Pollution Assessment Based on Element Concentration of Tree Leaves and Topsoil in Ayutthaya Province, Thailand
Abstract
1. Introduction
2. Materials and Methods
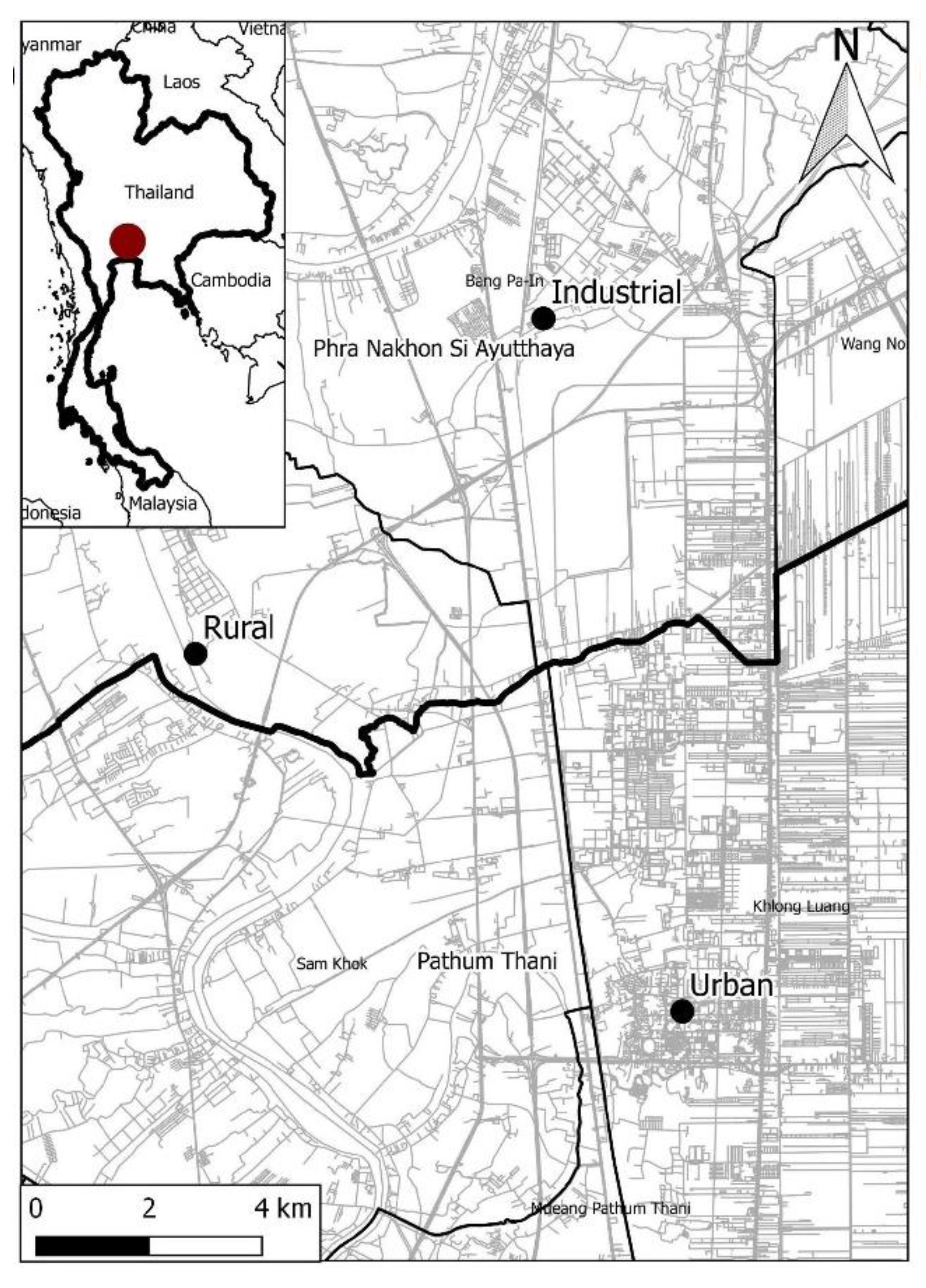
2.1. Study Sites and Sample Collection
2.2. Elemental Analysis in Leaves and Soil Samples
2.3. Determination of Soil Organic Matter Quantity and Humus Quality
2.4. Calculation of Bioaccumulation Factor (BAF)
2.5. Statistical Analysis
3. Results and Discussion
3.1. Elemental Concentration in Leaves
3.2. Elemental Concentrations of Soil
3.3. BAF
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Egedy, T.; Kovács, Z.; Szabó, B. Changing geography of the creative economy in Hungary at the beginning of the 21st century. Hung. Geogr. Bull. 2018, 67, 275–291. [Google Scholar] [CrossRef]
- United Nations. World Urbanization Prospects: The 2018 Revision. 2018. Available online: https://population.un.org/wup/Publications/Files/WUP2018-KeyFacts.pdf (accessed on 6 August 2019).
