Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation
2.2. Characterization
2.3. Adsorption Experiments
3. Results
3.1. Characterization of the Sorbent
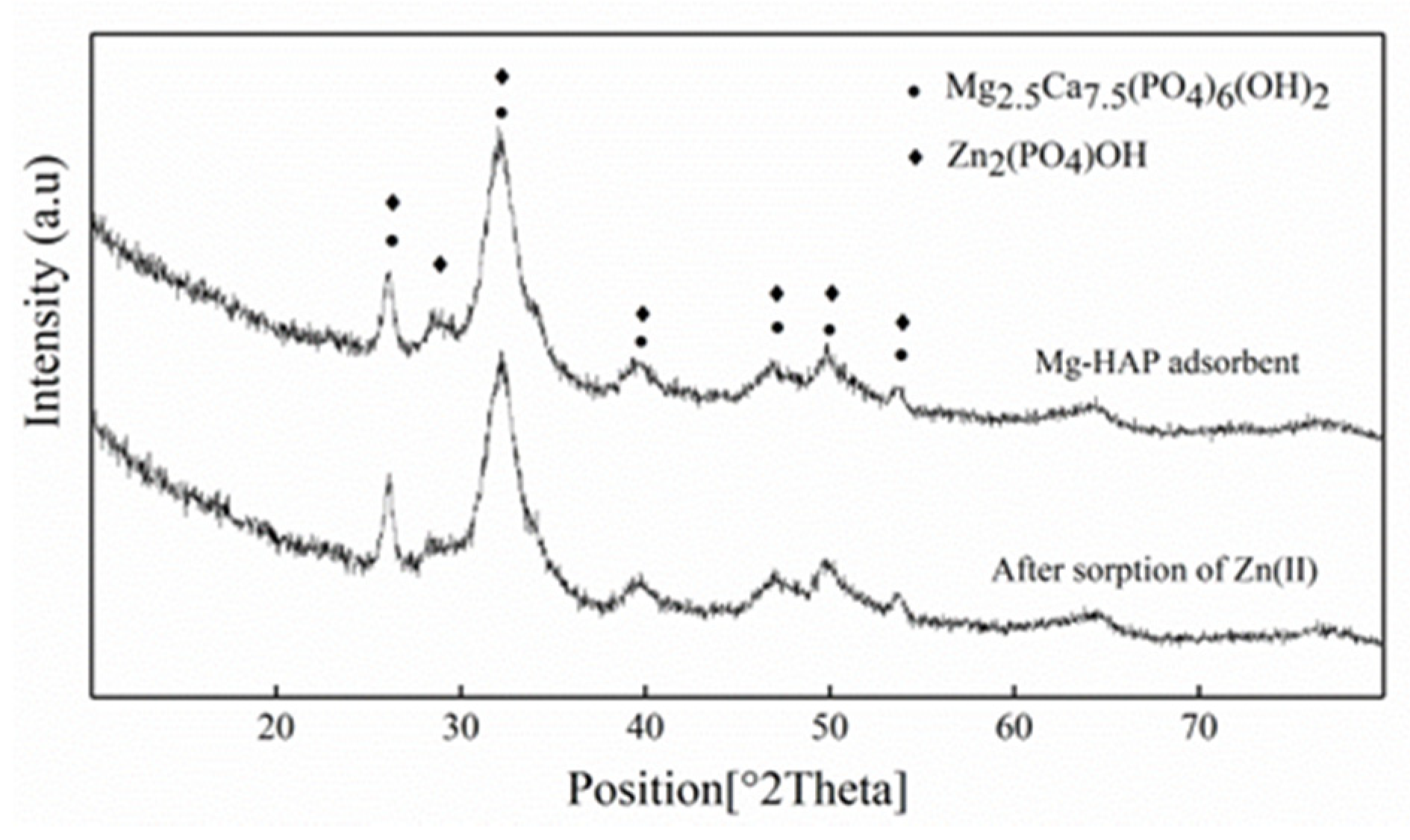
3.1.1. Powder X-ray Diffraction
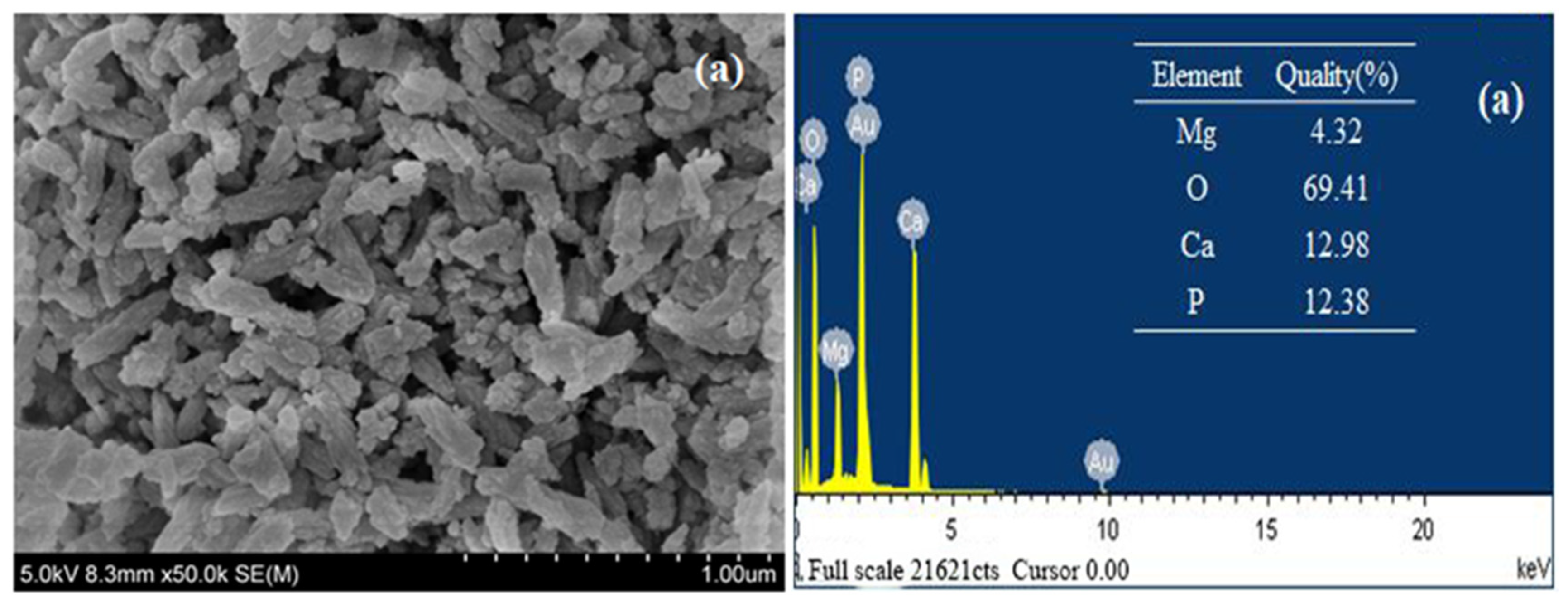
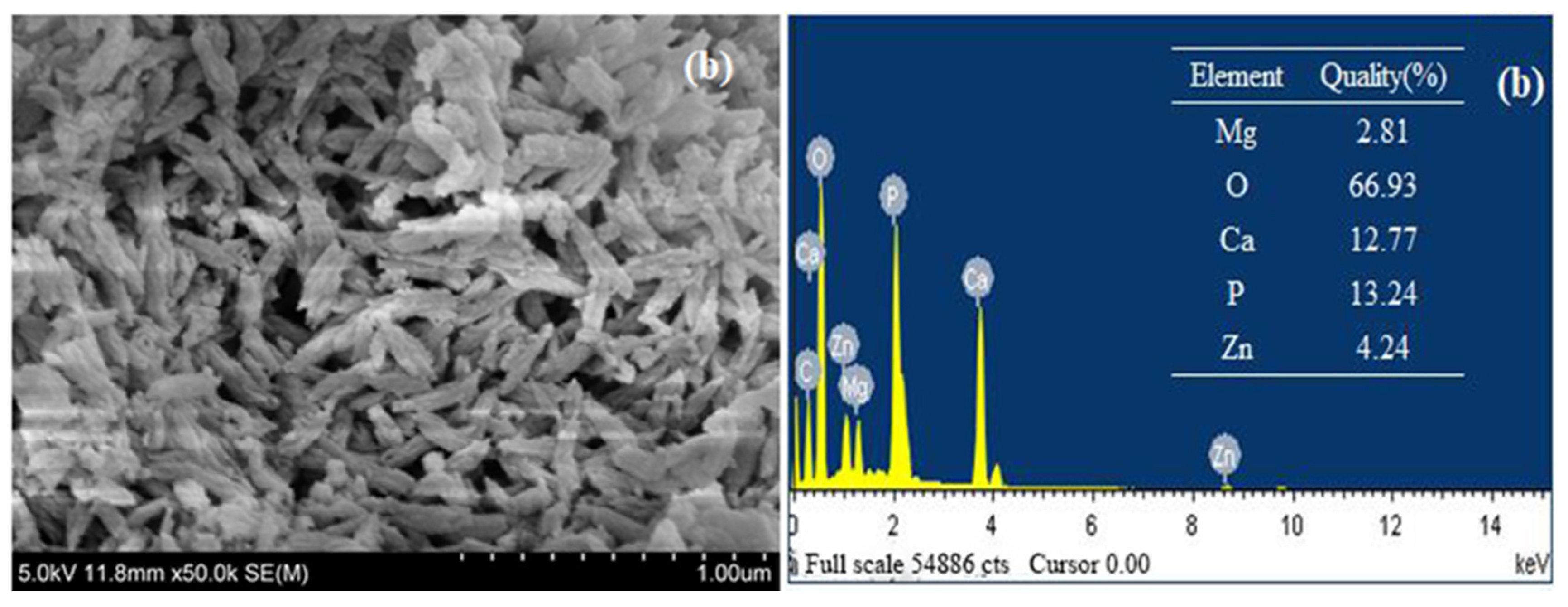
3.1.2. Scanning Electron Microscope (SEM) and Energy Dispersive Spectrum (EDS)
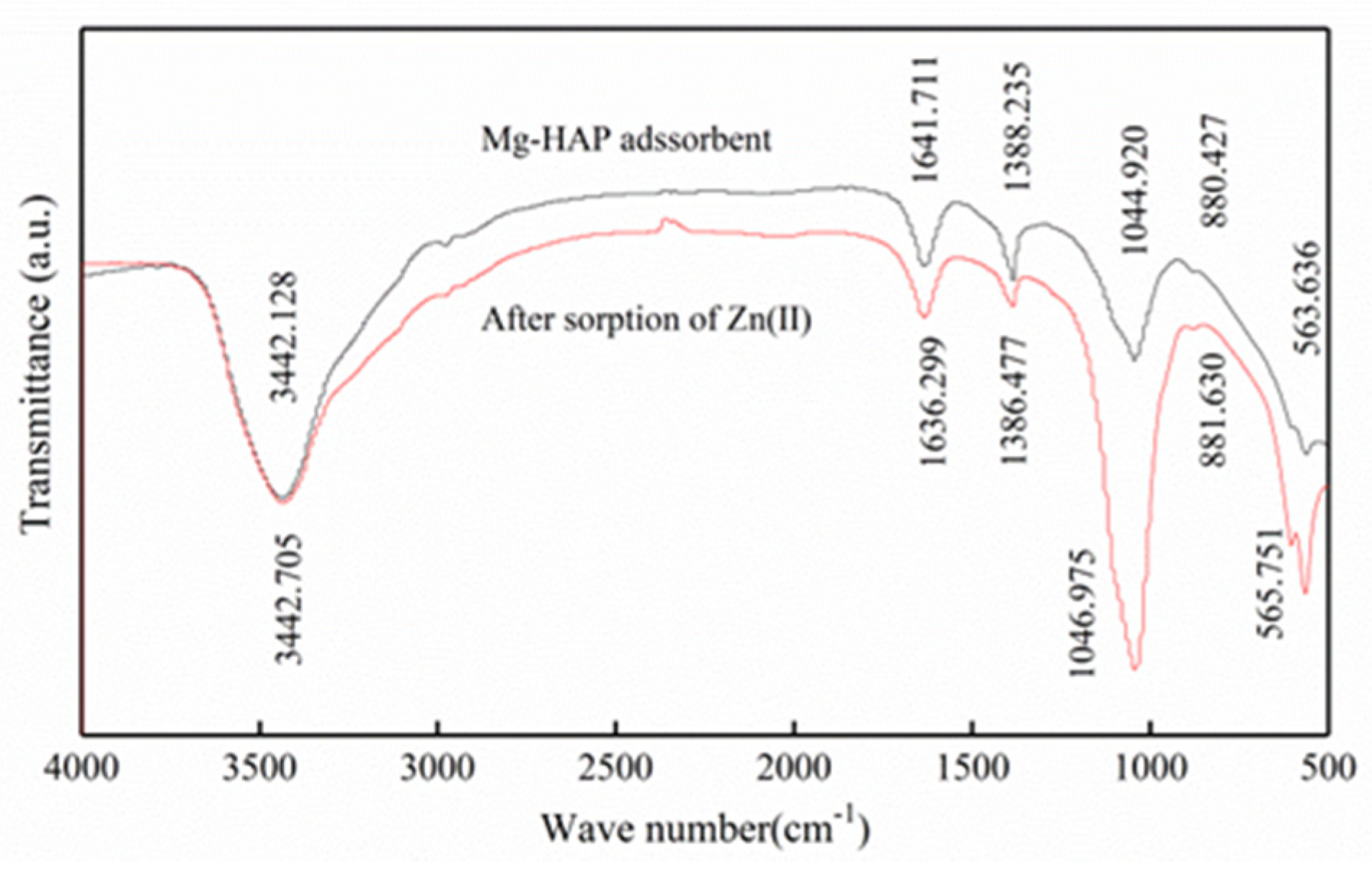
3.1.3. Fourier Transform Infrared Spectrometer (FTIR)
3.2. Influences of Adsorption Conditions
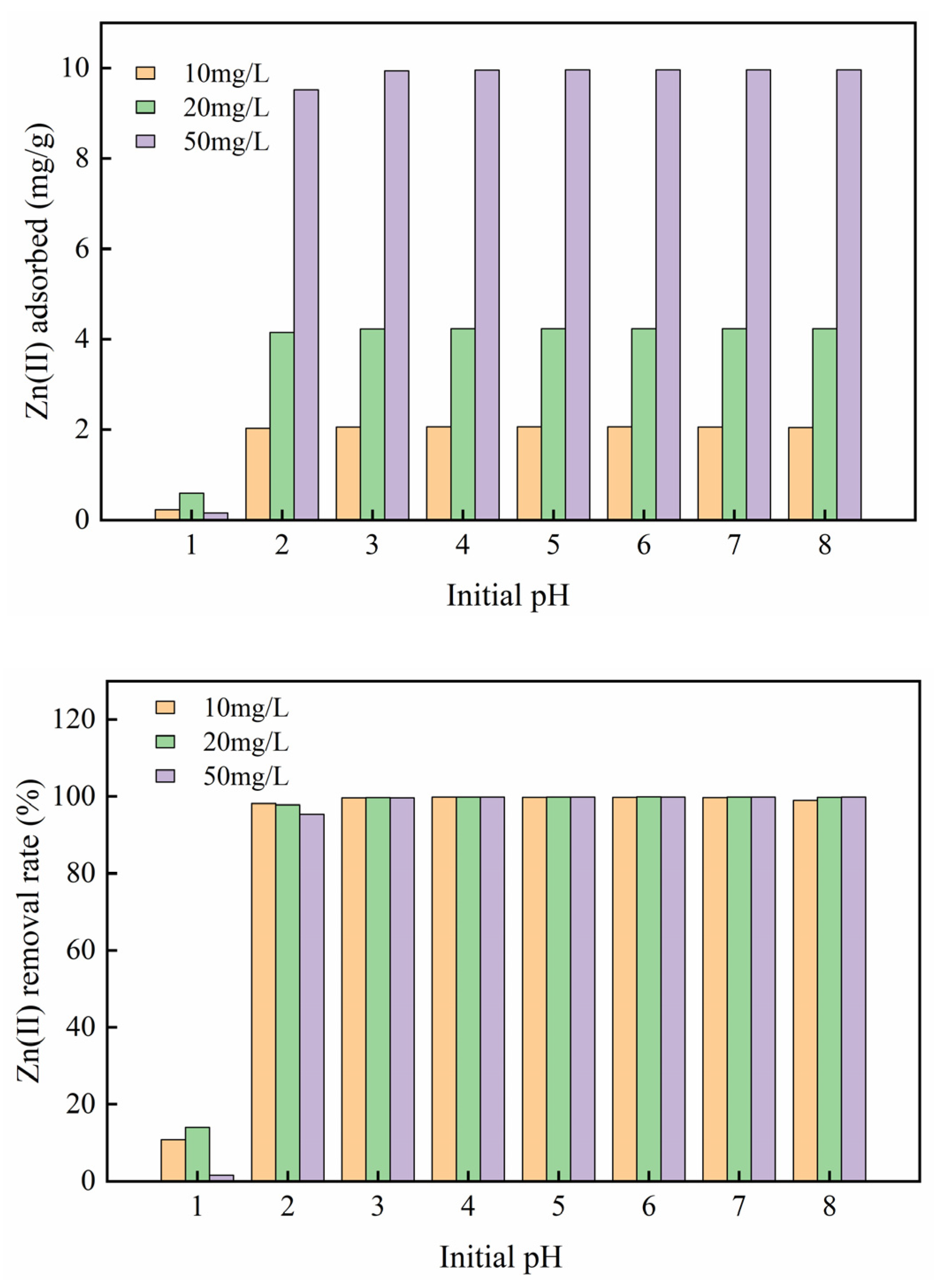
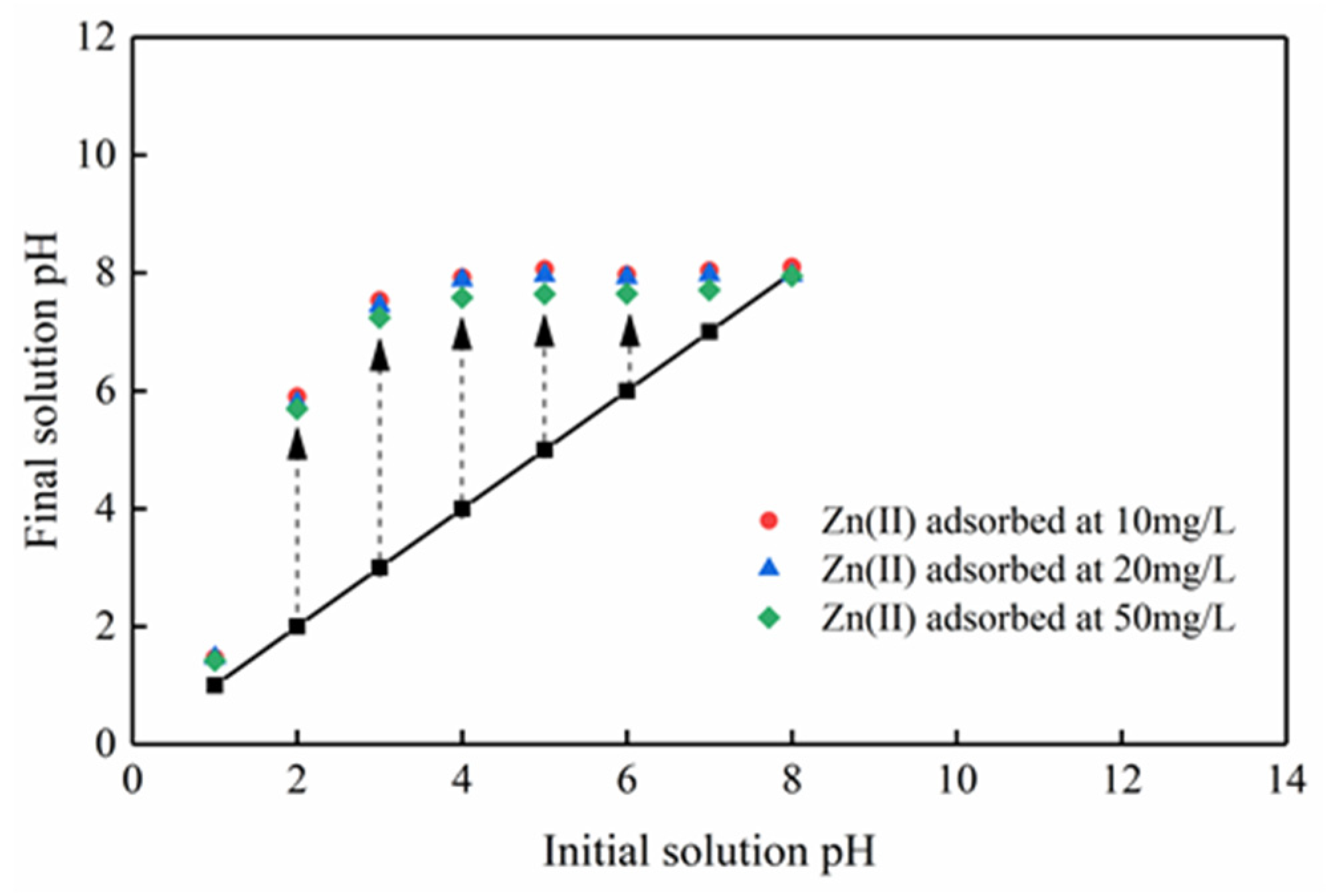
3.2.1. Influence of Initial Solution pH
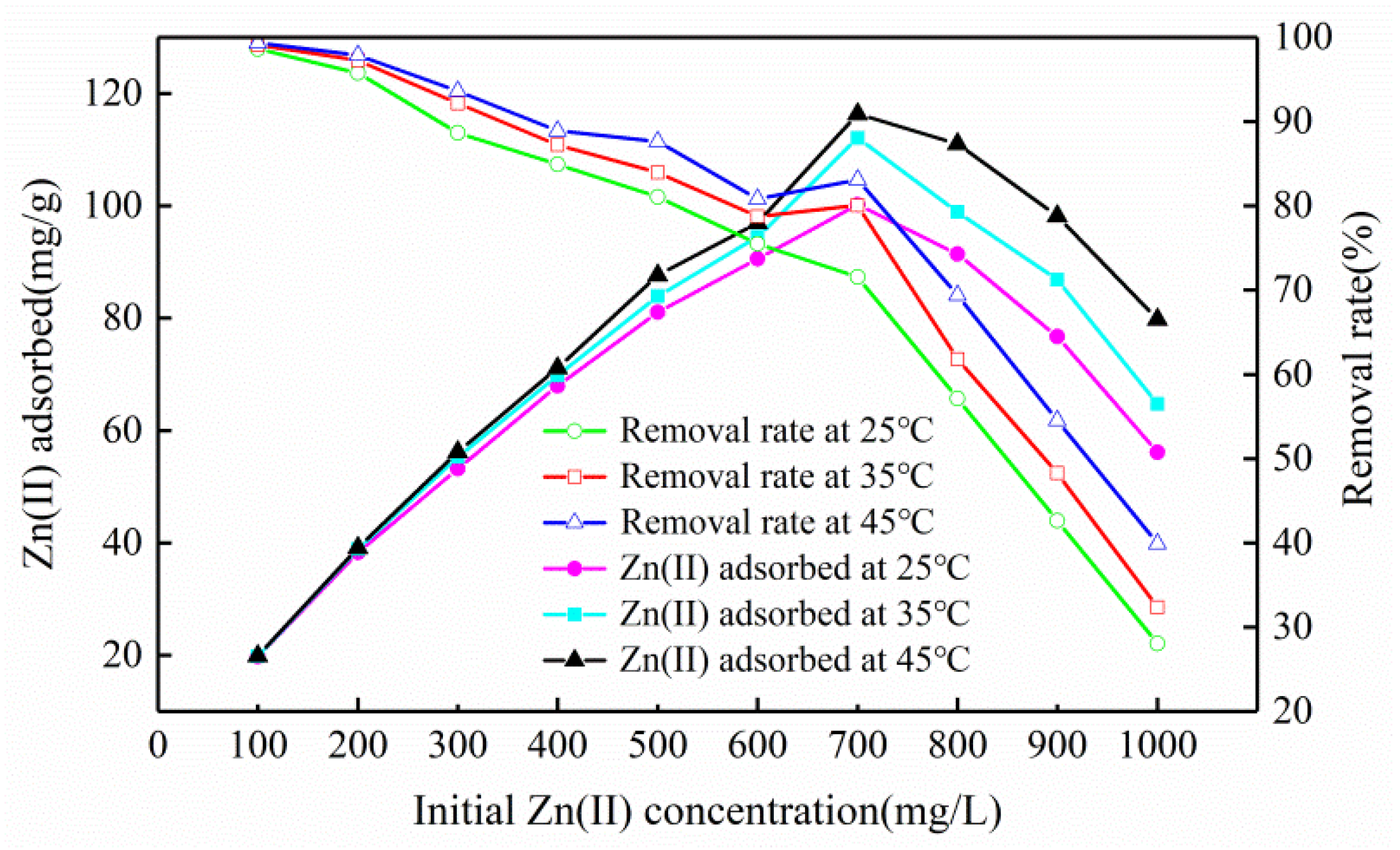
3.2.2. Influence of Initial Concentration and Temperature
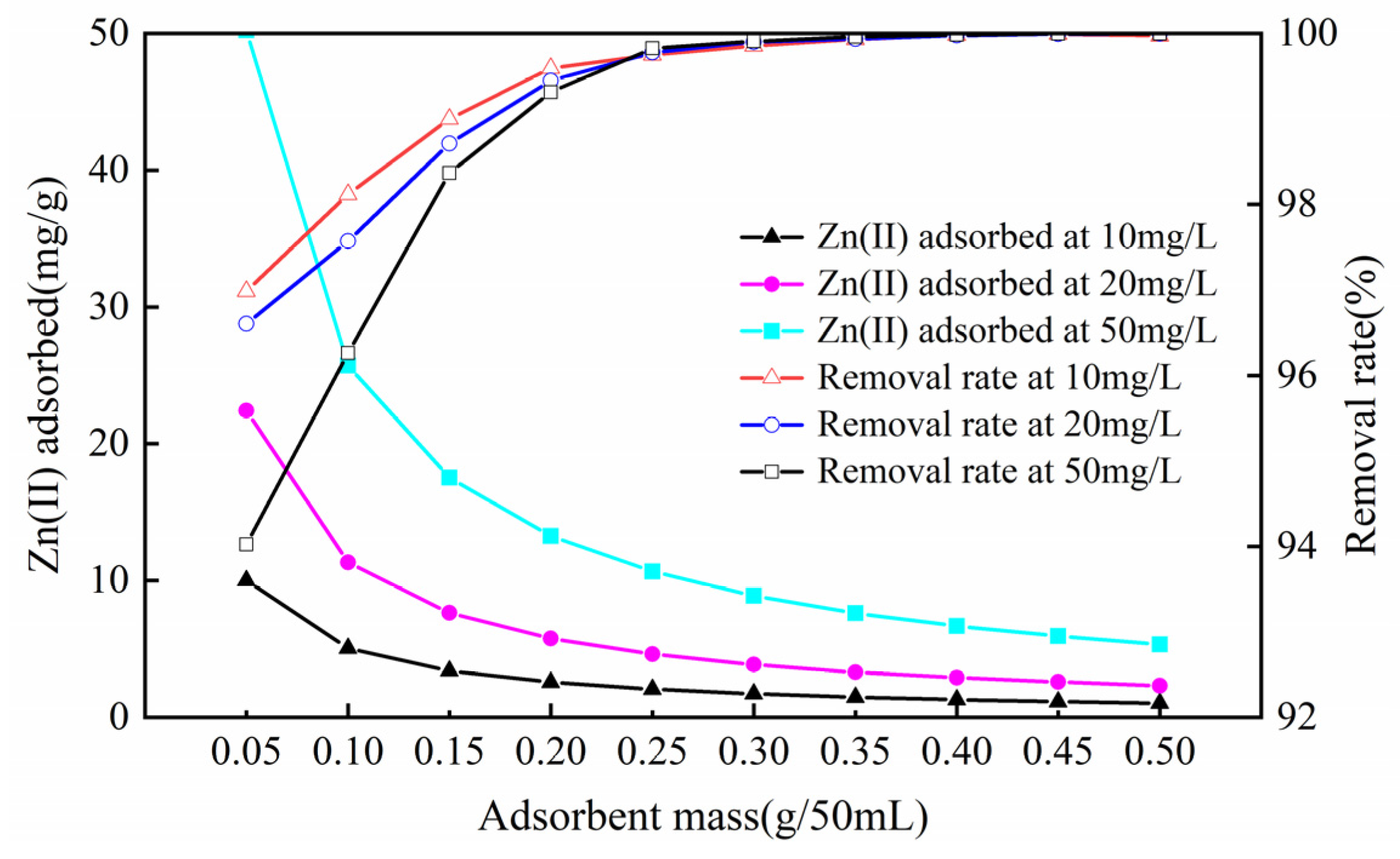
3.2.3. Influence of Sorbent Dosage
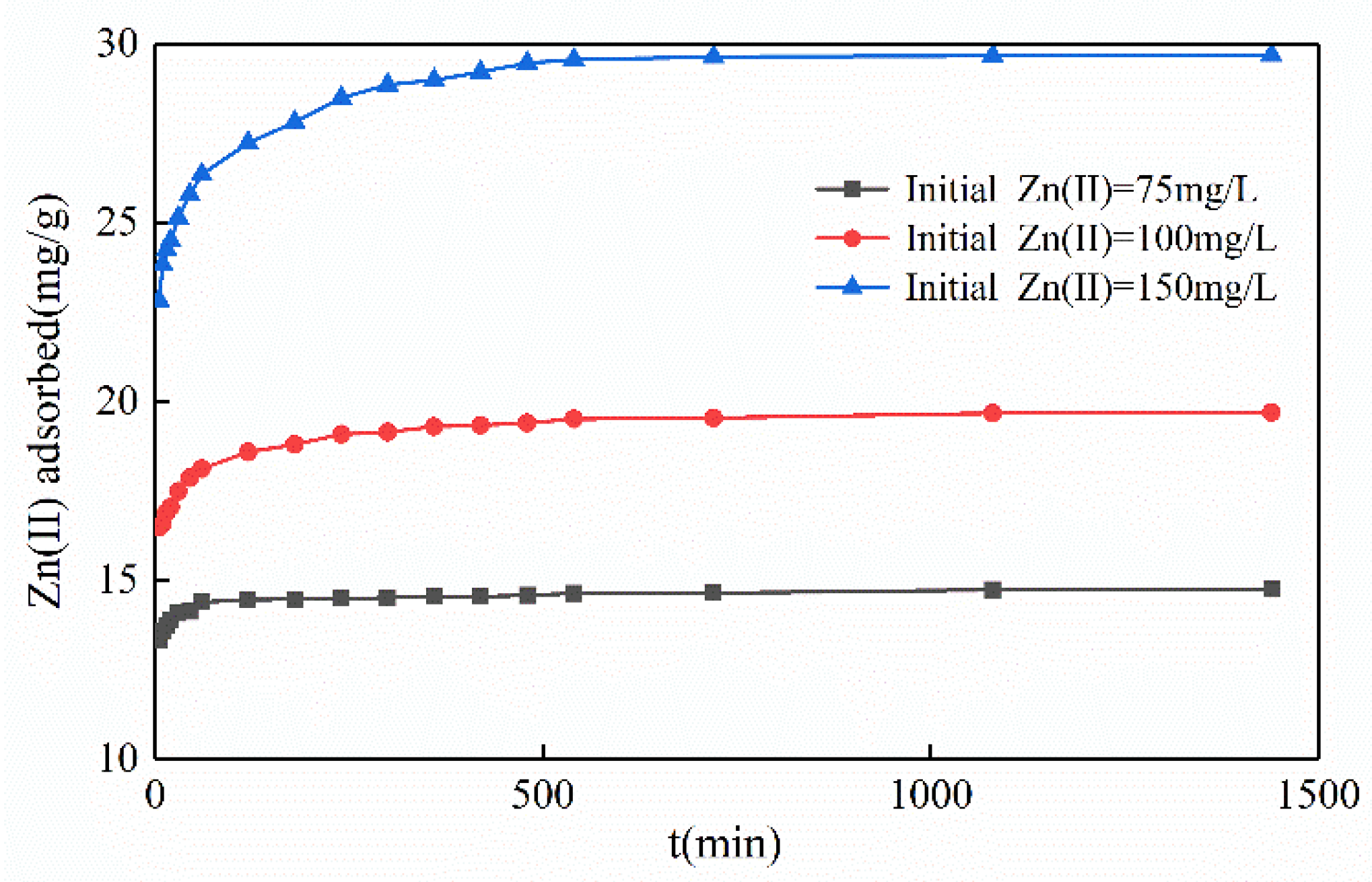
3.2.4. Influence of Contact Time
3.3. Sorption Dynamics and Isotherm
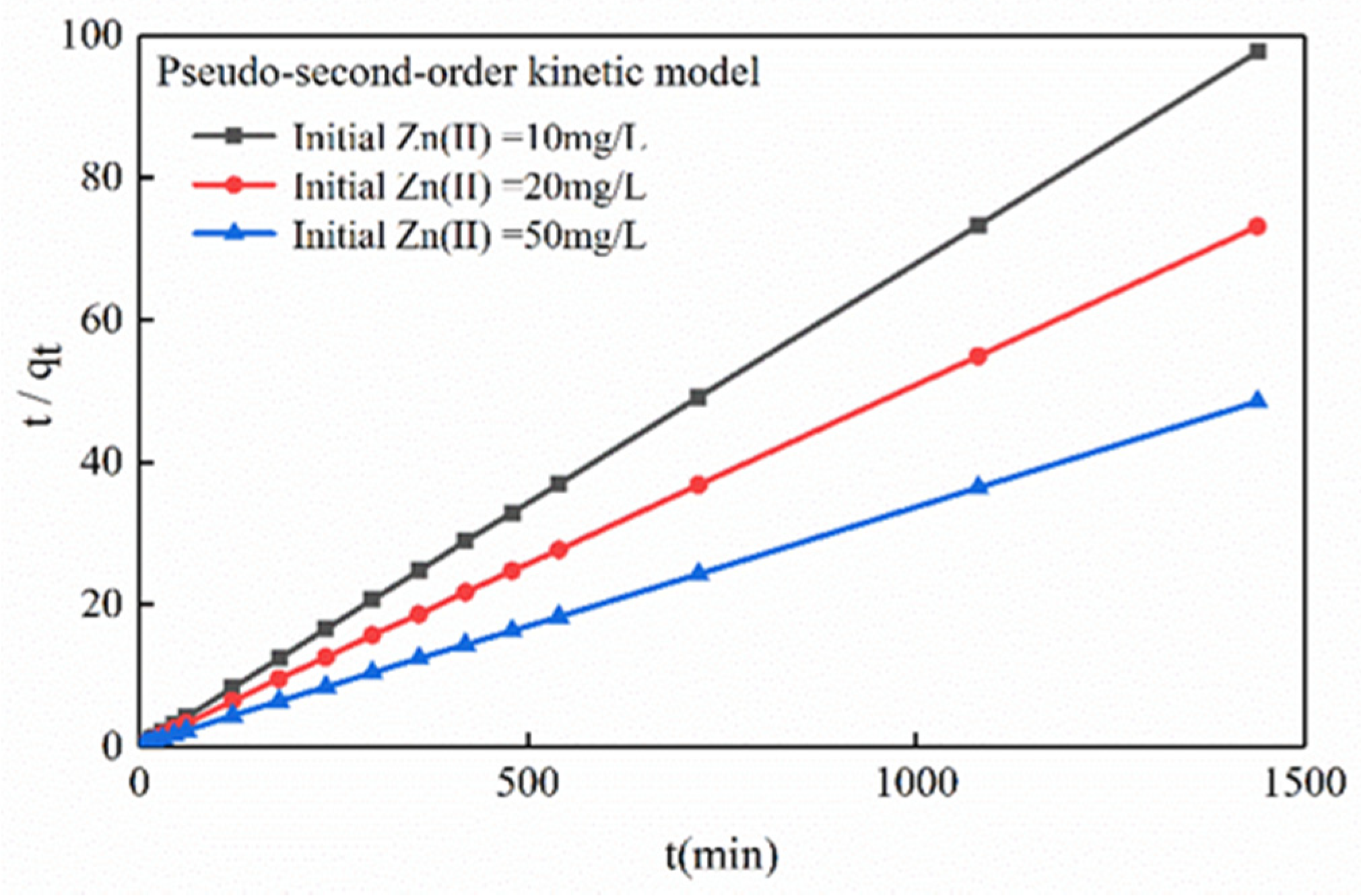
3.3.1. Sorption Dynamics
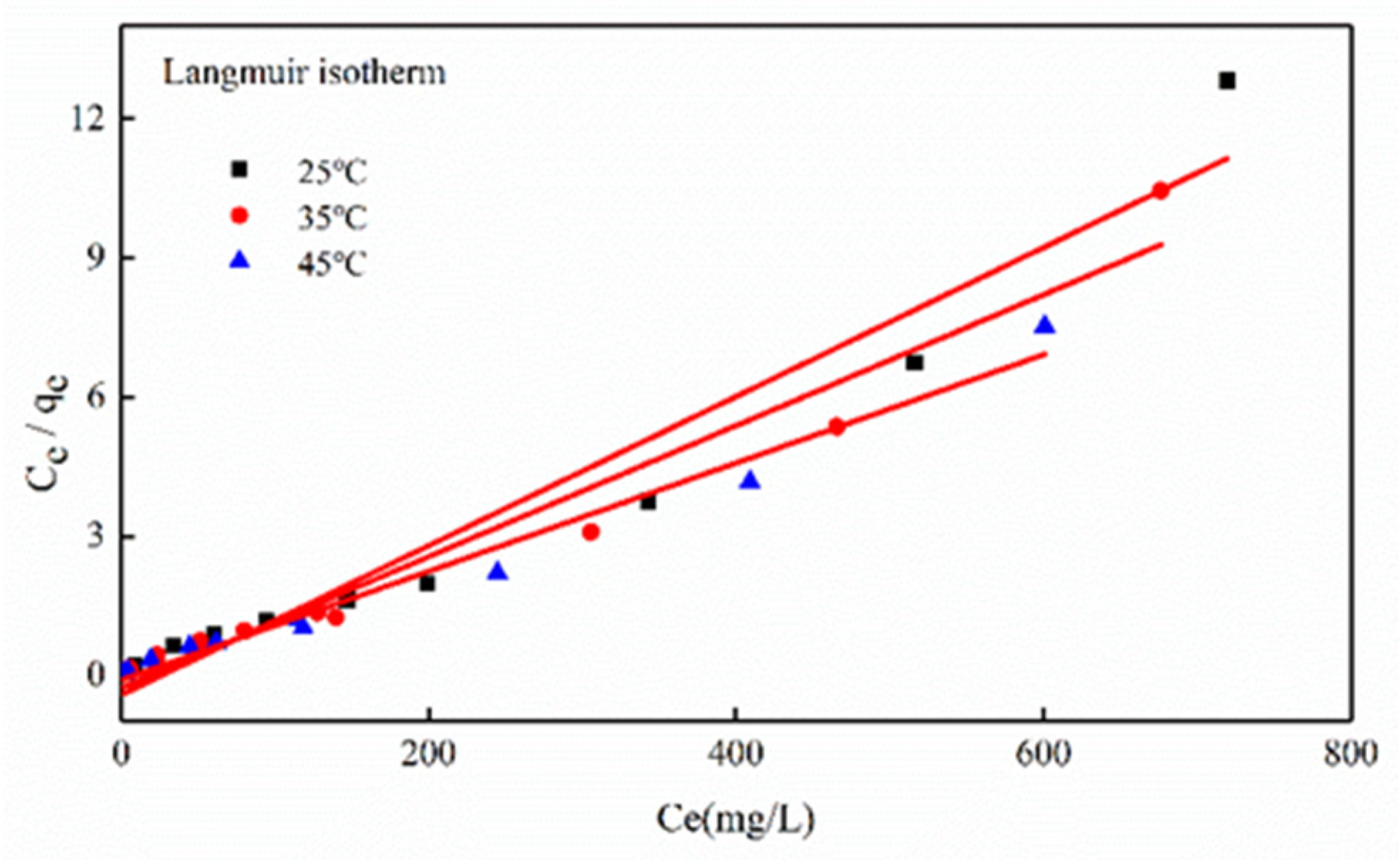
3.3.2. Adsorption Isotherm
3.4. Adsorption Thermodynamics
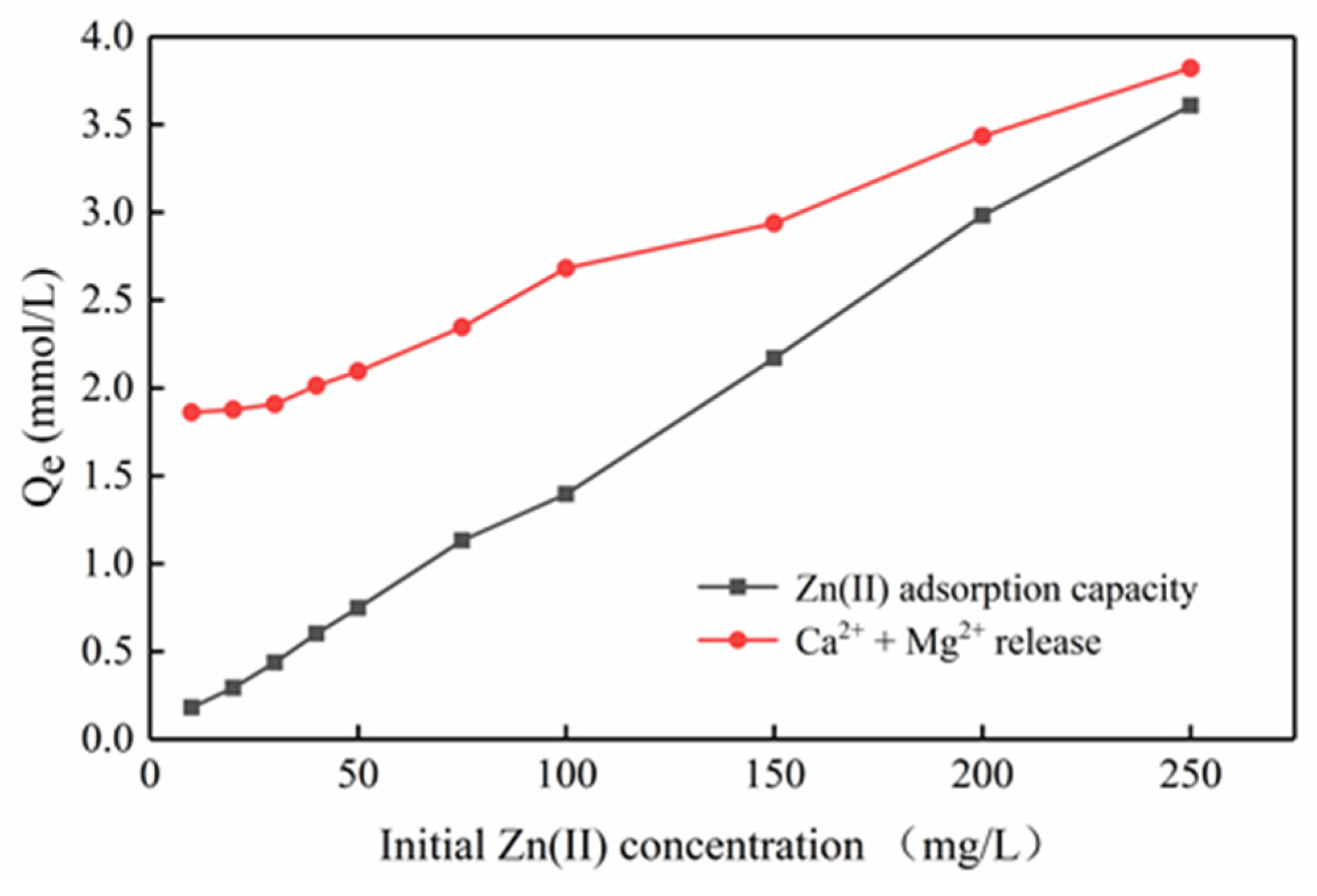
3.5. Adsorption Mechanisms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Du, X.D.; Wang, C.C.; Zhong, J.; Liu, J.G.; Li, Y.X.; Wang, P. Highly efficient removal of Pb2+ by a polyoxomolybdate-based organic-inorganic hybrid material {(4-Hap)4[Mo8O26]}. J. Environ. Chem. Eng. 2017, 5, 1866–1873. [Google Scholar] [CrossRef]
