognitive impairment and falls in older people with and without Parkinson’s disease: 1-Year Prospective Cohort Study. Gait Posture 2010, 32, 87–91. [Google Scholar] [CrossRef]
- Allcock, L.M.; Rowan, E.N.; Steen, I.N.; Wesnes, K.; Kenny, R.A.; Burn, D.J. Impaired attention predicts falling in Parkinson’s disease. Parkinsonism Relat. Disord. 2009, 15, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Domingos, J.M.; Godinho, C.; Dean, J.; Coelho, M.; Pinto, A.; Bloem, B.R.; Ferreira, J.J. Cognitive Impairment in Fall-Related Studies in Parkinson’s Disease. J. Parkinsons Dis. 2015, 5, 453–469. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.E.; Schwarzel, A.K.; Canning, C.G. Recurrent falls in Parkinson’s disease: A systematic review. Parkinsons Dis. 2013, 2013, 906274. [Google Scholar] [CrossRef] [PubMed]
- Matinolli, M.; Korpelainen, J.T.; Sotaniemi, K.A.; Myllylä, V.V.; Korpelainen, R. Recurrent falls and mortality in Parkinson’s disease: A prospective two-year follow-up study. Acta Neurol. Scand. 2011, 123, 193–200. [Google Scholar] [CrossRef]
- Ramirez-Ruiz, B.; Marti, M.J.; Tolosa, E.; Bartres-Faz, D.; Summerfield, C.; Salgado-Pineda, P.; Gomez-Anson, B.; Junque, C. Longitudinal evaluation of cerebral morphological changes in Parkinson’s disease with and without dementia. J. Neurol. 2005, 252, 1345–1352. [Google Scholar] [CrossRef]
- Burton, E.J.; McKeith, I.G.; Burn, D.J.; Williams, E.D.; O’Brien, J.T. Cerebral atrophy in Parkinson’s disease with and without dementia: A comparison with Alzheimer’s disease, dementia with Lewy bodies and controls. Brain 2004, 127, 791–800. [Google Scholar] [CrossRef]
- Ramirez-Ruiz, B.; Marti, M.J.; Tolosa, E.; Gimenez, M.; Bargallo, N.; Valldeoriola, F.; Junque, C. Cerebral atrophy in Parkinson’s disease patients with visual hallucinations. Eur. J. Neurol. 2007, 14, 750–756. [Google Scholar] [CrossRef]
- Beyer, M.K.; Janvin, C.C.; Larsen, J.P.; Aarsland, D. A magnetic resonance imaging study of patients with Parkinson’s disease with mild cognitive impairment and dementia using voxel-based morphometry. J. Neurol. Neurosurg. Psychiatry 2007, 78, 254–259. [Google Scholar] [CrossRef]
- Guo, T.; Guan, X.; Zeng, Q.; Xuan, M.; Gu, Q.; Huang, P.; Xu, X.; Zhang, M. Alterations of Brain Structural Network in Parkinson’s Disease With and Without Rapid Eye Movement Sleep Behavior Disorder. Front. Neurol. 2018, 9, 334. [Google Scholar] [CrossRef]
