Maternal Anthropometric Factors and Circulating Adipokines as Predictors of Birth Weight and Length
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Group
2.2. Anthropometric Measurements
2.3. Biological Material
2.4. Statistics
3. Results
3.1. Anthropometric Parameters of the Mother
3.2. Modulators of Metabolism
4. Discussion
5. Strengths and Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Neufeld, L.M.; Haas, J.D.; Grajeda, R.; Martorell, R. Changes in maternal weight from the first to second trimester of pregnancy are associated with fetal growth and infant length at birth. Am. J. Clin. Nutr. 2004, 79, 646–652. [Google Scholar] [CrossRef]
- Yin, Y.Z.; Chen, X.W.; Li, X.M.; Hou, H.Y.; Zhou, S.S. Relations of pregnant weight and weight gain during pregnancy with pregnancy-induced hypertension and birth weight. J. First Mil. Med. Univ. 2005, 25, 226. [Google Scholar]
- Moll, U.; Olsson, H.; Landin-Olsson, M. Impact of pregestational weight and weight gain during pregnancy on long-term risk for diseases. PLoS ONE. 2017, 12, 1–12. [Google Scholar] [CrossRef]
- O’Brien, T.; Ray, J.; Chan, W. Maternal body mass index and the risk of preeclampsia: A systematic overview. Epidemiology. 2003, 14, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Hedderson, M.; Williams, M.; Holt, V.; Weiss, N.S.; Ferrara, A. Body mass index and weight gain prior to pregnancy and risk of gestational diabetes mellitus. Am. J. Obstet. Gynecol. 2008, 198. [Google Scholar] [CrossRef] [PubMed]
- Przybyłowicz, K.; Przybyłowicz, M.; Grzybiak, M.; Janiszewska, K. Effects of physical activity during pregnancy and gestational weight gain on the nutritional status of newborn in Warmińsko-Mazurskie voivodeship. Acta Sci. Pol. Technol. Aliment. 2014, 13, 203–211. [Google Scholar] [CrossRef]
- Vidakovic, A.J.; Jaddoe, V.; Gishti, O.; Felix, J.; Williams, M.; Hofman, A.; Demmelmair, H.; Koletzko, B.; Tiemeier, H.; Gaillard, R. Body mass index, gestational weight gain and fatty acid concentrations during pregnancy: The Generation R Study. Eur. J. Epidemiol. 2015, 30, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Sámano, R.; Chico-Barba, G.; Martínez-Rojano, H.; Godínez, E.; Rodríguez-Ventura, A.; Ávila-Koury, G.; Aguilar-Sánchez, K. Prepregnancy body mass index classification and gestational weight gain on neonatal outcomes in adolescent mothers: A follow-Up study. PLoS ONE 2018, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Perichart-Perera, O.; Muñoz-Manrique, C.; Reyes-López, A.; Tolentino-Dolores, M.; Espino y Sosa, S.; Ramírez-González, C. Metabolic Markers during pregnancy and their association with maternal and newborn weight status. PLoS ONE 2017, 12, 1–12. [Google Scholar] [CrossRef]
