The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
- Chronotype OR circadian preference OR morningness OR eveningness OR circadian timing OR chronobiological
- Dietary behaviour OR eating habit OR dietary intake OR food intake OR meal consumption OR diet quality OR macronutrient OR micronutrient OR meal timing OR food preference OR portion size OR appetite OR craving OR night eating syndrome OR binge eating
- 1 AND 2
2.2. Selection Criteria
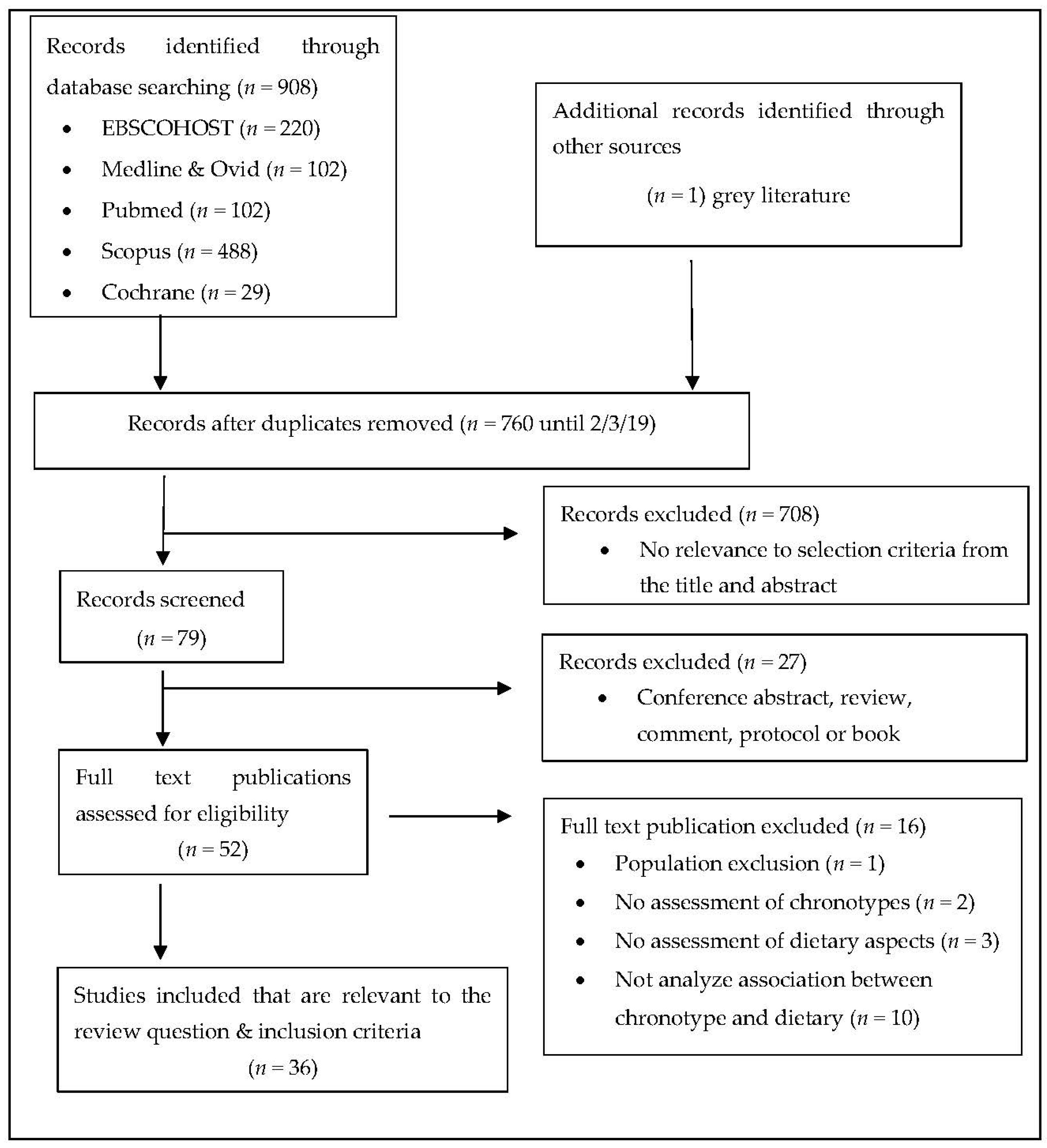
2.3. Selection of Included Publications
3. Results
3.1. Chronotype Assessment
3.2. Dietary Assessment
3.3. The Association between Chronotype and Dietary Habit
3.3.1. Eating behavior
3.3.2. Macro and Micro-Nutrients
3.3.3. Specific food group
4. Discussion
4.1. Dietary Behaviour
4.2. Macro and Micro Nutrients and Food Group
4.3. In Relation to Obesity and Weight Loss
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baron, K.G.; Reid, K.J. Relationship between Circadian Rhythms, Feeding, and Obesity. In Modulation of Sleep by Obesity, Diabetes, Age, and Diet; Academic Press: Cambridge, MA, USA, 2014; pp. 243–253. ISBN 9780124202405. [Google Scholar]
- Cagampang, F.R.; Bruce, K.D. The role of the circadian clock system in nutrition and metabolism. Br. J. Nutr. 2012, 108, 381–392. [Google Scholar] [CrossRef]
- Pagani, L.; Semenova, E.A.; Moriggi, E.; Revell, V.L.; Hack, L.M.; Lockley, S.W.; Arendt, J.; Skene, D.J.; Meier, F.; Izakovic, J.; et al. The physiological period length of the human circadian clock In Vivo is directly proportional to period in human fibroblasts. PLoS ONE 2010, 5, e13376. [Google Scholar] [CrossRef]
- Roenneberg, T.; Kuehnle, T.; Juda, M.; Kantermann, T.; Allebrandt, K.; Gordijn, M.; Merrow, M. Epidemiology of the human circadian clock. Sleep Med. Rev. 2007, 11, 429–438. [Google Scholar] [CrossRef]
- Wirz-Justice, A. How to measure circadian rhythms in humans. Medicographia 2007, 29, 84–90. [Google Scholar]
- Hofstra, W.A.; de Weerd, A.W. How to assess circadian rhythm in humans: A review of literature. Epilepsy Behav. 2008, 13, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Haraszti, R.Á.; Purebl, G.; Salavecz, G.; Poole, L.; Dockray, S.; Steptoe, A. Morningness-eveningness interferes with perceived health, physical activity, diet and stress levels in working women: A cross-sectional study. Chronobiol. Int. 2014, 31, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Patterson, F.; Malone, S.K.; Lozano, A.; Grandner, M.A.; Hanlon, A.L. Smoking, Screen-Based Sedentary Behavior, and Diet Associated with Habitual Sleep Duration and Chronotype: Data from the UK Biobank. Ann. Behav. Med. 2016, 50, 715–726. [Google Scholar] [CrossRef]
- Horne, J. a.; Ostberg, O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int. J. Chronobiol. 1976, 4, 97–110. [Google Scholar]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Kronholm, E.; Konttinen, H.; Wennman, H.; Männistö, S. The associations between chronotype, a healthy diet and obesity. Chronobiol. Int. 2016, 33, 972–981. [Google Scholar] [CrossRef]
