The Exponential Phase of the Covid-19 Pandemic in Central Italy: An Integrated Care Pathway
Abstract
1. Introduction
2. Materials and Methods
3. Results
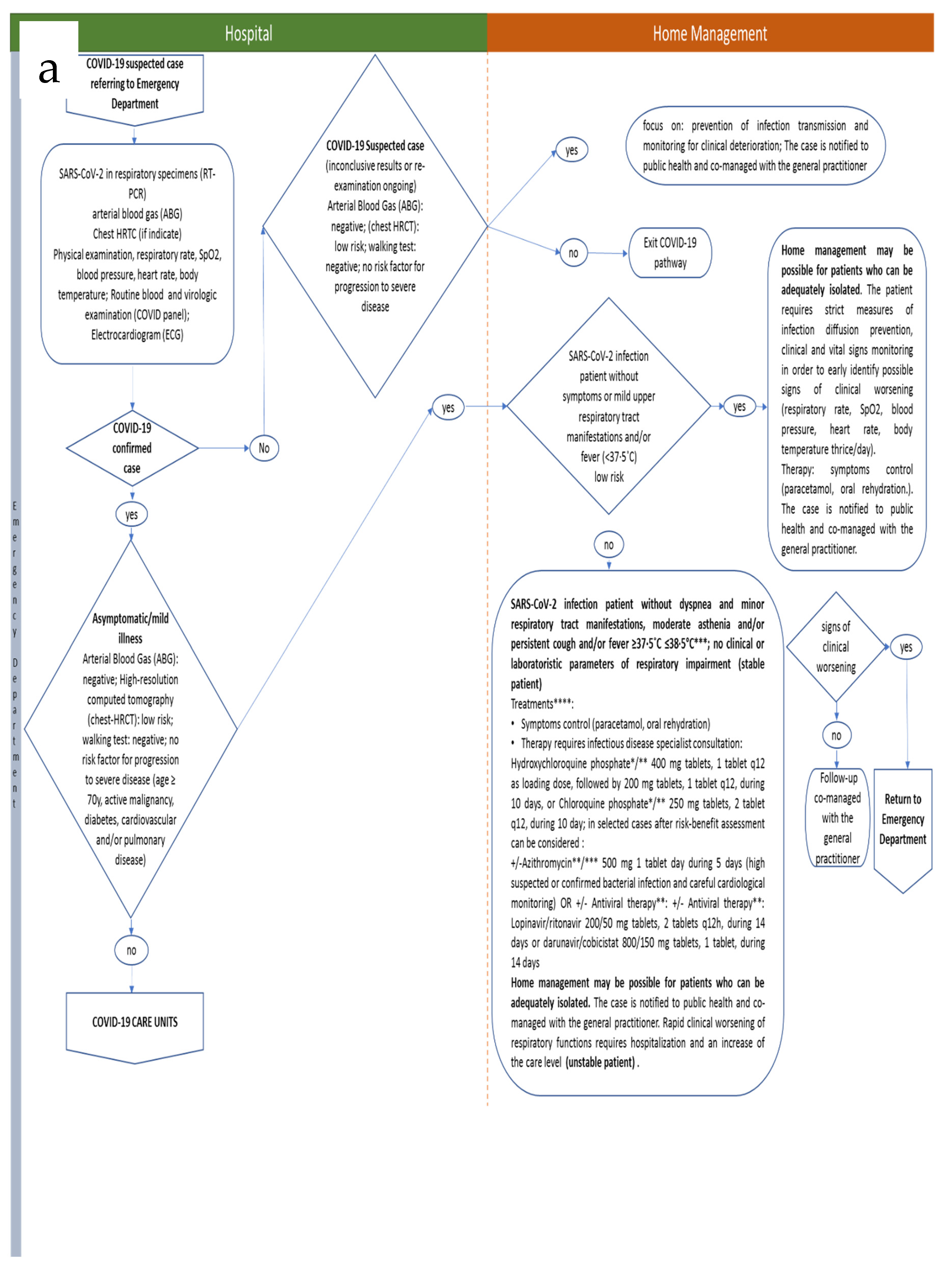
3.1. Integrated Care Pathway
3.2. Preliminary Assessment of the Integrated Care Pathway
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.S.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, X.; Wu, P.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Grasselli, G.; Pesenti, A.; Cecconi, M. Critical Care Utilization for the COVID-19 Outbreak in Lombardy, Italy: Early Experience and Forecast During an Emergency Response. JAMA 2020, 323, 1545–1546. [Google Scholar] [CrossRef] [PubMed]
- Onder, G.; Rezza, G.; Brusaferro, S. Case-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in Italy. JAMA 2020, 323, 1775–1776. [Google Scholar] [CrossRef] [PubMed]
- Nuovo Coronavirus. Available online: www.salute.gov.it (accessed on 8 April 2020).
