Prevalence of Insulin Resistance in the Hungarian General and Roma Populations as Defined by Using Data Generated in a Complex Health (Interview and Examination) Survey
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Representative of the Hungarian Roma (HR) Population Living in Segregated Colonies in Northeast Hungary
2.2. Sample Representative of the Hungarian General (HG) Population Living in Northeast Hungary
2.3. Pillars of the Complex (Health Interview and Examination) Survey
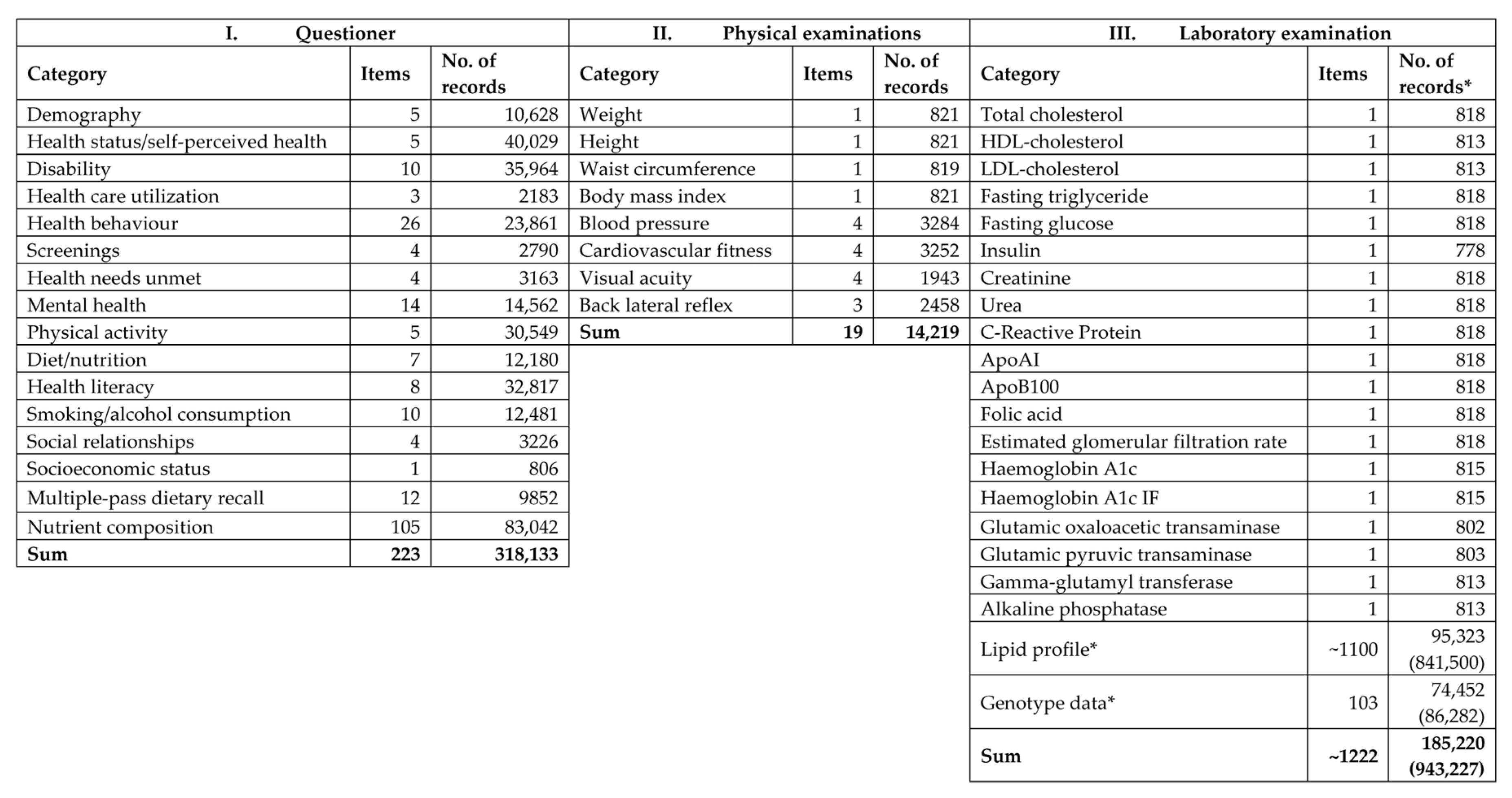
2.3.1. I. Questionnaire-Based Interviews
2.3.2. II. Physical Examinations
2.3.3. III. Laboratory Examinations
2.4. Creation of the Database
2.5. Determination of the Prevalence of Metabolic Syndrome and Its Components
2.6. Insulin Sensitivity/Resistance Indices Calculated by Using Fasting Serum/Plasma Concentrations of Insulin, Glucose, HDL-C and Triglycerides
2.6.1. Determination of the Cut-Off Values of Surrogate Measures for Insulin Resistance
2.6.2. Determination of the Prevalence of IR by Using Different Surrogate Indices
2.7. Statistical Analysis
2.8. Ethical Statement
3. Results
3.1. Demographic and Anthropometric Characteristics of the Samples
3.2. The Prevalence of MetS and Its Components in the Study Populations
3.3. Findings Used to Estimate the Risk of IR for MetS
3.4. Determination of Cut-Off Points for IR Surrogate Indices
3.5. Prevalence of IR in the Hungarian General and Hungarian Roma Populations
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Commission. Health Status of the Roma Population. Data Collection in the Member States of the European Union; European Commission: Brussels, Belgium, 2014; ISBN 978-92-79-37904-8. [Google Scholar]
- Adany, R. Roma health is global ill health. Eur. J. Public Health 2014, 24, 702–703. [Google Scholar] [CrossRef] [PubMed]
- Farkas, L. Data Collection in the Field of Ethnicity; Publications Office of the European Union, European Commission: Luxembourg, 2017; ISBN 978-92-79-66084-9. [Google Scholar]
- Fernandez-Feito, A.; Pesquera-Cabezas, R.; Gonzalez-Cobo, C.; Prieto-Salceda, M.D. What do we know about the health of Spanish Roma people and what has been done to improve it? A scoping review. Ethn. Health 2019, 24, 224–243. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.S.; Azmanov, D.N.; Ivanova, M.B.; Chamova, T.; Pacheva, I.H.; Panova, M.V.; Song, S.; Morar, B.; Yordanova, R.V.; Galabova, F.K.; et al. Founder p.Arg 446* mutation in the PDHX gene explains over half of cases with congenital lactic acidosis in Roma children. Mol. Genet. Metab. 2014, 113, 76–83. [Google Scholar] [CrossRef]
- Dotcho, M. The Health Situation of Roma Communities: Analysis of the Data from the UNDP/World Bank/EC Regional Roma Survey; UNDP Europe and the CIS, Bratislava Regional Centre, Valeur, s. r. o.: Dunajská Streda, Slovak Republic, 2012; ISBN 978-92-95092-60-0. [Google Scholar]
- Ekmekci, P.E. Health and Roma People in Turkey. Balkan Med. J. 2016, 33, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Sandor, J.; Kosa, Z.; Boruzs, K.; Boros, J.; Tokaji, I.; McKee, M.; Adany, R. The decade of Roma Inclusion: Did it make a difference to health and use of health care services? Int. J. Public Health 2017, 62, 803–815. [Google Scholar] [CrossRef] [PubMed]
- Belak, A.; Madarasova Geckova, A.; van Dijk, J.P.; Reijneveld, S.A. Health-endangering everyday settings and practices in a rural segregated Roma settlement in Slovakia: A descriptive summary from an exploratory longitudinal case study. BMC Public Health 2017, 17, 128. [Google Scholar] [CrossRef]