- Sato-Mito, N.; Sasaki, S.; Murakami, K.; Okubo, H.; Takahashi, Y.; Shibata, S.; Yamada, K.; Sato, K. The midpoint of sleep is associated with dietary intake and dietary behavior among young Japanese women. Sleep Med. 2011, 12, 289–294. [Google Scholar] [CrossRef]
- Reutrakul, S.; Hood, M.M.; Crowley, S.J.; Morgan, M.K.; Teodori, M.; Knutson, K.L. The relationship between breakfast skipping, chronotype, and glycemic control in type 2 diabetes. Chronobiol. Int. 2014, 31, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Kanerva, N.; Kronholm, E.; Partonen, T.; Ovaskainen, M.-L.L.; Kaartinen, N.E.; Konttinen, H.; Broms, U.; Männistö, S.; Männist, S.; Männistö, S. Tendency toward eveningness is associated with unhealthy dietary habits. Chronobiol. Int. 2012, 29, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.M.; Reedy, J.; Krebs-Smith, S.M. American Diet Quality: Where It Is, Where It Is Heading, and What It Could Be. J. Acad. Nutr. Diet. 2016, 116, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Wright, W.L.; Zelman, K.M. Maximizing your “nutrition minute”: Bridging nutritional gaps across the life span. J. Am. Assoc. Nurse Pract. 2018, 30, 160–177. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Musso, G.; Beccuti, G.; Fadda, M.; Fedele, D.; Gambino, R.; Gentile, L.; Durazzo, M.; Ghigo, E.; Cassader, M. Consuming more of daily caloric intake at dinner predisposes to obesity. A 6-year population-based prospective cohort study. PLoS ONE 2014, 9, e108467. [Google Scholar] [CrossRef] [PubMed]
- Coulthard, J.D.; Pot, G.K. The timing of the evening meal: How is this associated with weight status in UK children? Br. J. Nutr. 2016. [Google Scholar] [CrossRef]
- Nas, A.; Mirza, N.; Hägele, F.; Kahlhöfer, J.; Keller, J.; Rising, R.; Kufer, T.A.; Bosy-Westphal, A. Impact of breakfast skipping compared with dinner skipping on regulation of energy balance and metabolic risk. Am. J. Clin. Nutr. 2017, 105, 1351–1361. [Google Scholar] [CrossRef]
- Nimitphong, H.; Siwasaranond, N.; Saetung, S.; Thakkinstian, A.; Ongphiphadhanakul, B.; Reutrakul, S. The relationship among breakfast time, morningness–eveningness preference and body mass index in Type 2 diabetes. Diabet. Med. 2018, 35, 964–971. [Google Scholar] [CrossRef]
- Reutrakul, S.; Hood, M.M.; Crowley, S.J.; Morgan, M.K.; Teodori, M.; Knutson, K.L.; Van Cauter, E.V.E. Chronotype Is Independently Associated With Glycemic Control in Type 2 Diabetes. Diabetes Care 2013, 36, 2523–2529. [Google Scholar] [CrossRef]
- Garaulet, M.; Gómez-Abellán, P.; Alburquerque-Béjar, J.J.; Lee, Y.C.; Ordovás, J.M.; Scheer, F.A.J.L. Timing of food intake predicts weight loss effectiveness. Int. J. Obes. 2013, 37, 604–611. [Google Scholar] [CrossRef]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. Theory Pract. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Ishihara, K.; Miyasita, A.; Inugami, M.; Fukuda, K.; Yamazaki, K.; Miyata, Y. Differences in the Time or Frequency of Meals, Alcohol and Caffeine Ingestion, and Smoking Found between ‘Morning’ and ‘Evening’ Types. Psychol. Rep. 1985, 57, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Adan, A. Chronotype and personality factors in the daily consumption of alcohol and psychostimulants. Addiction 1994, 89, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Schubert, E.; Randler, C. Association between chronotype and the constructs of the Three-Factor-Eating-Questionnaire. Appetite 2008, 51, 501–505. [Google Scholar] [CrossRef]