- Cherfi, A.; Abdoun, S.; Gaci, O. Food survey: Levels and potential health risks of chromium, lead, zinc and copper content in fruits and vegetables consumed in Algeria. Food Chem. Toxicol. 2014, 70, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Cherasse, Y.; Urade, Y. Dietary Zinc Acts as a Sleep Modulator. Int. J. Mol. Sci. 2017, 18, 2334. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Chang, W.; Wang, W.; Wu, H.; Pu, K.; Wu, A.; Qin, Z.; Tao, Y.; Yue, Z.; Wang, P.; et al. A novel tetrapeptide fluorescence sensor for early diagnosis of prostate cancer based on imaging Zn2+ in healthy versus cancerous cells. J. Adv. Res. 2020. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Hanauer, T.; Felix-Henningsen, P.; Steffens, D.; Kalandadze, B.; Navrozashvili, L.; Urushadze, T. In situ stabilization of metals (Cu, Cd, and Zn) in contaminated soils in the region of Bolnisi, Georgia. Plant Soil 2010, 341, 193–208. [Google Scholar] [CrossRef]
- Hu, C.; Liu, F.; Lan, H.; Liu, H.; Qu, J. Preparation of a manganese dioxide/carbon fiber electrode for electrosorptive removal of copper ions from water. J. Colloid Interface Sci. 2015, 446, 359–365. [Google Scholar] [CrossRef]
- Alqadami, A.A.; Naushad, M.; Abdalla, M.A.; Ahamad, T.; Alothman, Z.A.; Alshehri, S.M.; Ghfar, A.A. Efficient removal of toxic metal ions from wastewater using a recyclable nanocomposite: A study of adsorption parameters and interaction mechanism. J. Clean. Prod. 2017, 156, 426–436. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Siafaka, P.I.; Pavlidou, E.G.; Chrissafis, K.J.; Bikiaris, D.N. Synthesis and adsorption application of succinyl-grafted chitosan for the simultaneous removal of zinc and cationic dye from binary hazardous mixtures. Chem. Eng. J. 2015, 259, 438–448. [Google Scholar] [CrossRef]
- Prabhu, S.M.; Elanchezhiyan, S.S.; Lee, G.; Khan, A.; Meenakshi, S. Assembly of nano-sized hydroxyapatite onto graphene oxide sheets via in-situ fabrication method and its prospective application for defluoridation studies. Chem. Eng. J. 2016, 300, 334–342. [Google Scholar] [CrossRef]
- Gayathri, B.; Muthukumarasamy, N.; Velauthapillai, D.; Santhosh, S.B.; Vijayshankar, A. Magnesium incorporated hydroxyapatite nanoparticles: Preparation, characterization, antibacterial and larvicidal activity. Arab. J. Chem. 2018, 11, 645–654. [Google Scholar] [CrossRef]
- Gong, L.; Zhang, W.; Shen, Y. Magnesium substituted hydroxyapatite whiskers synthesis, characterization and bioactivity evaluation. R. Soc. Chem. Adv. 2016, 6, 114707–114713. [Google Scholar]
- Chen, L.; Zhang, K.S.; He, J.Y.; Xu, W.H.; Huang, X.J.; Liu, J.H. Enhanced fluoride removal from water by sulfate-doped hydroxyapatite hierarchical hollow microspheres. Chem. Eng. J. 2016, 285, 616–624. [Google Scholar] [CrossRef]