- Weiss, E.; Japie, C.; Balahura, A.M.; Bartos, D.; Badila, E. Cardiovascular risk factors in a Roma sample population from Romania. Rom. J. Intern. Med. 2018, 56, 193–202. [Google Scholar] [CrossRef]
- Babinska, I.; Veselska, Z.D.; Bobakova, D.; Pella, D.; Panico, S.; Reijneveld, S.A.; Jarcuska, P.; Jarcuska, P.; Zezula, I.; Geckova, A.M.; et al. Is the cardiovascular risk profile of people living in Roma settlements worse in comparison with the majority population in Slovakia? Int. J. Public Health 2013, 58, 417–425. [Google Scholar] [CrossRef]
- Zeljko, H.M.; Skaric-Juric, T.; Narancic, N.S.; Baresic, A.; Tomas, Z.; Petranovic, M.Z.; Milicic, J.; Salihovic, M.P.; Janicijevic, B. Age trends in prevalence of cardiovascular risk factors in Roma minority population of Croatia. Econ. Hum. Biol. 2013, 11, 326–336. [Google Scholar] [CrossRef]
- Nunes, M.A.; Kucerova, K.; Lukac, O.; Kvapil, M.; Broz, J. Prevalence of Diabetes Mellitus among Roma Populations-A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 2607. [Google Scholar] [CrossRef]
- Janevic, T.; Jankovic, J.; Bradley, E. Socioeconomic position, gender, and inequalities in self-rated health between Roma and non-Roma in Serbia. Int. J. Public Health 2012, 57, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Sarvary, A.; Kosa, Z.; Javorne, R.E.; Gyulai, A.; Takacs, P.; Sandor, J.; Sarvary, A.; Nemeth, A.; Halmai, R.; Adany, R. Socioeconomic status, health related behaviour, and self-rated health of children living in Roma settlements in Hungary. Cent. Eur. J. Public Health 2019, 27, 24–31. [Google Scholar] [CrossRef] [PubMed]
- La Parra-Casado, D.; Mosquera, P.A.; Vives-Cases, C.; San Sebastian, M. Socioeconomic Inequalities in the Use of Healthcare Services: Comparison between the Roma and General Populations in Spain. Int. J. Environ. Res. Public Health 2018, 15, 121. [Google Scholar] [CrossRef] [PubMed]
- Voko, Z.; Csepe, P.; Nemeth, R.; Kosa, K.; Kosa, Z.; Szeles, G.; Adany, R. Does socioeconomic status fully mediate the effect of ethnicity on the health of Roma people in Hungary? J. Epidemiol. Community Health 2009, 63, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Duval, L.; Wolff, F.C.; McKee, M.; Roberts, B. The Roma vaccination gap: Evidence from twelve countries in Central and South-East Europe. Vaccine 2016, 34, 5524–5530. [Google Scholar] [CrossRef] [PubMed]
- Fiatal, S.; Piko, P.; Kosa, Z.; Sandor, J.; Adany, R. Genetic profiling revealed an increased risk of venous thrombosis in the Hungarian Roma population. Thromb. Res. 2019, 179, 37–44. [Google Scholar] [CrossRef]
- Piko, P.; Fiatal, S.; Kosa, Z.; Sandor, J.; Adany, R. Genetic factors exist behind the high prevalence of reduced high-density lipoprotein cholesterol levels in the Roma population. Atherosclerosis 2017, 263, 119–126. [Google Scholar] [CrossRef]
- Sipeky, C.; Weber, A.; Szabo, M.; Melegh, B.I.; Janicsek, I.; Tarlos, G.; Szabo, I.; Sumegi, K.; Melegh, B. High prevalence of CYP2C19*2 allele in Roma samples: Study on Roma and Hungarian population samples with review of the literature. Mol. Biol. Rep. 2013, 40, 4727–4735. [Google Scholar] [CrossRef]
- Grundy, S.M.; Brewer, H.B., Jr.; Cleeman, J.I.; Smith, S.C., Jr.; Lenfant, C.; American Heart, A.; National Heart, L.; Blood, I. Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 2004, 109, 433–438. [Google Scholar] [CrossRef]
- WHO: Non communicable diseases. Key Facts. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases (accessed on 17 June 2020).