- Frederick, I.; Williams, M.; Sales, A.; Martin, D.; Killien, M. Prepregnancy body mass index, gestational weight gain, and other maternal characteristics in relation to infant birth weight. Matern. Child. Health J. 2008, 12, 557–567. [Google Scholar] [CrossRef]
- Contu, L.; Hawkes, C.A. A review of the impact of maternal obesity on the cognitive function and mental health of the offspring. Int. J. Mol. Sci. 2017, 18, 1093. [Google Scholar] [CrossRef] [PubMed]
- Hendler, I.; Blackwell, S.C.; Mehta, S.H.; Whitty, J.E.; Russell, E.; Sorokin, Y.; Cotton, D.B. The levels of leptin, adiponectin, and resistin in normal weight, overweight, and obese pregnant women with and without pre-eclampsia. Am. J. Obstet. Gynecol. 2005, 193, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M.; Hoegh, M.; Minimum, J.; Huston-Presley, L.; Bernard, S.; Kalhan, S. Adiponectin in human pregnancy: Implications for regulation of glucose and lipid metabolism. Diabetologia 2006, 49, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, B.; Bobby, Z.; Dorairajan, G.; Vinayagam, V.; Muthu, R. Adipokine levels and their association with insulin resistance and fetal outcomes among the newborns of Indian gestational diabetic mothers. Saudi Med. J. 2019, 40, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewska-Ulfik, D.; Godula-Stuglik, U.; Szymańska, A.; Nawrat, A. Serum adiponectin concentration in healthy, eutrophic full-term neonates born by cesarean section. Pediatr. Endocrinol. 2016, 15, 43–50. (In Polish) [Google Scholar] [CrossRef]
- Teler, J.; Dąbrowska-Zamojcin, E.; Prystacki, T.; Towpik, I. The role of adiponectin and leptin in gestational diabetes mellitus. Perinatol. Neonatol. Ginekol. 2013, 6, 168–171. (In Polish) [Google Scholar]
- Larqué, E.; Ruiz-Palacios, M.; Koletzko, B. Placental regulation of fetal nutrient supply. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 292–297. [Google Scholar] [CrossRef]
- Fang, H.; Judd, R. Adiponectin regulation and function. Compr. Physiol. 2018, 8, 1031–1063. [Google Scholar]