- Nakade, M.; Takeuchi, H.; Kurotani, M.; Harada, T. Effects of Meal Habits and Alcohol/Cigarette Consumption on Morningness-Eveningness Preference and Sleep Habits by Japanese Female Students Aged 18–29. J. Physiol. Anthropol. 2009, 28, 83–90. [Google Scholar] [CrossRef]
- Sato-Mito, N.; Shibata, S.; Sasaki, S.; Sato, K. Dietary intake is associated with human chronotype as assessed by both morningnesseveningness score and preferred midpoint of sleep in young Japanese women. Int. J. Food Sci. Nutr. 2011, 62, 525–532. [Google Scholar] [CrossRef]
- Harb, A.; Levandovski, R.; Oliveira, C.; Caumo, W.; Allison, K.C.; Stunkard, A.; Hidalgo, M.P. Night eating patterns and chronotypes: A correlation with binge eating behaviors. Psychiatry Res. 2012, 200, 489–493. [Google Scholar] [CrossRef]
- Lai, P.P.; Say, Y.H. Associated factors of sleep quality and behavior among students of two tertiary institutions in Northern Malaysia. Med. J. Malaysia 2013, 68, 196–203. [Google Scholar]
- Meule, A.; Roeser, K.; Randler, C.; Kübler, A. Skipping breakfast: Morningness-eveningness preference is differentially related to state and trait food cravings. Eat. Weight Disord. 2012, 17, 304–308. [Google Scholar]
- Lucassen, E.A.; Zhao, X.; Rother, K.I.; Mattingly, M.S.; Courville, A.B.; de Jonge, L.; Csako, G.; Cizza, G.; de Jonge, L.; Csako, G.; et al. Evening Chronotype Is Associated with Changes in Eating Behavior, More Sleep Apnea, and Increased Stress Hormones in Short Sleeping Obese Individuals. PLoS ONE 2013, 8, e56519. [Google Scholar] [CrossRef]
- Culnan, E.; Kloss, J.D.; Grandner, M. A prospective study of weight gain associated with chronotype among college freshmen. Chronobiol. Int. 2013, 30, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Mota, M.C.; Waterhouse, J.; De-Souza, D.A.; Rossato, L.T.; Silva, C.M.; Araújo, M.B.J.; Tufik, S.; de Mello, M.T.; Crispim, C.A. Association between chronotype, food intake and physical activity in medical residents. Chronobiol. Int. 2016, 33, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.S.G.; Cañavate, R.; Hernández, C.M.; Cara-Salmerón, V.; Morante, J.J.H. The association among chronotype, timing of food intake and food preferences depends on body mass status. Eur. J. Clin. Nutr. 2017, 71, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Ruiz - Lozano, T.; Vidal, J.; De Hollanda, A.; Canteras, M.; Garaulet, M.; Izquierdo-Pulido, M. Evening chronotype associates with obesity in severely obese subjects: Interaction with CLOCK 3111T/C. Int. J. Obes. 2016, 40, 1550–1557. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.M.; Mota, M.C.; Miranda, M.T.; Paim, S.L.; Waterhouse, J.; Crispim, C.A. Chronotype, social jetlag and sleep debt are associated with dietary intake among Brazilian undergraduate students. Chronobiol. Int. 2016, 33, 740–748. [Google Scholar] [CrossRef]