- Nie, Y.; Hu, C.; Kong, C. Enhanced fluoride adsorption using Al (III) modified calcium hydroxyapatite. J. Hazard. Mater. 2012, 233, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Leyva, A.; Marrero, J.; Michowski, P.; Daniel, C. Sorption of Antimony onto Hydroxyapatite. Environ. Sci. Technol. 2001, 35, 3669–3675. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Liu, Y.X.; Lu, H.H.; Yang, R.Q.; Yang, S.M. Competitive adsorption of Pb(II), Cu(II), and Zn(II) ions onto hydroxyapatite-biochar nanocomposite in aqueous solutions. J. Solid State Chem. 2018, 261, 53–61. [Google Scholar] [CrossRef]
- Zhu, Y.N.; Zhu, Z.Q.; Zhao, X.; Liang, Y.P.; Dai, L.Q.; Huang, Y.H. Characterization, dissolution and solubility of cadmium–calcium hydroxyapatite solid solutions at 25 °C. Chem. Geol. 2016, 423, 34–48. [Google Scholar] [CrossRef]
- Yasukawa, A.; Yokoyama, T.; Kandori, K.; Ishikawa, T. Ion-exchange of magnesium–calcium hydroxyapatite solid solution particles with Cd2+ ion. Colloids Surf. A Physicochem. Eng. Asp. 2008, 317, 123–128. [Google Scholar] [CrossRef]
- Feng, Y.; Gong, J.L.; Zeng, G.M.; Niu, Q.Y.; Zhang, H.Y.; Niu, C.G.; Deng, J.H.; Yan, M. Adsorption of Cd (II) and Zn (II) from aqueous solutions using magnetic hydroxyapatite nanoparticles as adsorbents. Chem. Eng. J. 2010, 162, 487–494. [Google Scholar] [CrossRef]
- Yasukawa, A.; Yokoyama, T.; Kandori, K.; Ishikawa, T. Ion-exchange property and mechanism of magnesium–calcium hydroxyapatite solid solutions. Colloids Surf. A Physicochem. Eng. Asp. 2004, 238, 133–139. [Google Scholar] [CrossRef]