- Retnakaran, R. Adiponectin and beta-Cell adaptation in pregnancy. Diabetes 2017, 66, 1121–1122. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.C.; Nuyt, A.M.; Delvin, E.; Fraser, W.D.; Julien, P.; Audibert, F.; Girard, I.; Shatenstein, B.; Deal, C.; Grenier, E.; et al. Maternal and fetal leptin, adiponectin levels and associations with fetal insulin sensitivity. Obesity 2013, 21, 210–216. [Google Scholar] [CrossRef]
- Retnakaran, R.; Ye, C.; Hanley, A.J.; Connelly, P.W.; Sermer, M.; Zinman, B.; Hamilton, J.K. Effect of maternal weight, adipokines, glucose intolerance and lipids on infant birth weight among women without gestational diabetes mellitus. Can. Med. Assoc. J. 2012, 184, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Markowska, A.; Drews, K.; Malendowicz, K. Role of leptin in pathophysiology of pregnancy. Adv. Clin. Exp. Med. 2003, 12, 811–816. [Google Scholar]
- Lacroix, M.; Battista, M.C.; Doyon, M.; Moreau, J.; Patenaude, J.; Guillemette, L.; Ménard, J.; Ardilouze, J.L.; Perron, P.; Hivert, M.F. Higher maternal leptin levels at second trimester are associated with subsequent greater gestational weight gain in late pregnancy. BMC Pregnancy Childbirth. 2016, 16, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Goldenberg, R.; Johnston, K.; Cliver, S. Serum leptin concentrations during pregnancy and their relationship to fetal growth. Obstet. Gynecol. 1988, 91, 389–395. [Google Scholar] [CrossRef]
- Perez-Perez, A.; Toro, A.; Vilarino-Garcia, T.; Maymo, J.; Guadix, P.; Duenas, J.; Fernandez-Sanchez, M.; Varone, C.; Sanchez-Margalet, V. Leptin action in normal and pathological pregnancies. J. Cell. Mol. Med. 2018, 22, 716–727. [Google Scholar] [CrossRef]
- Aaltonen, J.; Ojala, T.; Laitinen, K.; Poussa, T.; Ozanne, S.; Isolauri, E. Impact of maternal diet during pregnancy and breastfeeding on infant metabolic programming: A prospective randomized controlled study. Eur. J. Clin. Nutr. 2011, 65, 10–19. [Google Scholar] [CrossRef]
- Mardolkar, M. Body mass index (BMI) data analysis and classification. Int. J. Comput. Sci. Mob. Comput. 2017, 6, 8–16. [Google Scholar]
- Merchant, S.S.; Momin, I.A.; Sewani, A.A.; Zuberi, N.F. Effect of prepregnancy body mass index and gestational weight gain on birth weight. J. Pak. Med. Assoc. 1999, 49, 23–25. [Google Scholar] [PubMed]
- Kaźmierczak, J.; Reszczyńska, M.; Szymański, W.; Daniłko, M. The value of mother’s body weight for delivery and newborn’s adaptation. Perinatol. Neonatol. Ginekol. 2009, 2, 266–273. (In Polish) [Google Scholar]