- Ciarkowska, K.; Gambuś, F.; Antonkiewicz, J.; Koliopoulos, T. Polycyclic aromatic hydrocarbon and heavy metal contents in the urban soils in southern Poland. Chemosphere 2019, 229, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Serbula, S.M.; Miljkovic, D.D.; Kovacevic, R.M.; Ilic, A.A. Assessment of airborne heavy metal pollution using plant parts and topsoil. Ecotoxicol. Environ. Saf. 2012, 76, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Zhou, W.; Li, W.; Li, L. Impact of urbanization level on urban air quality: A case of fine particles (PM 2.5) in Chinese cities. Environ. Pollut. 2014, 194, 163–170. [Google Scholar] [CrossRef]
- Feng, W.; Guo, Z.; Xiao, X.; Peng, C.; Shi, L.; Ran, H.; Xu, W. Atmospheric deposition as a source of cadmium and lead to soil-rice system and associated risk assessment. Ecotoxicol. Environ. Saf. 2019, 180, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Wan, Q.; Liu, H.; Liu, H.; Kapsar, K.; Peng, J.; Kasper, K. How does urbanization influence PM2.5 concentrations? Perspective of spillover effect of multi-dimensional urbanization impact. J. Clean. Prod. 2019, 220, 974–983. [Google Scholar] [CrossRef]
- Liacos, J.W.; Kam, W.; Delfino, R.J.; Schauer, J.J.; Sioutas, C. Characterization of organic, metal and trace element PM2.5 species and derivation of freeway-based emission rates in Los Angeles, CA. Sci. Total. Environ. 2012, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Notardonato, I.; Manigrasso, M.; Pierno, L.; Settimo, G.; Protano, C.; Vitali, M.; Mattei, V.; Martellucci, S.; Di, F.C.; Boccia, P.; et al. The importance of measuring ultrafine particles in urban air quality monitoring in small cities. Geogr. Pannonica 2019, 23, 347–358. [Google Scholar] [CrossRef]
- Maeaba, W.; Prasad, S.; Chandra, S. First Assessment of Metals Contamination in Road Dust and Roadside Soil of Suva City, Fiji. Arch. Environ. Contam. Toxicol. 2019, 77, 249–262. [Google Scholar] [CrossRef]
- Kharkan, J.; Sayadi, M.H.; Rezaei, M.R. Investigation of heavy metals accumulation in the soil and pine trees. Environ. Health Eng. Manag. 2019, 6, 17–25. [Google Scholar] [CrossRef]
- Szolnoki, Z.; Farsang, A.; Puskas, I. Cumulative impacts of human activities on urban garden soils: Origin and accumulation of metals. Environ. Pollut. 2013, 177, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Kandic, S.; Tepe, S.J.; Blanch, E.W.; De Silva, S.; Mikkonen, H.G.; Reichman, S.M. Quantifying factors related to urban metal contamination in vegetable garden soils of the west and north of Melbourne, Australia. Environ. Pollut. 2019, 251, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Gajbhiye, T.; Pandey, S.K.; Lee, S.S.; Kim, K.-H. Size fractionated phytomonitoring of airborne particulate matter (PM) and speciation of PM bound toxic metals pollution through Calotropis procera in an urban environment. Ecol. Indic. 2019, 104, 32–40. [Google Scholar] [CrossRef]
- He, C.; Qiu, K.; Alahmad, A.; Pott, R. Particulate matter capturing capacity of roadside evergreen vegetation during the winter season. Urban For. Urban Green. 2020, 48, 126510. [Google Scholar] [CrossRef]
- Molnár, V.É.; Simon, E.; Tóthmérész, B.; Ninsawat, S.; Szabó, S. Air pollution induced vegetation stress—The Air Pollution Tolerance Index as a quick tool for city health evaluation. Ecol. Ind. 2020, 113, 106234. [Google Scholar]
- Simon, E.; Baranyai, E.; Braun, M.; Cserhati, C.; Fabian, I.; Tóthmérész, B. Elemental concentrations in deposited dust on leaves along an urbanization gradient. Sci. Total. Environ. 2014, 490, 514–520. [Google Scholar] [CrossRef]