- Spindler, M.; Gooneratne, N.S.; Siderowf, A.; Duda, J.E.; Cantor, C.; Dahodwala, N. Daytime sleepiness is associated with falls in Parkinson’s disease. J. Parkinsons Dis. 2013, 3, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Hellendoorn, A.; Wijnroks, L.; van Daalen, E.; Dietz, C.; Buitelaar, J.K.; Leseman, P. Motor functioning, exploration, visuospatial cognition and language development in preschool children with autism. Res. Dev. Disabil. 2015, 39, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Richards, M.; Cote, L.J.; Stern, Y. The relationship between visuospatial ability and perceptual motor function in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1993, 56, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Dukart, J.; Sambataro, F.; Bertolino, A. Distinct Role of Striatal Functional Connectivity and Dopaminergic Loss in Parkinson’s Symptoms. Front. Aging Neurosci. 2017, 9, 151. [Google Scholar] [CrossRef]
- Stuart, S.; Galna, B.; Delicato, L.S.; Lord, S.; Rochester, L. Direct and indirect effects of attention and visual function on gait impairment in Parkinson’s disease: Influence of task and turning. Eur. J. Neurosci. 2017, 46, 1703–1716. [Google Scholar] [CrossRef]
- Hampshire, A.; Chamberlain, S.R.; Monti, M.M.; Duncan, J.; Owen, A.M. The role of the right inferior frontal gyrus: Inhibition and attentional control. Neuroimage 2010, 50, 1313–1319. [Google Scholar] [CrossRef]
- Cereda, E.; Cilia, R.; Klersy, C.; Siri, C.; Pozzi, B.; Reali, E.; Colombo, A.; Zecchinelli, A.L.; Mariani, C.B.; Tesei, S.; et al. Dementia in Parkinson’s disease: Is male gender a risk factor? Parkinsonism Relat. Disord. 2016, 26, 67–72. [Google Scholar] [CrossRef]
| Demographics | Normal Group | PDD | F + | p-Value + | ||
|---|---|---|---|---|---|---|
| With Fall | Without Fall | All Patients | ||||
| Number of cases | 37 | 17 | 18 | 35 | ||
| Sex (n = men/women) | 10/27 | 5 / 12 | 3 / 15 | 8/27 | 0.630 | |
| Ages (years) | 63.1 ± 5.34 | 65.3 ± 5.99 | 62.8 ± 5.61 | 64.0 ± 5.85 | 1.112 | 0.335 |
| Education | 9.95 ± 4.73 # | 5.53 ± 4.14 # | 6.72 ± 4.78 | 6.14 ± 4.45 | 6.416 | 0.003 |
| MMSE | 26.7 ± 2.22§# | 19.2 ± 4.10 # | 21.1 ± 4.30 § | 20.1 ± 4.25 | 36.481 | <0.001 |
| Neuropsychological domains | ||||||
| Attention | 0.46 ± 0.48 § # | −0.70 ±-0.66 # | −0.30 ± 0.72 § | −0.49 ± 0.71 | 5.871 | <0.001 |
| Executive function | 0.43 ± 0.76 § # | −0.54 ± 0.62 # | −0.41 ± 0.65 § | −0.48 ± 0.63 | 1.461 | <0.001 |