- Woynarowska, B.; Palczewska, I.; Oblacińska, A. WHO child growth standards for children 0–5 years. Percentile charts of length/height, weight, body mass index and head circumference. Med. Wieku Rozwoj. 2012, 16, 232–239. (In Polish) [Google Scholar]
- Przybyłowicz, K.; Kalinowska, K. Pregnant women nutrition and newborn status expressed as ponderal index. Probl. Hig. Epidemiol. 2011, 92, 208–511. (In Polish) [Google Scholar]
- Oluwafemi, O.R.; Njokanma, F.O.; Disu, E.A.; Ogunlesi, T.A. Current pattern of ponderal indices of term small-for-gestational age in a population of Nigerian babies. BMC Pediatr. 2013, 13, 110–117. [Google Scholar] [CrossRef]
- Indrayan, A. Medical Biostatistics; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2012. [Google Scholar]
- Gala, U.M.; Godhia, M.L.; Nandanwar, Y.S. Effect of maternal nutritional status on birth outcome. Int. J. Adv. Nutr. Health Sci. 2016, 4, 226–233. [Google Scholar] [CrossRef]
- Voerman, E.; Santos, S.; Patro Golab, B.; Amiano, P.; Ballester, F.; Barros, H.; Bergström, A.; Charles, M.A.; Chatzi, L.; Chevrier, C.; et al. Maternal body mass index, gestational weight gain, and the risk of overweight and obesity across childhood: An individual participant data meta-analysis. PLOS Med. 2019, 16, e1002744. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.F.; Abell, S.K.; Ranasinha, S.; Misso, M.L.; Boyle, J.A.; Harrison, C.L.; Black, M.H.; Li, N.; Hu, G.; Corrado, F.; et al. Gestational weight gain across continents and ethnicity: Systematic review and meta-analysis of maternal and infant outcomes in more than one million women. BMC Med. 2018, 16, 1–14. [Google Scholar] [CrossRef]
- Ceddia, R.B.; Somwar, R.; Maida, A.; Fang, X.; Bikopoulos, G.; Sweeney, G. Globular adiponectin increases GLUT4 translocation and glucose uptake but reduces glycogen synthesis in rat skeletal muscle cells. Diabetologia 2005, 48, 132–139. [Google Scholar] [CrossRef]
- Aye, I.; Rosario, F.; Powell, T.L.; Jansson, T. Adiponectin supplementation in pregnant mice prevents the adverse effects of maternal obesity on placental function and fetal growth. Proc. Natl. Acad. Sci. USA 2015, 112, 12858–12863. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, B.; Lof, M.; Olausson, H.; Forsum, E. Body fat, insulin resistance, energy expenditure and serum concentrations of leptin, adiponectin and resistin before, during and after pregnancy in healthy Swedish women. Br. J. Nutr. 2010, 103, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Naghshineh, E.; Faryadras, N.; Rouholamin, S. Is there any relation between adiponectin levels in the first trimester of pregnancy and gestational diabetes? Womens Health Gynecol. 2016, 2, 1–4. [Google Scholar]