- Suh, S.; Yang, H.-C.H.C.; Kim, N.; Yu, J.H.; Choi, S.; Yun, C.-H.C.H.; Shin, C. Chronotype Differences in Health Behaviors and Health-Related Quality of Life: A Population-Based Study Among Aged and Older Adults. Behav. Sleep Med. 2017, 15, 361–376. [Google Scholar] [CrossRef]
- Yoshizaki, T.; Kawano, Y.; Noguchi, O.; Onishi, J.; Teramoto, R.; Sunami, A.; Yokoyama, Y.; Tada, Y.; Hida, A.; Togo, F. Association of eating behaviours with diurnal preference and rotating shift work in Japanese female nurses: A cross-sectional study. BMJ Open 2016, 6, e011987. [Google Scholar] [CrossRef]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Kronholm, E.; Tapanainen, H.; Kontto, J.; Männistö, S.; M??nnist??, S.; Männistö, S. Chronotype differences in timing of energy and macronutrient intakes: A population-based study in adults. Obesity 2017, 25, 608–615. [Google Scholar] [CrossRef]
- Teixeira, G.P.; Mota, M.C.; Crispim, C.A. Eveningness is associated with skipping breakfast and poor nutritional intake in Brazilian undergraduate students. Chronobiol. Int. 2018, 35, 358–367. [Google Scholar] [CrossRef]
- Vera, B.; Dashti, H.S.; Gómez-Abellán, P.; Hernández-Martínez, A.M.; Esteban, A.; Scheer, F.A.J.L.; Saxena, R.; Garaulet, M. Modifiable lifestyle behaviors, but not a genetic risk score, associate with metabolic syndrome in evening chronotypes. Sci. Rep. 2018, 8, 945. [Google Scholar] [CrossRef]
- Toktas, N.; Erman, K.A.; Mert, Z. Nutritional Habits According to Human Chronotype and Nutritional Status of Morningness and Eveningness. J. Educ. Train. Stud. 2018, 6, 61–67. [Google Scholar] [CrossRef]
- Yoshizaki, T.; Komatsu, T.; Tada, Y.; Hida, A.; Kawano, Y.; Togo, F. Association of habitual dietary intake with morningness-eveningness and rotating shift work in Japanese female nurses. Chronobiol. Int. 2018, 35, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, D.; Sheng, L.; Xiao, H.; Yao, M.; Chao, Y.; Zhao, Y. Chronotype and sleep duration are associated with stimulant consumption and BMI among Chinese undergraduates. Sleep Biol. Rhythms 2018, 16, 211–222. [Google Scholar] [CrossRef]
- Gangwar, A.; Tiwari, S.; Rawat, A.; Verma, A.; Singh, K.; Kant, S.; Garg, R.K.; Singh, P.K. Circadian Preference, Sleep Quality, and Health-impairing Lifestyles Among Undergraduates of Medical University. Cureus 2018, 10, e2856. [Google Scholar] [CrossRef]
- Kandeger, A.; Egilmez, U.; Sayin, A.A.; Selvi, Y. The relationship between night eating symptoms and disordered eating attitudes via insomnia and chronotype differences. Psychiatry Res. 2018, 268, 354–357. [Google Scholar] [CrossRef]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Männistö, S. Chronotype and energy intake timing in relation to changes in anthropometrics: a 7-year follow-up study in adults. Chronobiol. Int. J. Biol. Med. Rhythm Res. 2019, 36, 27–41. [Google Scholar] [CrossRef]