- Simon, E.; Harangi, S.; Baranyai, E.; Fábián, I.; Tóthmérész, B. Influence of past industry and urbanization on elemental concentrations in deposited dust and tree leaf tissue. Urban Forest. Urban Green. 2016, 20, 12–19. [Google Scholar] [CrossRef]
- Simon, E.; Molnár, V.É.; Tóthmérész, B.; Szabó, S. Ecological Assessment of Particulate Material (PM5 and PM10) in Urban Habitats. Atmosphere 2020, 11, 559. [Google Scholar] [CrossRef]
- Mirecki, N.; Agic, R.; Šunić, L.; Milenkovic, L.; Ilic, Z. Transfer factor as indicator of heavy metals content in plants. Fresen Environ. Bull. 2015, 24, 4212–4219. [Google Scholar]
- Bhattacharya, T.; Bhumika, F.; Bhattacharya, P. Heavy metal concentrations in street and leaf deposited dust in Anand City, India. Res. J. Chem. Sci. 2011, 1, 61–66. [Google Scholar]
- Braun, M.; Margitai, Z.; Tóth, A.; Leermakers, M. Environmental monitoring using linden tree leaves as natural traps of atmospheric deposition: A pilot study in Transilvania, Romania. Acta Geogr. Debrecina Lands. Environ. 2007, 1, 24–35. [Google Scholar]
- Kosiorek, M.; Modrzewska, B.; Wyszkowski, M. Levels of selected trace elements in Scots pine (Pinus sylvestris L.), silver birch (Betula pendula L.), and Norway maple (Acer platanoides L.) in an urbanized environment. Environ. Monit. Assess. 2016, 188, 598. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Margitai, Z.; Simon, E.; Fábián, I.; Braun, M. Inorganic chemical composition of dust deposited on oleander (Nerium oleander L.) leaves. Air Qual. Atmosphere Health 2016, 10, 339–347. [Google Scholar] [CrossRef]
- Zakrzewska, M.; Klimek, B. Trace Element Concentrations in Tree Leaves and Lichen Collected Along a Metal Pollution Gradient Near Olkusz (Southern Poland). Bull. Environ. Contam. Toxicol. 2017, 100, 245–249. [Google Scholar] [CrossRef]
- Filep, T.; Draskovits, E.; Szabó, J.; Koós, S.; László, P.; Szalai, Z. The dissolved organic matter as a potential soil quality indicator in arable soils of Hungary. Environ. Monit. Assess. 2015, 187, 220. [Google Scholar] [CrossRef]
- Paul, E.A. The nature and dynamics of soil organic matter: Plant inputs, microbial transformations, and organic matter stabilization. Soil Boil. Biochem. 2016, 98, 109–126. [Google Scholar] [CrossRef]
- Szabó, S.; Ágoston, C.; Braun, M.; Keresztúri, P.; Szabó, G. Cadmium and zinc uptake of rye-grass as related to soil type and different land use. Cereal Res.Commun. 2008, 36, 427–430. [Google Scholar]
- Lasota, J.; Błońska, E.; Łyszczarz, S.; Tibbett, M. Forest Humus Type Governs Heavy Metal Accumulation in Specific Organic Matter Fractions. Water Air Soil Pollut. 2020, 231, 1–13. [Google Scholar] [CrossRef]
- Horváth, A.; Szucs, P.; Bidló, A. Soil condition and pollution in urban soils: Evaluation of the soil quality in a Hungarian town. J. Soils Sediments 2014, 15, 1825–1835. [Google Scholar] [CrossRef]
- Oktaba, L.; Odrobińska, D.; Uzarowicz, Ł. The impact of different land uses in urban area on humus quality. J. Soils Sediments 2018, 18, 2823–2832. [Google Scholar] [CrossRef]
- Uttamang, P.; Aneja, V.P.; Hanna, A.F. Assessment of gaseous criteria pollutants in the Bangkok Metropolitan Region, Thailand. Atmospheric Chem. Phys. Discuss. 2018, 18, 12581–12593. [Google Scholar] [CrossRef]
- Langkulsen, U.; Rwodzi, D. Climate Change, Air Pollution, and Human Health in Bangkok. In Springer Climate; Rais, A., Cosimo, P., Eds.; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 255–271. [Google Scholar]
- Hajizadeh, Y.; Mokhtari, M.; Faraji, M.; Abdolahnejad, A.; Mohammadi, A. Biomonitoring of airborne metals using tree leaves: Protocol for biomonitor selection and spatial trend. MethodsX 2019, 6, 1694–1700. [Google Scholar] [CrossRef] [PubMed]