- Roberts, C.K.; Hevener, A.L.; Barnard, R.J. Metabolic syndrome and insulin resistance: Underlying causes and modification by exercise training. Compr. Physiol. 2013, 3, 1–58. [Google Scholar] [CrossRef]
- Reaven, G.M. Insulin resistance and compensatory hyperinsulinemia: Role in hypertension, dyslipidemia, and coronary heart disease. Am. Heart J. 1991, 121, 1283–1288. [Google Scholar] [CrossRef]
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E.; Haffner, S.M.; Mitchell, B.D.; Stern, M.P. Hyperinsulinaemia: The key feature of a cardiovascular and metabolic syndrome. Diabetologia 1991, 34, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: A summary of the evidence. Diabetes Care 2005, 28, 1769–1778. [Google Scholar] [CrossRef]
- Wang, W.; Wang, K.; Li, T.; Xiang, H.; Ma, L.; Fu, Z.; Chen, J.; Liu, Z.; Bai, J.; Feng, J.; et al. A discussion on utility and purposed value of obesity and abdomen obesity when body mass index, waist circumference, waist to hip ratio used as indexes predicting hypertension and hyper-blood glucose. Zhonghua Liu Xing Bing Xue Za Zhi 2002, 23, 16–19. [Google Scholar]
- Fan, H.; Zhang, P.R.; Xu, Y. Relationship between non-alcohol fatty liver disease and insulin resistance/cardiovascular diseases in patients with type 2 diabetes mellitus. Chin. Gen. Pract. 2011, 14, 147–150. [Google Scholar]
- Andrew, J.K.; Graham, A.H. The Expanding Pathogenic Role of Insulin Resistance in Human Disease. Diabet. Med. 2014. [Google Scholar] [CrossRef]
- Semenkovich, C.F. Insulin Resistance and a Long, Strange Trip. N. Engl. J. Med. 2016, 374, 1378–1379. [Google Scholar] [CrossRef]
- Zhou, M.S.; Wang, A.; Yu, H. Link between insulin resistance and hypertension: What is the evidence from evolutionary biology? Diabetol. Metab. Syndr. 2014, 6, 12. [Google Scholar] [CrossRef]
- Cho, J.; Hong, H.; Park, S.; Kim, S.; Kang, H. Insulin Resistance and Its Association with Metabolic Syndrome in Korean Children. Biomed. Res. Int. 2017, 2017, 8728017. [Google Scholar] [CrossRef]
- Brown, A.E.; Walker, M. Genetics of Insulin Resistance and the Metabolic Syndrome. Curr. Cardiol. Rep. 2016, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Brede, S.; Serfling, G.; Klement, J.; Schmid, S.M.; Lehnert, H. Clinical Scenario of the Metabolic Syndrome. Visc. Med. 2016, 32, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Mansyur, M.A.; Bakri, S.; Patellongi, I.J.; Rahman, I.A. The association between metabolic syndrome components, low-grade systemic inflammation and insulin resistance in non-diabetic Indonesian adolescent male. Clin. Nutr. ESPEN 2020, 35, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Kosa, K.; Darago, L.; Adany, R. Environmental survey of segregated habitats of Roma in Hungary: A way to be empowering and reliable in minority research. Eur. J. Public Health 2011, 21, 463–468. [Google Scholar] [CrossRef]
- Szeles, G.; Voko, Z.; Jenei, T.; Kardos, L.; Pocsai, Z.; Bajtay, A.; Papp, E.; Pasti, G.; Kosa, Z.; Molnar, I.; et al. A preliminary evaluation of a health monitoring programme in Hungary. Eur. J. Public Health 2005, 15, 26–32. [Google Scholar] [CrossRef]
- European Health Interview Survey (EHIS wave 2) Methodological Manual; Publications Office of the European Union, European Commission Eurostat: Luxembourg, 2013; ISBN 978-92-79-29424-2.
- Goldberg, D.P.; Gater, R.; Sartorius, N.; Ustun, T.B.; Piccinelli, M.; Gureje, O.; Rutter, C. The validity of two versions of the GHQ in the WHO study of mental illness in general health care. Psychol. Med. 1997, 27, 191–197. [Google Scholar] [CrossRef]
- Örkényi, Á. Lelki Egészség (Mental Health); Országos Epidemiológiai Központ: Budapest, Hungary, 2005. [Google Scholar]
- Saunders, J.B.; Aasland, O.G.; Babor, T.F.; de la Fuente, J.R.; Grant, M. Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption--II. Addiction 1993, 88, 791–804. [Google Scholar] [CrossRef]
- Babor, T.F.; Higgins-Biddle, J.C.; Saunders, J.B.; Monteiro, M.G. AUDIT: The Alcohol Use Disorders Identification Test: Guidelines for Use in Primary Care, 2nd ed.; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Reinert, D.F.; Allen, J.P. The alcohol use disorders identification test: An update of research findings. Alcohol. Clin. Exp. Res. 2007, 31, 185–199. [Google Scholar] [CrossRef]