- Nanda, S.; Akolekar, R.; Sodre, D.; Vaikousi, E.; Nicolaides, K. Maternal serum adiponectin at 11–13 weeks of gestation in pregnancies delivering small for gestation neonates. Fetal Diagn. Ther. 2011, 29, 274–279. [Google Scholar] [CrossRef]
- Vernini, J.M.; Moreli, J.B.; Costa, R.A.A.; Negrato, C.A.; Rudge, A.V.C.; Calderon, I.M.P. Maternal adipokines and insulin as biomarkers of pregnancies complicated by overweight and obesity. Diabetolog Metab Syndr. 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Vahamiko, S.; Isolauri, E.; Pesonen, U.; Koskinen, P.; Ekblad, U.; Laitinen, K. Dietary sucrose intake is related to serum leptin concentration in overweight pregnant women. Eur. J. Nutr. 2010, 49, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Masuzaki, H.; Ogawa, Y.; Sagawa, N.; Hosoda, K.; Matsumoto, T.; Mise, H.; Nishimura, H.; Yoshimasa, Y.; Tanaka, I.; Mori, T.; et al. Nonadipose tissue production of leptin: Leptin as a novel placenta-Derived hormone in humans. Nat. Med. 1997, 3, 1029–1033. [Google Scholar] [CrossRef]
- Huras, H.; Spaczyńska, J.; Gadamer, A.; Radoń-Pokracka, M. An influence of changes in the levels of leptin on the risk of preterm delivery in patients with excessive BMI. Przegl. Lek. 2016, 73, 293–295. (In Polish) [Google Scholar] [PubMed]
- Samano, R.; Martinez-Rojano, H.; Chico-Barba, G.; Godinez-Martinez, E.; Sanchez-Jimenez, B.; Montiel-Ojeda, D.; Tolentino, M. Serum concentration of leptin in pregnant adolescents correlated with gestational weight gain, postpartum weight retention and newborn weight/length. Nutrients 2017, 9, 1067. [Google Scholar] [CrossRef] [PubMed]
- Hinkle, S.N.; Rawal, S.; Liu, D.; Chen, J.; Tsai, M.Y.; Zhang, C. Maternal adipokines longitudinally measured across pregnancy and their associations with neonatal size, length, and adiposity. Int. J. Obes. 2019, 43, 1422–1434. [Google Scholar] [CrossRef] [PubMed]
- Solis-Paredes, M.; Espino y Sosa, S.; Estrada-Gutierrez, G.; Nava-Salazar, S.; Ortega-Castillo, V.; Rodriguez-Bosch, M.; Bravo-Flores, E.; Espejel-Nuñez, A.; Tolentino-Dolores, M.; Gaona-Estudillo, R.; et al. Maternal and Fetal Lipid and Adipokine Profiles and Their Association with Obesity. Int J. Endocrinol. 2016. [Google Scholar] [CrossRef]
| Weight Gain during Pregnancy | |||
|---|---|---|---|