- COVID-19 Dashboard. Available online: https://coronaboard.kr/en/ (accessed on 13 April 2020).
- Ministero Della Salute. Available online: www.salute.gov.it/imgs/C_17_notizie_4454_1_update (accessed on 8 April 2020).
- Transformer. Available online: http://bioinformatics.charite.de/transformer/ (accessed on 13 April 2020).
- COVID-19 Drug Interactions. Available online: www.covid19-druginteractions (accessed on 13 April 2020).
- Drug-PIN Personalized Interacions Network. Available online: www.drug-pin.com (accessed on 13 April 2020).
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 13. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruan, R.; Song, B.; Cai, Y.; Wei, M.; et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe Covid-19. N. Engl. J. Med. 2020, 382, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.S.; Zhou, M.; Dong, X.; Qu, P.J.; Gong, F.; Han, Y.; Qiu, P.Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Barnaby, D.P.; Becker, L.B.; Chelico, G.D.; Choen, S.L.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 22. [Google Scholar] [CrossRef] [PubMed]
- Du, R.H.; Liang, L.R.; Yang, C.Q.; Wang, W.; Cao, T.Z.; Li, M.; Guo, J.Y.; Du, J.; Zheng, C.L.; Zhu, Q.; et al. Predictors of Mortality for Patients with COVID-19 Pneumonia Caused by SARS-CoV-2: A Prospective Cohort Study. Eur. Respir. J. 2020, 55. [Google Scholar] [CrossRef] [PubMed]

| Action Area | Indication (To Support Clinician Activities) |
|---|---|
| Home Management (Asymptomatic Or Mild Infection) | SARS-CoV-2 infection patient without symptoms or mild upper respiratory tract manifestations and/or fever (<37.5 °C) and/or minor clinical manifestation (malaise, muscle pain, nasal congestion, or headache); arterial blood gas (ABG): negative; high-resolution computed tomography (chest-HRCT): low risk; walking test: negative; no risk factor for progression to severe disease (age ≥ 70 y, active malignancy, diabetes, cardiovascular and/or pulmonary disease). Team leader: emergency physician (Responsible); infectious disease specialist and pneumologist (Involved).
Treatments: symptoms control (paracetamol, oral rehydration). |
| SARS-CoV-2 infection patient without dyspnea and minor respiratory tract manifestations, moderate asthenia and/or persistent cough and/or fever ≥37.5 °C ≤ 38.5 °C ***; no clinical or laboratory parameters of respiratory impairment. Arterial blood gas (ABG): negative; high-resolution computed tomography (chest-HRCT): low risk; walking test: negative; no risk factor for progression to severe disease (age ≥ 70 y, active malignancy, diabetes, cardiovascular and/or pulmonary disease); stable patient. Team leader: emergency physician (Responsible); infectious disease specialist and pneumologist (Involved).
Treatments:
In selected cases after risk–benefit assessment can be considered:
or
* ECG (QT interval) and glucose-6-phosphate dehydrogenase (G6PD) deficiency test should be considered ** ECG should be performed *** In patient with fever perform at least 2 blood cultures and consider antimicrobial therapies. | |
| Hospitalized Patients Low-Intensity Care Unit | Hospitalized patients with mild illness (no hypoxia) and risk factors for progression to severe disease OR hospitalized patients with hypoxia or radiographic evidence of pneumonia but not critically ill (clinically stable). Team leader: infectious disease specialist (Responsible); internal medicine specialist and pneumologist (Involved).