- Angelova, V.; Akova, V.; Ivanov, K.; Licheva, P.A. Comparative study of titrimetric methods for determination of organic carbon in soils, compost and sludge. J. Int. Sci. Public. Ecol. Safe. 2014, 8, 430–440. [Google Scholar]
- Molnár, S.; Barna, G.; Draskovits, E.; Földényi, R.; Hernádi, H.; Bakacsi, Z.; Makó, A. Analysis of correlations between BET-specific surface area, humus materials and other soil properties on typical Hungarian soil types. Agrokémia és Talajtan 2019, 68, 57–77. [Google Scholar] [CrossRef]
- Sipos, S.; Sipos, É.; Nagy, Á. Infrared spectroscopyc examination of humic acids II. Acta Phys. Chem. 1980, 25, 103–110. [Google Scholar]
- Ivanciuc, T.; Ivanciuc, O.; Klein, D.J. Modeling the bioconcentration factors and bioaccumulation factors of polychlorinated biphenyls with posetic quantitative super-structure/activity relationships (QSSAR). Mol. Divers. 2006, 10, 133–145. [Google Scholar] [CrossRef]
- Van Gestel, C.A.M.; Ortiz, M.D.; Borgman, E.; Verweij, R.A. The bioaccumulation of Molybdenum in the earthworm Eisenia andrei: Influence of soil properties and ageing. Chemosphere 2011, 82, 1614–1619. [Google Scholar] [CrossRef]
- De Sá, J.P.M. Applied Statistics Using SPSS, STATISTICA, MATLAB and R, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 978-3-540-71971-7. [Google Scholar]
- Boehmke, B.; Greenwell, B.M. Hands-On Machine Learning with R; Informa UK Limited: London, UK, 2019. [Google Scholar]
- Bruce, P.; Bruce, A. Practical Statistics for Data Scientists: 50 Essential Concepts; O’Reilly Media, Inc.: Sebastopol, CA, USA, 2017. [Google Scholar]
- Singkran, N.; Anantawong, P.; Intharawichian, N.; Kunta, K. The Chao Phraya River Basin: Water quality and anthropogenic influences. Water Supply 2018, 19, 1287–1294. [Google Scholar] [CrossRef]
- Jansen, S.; Watanabe, T.; Smets, E. Aluminium Accumulation in Leaves of 127 Species in Melastomataceae, with Comments on the Order Myrtales. Ann. Bot. 2002, 90, 53–64. [Google Scholar] [CrossRef]
- Briat, J.F. Le fer du sol aux produits végétaux [Iron from soil to plant products]. Bull Acad Natl Med. 2005, 189, 1609–1621. [Google Scholar]
- McBride, M.B.; Shayler, H.A.; Spliethoff, H.M.; Mitchell, R.G.; Marquez-Bravo, L.G.; Ferenz, G.S.; Russell-Anelli, J.M.; Casey, L.; Bachman, S. Concentrations of lead, cadmium and barium in urban garden-grown vegetables: The impact of soil variables. Environ. Pollut. 2014, 194, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Nandy, A.; Talapatra, S.N.; Bhattacharjee, P.; Chaudhuri, P.; Mukhopadhyay, A. Assessment of Morphological Damages of Leaves of Selected Plant Species due to Vehicular Air Pollution, Kolkata, India. Int. Lett. Nat. Sci. 2014, 9, 76–91. [Google Scholar] [CrossRef]
- El-Khatib, A.A.; Abd El-Rahman, A.M.; Elsheikh, O.M. Leaf geometric design of urban trees: Potentiality to capture airborne particle pollutants. J. Environ. Studies. 2011, 7, 49–59. [Google Scholar]
- Prajapati, S.K.; Tripathi, B. Seasonal Variation of Leaf Dust Accumulation and Pigment Content in Plant Species Exposed to Urban Particulates Pollution. J. Environ. Qual. 2008, 37, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Madhak, S.A.; Savsani, J.D.; Pandya, D.J. Comparative pharmacognostical and phytochemical study of leaves of different species of Mimusops. Int. J. Pharm. Sci. Res. 2012, 4, 1074–1078. [Google Scholar]
- FAO. Food and Agricultural Organisation Coping with Water Scarcity. In 2007 World Water Day. 22 March 2007. Available online: http://www.fao.org/nr/water7docs/wwd07brochure.pdf (accessed on 22 March 2007).