- Gontijo, C.A.; Balieiro, L.C.T.; Teixeira, G.P.; Maia, Y.C.D.P.; Crispim, C.A.; Cabral, B.B.M.; Fahmy, W.M. Time-related eating patterns and chronotype are associated with diet quality in pregnant women. Chronobiol. Int. 2019, 36, 75–84. [Google Scholar] [CrossRef]
- Kandeger, A.; Selvi, Y.; Tanyer, D.K. The effects of individual circadian rhythm differences on insomnia, impulsivity, and food addiction. Eat. Weight Disord. EWD 2019, 24, 47–55. [Google Scholar] [CrossRef]
- Wolfson, A.R.; Montgomery-Downs, H.E. The Oxford Handbook of Infant, Child and Adolescent Sleep and Behavior; Oxford University Press: Cary, NC, USA, 2013. [Google Scholar]
- Burgess, H.J.; Wyatt, J.K.; Park, M.; Fogg, L.F. Home Circadian Phase Assessments with Measures of Compliance Yield Accurate Dim Light Melatonin Onsets. Sleep 2015, 38, 889–897. [Google Scholar] [CrossRef]
- Kantermann, T.; Sung, H.; Burgess, H.J. Comparing the Morningness-Eveningness Questionnaire and Munich ChronoType Questionnaire to the Dim Light Melatonin Onset. J. Biol. Rhythms 2015, 30, 449–453. [Google Scholar] [CrossRef]
- Adan, A.; Archer, S.N.; Hidalgo, M.P.; Di Milia, L.; Natale, V.; Randler, C. Circadian typology: A comprehensive review. Chronobiol. Int. 2012, 29, 1153–1175. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, S.; Hida, A.; Aritake, S.; Higuchi, S.; Enomoto, M.; Kato, M.; Vetter, C.; Roenneberg, T.; Mishima, K. Validity of the Japanese version of the Munich ChronoType Questionnaire. Chronobiol. Int. 2014, 31, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Torsvall, L.; Akerstedt, T. A diurnal type scale. Construction consistency and validation in shift work. Scand. J. Work. Environ. Heal. 1980, 6, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.S.; Reilly, C.; Midkiff, K. Evaluation of Three Circadian Rhythm Questionnaires With Suggestions for an Improved Measure of Morningness. J. Appl. Psychol. 1989, 74, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Roßbach, S.; Diederichs, T.; Nöthlings, U.; Buyken, A.E.; Alexy, U. Relevance of chronotype for eating patterns in adolescents. Chronobiol. Int. 2018, 35, 336–347. [Google Scholar] [CrossRef]
- Fleig, D.; Randler, C. Association between chronotype and diet in adolescents based on food logs. Eat. Behav. 2009, 10, 115–118. [Google Scholar] [CrossRef]
- Lopez-Minguez, J.; Dashti, H.S.; Madrid-Valero, J.J.; Madrid, J.A.; Saxena, R.; Scheer, F.A.; Ordonana, J.R.; Garaulet, M. Heritability of the timing of food intake. Clin. Nutr. 2018, 38, 767–773. [Google Scholar] [CrossRef]
- Xiao, Q.; Garaulet, M.; Scheer, F.A.J.L. Meal timing and obesity: interactions with macronutrient intake and chronotype. Int. J. Obes. 2019, 43, 1701–1711. [Google Scholar] [CrossRef]
- Yasuda, J.; Asako, M.; Arimitsu, T.; Fujita, S. Skipping breakfast is associated with lower fat-free mass in healthy young subjects: a cross-sectional study. Nutr. Res. 2018, 60, 26–32. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Wainstein, J.; Landau, Z.; Raz, I.; Ahren, B.; Chapnik, N.; Ganz, T.; Menaged, M.; Barnea, M.; Bar-Dayan, Y.; et al. Influences of breakfast on clock gene expression and postprandial glycemia in healthy individuals and individuals with diabetes: A randomized clinical trial. Diabetes Care 2017, 40, 1573–1579. [Google Scholar] [CrossRef]