| Below Standard (n = 28) | Standard (n = 90) | Above Standard (n = 50) | |
| BMI | <12.4 kg | 12.4–18.0 kg | >18.0 kg |
| Underweight (<18.5 kg/m2) | 4 | 12 | 2 |
| <11.5 kg | 11.5–16.0 kg | >16.0 kg | |
| Standard (18.5–24.9 kg/m2) | 22 | 52 | 26 |
| <7.0 kg | 7–11.5 kg | >11.5 kg | |
| Overweight (25.0–29.9 kg/m2) | 2 | 20 | 14 |
| ≤7.0 kg | >7.0 kg | ||
| Obese (≥30.0 kg/m2) | 6 | 8 | |
| Pregnant Women | |
|---|---|
| Median (Q1; Q2) | |
| Age (years) | 30.5 (28; 34) |
| Body height (cm) | 165.0 (161; 170) |
| Body weight before pregnancy (kg) | 62.0 (54; 71) |
| Body mass index (BMI) before pregnancy (kg/m2) | 22.3 (20.4; 25.4) |
| <18.5 (kg/m2) (n = 18) | 17.5 (17.4; 17.7) |
| 18.5–24.9 (kg/m2) (n = 100) | 21.5 (20.4; 23.3) |
| 25.0–29.9 (kg/m2) (n =36) | 26.7 (25.8; 28.3) |
| ≥30.0 (kg/m2) (n = 14) | 32.7 (31.6; 37.9) |
| Body weight before delivery (kg) | 78.0 (69; 86) |
| Weight gain during pregnancy (kg) | 14 (10; 18) |
| Below standard (n = 28) | 7.5 (4; 10) |
| Standard (n = 90) | 13.5 (11; 14) |
| Above standard (n = 50) | 19.5 (18; 22) |
| (%) | |
| Place of residence | |
| City | 71 |
| Small town | 14 |
| Village | 15 |
| Educational level | |
| Basic education | 2 |
| Vocational education | 8 |
| Secondary education | 24 |
| Higher education | 66 |
| Marital status | |
| Married | 76 |
| In a relationship | 23 |
| Single | 1 |
| Neonates | |
|---|---|
| Median (Q1; Q2) | |
| Birth length (cm) | 54.0 (52; 56) |
| Birth weight (g) | 3350 (3060; 3790) |
| Average ± SD | |
| Ponderal index (g/cm3) | 2.18 ± 0.32 |
| BMI | BMI < 18.5 (kg/m2) (n = 18) | BMI 18.5–24.9 (kg/m2) (n = 100) | BMI 25–29.9 (kg/m2) (n = 36) | BMI ≥ 30 (kg/m2) (n = 14) | p | ||||
|---|---|---|---|---|---|---|---|---|---|
| Birth parameters | Me | (Q1; Q3) | Me | (Q1; Q3) | Me | (Q1; Q3) | Me | (Q1; Q3) | |
| Birth weight (g) * | 3210 a | (3040; 3450) | 3300 b,c | (2950; 3600) | 3635 b | (3120; 4010) | 4005 a,c | (3950; 4190) | pa = 0.002 p b = 0.04 p c < 0.001 |
| Birth length (cm) * | 55 | (52; 57) | 54 | (51; 56) | 54 | (52; 56) | 55 | (53; 58) | p > 0.05 |
| Average | ± SD | Average | ± SD | Average | ± SD | Average | ± SD | p | |
| Ponderal index (g/cm3) ** | 1.99 d,e | 0.5 | 2.15 f | 0.3 | 2.28 d,f | 0.3 | 2.45 e | 0.4 | pd = 0.03 p e = 0.04 p f = 0.04 |
| Weight Gain during Pregnancy | Below Standard (n = 34) | Standard (n = 78) | Above Standard (n = 56) | p | |||
|---|---|---|---|---|---|---|---|