| Memory | 0.41 ± 0.53 § # | −0.54 ± 0.85 # | −0.34 ± 0.65 § | −0.43 ± 0.75 | 2.140 | 0.001 |
| Speech and language | 0.46 ± 0.71 § # | −0.52 ± 0.58 # | −0.50 ± 0.69 § | −0.51 ± 0.63 | 0.317 | <0.001 |
| Visuospatial function | 0.46 ± 0.62 § # | −0.45 ± 0.77 # | −0.53 ± 0.74 § | −0.49 ± 0.74 | 1.054 | <0.001 |
| Disease Severity | PD with Dementia | F + | p-Value + | ||
|---|---|---|---|---|---|
| With Fall | Without Fall | All Patients | |||
| Number of Cases | 17 | 18 | 35 | ||
| Unified Parkinson’s Disease Rating Scale (UPDRS) a | 57.0 ± 28.3 | 42.6 ± 22.9 | 49.6 ± 26.3 | 0.559 | 0.106 |
| UPDRS I b | 4.88 ± 2.91 | 3.72 ± 2.95 | 4.28 ± 2.95 | 0.040 | 0.250 |
| UPDRS-II c | 16.7 ± 9.98 | 10.5 ± 5.54 | 13.5 ± 8.49 | 2.540 | 0.028 * |
| UPDRS III d | 35.4 ± 16.8 | 28.3 ± 16.1 | 31.8 ± 16.6 | 0.010 | 0.213 |
| Hoehn and Yahr staging | 2.53 ± 0.89 | 2.14 ± 0.98 | 2.33 ± 0.95 | 0.294 | 0.228 |
| Activity of daily living | 76.5 ± 15.0 | 80.6 ± 16.3 | 78.6 ± 15.6 | 0.022 | 0.446 |
| Morse fall scale | 62.9 ± 27.7 | 49.7 ± 26.8 | 56.1 ± 27.7 | 0.102 | 0.161 |
| Gray Matter Volume | Anatomical Regions | x | y | z | Cluster Size | T-Value |
|---|---|---|---|---|---|---|
| Normal > all PDD | ||||||
| R superior temporal gyrus * | 59 | −11 | 0 | 14770 | 5.90 | |
| L putamen * | −27 | 15 | 6 | 10899 | 5.68 | |
| R cerebellum * | 20 | −74 | −47 | 14753 | 5.54 | |
| R middle frontal gyrus * | 29 | 50 | 8 | 591 | 5.40 | |
| L middle frontal gyrus * | −29 | 42 | 30 | 304 | 4.94 | |
| R fusiform | 41 | −57 | −18 | 289 | 4.55 | |
| R precentral gyrus | 53 | -8 | 47 | 711 | 4.51 | |
| L precentral gyrus | −44 | 6 | 47 | 646 | 4.42 | |
| L superior frontal gyrus | −18 | 60 | 8 | 445 | 4.26 | |
| L middle occipital gyrus | −33 | −81 | 14 | 195 | 4.20 | |
| L superior medial frontal gyrus | −11 | 4 | −20 | 238 | 4.11 | |
| L cerebellum | −30 | −68 | -36 | 648 | 3.52 | |
| Normal > PDD with fall | ||||||
| L middle temporal gyrus * | −47 | 2 | −35 | 53327 | 7.82 | |
| L cuneus * | −5 | −86 | 3 | 28263 | 7.14 | |
| L medial frontal gyrus * | −12 | 39 | 18 | 2027 | 5.34 | |
| L middle occipital gyrus | −33 | −81 | 14 | 641 | 6.10 | |
| R inferior parietal gyrus | 57 | −56 | 44 | 270 | 5.23 | |
| L inferior parietal gyrus | −59 | −47 | 45 | 281 | 4.79 | |
| R middle occipital gyrus | 42 | −80 | 8 | 532 | 4.67 | |
| L middle temporal gyrus | −41 | −65 | 12 | 441 | 4.40 | |
| R cingulate gyrus | 14 | 11 | 44 | 165 | 3.92 | |
| R anterior cingulate | 14 | 44 | 3 | 234 | 3.87 | |