- WHO. Permissible Limits of Heavy Metals in Soil and Plants; World Health Organization: Geneva, Switzerland, 1996. [Google Scholar]
- Changlian, P.; Dazhi, W.; Zijian, S.; Guizhu, L.; Zhifang, L.; Guohui, K. Response of some plants for municipal greening to air pollutants. J. Trop. Subtrop. Bot. 2002, 10, 321–327. [Google Scholar]
- Thakar, B.K.; Mishra, P.C. Dust collection potential and air pollution tolerance index of tree vegetation around vedanta aluminium limited, Jharsuguda. Biascan 2010, 3, 603–612. [Google Scholar]
- Chattaong, P.; Jutamas, M. Heavy metal accumulation in scallion (Allium cepa var. aggregatum) fields in Uttaradit Province, Thailand. Int. J. Agric. Technol. 2020, 16, 27–36. [Google Scholar]
- Simasuwannarong, B.; Satapanajaru, T.; Khuntong, S.; Pengthamkeerati, P. Spatial Distribution and Risk Assessment of As, Cd, Cu, Pb, and Zn in Topsoil at Rayong Province, Thailand. Water Air Soil Pollut. 2011, 223, 1931–1943. [Google Scholar] [CrossRef]
- Krailertrattanachai, N.; Ketrot, D.; Wisawapipat, W. The Distribution of Trace Metals in Roadside Agricultural Soils, Thailand. Int. J. Environ. Res. Public Health 2019, 16, 714. [Google Scholar] [CrossRef]
- Sowana, A.; Shrestha, R.P.; Parkpian, P.; Pongquan, S. Influence of Coastal Land Use on Soil Heavy-Metal Contamination in Pattani Bay, Thailand. J. Coast. Res. 2011, 272, 252–262. [Google Scholar] [CrossRef]
- Damrongsiri, S.; Vassanadumrongdee, S.; Tanwattana, P. Heavy metal contamination characteristic of soil in WEEE (waste electrical and electronic equipment) dismantling community: A case study of Bangkok, Thailand. Environ. Sci. Pollut. Res. 2016, 23, 17026–17034. [Google Scholar] [CrossRef] [PubMed]
- Mingkhwan, R.; Worakhunpiset, S. Heavy Metal Contamination Near Industrial Estate Areas in Phra Nakhon Si Ayutthaya Province, Thailand and Human Health Risk Assessment. Int. J. Environ. Res. Public Health 2018, 15, 1890. [Google Scholar] [CrossRef] [PubMed]
- Chaiyarat, R.; Ngoendee, M.; Kruatrachue, M. Accumulation of Cd, Cu, Pb, and Zn in water, sediments, and mangrove crabs (Sesarma mederi) in the upper Gulf of Thailand. Science 2013, 39, 376. [Google Scholar] [CrossRef]
- Gustafsson, J.P.; Pechová, P.; Berggren, D. Modeling Metal Binding to Soils: The Role of Natural Organic Matter. Environ. Sci. Technol. 2003, 37, 2767–2774. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.; Chen, W.; Hu, X.; Cai, P.; Chen, C.; Yu, X.-Y.; Huang, Q. Heavy metal behaviour at mineral-organo interfaces: Mechanisms, modelling and influence factors. Environ. Int. 2019, 131, 104995. [Google Scholar] [CrossRef]
- Rieuwerts, J.S.; Thornton, I.; Farago, M.E.; Ashmore, M.R. Factors influencing metal bioavailability in soils: Preliminary investigations for the development of a critical loads approach for metals. Chem. Speciat. Bioavailab. 1998, 10, 61–75. [Google Scholar] [CrossRef]
- Micó, C.; Peris, M.; Recatalá, L.; Sánchez, J. Baseline values for heavy metals in agricultural soils in an European Mediterranean region. Sci. Total. Environ. 2007, 378, 13–17. [Google Scholar] [CrossRef]
- Nodelkoska, T.V.; Doran, P.M. Characteristics of heavy metal by plant species with potential for phytoremediation and phyto mining. Miner. Engin. 2000, 5, 549–556. [Google Scholar] [CrossRef]
- Santos, E.F.; Santini, J.M.K.; Paixão, A.P.; Júnior, E.F.; Lavres, J.; Al., S.C.E.; Dos Reis, A.R. Physiological highlights of manganese toxicity symptoms in soybean plants: Mn toxicity responses. Plant Physiol. Biochem. 2017, 113, 6–19. [Google Scholar] [CrossRef]
- Ghori, N.-H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Javidan, N.; Kavian, A.; Ly, H.-B.; Conoscenti, C.; Jafarian, Z. Gully Erosion Susceptibility Mapping Using Multivariate Adaptive Regression Splines—Replications and Sample Size Scenarios. Water 2019, 11, 2319. [Google Scholar] [CrossRef]
- Kilinc, K.B.; Malkoc, S.; Koparal, A.S.; Yazici, B. Using multivariate adaptive regression splines to estimate pollution in soil. Int. J. Adv. Appl. Sci. 2017, 4, 10–16. [Google Scholar] [CrossRef][Green Version]
 = Ficus religiosa from industrial,
= Ficus religiosa from industrial,  = F. religiosa from urban,
= F. religiosa from urban,  = F. religiosa from rural,
= F. religiosa from rural,  = Mimusops elengi from industrial,
= Mimusops elengi from industrial,  = M. elengi from urban,
= M. elengi from urban,  = M. elengi from rural site.
= M. elengi from rural site.
 = Ficus religiosa from industrial,
= Ficus religiosa from industrial,  = F. religiosa from urban,
= F. religiosa from urban,  = F. religiosa from rural,
= F. religiosa from rural,  = Mimusops elengi from industrial,
= Mimusops elengi from industrial,  = M. elengi from urban,
= M. elengi from urban,  = M. elengi from rural site.
= M. elengi from rural site.