- Ramanan, S.R.; Venkatesh, R. A study of hydroxyapatite fibers prepared via sol–gel route. Mater. Lett. 2004, 58, 3320–3323. [Google Scholar] [CrossRef]
- Jia, J.F.; Zhou, H.J.; Wei, J.; Jiang, X.; Hua, H.; Chen, F.P.; Wei, S.C.; Shin, J.W.; Liu, C.S. Development of magnesium calcium phosphate biocement for bone regeneration. J. R. Soc. Interface 2010, 7, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Lv, Y.; Song, H. The morphological evolution of hydroxyapatite on high-efficiency Pb2+ removal and antibacterial activity. Microchem. J. 2017, 135, 16–25. [Google Scholar] [CrossRef]
- Zhu, Y.N.; Jiang, Y.H.; Zhu, Z.Q.; Deng, H.; Ding, H.; Li, Y.H.; Zhang, L.H.; Lin, J. Preparation of a porous hydroxyapatite-carbon composite with the bio-template of sugarcane top stems and its use for the Pb(II) removal. J. Clean. Prod. 2018, 187, 650–661. [Google Scholar] [CrossRef]
- Mobasherpour, I.; Salahi, E.; Pazouki, M. Removal of divalent cadmium cations by means of synthetic nano crystallite hydroxyapatite. Desalination 2011, 266, 142–148. [Google Scholar] [CrossRef]
- Deng, H.; Zhu, Z.Q.; Zhu, Y.N.; Ding, H.; Li, Y.H.; Lin, J.; Zhang, L.H. Removal of Cd(II) from aqueous solution by a hierarchical porous hydroxylapatite-carbon composite prepared with the biotemplate of stalk internodes of sugarcane tops. Desalin. Water Treat. 2018, 136, 341–355. [Google Scholar] [CrossRef]