- Hagstromer, M.; Oja, P.; Sjostrom, M. The International Physical Activity Questionnaire (IPAQ): A study of concurrent and construct validity. Public Health Nutr. 2006, 9, 755–762. [Google Scholar] [CrossRef]
- Simental-Mendia, L.E.; Rodriguez-Moran, M.; Guerrero-Romero, F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat. Disord. 2008, 6, 299–304. [Google Scholar] [CrossRef]
- Hubkova, B.; Maslankova, J.; Stupak, M.; Guzy, J.; Kovacova, A.; Pella, D.; Jarcuska, P.; Marekova, M.; HepaMeta, T. Assessment of clinical biochemical parameters in Roma minority residing in eastern Slovakia compared with the majority population. Cent. Eur. J. Public Health 2014, 22, S12–S17. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Tobin, J.D.; Andres, R. Glucose clamp technique: A method for quantifying insulin secretion and resistance. Am. J. Physiol. 1979, 237, E214–E223. [Google Scholar] [CrossRef] [PubMed]
- Bonora, E.; Targher, G.; Alberiche, M.; Bonadonna, R.C.; Saggiani, F.; Zenere, M.B.; Monauni, T.; Muggeo, M. Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity: Studies in subjects with various degrees of glucose tolerance and insulin sensitivity. Diabetes Care 2000, 23, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Saxena, A. Surrogate markers of insulin resistance: A review. World J. Diabetes 2010, 1, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative insulin sensitivity check index: A simple, accurate method for assessing insulin sensitivity in humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410. [Google Scholar] [CrossRef]
- McAuley, K.A.; Williams, S.M.; Mann, J.I.; Walker, R.J.; Lewis-Barned, N.J.; Temple, L.A.; Duncan, A.W. Diagnosing insulin resistance in the general population. Diabetes Care 2001, 24, 460–464. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Simental-Mendia, L.E.; Gonzalez-Ortiz, M.; Martinez-Abundis, E.; Ramos-Zavala, M.G.; Hernandez-Gonzalez, S.O.; Jacques-Camarena, O.; Rodriguez-Moran, M. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J. Clin. Endocrinol. Metab. 2010, 95, 3347–3351. [Google Scholar] [CrossRef]
- Roa Barrios, M.; Arata-Bellabarba, G.; Valeri, L.; Velazquez-Maldonado, E. Relationship between the triglyceride/high-density lipoprotein-cholesterol ratio, insulin resistance index and cardiometabolic risk factors in women with polycystic ovary syndrome. Endocrinol. Nutr. 2009, 56, 59–65. [Google Scholar] [CrossRef]
- McLaughlin, T.; Reaven, G.; Abbasi, F.; Lamendola, C.; Saad, M.; Waters, D.; Simon, J.; Krauss, R.M. Is there a simple way to identify insulin-resistant individuals at increased risk of cardiovascular disease? Am. J. Cardiol. 2005, 96, 399–404. [Google Scholar] [CrossRef]
- Ruopp, M.D.; Perkins, N.J.; Whitcomb, B.W.; Schisterman, E.F. Youden Index and optimal cut-point estimated from observations affected by a lower limit of detection. Biom. J. 2008, 50, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.; Park, J.H.; Jang, E.J.; Park, Y.K.; Yu, J.M.; Park, J.S.; Ahn, Y.; Choi, S.H.; Yoo, H.J. The Cut-off Values of Surrogate Measures for Insulin Sensitivity in a Healthy Population in Korea according to the Korean National Health and Nutrition Examination Survey (KNHANES) 2007-2010. J. Korean Med. Sci. 2018, 33, e197. [Google Scholar] [CrossRef] [PubMed]
- Soltesz, B.; Piko, P.; Sandor, J.; Kosa, Z.; Adany, R.; Fiatal, S. The genetic risk for hypertension is lower among the Hungarian Roma population compared to the general population. PLoS ONE 2020, 15, e0234547. [Google Scholar] [CrossRef] [PubMed]
- Kuhlbrandt, C.; Footman, K.; Rechel, B.; McKee, M. An examination of Roma health insurance status in Central and Eastern Europe. Eur. J. Public Health 2014, 24, 707–712. [Google Scholar] [CrossRef] [PubMed]
- OECD Economic Surveys: Slovak Republic 2019; OECD Publishing: Paris, France, 2019. [CrossRef]