Treatments:
In selected cases after risk–benefit assessment can be considered:
* ECG (QT interval) and G6PD deficiency test should be considered ** ECG should be performed *** In patient with fever perform at least 2 blood cultures and consider antimicrobial therapies. |
| Hospitalized Patients Sub-Intensive Care Unit | Hospitalized patients with hypoxia or radiographic evidence of pneumonia but not critically ill (clinically stable) with need for high flow oxygen/Non Invasive Ventilation (NIV). Team leader: pneumologist (Responsible); infectious disease specialist (Involved).
Treatments:
In selected cases, after risk–benefit assessment together with the intensive care specialist, can be considered:
+/− Antimicrobial therapy (preferentially based on microbiological culture results; requires infectious diseases specialist consultation); * ECG and G6PD deficiency test should be considered ** ECG should be performed *** In patient with fever perform at least 2 blood cultures and consider antimicrobial therapies. |
| Hospitalized Patients Intensive Care Unit | Hospitalized patients critically ill affected by a very severe illness, due to moderate respiratory failure (paO2/FiO2 < 200 mmHg) or severe respiratory failure (PaO2/FiO2 < 100 mmHg) and/or severe impairment of other vital functions. Team leader: intensive care specialist (Responsible); infectious disease specialist (involved). Treatments:
In selected cases after risk–benefit assessment can be considered:
** ECG should be performed |
| |
| SpO2: peripheral oxygen saturation; ECG: electrocardiogram; G6PD: glucose-6-phosphate dehydrogenase; ABG: arterial blood gas; IL-6: Interleukine 6; SIC: sepsis-induced coagulopathy score |
| Indicator | Hospital Fatality Risk (%) | p-Value ** | |
|---|---|---|---|
| Sant’Andrea Hospital | Reference indicatorWuhan Hospitals | 0.0253 | |
| (fatal cases)/(fatal cases + recovered cases) | 22/182 (12.1%) | 54/245 < 22% * | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capalbo, C.; Aceti, A.; Simmaco, M.; Bonfini, R.; Rocco, M.; Ricci, A.; Napoli, C.; Rocco, M.; Alfonsi, V.; Teggi, A.; et al. The Exponential Phase of the Covid-19 Pandemic in Central Italy: An Integrated Care Pathway. Int. J. Environ. Res. Public Health 2020, 17, 3792. https://doi.org/10.3390/ijerph17113792
Capalbo C, Aceti A, Simmaco M, Bonfini R, Rocco M, Ricci A, Napoli C, Rocco M, Alfonsi V, Teggi A, et al. The Exponential Phase of the Covid-19 Pandemic in Central Italy: An Integrated Care Pathway. International Journal of Environmental Research and Public Health. 2020; 17(11):3792. https://doi.org/10.3390/ijerph17113792
Chicago/Turabian StyleCapalbo, Carlo, Antonio Aceti, Maurizio Simmaco, Rita Bonfini, Monica Rocco, Alberto Ricci, Christian Napoli, Matteo Rocco, Valeria Alfonsi, Antonella Teggi, and et al. 2020. "The Exponential Phase of the Covid-19 Pandemic in Central Italy: An Integrated Care Pathway" International Journal of Environmental Research and Public Health 17, no. 11: 3792. https://doi.org/10.3390/ijerph17113792
APA StyleCapalbo, C., Aceti, A., Simmaco, M., Bonfini, R., Rocco, M., Ricci, A., Napoli, C., Rocco, M., Alfonsi, V., Teggi, A., Orsi, G. B., Borro, M., Santino, I., Preissner, R., Marchetti, P., Marcolongo, A., & Anibaldi, P. (2020). The Exponential Phase of the Covid-19 Pandemic in Central Italy: An Integrated Care Pathway. International Journal of Environmental Research and Public Health, 17(11), 3792. https://doi.org/10.3390/ijerph17113792