- Zhu, H.Y.; Jiang, R.; Xiao, L.; Zeng, G.M. Preparation, characterization, adsorption kinetics and thermodynamics of novel magnetic chitosan enwrapping nanosized gamma-Fe2O3 and multi-walled carbon nanotubes with enhanced adsorption properties for methyl orange. Bioresour. Technol. 2010, 101, 5063–5069. [Google Scholar] [CrossRef]
- Pavagadhi, S.; Tang, A.L.L.; Sathishkumar, M.; PingLoh, k.; Balasubramanian, R. Removal of microcystin-LR and microcystin-RR by graphene oxide: Adsorption and kinetic experiments. Water Res. 2013, 47, 4621–4629. [Google Scholar] [CrossRef]
- Singh, R.; Misra, V.; Singh, R.P. Removal of hexavalent chromium from contaminated ground water using zero-valent iron nanoparticles. Environ. Monit. Assess. 2011, 184, 3643–3651. [Google Scholar] [CrossRef]
- Alshameri, A.; Yan, C.; Al-Ani, Y.; Dawood, A.S.; Ibrahim, A.; Zhou, C.; Wang, H. An investigation into the adsorption removal of ammonium by salt activated Chinese (Hulaodu) natural zeolite: Kinetics, isotherms, and thermodynamics. J. Taiwan Inst. Chem. Eng. 2014, 45, 554–564. [Google Scholar] [CrossRef]
- Chung, H.K.; Kim, W.H.; Park, J.; Cho, J.; Jeong, T.Y.; Park, P.K. Application of Langmuir and Freundlich isotherms to predict adsorbate removal efficiency or required amount of adsorbent. J. Ind. Eng. Chem. 2015, 28, 241–246. [Google Scholar] [CrossRef]
- Chen, H.; Zhao, J.; Dai, G.L.; Wu, J.Y.; Yan, H. Adsorption characteristics of Pb(II) from aqueous solution onto a natural biosorbent, fallen Cinnamomum camphora leaves. Desalination 2010, 262, 174–182. [Google Scholar] [CrossRef]