- Pasztor, I.; Penzes, J.; Tatrai, P.; Pálóczi, Á. The number and spatial distribution of the Roma population in Hungary—in the light of different approaches. Folia Geogr. 2016, 58, 5–21. [Google Scholar]
- Kosa, Z.; Szeles, G.; Kardos, L.; Kosa, K.; Nemeth, R.; Orszagh, S.; Fesus, G.; McKee, M.; Adany, R.; Voko, Z. A comparative health survey of the inhabitants of Roma settlements in Hungary. Am. J. Public Health 2007, 97, 853–859. [Google Scholar] [CrossRef]
- Toth, M.D.; Adam, S.; Zonda, T.; Birkas, E.; Purebl, G. Risk factors for multiple suicide attempts among Roma in Hungary. Transcult. Psychiatry 2018, 55, 55–72. [Google Scholar] [CrossRef]
- Paulik, E.; Nagymajtenyi, L.; Easterling, D.; Rogers, T. Smoking behaviour and attitudes of Hungarian Roma and non-Roma population towards tobacco control policies. Int. J. Public Health 2011, 56, 485–491. [Google Scholar] [CrossRef]
- Vincze, F.; Foldvari, A.; Palinkas, A.; Sipos, V.; Janka, E.A.; Adany, R.; Sandor, J. Prevalence of Chronic Diseases and Activity-Limiting Disability among Roma and Non-Roma People: A Cross-Sectional, Census-Based Investigation. Int. J. Environ. Res. Public Health 2019, 16, 3620. [Google Scholar] [CrossRef]
- Arora, V.S.; Kuhlbrandt, C.; McKee, M. An examination of unmet health needs as perceived by Roma in Central and Eastern Europe. Eur. J. Public Health 2016, 26, 737–742. [Google Scholar] [CrossRef]
- Sandor, J.; Nagy, A.; Foldvari, A.; Szabo, E.; Csenteri, O.; Vincze, F.; Sipos, V.; Kovacs, N.; Palinkas, A.; Papp, M.; et al. Delivery of cardio-metabolic preventive services to Hungarian Roma of different socio-economic strata. Fam. Pract. 2017, 34, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Sudzinova, A.; Nagyova, I.; Studencan, M.; Rosenberger, J.; Skodova, Z.; Vargova, H.; Middel, B.; Reijneveld, S.A.; van Dijk, J.P. Roma coronary heart disease patients have more medical risk factors and greater severity of coronary heart disease than non-Roma. Int. J. Public Health 2013, 58, 409–415. [Google Scholar] [CrossRef]
- Kolvek, G.; Rosicova, K.; Rosenberger, J.; Podracka, L.; Stewart, R.E.; Nagyova, I.; Reijneveld, S.A.; van Dijk, J.P. End-stage renal disease among Roma and non-Roma: Roma are at risk. Int. J. Public Health 2012, 57, 751–754. [Google Scholar] [CrossRef] [PubMed]
- Werissa, N.A.; Piko, P.; Fiatal, S.; Kosa, Z.; Sandor, J.; Adany, R. SNP-Based Genetic Risk Score Modeling Suggests No Increased Genetic Susceptibility of the Roma Population to Type 2 Diabetes Mellitus. Genes 2019, 10, 942. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Fiatal, S.; Sandor, J.; Adany, R. Distinct Penetrance of Obesity-Associated Susceptibility Alleles in the Hungarian General and Roma Populations. Obes. Facts 2017, 10, 444–457. [Google Scholar] [CrossRef]
- Melegh, B.I.; Banfai, Z.; Hadzsiev, K.; Miseta, A.; Melegh, B. Refining the South Asian Origin of the Romani people. BMC Genet. 2017, 18, 82. [Google Scholar] [CrossRef]
- Reid, T. Essays on the Intellectual Powers of Man; J. Bartlett: Edinburgh, UK, 1785. [Google Scholar]
- Antoniolli, L.P.; Nedel, B.L.; Pazinato, T.C.; de Andrade Mesquita, L.; Gerchman, F. Accuracy of insulin resistance indices for metabolic syndrome: A cross-sectional study in adults. Diabetol. Metab. Syndr. 2018, 10, 65. [Google Scholar] [CrossRef]
- Kim, T.J.; Kim, H.J.; Kim, Y.B.; Lee, J.Y.; Lee, H.S.; Hong, J.H.; Lee, J.W. Comparison of Surrogate Markers as Measures of Uncomplicated Insulin Resistance in Korean Adults. Korean J. Fam. Med. 2016, 37, 188–196. [Google Scholar] [CrossRef][Green Version]
- Kouis, P.; Griffin, M.; Nicolaides, A.; Panayiotou, A. A comparison of different Insulin Resistance indices for the prediction of the Metabolic Syndrome. Eur. J. Public Health 2016, 26. [Google Scholar] [CrossRef]
- Yu, X.; Wang, L.; Zhang, W.; Ming, J.; Jia, A.; Xu, S.; Li, Q.; Ji, Q. Fasting triglycerides and glucose index is more suitable for the identification of metabolically unhealthy individuals in the Chinese adult population: A nationwide study. J. Diabetes Investig. 2019, 10, 1050–1058. [Google Scholar] [CrossRef]