| Birth parameters | Me | (Q1; Q3) | Me | (Q1; Q3) | Me | (Q1; Q3) | |
| Birth weight (g) * | 3255 a | (2875; 3440) | 3360 | (2960; 3725) | 3500 a | (3200; 4000) | pa = 0.004 |
| Birth length (cm) * | 54 | (50.5; 55) | 54 | (52; 56) | 54 | (52; 56) | p > 0.05 |
| Average | ± SD | Average | ± SD | Average | ± SD | p | |
| Ponderal index (g/cm3) ** | 2.12 | 0.25 | 2.14 | 0.35 | 2.29 | 0.3 | p > 0.05 |
| Adiponectin (ng/mL) | |||||
|---|---|---|---|---|---|
| n | Me | (Q1; Q3) | p | ||
| Mothers | |||||
| Age * | <30 years | 74 | 4439 | (3191; 6503) | p = 0.046 |
| ≥30 years | 94 | 5391 | (3769; 8280) | ||
| BMI ** | <18.5 kg/m2 | 18 | 5681 | (3830; 8360) | p > 0.05 |
| 18.5–24.9 kg/m2 | 100 | 5101 | (3342; 7028) | ||
| 24.9–29.9 kg/m2 | 36 | 4299 | 2974; 5862) | ||
| ≥ 30 kg/m2 | 14 | 6445 | (5189; 8654) | ||
| Weight gain ** | Below standard | 34 | 5895 a,b | (4447; 9460) | pa = 0.032 p b = 0.031 |
| Standard | 78 | 4823 a | (3036; 7576) | ||
| Above standard | 56 | 4561 b | (3314; 6122) | ||
| Neonates | |||||
| Birth weight ** | <2500 g | 16 | 5379 | (3791; 10172) | p > 0.05 |
| 2500–3999 g | 123 | 5089 | (3211; 7826) | ||
| ≥4000 g | 29 | 5436 | (3830; 6682) | ||
| Birth length ** | <46 cm | 10 | 5379 | (4398; 14287) | p > 0.05 |
| 46–54 cm | 91 | 5193 | (3530; 7833) | ||
| >54 cm | 67 | 4869 | (3192; 6962) | ||
| Ponderal index ** | <Standard | 51 | 5700 | (2752; 8555) | p > 0.05 |
| <Standard, acceptable | 87 | 5101 | (3329; 6794) | ||
| Standard | 30 | 4515 | (4193; 5982) | ||
| Leptin (ng/mL) | |||||
|---|---|---|---|---|---|
| n | Me | (Q1; Q3) | p | ||
| Mothers | |||||
| Age * | <30 years | 74 | 29.0 | (11.3; 52.1) | p > 0.05 |
| ≥30 years | 94 | 25.9 | (11.8; 41.5) | ||
| BMI ** | <18.5 kg/m2 | 18 | 9.5 a,b | (5.7; 17.2) | pa = 0.013 p b = 0.000 p c = 0.007 |
| 18.5–24.9 kg/m2 | 100 | 25.8 c | (12.5; 41.5) | ||
| 25–29.9 kg/m2 | 36 | 35.5 a | 21.4; 48.4) | ||
| ≥30 kg/m2 | 14 | 47.4 b,c | (38.3; 97.1) | ||
| Weight gain ** | Below standard | 34 | 13.7 d | (7.9; 37.7) | pd = 0.001 p e = 0.003 |
| Standard | 78 | 23.6 e | (11.5; 40.9) | ||
| Above standard | 56 | 36.9 d,e | (25.7; 52.2) | ||
| Neonates | |||||
| Birth weight ** | <2500 g | 16 | 29.3 | (13.1; 52.8) | p > 0.05 |
| 2500–3999 g | 123 | 25.7 | (11.3; 42.6) | ||
| ≥4000 g | 29 | 36.8 | (23.2; 50.0) | ||
| Birth length ** | <46 cm | 10 | 26.8 | (6.8; 57.1) | p > 0.05 |
| 46–54 cm | 91 | 27.3 | (12.3; 41.5) | ||
| >54 cm | 67 | 26.7 | (11.8; 47.7) | ||
| Ponderal index ** | <Standard | 51 | 28.9 | (10.4; 44.6) | p > 0.05 |