| Normal > PDD without fall | ||||||
| R cerebellum | 17 | −74 | −45 | 1860 | 4.00 | |
| R superior temporal gyrus | 62 | 2 | −3 | 236 | 3.63 | |
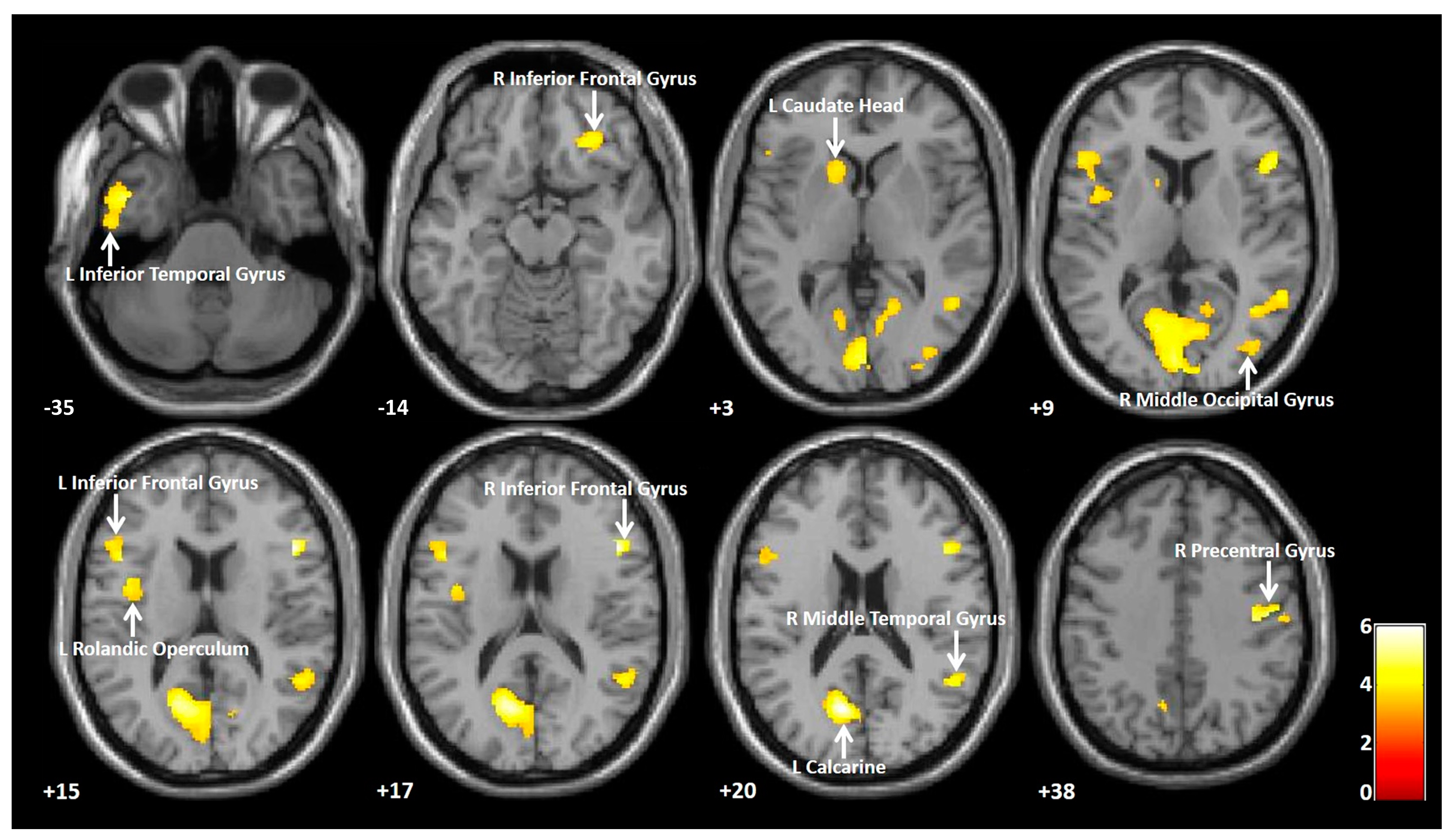
| PDD with fall < PDD without fall | ||||||
| L calcarine * | −9 | −68 | 20 | 3735 | 5.97 | |
| R inferior frontal gyrus *(pars triangularis) | 48 | 20 | 17 | 333 | 6.01 | |
| L inferior temporal gyrus | −47 | 3 | −35 | 717 | 4.97 | |
| R precentral gyrus | 41 | −15 | 38 | 363 | 4.77 | |
| L inferior frontal gyrus (pars opercularis) | −47 | 15 | 15 | 444 | 4.73 | |
| R middle temporal gyrus | 50 | -53 | 20 | 390 | 4.52 | |
| R inferior frontal gyrus (pars orbitalis) | 30 | 35 | −14 | 385 | 4.32 | |
| L Rolandic operculum | −42 | 2 | 15 | 312 | 4.06 | |
| L caudate head | −14 | 15 | 3 | 248 | 3.82 | |
| R middle occipital gyrus | 41 | −78 | 9 | 205 | 3.72 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, K.-L.; Lin, L.-H.; Chen, P.-C.; Chiang, P.-L.; Chen, Y.-S.; Chen, H.-L.; Chen, M.-H.; Chou, K.-H.; Li, S.-H.; Lu, C.-H.; et al. Reduced Gray Matter Volume and Risk of Falls in Parkinson’s Disease with Dementia Patients: A Voxel-Based Morphometry Study. Int. J. Environ. Res. Public Health 2020, 17, 5374. https://doi.org/10.3390/ijerph17155374
Cheng K-L, Lin L-H, Chen P-C, Chiang P-L, Chen Y-S, Chen H-L, Chen M-H, Chou K-H, Li S-H, Lu C-H, et al. Reduced Gray Matter Volume and Risk of Falls in Parkinson’s Disease with Dementia Patients: A Voxel-Based Morphometry Study. International Journal of Environmental Research and Public Health. 2020; 17(15):5374. https://doi.org/10.3390/ijerph17155374
Chicago/Turabian StyleCheng, Kai-Lun, Li-Han Lin, Po-Cheng Chen, Pi-Ling Chiang, Yueh-Sheng Chen, Hsiu-Ling Chen, Meng-Hsiang Chen, Kun-Hsien Chou, Shau-Hsuan Li, Cheng-Hsien Lu, and et al. 2020. "Reduced Gray Matter Volume and Risk of Falls in Parkinson’s Disease with Dementia Patients: A Voxel-Based Morphometry Study" International Journal of Environmental Research and Public Health 17, no. 15: 5374. https://doi.org/10.3390/ijerph17155374
APA StyleCheng, K.-L., Lin, L.-H., Chen, P.-C., Chiang, P.-L., Chen, Y.-S., Chen, H.-L., Chen, M.-H., Chou, K.-H., Li, S.-H., Lu, C.-H., & Lin, W.-C. (2020). Reduced Gray Matter Volume and Risk of Falls in Parkinson’s Disease with Dementia Patients: A Voxel-Based Morphometry Study. International Journal of Environmental Research and Public Health, 17(15), 5374. https://doi.org/10.3390/ijerph17155374