- Navarro-Gonzalez, D.; Sanchez-Inigo, L.; Pastrana-Delgado, J.; Fernandez-Montero, A.; Martinez, J.A. Triglyceride-glucose index (TyG index) in comparison with fasting plasma glucose improved diabetes prediction in patients with normal fasting glucose: The Vascular-Metabolic CUN cohort. Prev. Med. 2016, 86, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.H.; Sobia, F.; Niazi, N.K.; Manzoor, S.M.; Fazal, N.; Ahmad, F. Metabolic clustering of risk factors: Evaluation of Triglyceride-glucose index (TyG index) for evaluation of insulin resistance. Diabetol. Metab. Syndr. 2018, 10, 74. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, F.; Reaven, G.M. Comparison of two methods using plasma triglyceride concentration as a surrogate estimate of insulin action in nondiabetic subjects: Triglycerides x glucose versus triglyceride/high-density lipoprotein cholesterol. Metabolism 2011, 60, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Kosa, Z.; Moravcsik-Kornyicki, A.; Dioszegi, J.; Roberts, B.; Szabo, Z.; Sandor, J.; Adany, R. Prevalence of metabolic syndrome among Roma: A comparative health examination survey in Hungary. Eur. J. Public Health 2015, 25, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Geckova, A.M.; Jarcuska, P.; Marekova, M.; Pella, D.; Siegfried, L.; Jarcuska, P.; Halanova, M.; HepaMeta, T. Hepameta—Prevalence of hepatitis B/C and metabolic syndrome in population living in separated and segregated Roma settlements: A methodology for a cross-sectional population-based study using community-based approach. Cent. Eur. J. Public Health 2014, 22, S6–S11. [Google Scholar] [CrossRef]
- Macejova, Z.; Kristian, P.; Janicko, M.; Halanova, M.; Drazilova, S.; Antolova, D.; Marekova, M.; Pella, D.; Madarasova-Geckova, A.; Jarcuska, P.; et al. The Roma Population Living in Segregated Settlements in Eastern Slovakia Has a Higher Prevalence of Metabolic Syndrome, Kidney Disease, Viral Hepatitis B and E, and Some Parasitic Diseases Compared to the Majority Population. Int. J. Environ. Res. Public Health 2020, 17, 3112. [Google Scholar] [CrossRef]

| Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | p-Value | ||
|---|---|---|---|---|
| Prevalence % (n) | Prevalence % (n) | |||
| Sex | Male | 44.58 (177) | 26.36 (97) | <0.001 |
| Female | 55.42 (220) | 73.64 (271) | ||
| Age groups | 20–34 | 24.69 (98) | 28.80 (106) | 0.434 |
| 35–49 | 39.04 (155) | 37.23 (137) | ||
| 50–64 | 36.27 (144) | 33.97 (125) | ||
| Mean (95% CI) | Mean (95% CI) | p-Value | ||
| Age (years) | 44.18 (42.97–45.38) | 42.82 (41.57–44.07) | 0.123 | |
| Weight (kg) | 77.80 (76.12–79.47) | 71.91 (69.93–73.89) | <0.001 | |
| Height (cm) | 168.73 (167.79–169.68) | 161.15 (160.20–162.11) | <0.001 | |
| BMI (kg/m2) | 27.27 (26.74–27.81) | 27.61 (26.90–28.31) | 0.918 | |
| Metabolic Syndrome and Its Components | Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | p-Value | Males in the HG Population (n = 177) | Males in the HR Population (n = 97) | p-Value | Females in the HG Population (n = 220) | Females in the HR Population (n = 271) | p-Value |
|---|---|---|---|---|---|---|---|---|---|
| Prevalence % (95% CI) | Prevalence % (95% CI) | Prevalence % (95% CI) | Prevalence % (95% CI) | Prevalence % (95% CI) | Prevalence % (95% CI) | ||||
| Central obesity | 75.56 (70.11–78.65) | 73.37 (68.68–77.69) | 0.719 | 64.41 (57.17–71.18) | 58.76 (48.83–68.18) | 0.356 | 82.73 (77.32–87.28) | 78.60 (73.43–83.16) | 0.251 |
| Raised blood pressure or treated hypertension | 57.18 (52.27–61.98) | 54.08 (48.97–59.12) | 0.378 | 60.45 (53.13–67.44) | 57.73 (47.79–67.22) | 0.661 | 54.55 (47.94–61.03) | 52.77 (46.82–58.66) | 0.694 |
| Raised fasting plasma glucose concentration or previously diagnosed diabetes mellitus | 25.19 (21.11–29.63) | 23.91 (19.77–28.46) | 0.666 | 27.12 (20.98–34.00) | 31.96 (23.31–41.66) | 0.398 | 23.64 (18.39–29.57) | 21.03 (16.50–26.18) | 0.490 |
| Raised triglyceride level or treated lipid disorder | 37.28 (32.63–42.11) | 37.77 (32.93–42.81) | 0.767 | 42.94 (35.81–50.30) | 51.55 (41.68–61.32) | 0.172 | 32.73 (26.78–39.12) | 32.84 (27.46–38.59) | 0.979 |