- Baltar, V.T.; Cunha, D.B.; Santos, R.O.; Marchioni, D.M.; Sichieri, R. Breakfast patterns and their association with body mass index in Brazilian adults. Cad. Saude Publica 2018, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chatelan, A.; Castetbon, K.; Pasquier, J.; Allemann, C.; Zuber, A.; Camenzind-Frey, E.; Zuberbuehler, C.A.; Bochud, M. Association between breakfast composition and abdominal obesity in the Swiss adult population eating breakfast regularly. Int. J. Behav. Nutr. Phys. Act. 2018, 15, 115. [Google Scholar] [CrossRef] [PubMed]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and Peripheral Circadian Clocks in Mammals. Annu. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D. Physiological responses to food intake throughout the day. Nutr. Res. Rev. 2014, 27, 107–118. [Google Scholar] [CrossRef]
- Vetter, C.; Scheer, F.A.J.L. Circadian Biology: Uncoupling Human Body Clocks by Food Timing. Curr. Biol. 2017, 27, R656–R658. [Google Scholar] [CrossRef]
- Schibler, U.; Gotic, I.; Saini, C.; Gos, P.; Curie, T.; Emmenegger, Y.; Sinturel, F.; Gosselin, P.; Gerber, A.; Fleury-Olela, F.; et al. Clock-Talk: Interactions between central and peripheral circadian oscillators in mammals. Proc. Cold Spring Harb. Symp. Quant. Biol. 2016; 80, pp. 223–232. [Google Scholar]
- Carneiro, B.T.S.; Araujo, J.F. Food entrainment: major and recent findings. Front. Behav. Neurosci. 2012, 6, 83. [Google Scholar] [CrossRef]
- Kalsbeek, A.; La Fleur, S.; Fliers, E. Circadian control of glucose metabolism. Mol. Metab. 2014, 3, 372–383. [Google Scholar] [CrossRef]
- Wang, D.; Opperhuizen, A.L.; Reznick, J.; Turner, N.; Su, Y.; Cooney, G.J.; Kalsbeek, A. Effects of feeding time on daily rhythms of neuropeptide and clock gene expression in the rat hypothalamus. Brain Res. 2017, 1671, 93–101. [Google Scholar] [CrossRef]
- Mukherji, A.; Kobiita, A.; Damara, M.; Misra, N.; Meziane, H.; Champy, M.-F.; Chambon, P. Shifting eating to the circadian rest phase misaligns the peripheral clocks with the master SCN clock and leads to a metabolic syndrome. Proc. Natl. Acad. Sci. 2015, 112, E6691–E6698. [Google Scholar] [CrossRef]
- Oishi, K.; Hashimoto, C. Short-term time-restricted feeding during the resting phase is sufficient to induce leptin resistance that contributes to development of obesity and metabolic disorders in mice. Chronobiol. Int. 2018, 35, 1576–1594. [Google Scholar] [CrossRef]
- Reznick, J.; Preston, E.; Wilks, D.L.; Beale, S.M.; Turner, N.; Cooney, G.J. Altered feeding differentially regulates circadian rhythms and energy metabolism in liver and muscle of rats. Biochim. Biophys. Acta - Mol. Basis Dis. 2013, 1832, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Opperhuizen, A.L.; Wang, D.; Foppen, E.; Jansen, R.; Boudzovitch-Surovtseva, O.; de Vries, J.; Fliers, E.; Kalsbeek, A. Feeding during the resting phase causes profound changes in physiology and desynchronization between liver and muscle rhythms of rats. Eur. J. Neurosci. 2016, 44, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Strable, M.S.; Ntambi, J.M. Genetic control of de novo lipogenesis: Role in diet-induced obesity. Crit. Rev. Biochem. Mol. Biol. 2010, 45, 199–214. [Google Scholar] [CrossRef] [PubMed]