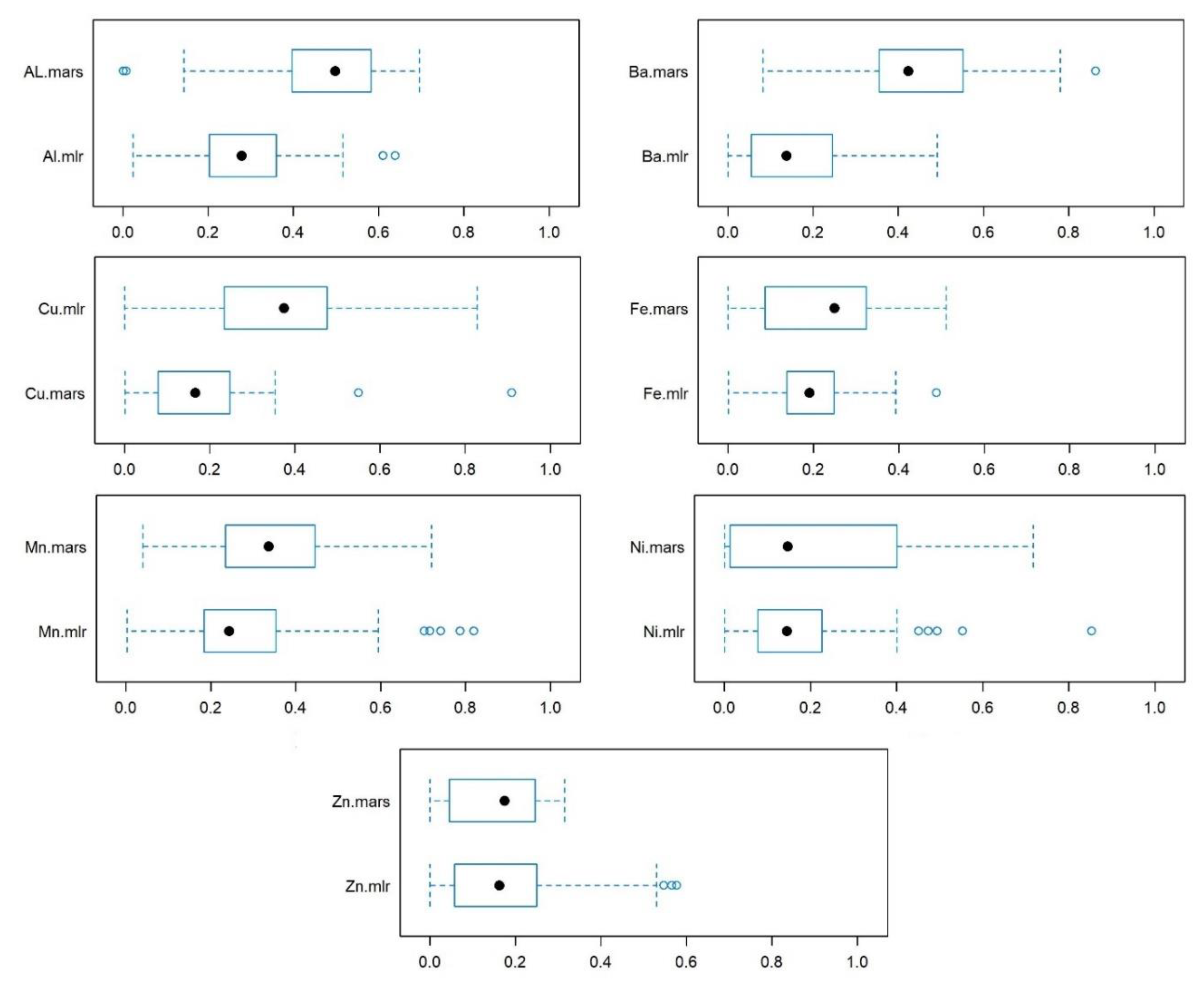
 = industrial site,
= industrial site,  = urban site,
= urban site,  = rural site.
= rural site.
 = industrial site,
= industrial site,  = urban site,
= urban site,  = rural site.