| Reduced HDL cholesterol level or treated lipid disorder | 36.27 (31.66–41.09) | 55.98 (50.88–60.99) | <0.001 | 32.20 (25.65–39.33) | 47.42 (37.68–57.31) | 0.013 | 39.55 (33.26–46.11) | 59.04 (53.12–64.77) | <0.001 |
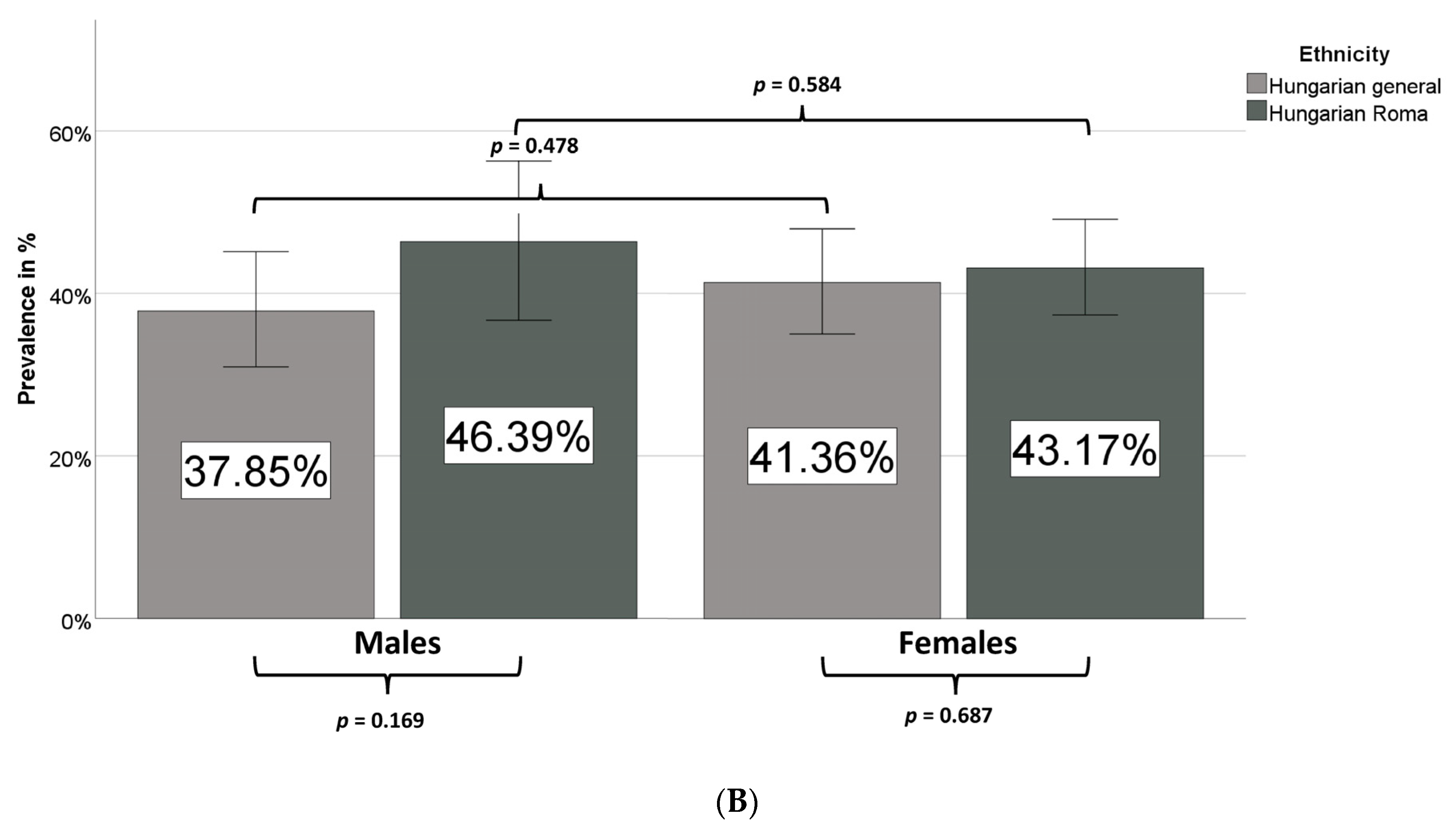
| Metabolic syndrome | 39.80 (35.07–44.67) | 44.02 (39.01–49.12) | 0.232 | 37.85 (30.95–45.15) | 46.39 (36.70–56.30) | 0.169 | 41.36 (35.00–47.95) | 43.17 (37.37–49.12) | 0.687 |
| Without Mets | With Mets | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| HG (n = 397) | HR (n = 368) | p-Value | HG (n = 239) | HR (n = 206) | p-Value | HG (n = 158) | HR (n = 162) | p-Value | |
| Mean | Mean | Mean | |||||||
| Fasting insulin (mU/L) | 15.82 | 16.67 | 0.892 | 11.02 | 11.27 | 0.371 | 23.10 | 23.54 | 0.736 |
| Fasting glucose (mmol/L) | 5.25 | 5.10 | 0.105 | 4.77 | 4.63 | 0.158 | 5.98 | 5.70 | 0.177 |
| Fasting TG (mmol/L) | 1.58 | 1.61 | 0.375 | 1.18 | 1.12 | 0.941 | 2.19 | 2.23 | 0.386 |
| HDL-C (mmol/L) | 1.37 | 1.26 | <0.001 | 1.49 | 1.37 | 0.001 | 1.19 | 1.12 | 0.006 |
| Waist circumference (cm) | 96.03 | 94.78 | 0.286 | 89.6 | 86.9 | 0.008 | 105.8 | 104.8 | 0.580 |
| Systolic blood pressure (mmHg) | 126.75 | 123.71 | < 0.001 | 122.4 | 119.3 | 0.007 | 133.4 | 129.3 | 0.001 |
| Diastolic blood pressure (mmHg) | 78.82 | 79.63 | 0.600 | 76.9 | 76.6 | 0.469 | 81.7 | 83.4 | 0.229 |
| Prevalence of antihypertensive treatment (%) | 28.46 | 31.52 | 0.356 | 12.97 | 13.11 | 0.966 | 51.90 | 54.94 | 0.586 |
| Prevalence of antidiabetic treatment (%) | 6.05 | 11.14 | 0.012 | 0.84 | 1.94 | 0.314 | 13.92 | 22.84 | 0.040 |
| Prevalence of lipid lowering therapy (%) | 6.80 | 11.68 | 0.019 | 1.67 | 1.46 | 0.845 | 14.56 | 24.69 | 0.023 |
| Indices | Without MetS | With MetS | |||||||
|---|---|---|---|---|---|---|---|---|---|
| HG (n = 397) | HR (n = 368) | p-Value | HG (n = 239) | HR (n = 206) | p-Value | HG (n = 158) | HR (n = 162) | p-Value | |
| Mean | Mean | Mean | |||||||
| HOMA-IR | 4.07 | 4.32 | 0.722 | 2.51 | 2.47 | 0.321 | 6.44 | 6.68 | 0.568 |
| QUICKI | 0.34 | 0.35 | 0.722 | 0.36 | 0.37 | 0.320 | 0.32 | 0.32 | 0.568 |
| McAuley index | 6.96 | 7.00 | 0.746 | 7.94 | 8.20 | 0.375 | 5.48 | 5.47 | 0.881 |
| TG/HDL-C ratio | 1.36 | 1.49 | 0.033 | 0.90 | 0.90 | 0.110 | 2.05 | 2.24 | 0.085 |
| TyG index | 4.64 | 4.64 | 0.871 | 4.47 | 4.45 | 0.743 | 4.88 | 4.88 | 0.978 |
| A | ||||||||||||
| Variables | Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | Combined Population (n = 765) | |||||||||
| Cop | Sens./Spec. | YI | AUC | Cop | Sens./Spec. | YI | AUC | Cop | Sens./Spec. | YI | AUC | |
| Fasting insulin (mU/L) | 12.480 | 0.614/0.749 | 0.363 | 0.735 | 10.100 | 0.728/0.650 | 0.379 | 0.726 | 11.855 | 0.634/0.712 | 0.347 | 0.730 |
| Fasting glucose (mmol/L) | 5.350 | 0.506/0.828 | 0.335 | 0.696 | 5.550 | 0.364/0.927 | 0.291 | 0.674 | 5.350 | 0.450/0.852 | 0.302 | 0.684 |
| Fasting TG (mmol/L) | 1.590 | 0.709/0.828 | 0.537 | 0.826 | 1.750 | 0.673/0.932 | 0.605 | 0.857 | 1.590 | 0.713/0.849 | 0.562 | 0.841 |
| HDL-C (mmol/L) | 1.195 | 0.620/0.808 | 0.428 | 0.745 | 1.210 | 0.753/0.675 | 0.428 | 0.744 | 1.195 | 0.675/0.753 | 0.428 | 0.743 |
| B | ||||||||||||
| Indices | Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | Combined Population (n = 765) | |||||||||
| Cop | Sens./Spec. | YI | AUC | Cop | Sens./Spec. | YI | AUC | Cop | Sens./Spec. | YI | AUC | |
| HOMA-IR | 2.291 | 0.747/0.695 | 0.441 | 0.763 | 2.224 | 0.710/0.660 | 0.370 | 0.744 | 2.320 | 0.709/0.690 | 0.399 | 0.753 |
| QUICKI | 0.337 | 0.747/0.695 | 0.441 | 0.763 | 0.338 | 0.710/0.660 | 0.370 | 0.744 | 0.336 | 0.709/0.690 | 0.399 | 0.753 |
| McAuley index | 6.297 | 0.741/0.782 | 0.523 | 0.825 | 6.768 | 0.833/0.704 | 0.537 | 0.828 | 5.989 | 0.697/0.827 | 0.524 | 0.827 |
| TG/HDL-C ratio | 1.304 | 0.722/0.833 | 0.554 | 0.831 | 1.274 | 0.747/0.864 | 0.611 | 0.855 | 1.274 | 0.734/0.843 | 0.574 | 0.843 |
| TyG index | 4.694 | 0.791/0.820 | 0.611 | 0.858 | 4.685 | 0.759/0.869 | 0.628 | 0.867 | 4.694 | 0.772/0.843 | 0.615 | 0.862 |
| Indices | Prevalence of Insulin Resistance (%) Based on the Cut-Off Points Identified in the Study Populations (95% CI) | p-Value | Prevalence of Insulin Resistance (%) Based on the Cut-Off Points Identified in the Combined Population (95% CI) | p-Value | ||