- Anothaisintawee, T.; Lertrattananon, D.; Thamakaison, S.; Thakkinstian, A.; Reutrakul, S. The relationship among morningness-eveningness, sleep duration, social jetlag, and body mass index in Asian patients with prediabetes. Front. Endocrinol. 2018, 9, 435. [Google Scholar] [CrossRef]
- Zainuddin, N.S.; Shahar, S.; Safii, S.; Haron, H.; Omar, M.A. Sugar intake and metabolic syndrome among older adults in Peninsular Malaysia. Malays. J. Nutr. 2018, 24, 163–174. [Google Scholar]
- Rippe, J.M.; Angelopoulos, T.J. Sugars, obesity, and cardiovascular disease: results from recent randomized control trials. Eur. J. Nutr. 2016, 55, 45–53. [Google Scholar] [CrossRef]
- Jones, J.M.; Engleson, J. Whole Grains: Benefits and Challenges. Annu. Rev. Food Sci. Technol. 2010, 1, 19–40. [Google Scholar] [CrossRef]
- Villegas, R.; Takata, Y.; Murff, H.; Blot, W.J. Fish, omega-3 long-chain fatty acids, and all-cause mortality in a low-income US population: Results from the Southern Community Cohort Study. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 651–658. [Google Scholar] [CrossRef]
- Binnie, M.A.; Barlow, K.; Johnson, V.; Harrison, C. Red meats: Time for a paradigm shift in dietary advice. Meat Sci. 2014, 98, 445–451. [Google Scholar] [CrossRef]
- Peuhkuri, K.; Sihvola, N.; Korpela, R. Dietary factors and fluctuating levels of melatonin. Food Nutr. Res. 2012, 56, 17252. [Google Scholar] [CrossRef]
- Whittier, A.; Sanchez, S.E.; Castañeda, B.; Sanchez, E.; Gelaye, B.; Yanez, D.; Williams, M.A. Eveningness chronotype, daytime sleepiness, caffeine consumption, and use of other stimulants among peruvian university students. J. Caffeine Res. 2014, 4, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Watson, E.J.; Coates, A.M.; Kohler, M.; Banks, S. Caffeine consumption and sleep quality in Australian adults. Nutrients 2016, 8, 479. [Google Scholar] [CrossRef] [PubMed]
- Galli, G.; Piaggi, P.; Mattingly, M.S.; De Jonge, L.; Courville, A.B.; Pinchera, A.; Santini, F.; Csako, G.; Cizza, G. Inverse relationship of food and alcohol intake to sleep measures in obesity. Nutr. Diabetes 2013, 3, e58. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ehlers, C.L.; Wills, D.; Gilder, D.A. A history of binge drinking during adolescence is associated with poorer sleep quality in young adult Mexican Americans and American Indians. Psychopharmacology 2018, 235, 1775–1782. [Google Scholar] [CrossRef] [PubMed]
- Fucito, L.M.; Bold, K.W.; Van Reen, E.; Redeker, N.S.; O’Malley, S.S.; Hanrahan, T.H.; DeMartini, K.S. Reciprocal variations in sleep and drinking over time among heavy-drinking young adults. J. Abnorm. Psychol. 2018, 127, 92–103. [Google Scholar] [CrossRef]