= rural site.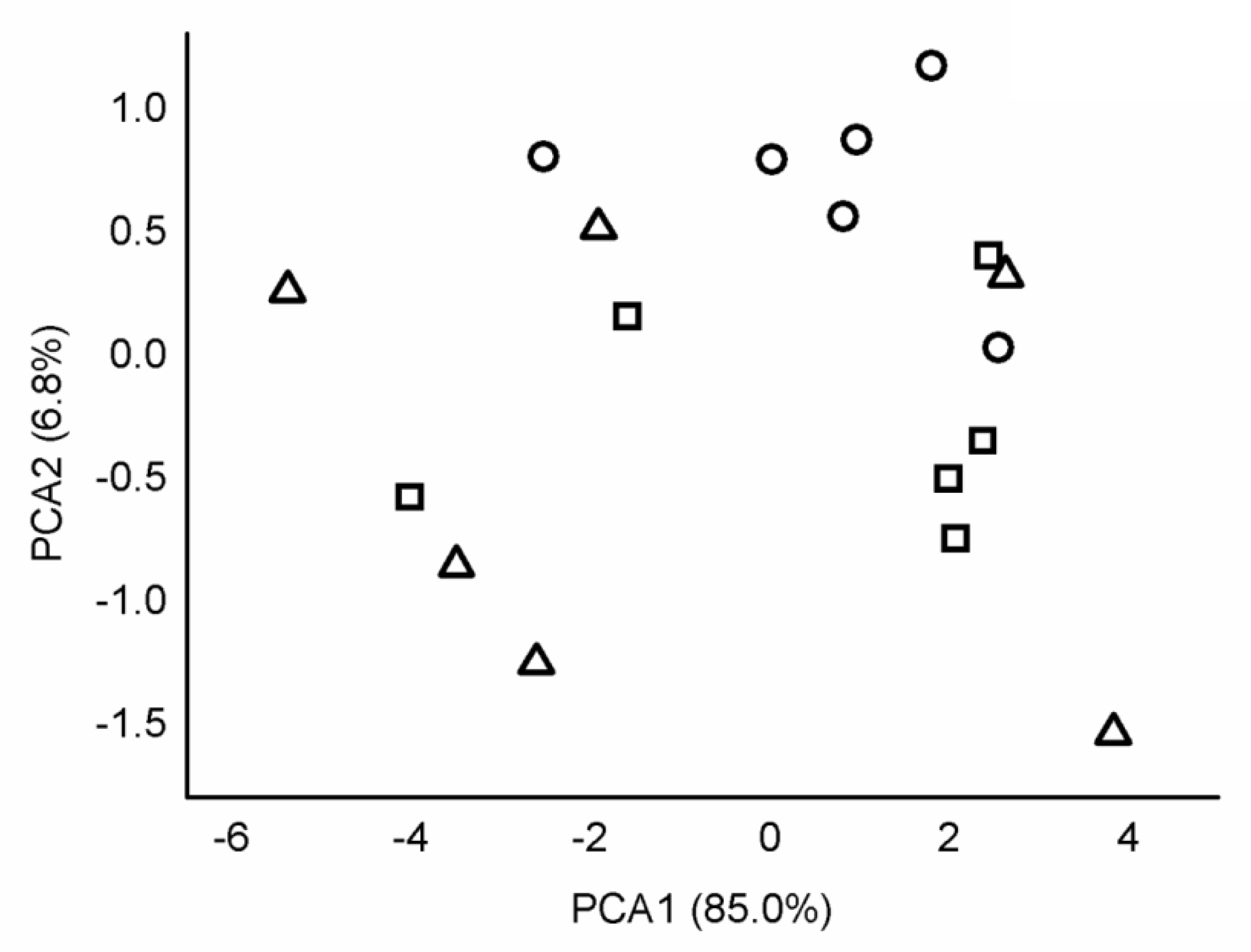
 = BAF value-based M. elengi,
= BAF value-based M. elengi,  = BAF value-based F. religiosa.
= BAF value-based F. religiosa.
 = BAF value-based M. elengi,
= BAF value-based M. elengi,  = BAF value-based F. religiosa.
= BAF value-based F. religiosa.