- Sheha, R. Sorption behavior of Zn(II) ions on synthesized hydroxyapatites. J. Colloid Interface Sci. 2007, 310, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulos, E.; Rossi, A.M.; Costa, A.M.; Perez, C.A.; Moreira, J.C.; Saldanha, M. Studies on the mechanisms of lead immobilization by hydroxyapatite. Environ. Sci. Technol. 2002, 36, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
| Model | Initial Concentration (mg/L) | Equation | qe (mg/g) | R2 |
|---|---|---|---|---|
| Pseudo-First-Order Dynamics | 10 | ln(qe − qt) = −0.0064t + 0.2832 | 1.3274 | 0.8729 |
| 20 | ln(qe − qt) = −0.0068t + 1.3503 | 3.8586 | 0.8695 | |
| 50 | ln(qe − qt) = −0.0017t + 1.7492 | 5.7500 | 0.8688 | |
| Pseudo-Second-Order Dynamics | 10 | t/q = 0.0678t + 0.1822 | 14.75 | 1.0000 |
| 20 | t/q = 0.0507t + 0.2270 | 19.72 | 1.0000 | |
| 50 | t/q = 0.0335t + 0.2313 | 29.85 | 0.9999 | |
| Morrist Particle Intimal Diffusion | 10 | qt = 0.0328t1/2 + 13.807 | - | 0.6803 |
| 20 | qt = 0.0953t1/2 + 17.027 | - | 0.7976 | |
| 50 | qt = 0.2357t1/2 + 23.888 | - | 0.7883 | |
| Elovich Equation | 10 | qt = 0.2348lnt + 13.165 | - | 0.9249 |
| 20 | qt = 0.6561lnt + 15.292 | - | 0.9769 | |
| 50 | qt = 1.3631 lnt + 20.677 | - | 0.9783 |
| Pseudo-Second-Order Constants | ||||
|---|---|---|---|---|
| Initial Zn(II) Concentration (mg/L) | k2 (g/(mg·min)) | h (mg/(g·min)) | qe (mg/g) | R2 |
| 10 | 0.1822 | 39.6399 | 14.75 | 1.0000 |
| 20 | 0.2770 | 107.7193 | 19.72 | 1.0000 |
| 50 | 0.2313 | 206.0935 | 29.85 | 0.9999 |