|---|---|---|---|---|---|---|
| Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | Hungarian General Population (n = 397) | Hungarian Roma Population (n = 368) | |||
| HOMA-IR | 48.11 (43.22–53.02) n = 191 | 49.73 (44.64–54.82) n = 185 | 0.550 | 47.61 (42.73–52.52) n = 189 | 47.83 (42.76–52.93) n = 176 | 0.952 |
| QUICKI | 48.11 (43.22–53.02) n = 191 | 49.46 (44.37–54.55) n = 182 | 0.710 | 47.10 (42.23–52.02) n = 187 | 47.83 (42.76–52.93) n = 176 | 0.841 |
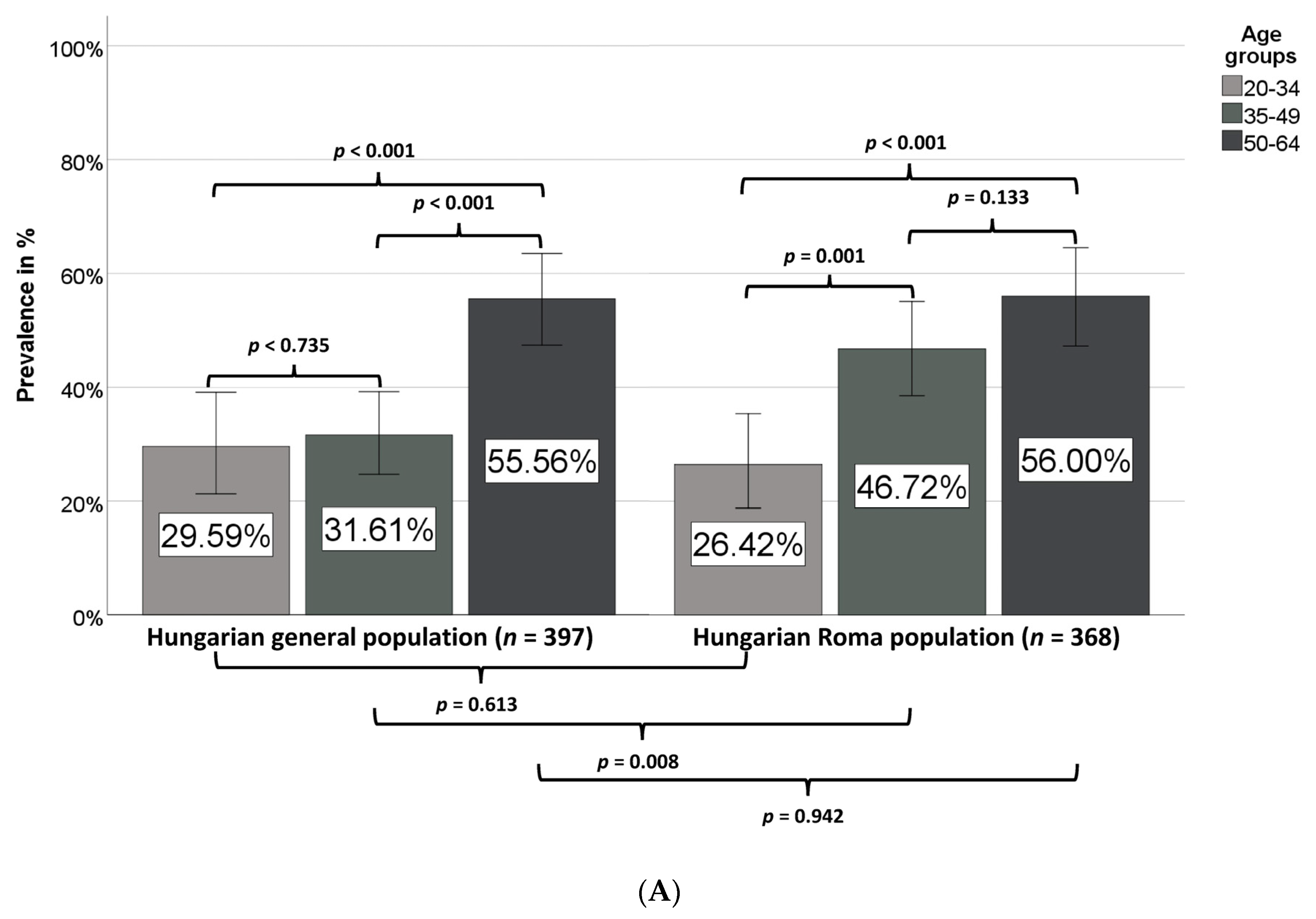
| McAuley index | 42.57 (37.77–47.47) n = 169 | 53.26 (48.15–58.32) n = 196 | 0.003 | 37.78 (33.12–42.63) n = 150 | 40.76 (35.83–45.84) n = 150 | 0.399 |
| TG/HDL-C ratio | 38.79 (34.09–43.65) n = 154 | 40.49 (35.56–45.56) n = 149 | 0.631 | 39.29 (34.58–44.16) n = 156 | 40.49 (35.56–45.56) n = 149 | 0.736 |
| TyG index | 42.32 (37.53–47.22) n = 168 | 40.76 (35.83–45.84) n = 150 | 0.663 | 42.32 (37.53–47.22) n = 168 | 40.49 (35.56–45.56) n = 149 | 0.608 |
| A | |||||||||
| HOMA-IR | McAuley Index | TyG Index | |||||||
| Age Groups | HG | HR | p-Value | HG | HR | p-Value | HG | HR | p-Value |
| Prevalence %; n | Prevalence %; n | Prevalence %; n | |||||||
| 20–34 | 44.90; n = 90 | 50.00; n = 53 | 0.466 | 32.65; n = 32 | 40.57; n = 43 | 0.242 | 32.65; n = 32 | 30.19; n = 32 | 0.705 |
| 35–49 | 37.42; n = 58 | 48.18; n = 66 | 0.064 | 33.55; n = 52 | 41.61; n = 57 | 0.155 | 38.06; n = 59 | 40.88; n = 56 | 0.624 |
| 50–64 | 60.42; n = 87 | 45.60; n = 57 | 0.015 | 45.83; n = 66 | 40.00; n = 50 | 0.335 | 53.47; n = 77 | 48.80; n = 61 | 0.444 |
| p for trend | 0.004 | 0.502 | 0.044 | 0.965 | 0.002 | 0.016 | |||
| B | |||||||||
| HOMA-IR | McAuley Index | TyG Index | |||||||
| Sex | HG | HR | p for Ethnicity | HG | HR | p for Ethnicity | HG | HR | p for Ethnicity |
| Prevalence %; n | Prevalence %; n | Prevalence %; n | |||||||
| Males | 49.72; n = 88 | 50.52; n = 49 | 0.899 | 44.07; n = 78 | 49.48; n = 48 | 0.021 | 49.15; n = 87 | 51.55; n = 50 | 0.705 |
| Females | 45.91; n = 101 | 46.86; n = 127 | 0.833 | 32.70; n = 72 | 37.64; n = 102 | 0.042 | 36.82; n = 81 | 36.53; n = 99 | 0.948 |
| p for sex | 0.450 | 0.537 | 0.390 | 0.258 | 0.013 | 0.010 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ádány, R.; Pikó, P.; Fiatal, S.; Kósa, Z.; Sándor, J.; Bíró, É.; Kósa, K.; Paragh, G.; Bácsné Bába, É.; Veres-Balajti, I.; et al. Prevalence of Insulin Resistance in the Hungarian General and Roma Populations as Defined by Using Data Generated in a Complex Health (Interview and Examination) Survey. Int. J. Environ. Res. Public Health 2020, 17, 4833. https://doi.org/10.3390/ijerph17134833
Ádány R, Pikó P, Fiatal S, Kósa Z, Sándor J, Bíró É, Kósa K, Paragh G, Bácsné Bába É, Veres-Balajti I, et al. Prevalence of Insulin Resistance in the Hungarian General and Roma Populations as Defined by Using Data Generated in a Complex Health (Interview and Examination) Survey. International Journal of Environmental Research and Public Health. 2020; 17(13):4833. https://doi.org/10.3390/ijerph17134833
Chicago/Turabian StyleÁdány, Róza, Péter Pikó, Szilvia Fiatal, Zsigmond Kósa, János Sándor, Éva Bíró, Karolina Kósa, György Paragh, Éva Bácsné Bába, Ilona Veres-Balajti, and et al. 2020. "Prevalence of Insulin Resistance in the Hungarian General and Roma Populations as Defined by Using Data Generated in a Complex Health (Interview and Examination) Survey" International Journal of Environmental Research and Public Health 17, no. 13: 4833. https://doi.org/10.3390/ijerph17134833
APA StyleÁdány, R., Pikó, P., Fiatal, S., Kósa, Z., Sándor, J., Bíró, É., Kósa, K., Paragh, G., Bácsné Bába, É., Veres-Balajti, I., Bíró, K., Varga, O., & Balázs, M. (2020). Prevalence of Insulin Resistance in the Hungarian General and Roma Populations as Defined by Using Data Generated in a Complex Health (Interview and Examination) Survey. International Journal of Environmental Research and Public Health, 17(13), 4833. https://doi.org/10.3390/ijerph17134833