| Elements | M. elengi | F. religiosa | ||||
|---|---|---|---|---|---|---|
| Industrial | Urban | Rural | Industrial | Urban | Rural | |
| Al, mg kg−1 | 160 ± 35 | 209 ± 53 | 123 ± 29 | 72 ± 16 | 124 ± 36 | 82 ± 25 |
| Ba, mg kg−1 | 5.4 ± 0.8 | 5.4 ± 2.4 | 7.9 ± 1.4 | 7.3 ± 4.9 | 7.0 ± 0.8 | 17.9 ± 6.2 |
| Ca, g kg−1 | 24 ± 3 | 20.0 ± 1.6 | 16.2 ± 1.0 | 37 ± 14 | 49 ± 5 | 36.0 ± 8.1 |
| Cr, mg kg−1 | 1.6 ± 0.4 | 1.7 ± 0.4 | 0.4 ± 0.4 | l.d. | 0.8 ± 0.4 | 0.4 ± 0.4 |
| Cu, mg kg−1 | 2.9 ± 0.4 | 2.5 ± 0.7 | 3.7 ± 0.4 | 13.9 ± 2.8 | 10.3 ± 0.3 | 13.2 ± 0.4 |
| Fe, mg kg−1 | 167 ± 43 | 220 ± 55 | 139 ± 32 | 82 ± 8 | 125 ± 16 | 109 ± 19 |
| K, g kg−1 | 8.7 ± 0.7 | 10.0 ± 2.3 | 12.1 ± 1.7 | 29 ± 2 | 30.7 ± 3.5 | 33.6 ± 2.3 |
| Mg, g kg−1 | 2.2 ± 0.2 | 2.1 ± 0.3 | 2.3 ± 0.3 | 3.4 ± 0.7 | 5.5 ± 1.3 | 5.1 ± 0.6 |
| Mn, mg kg−1 | 170 ± 81 | 168 ± 113 | 25 ± 6 | 23 ± 5 | 49 ± 23 | 24 ± 3 |
| Na, mg kg−1 | 336 ± 84 | 533 ± 130 | 1314 ± 894 | 178 ± 72 | 185 ± 20 | 280 ± 63 |
| Ni, mg kg−1 | 1.6 ± 0.4 | 1.6 ± 0.4 | 1.3 ± 0.1 | 1.2 ± 0.1 | 1.2 ± 0.1 | 1.2 ± 0.1 |
| Pb, mg kg−1 | 1.2 ± 0.1 | 0.8 ± 0.4 | 0.4 ± 0.4 | l.d. | 0.4 ± 0.4 | 0.4 ± 0.4 |
| Zn, mg kg−1 | 11.5 ± 2.0 | 9.5 ± 3.0 | 13.0 ± 2.3 | 23.6 ± 7.3 | 15.7 ± 3.9 | 26.0 ± 3.0 |
| Elements | Thailand | |||
|---|---|---|---|---|
| Industry | Urban | Rural | Critical Value | |
| Al, g kg−1 | 36 ± 10 | 44 ± 7 | 22 ± 8 | n.p. |
| Ba, mg kg−1 | 92 ± 17 | 83 ± 12 | 82 ± 33 | n.p. |
| Ca, g kg−1 | 19 ± 6 | 6 ± 2 | 11 ± 8 | n.p. |
| Cr, mg kg−1 | 33 ± 7 | 36 ± 12 | 21 ± 6 | 75–100 |
| Cu, mg kg−1 | 16 ± 4 | 14 ± 3 | 18 ± 9 | 60–125 |
| Fe, g kg−1 | 21 ± 4 | 27 ± 4 | 16 ± 4 | n.p. |
| K, g kg−1 | 5 ± 1 | 5 ± 1 | 3 ± 1 | n.p. |
| Mg, g kg−1 | 2 ± 0.5 | 2 ± 0.4 | 2 ± 1 | n.p. |
| Mn, mg kg−1 | 207 ± 36 | 236 ± 67 | 225 ± 92 | n.p. |
| Na, g kg−1 | 1 ± 0.2 | 1 ± 0.2 | 1 ± 0.3 | n.p. |
| Ni, mg kg−1 | 13 ± 2 | 16 ± 3 | 11 ± 3 | 100 |
| Pb, mg kg−1 | 11 ± 2 | 14 ± 2 | 13 ± 4 | 100–400 |
| Zn, mg kg−1 | 40 ± 11 | 40 ± 7 | 87 ± 42 | 50–100 |
| humus, % | 2.5 ± 1.5 | 2.5 ± 0.6 | 3.4 ± 1.7 | |
| stability coefficient of humus | 1.5 ± 0.9 | 0.8 ± 0.2 | 0.9 ± 0.5 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molnár, V.É.; Simon, E.; Ninsawat, S.; Tóthmérész, B.; Szabó, S. Pollution Assessment Based on Element Concentration of Tree Leaves and Topsoil in Ayutthaya Province, Thailand. Int. J. Environ. Res. Public Health 2020, 17, 5165. https://doi.org/10.3390/ijerph17145165
Molnár VÉ, Simon E, Ninsawat S, Tóthmérész B, Szabó S. Pollution Assessment Based on Element Concentration of Tree Leaves and Topsoil in Ayutthaya Province, Thailand. International Journal of Environmental Research and Public Health. 2020; 17(14):5165. https://doi.org/10.3390/ijerph17145165
Chicago/Turabian StyleMolnár, Vanda Éva, Edina Simon, Sarawut Ninsawat, Béla Tóthmérész, and Szilárd Szabó. 2020. "Pollution Assessment Based on Element Concentration of Tree Leaves and Topsoil in Ayutthaya Province, Thailand" International Journal of Environmental Research and Public Health 17, no. 14: 5165. https://doi.org/10.3390/ijerph17145165
APA StyleMolnár, V. É., Simon, E., Ninsawat, S., Tóthmérész, B., & Szabó, S. (2020). Pollution Assessment Based on Element Concentration of Tree Leaves and Topsoil in Ayutthaya Province, Thailand. International Journal of Environmental Research and Public Health, 17(14), 5165. https://doi.org/10.3390/ijerph17145165