| Langmuir Constants | ||||
|---|---|---|---|---|
| Temperature (°C) | qm (mg/g) | KL (L/mg) | RL | R2 |
| 25 | 62.11 | 0.0378 | 0.0065−0.1634 | 0.9677 |
| 35 | 70.92 | 0.0520 | 0.0047−0.1241 | 0.9608 |
| 45 | 85.47 | 0.1175 | 0.0039−0.1047 | 0.9821 |
| T (k) | ΔGθ (KJ/mol) | ΔHθ (KJ/mol) | ΔSθ (KJ/(mol·K)) |
|---|---|---|---|
| 298 | −10.399 | 20.2018 | 0.1028 |
| 308 | −11.573 | ||
| 318 | −12.456 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mo, N.; Zhu, Z.; Zhu, Y.; Liu, Y.; Wang, X.; Yang, H.; Zhao, N. Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation. Int. J. Environ. Res. Public Health 2020, 17, 3804. https://doi.org/10.3390/ijerph17113804
Mo N, Zhu Z, Zhu Y, Liu Y, Wang X, Yang H, Zhao N. Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation. International Journal of Environmental Research and Public Health. 2020; 17(11):3804. https://doi.org/10.3390/ijerph17113804
Chicago/Turabian StyleMo, Nan, Zongqiang Zhu, Yinian Zhu, Yang Liu, Xingxing Wang, Hongqu Yang, and Ningning Zhao. 2020. "Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation" International Journal of Environmental Research and Public Health 17, no. 11: 3804. https://doi.org/10.3390/ijerph17113804
APA StyleMo, N., Zhu, Z., Zhu, Y., Liu, Y., Wang, X., Yang, H., & Zhao, N. (2020). Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation. International Journal of Environmental Research and Public Health, 17(11), 3804. https://doi.org/10.3390/ijerph17113804