| <Standard, acceptable | 87 | 23.6 | (12.0; 39.5) | ||
| Standard | 30 | 35.9 | (25.8; 62.4) | ||
| Variable | Coefficient | p-Value * | R2 | |
|---|---|---|---|---|
| Adiponectin | BMI (kg/m2) | 0.093 | 0.753 | 0.05 |
| Body weight gain (kg) | −0.239 | 0.019 | ||
| Age | 0.416 | 0.186 | ||
| Leptin | BMI (kg/m2) | 2.280 | <0.001 | 0.19 |
| Body weight gain (kg) | 0.387 | 0.012 | ||
| Age | −0.162 | 0.732 |
| Variable | Coefficient | p-Value * | R2 | |
|---|---|---|---|---|
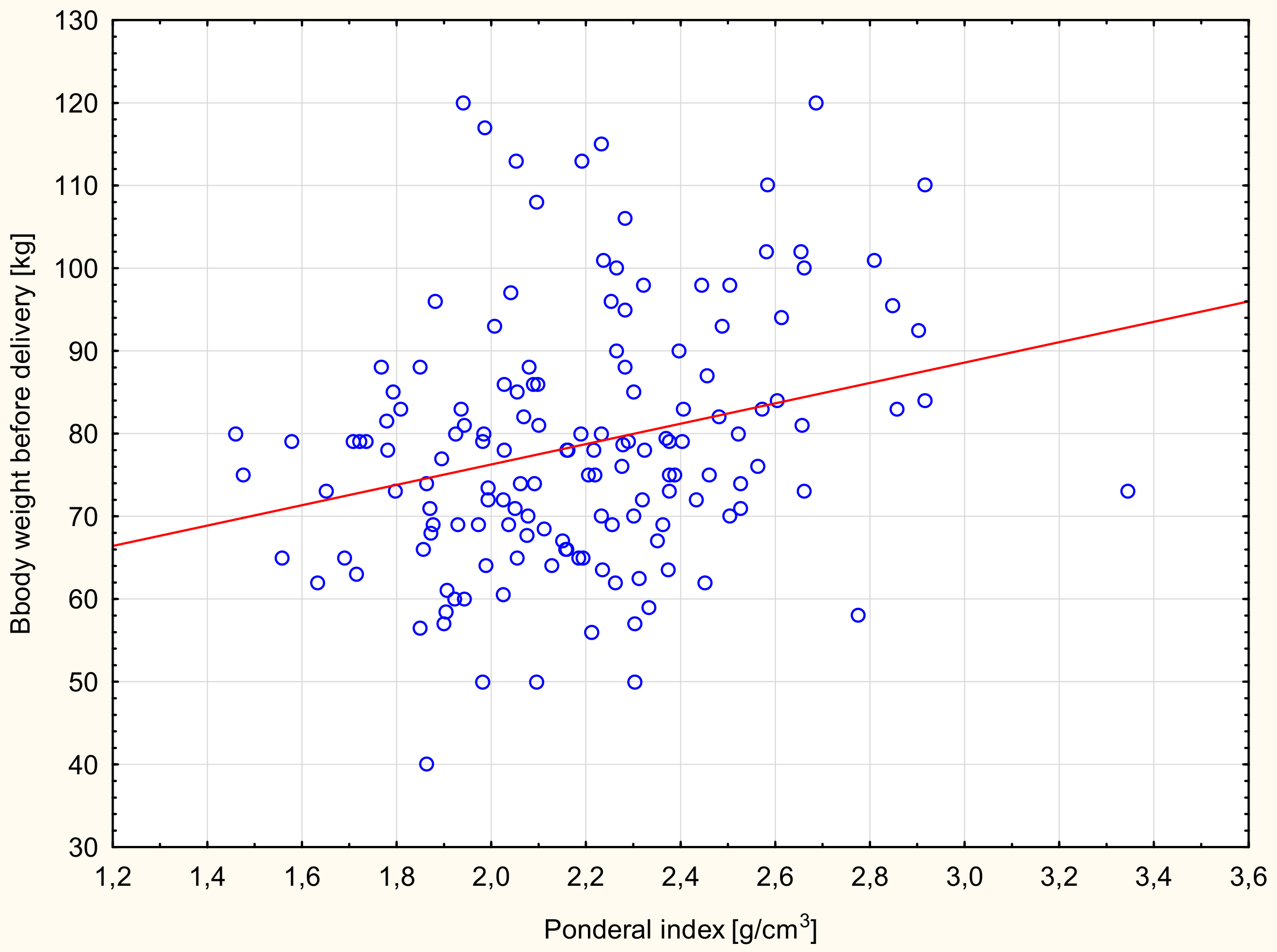
| Newborn weight (g) | BMI (kg/m2) | 0.449 | <0.001 | 0.14 |
| Body weight gain (kg) | 0.028 | 0.398 | ||
| Adiponectin (ng/mL) | −0.011 | 0.672 | ||
| Newborn weight (g) | BMI (kg/m2) | 0.510 | <0.001 | 0.15 |
| Body weight gain (kg) | 0.041 | 0.215 | ||
| Leptin (ng/mL) | −0.027 | 0.134 | ||
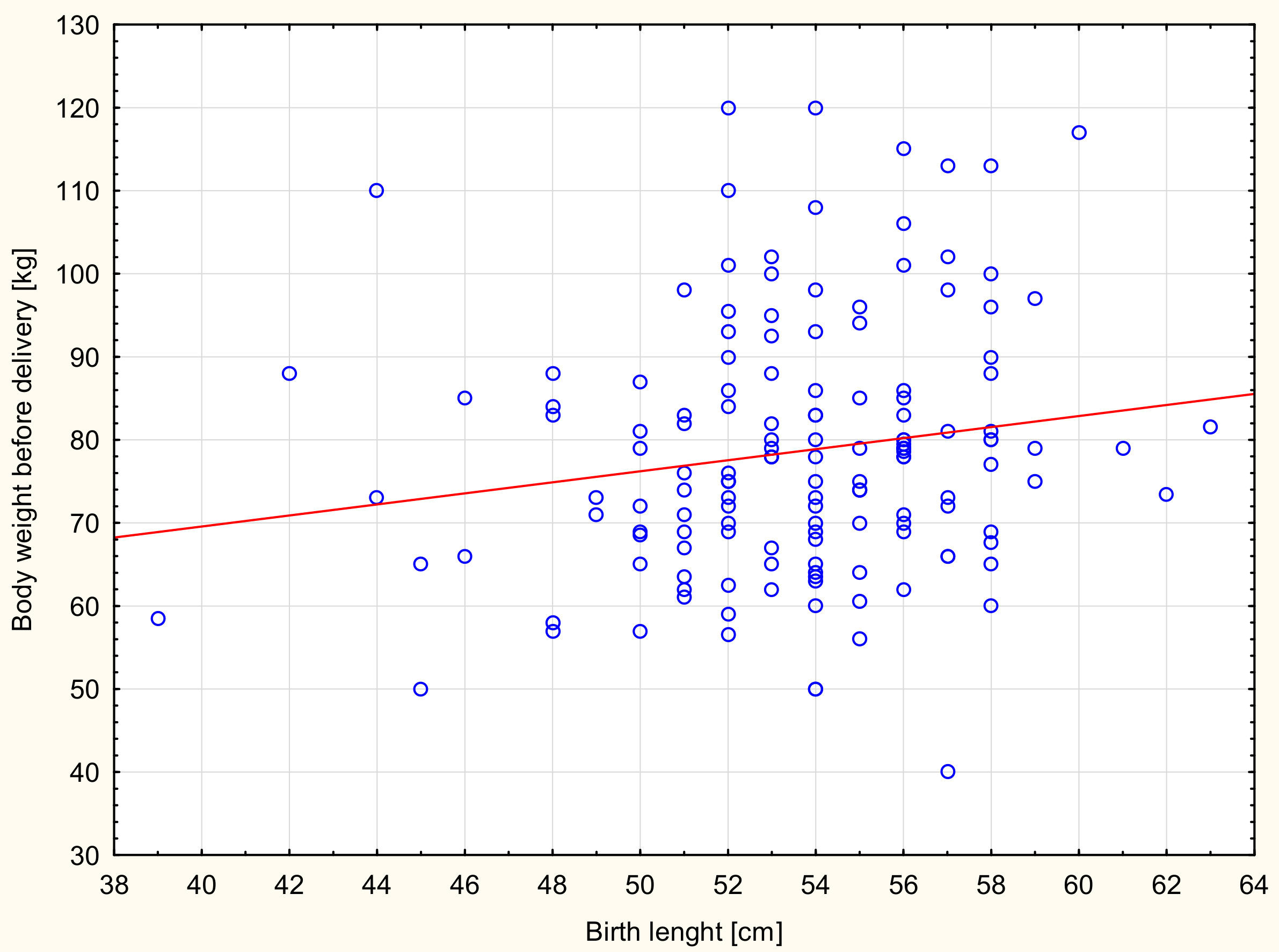
| Newborn length (cm) | Height (cm) | 0.208 | 0.168 | 0.02 |
| Adiponectin (ng/mL) | −0.004 | 0.645 | ||
| Leptin (ng/mL) | 0.005 | 0.422 | ||
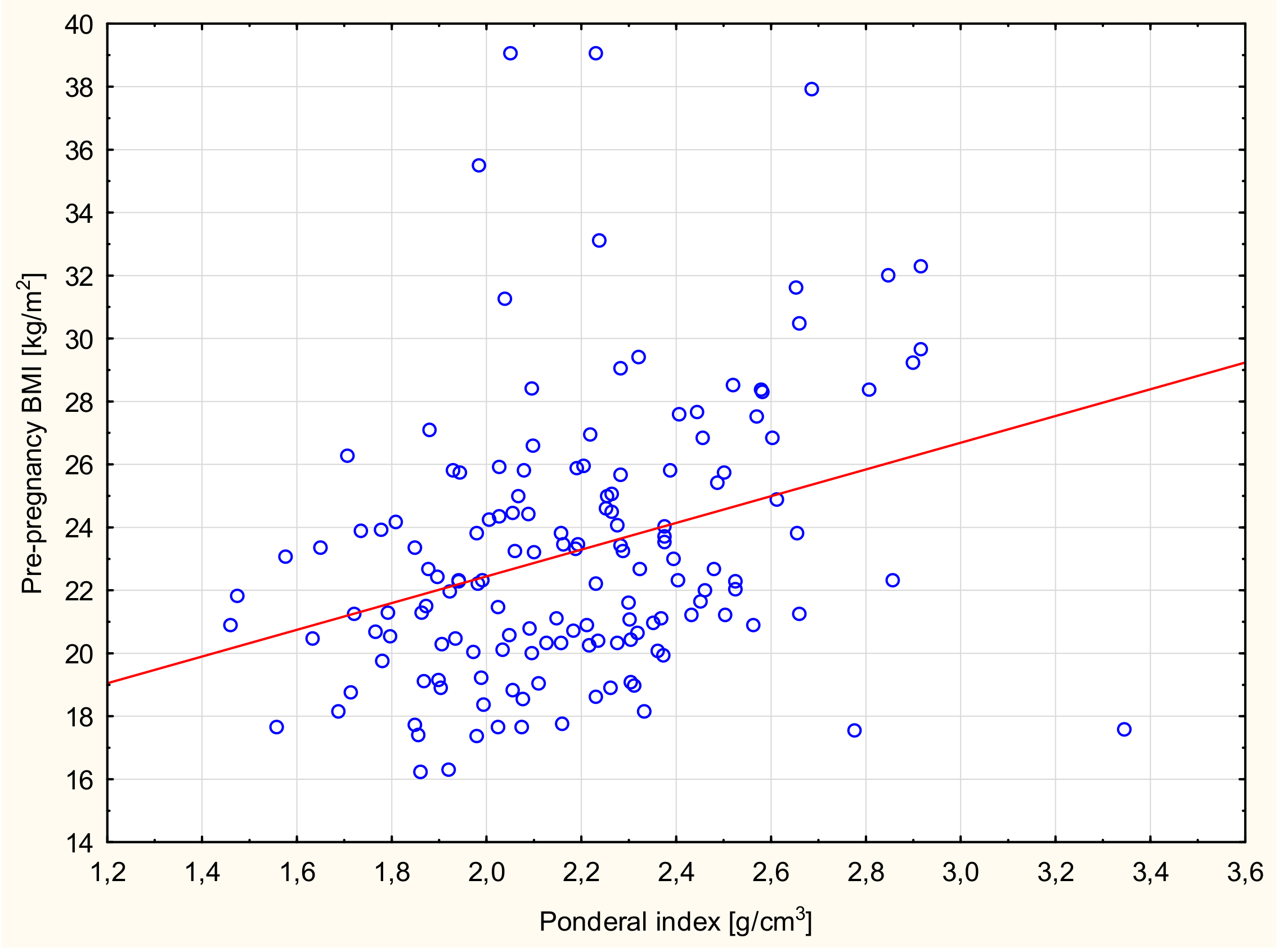
| Newborn Ponderal index | BMI (kg/m2) | 0.276 | <0.001 | 0.11 |
| Body weight gain (kg) | 0.025 | 0.296 | ||
| Adiponectin (ng/mL) | −0.009 | 0.639 | ||
| Newborn Ponderal index | BMI (kg/m2) | 0.335 | <0.001 | 0.14 |
| Body weight gain (kg) | 0.037 | 0.116 | ||
| Leptin (ng/mL) | −0.026 | 0.040 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazurek, D.; Bronkowska, M. Maternal Anthropometric Factors and Circulating Adipokines as Predictors of Birth Weight and Length. Int. J. Environ. Res. Public Health 2020, 17, 4799. https://doi.org/10.3390/ijerph17134799
Mazurek D, Bronkowska M. Maternal Anthropometric Factors and Circulating Adipokines as Predictors of Birth Weight and Length. International Journal of Environmental Research and Public Health. 2020; 17(13):4799. https://doi.org/10.3390/ijerph17134799
Chicago/Turabian StyleMazurek, Dominika, and Monika Bronkowska. 2020. "Maternal Anthropometric Factors and Circulating Adipokines as Predictors of Birth Weight and Length" International Journal of Environmental Research and Public Health 17, no. 13: 4799. https://doi.org/10.3390/ijerph17134799
APA StyleMazurek, D., & Bronkowska, M. (2020). Maternal Anthropometric Factors and Circulating Adipokines as Predictors of Birth Weight and Length. International Journal of Environmental Research and Public Health, 17(13), 4799. https://doi.org/10.3390/ijerph17134799