- Lydon-Staley, D.M.; Ram, N.; Brose, A.; Schmiedek, F. Reduced impact of alcohol use on next-day tiredness in older relative to younger adults: A role for sleep duration. Psychol. Aging 2017, 32, 642–653. [Google Scholar] [CrossRef]
- Robinson, D.; Gelaye, B.; Tadesse, M.G.; Williams, M.A.; Lemma, S.; Berhane, Y. Daytime Sleepiness, Circadian Preference, Caffeine Consumption and Khat Use among College Students in Ethiopia. J. Sleep Disord. Treat. Care 2013, 3. [Google Scholar] [CrossRef]
- Rupp, T.L.; Acebo, C.; Carskadon, M.A. Evening alcohol suppresses salivary melatonin in young adults. Chronobiol. Int. 2007, 24, 463–470. [Google Scholar] [CrossRef]
- Haynie, D.L.; Lewin, D.; Luk, J.W.; Lipsky, L.M.; O’Brien, F.; Iannotti, R.J.; Liu, D.; Simons-Morton, B.G. Beyond sleep duration: Bidirectional associations among chronotype, social jetlag, and drinking behaviors in a longitudinal sample of US high school students. Sleep 2018, 41, zsx202. [Google Scholar] [CrossRef]
- Wittmann, M.; Paulus, M.; Roenneberg, T. Decreased psychological well-being in late “chronotypes” is mediated by smoking and alcohol consumption. Subst. Use Misuse 2010, 45, 15–30. [Google Scholar] [CrossRef]
- Fogelholm, M.; Anderssen, S.; Gunnarsdottir, I.; Lahti-Koski, M. Dietary macronutrients and food consumption as determinants of long-term weight change in adult populations: a systematic literature review. Food Nutr. Res. 2012, 56, 19103. [Google Scholar] [CrossRef] [PubMed]
- Koebnick, C.; Black, M.H.; Wu, J.; Shu, Y.H.; MacKay, A.W.; Watanabe, R.M.; Buchanan, T.A.; Xiang, A.H. A diet high in sugar-sweetened beverage and low in fruits and vegetables is associated with adiposity and a pro-inflammatory adipokine profile. Br. J. Nutr. 2018, 120, 1230–1239. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.M.; Burch, J.B.; Youngstedt, S.D.; Wirth, M.D.; Hardin, J.W.; Hurley, T.G.; Blair, S.N.; Hand, G.A.; Shook, R.P.; Drenowatz, C.; et al. Relationships between chronotype, social jetlag, sleep, obesity and blood pressure in healthy young adults. Chronobiol. Int. 2019, 36, 493–509. [Google Scholar] [CrossRef] [PubMed]
- Saat, N.Z.M.; Ishak, I.; Lubis, S.H.; Huong Wen, S.; Mohd, S.N.L.S.; Zakaria, N.S.; Xin Ee, T.; Othman, M.; Hui Shein, T.; Zulkifli, Z.; et al. Stress And Its Relationship With Body Mass Index Among Biomedical Science Students In Kuala Lumpur, Malaysia. ASEAN J. Psychiatry 2010, 11, 190–197. [Google Scholar]
- Yeng, P.P.; Sedek, R. The body weight perception and weight control behaviors among undergraduate students in National University of Malaysia (UKM). Pakistan J. Nutr. 2012, 11, 1131–1138. [Google Scholar] [CrossRef]
- Ross, K.M.; Graham Thomas, J.; Wing, R.R. Successful weight loss maintenance associated with morning chronotype and better sleep quality. J. Behav. Med. 2016, 39, 465–471. [Google Scholar] [CrossRef]
- Almoosawi, S.; Vingeliene, S.; Gachon, F.; Voortman, T.; Palla, L.; Johnston, J.D.; Van Dam, R.M.; Darimont, C.; Karagounis, L.G. Chronotype: Implications for Epidemiologic Studies on Chrono-Nutrition and Cardiometabolic Health. Adv. Nutr. 2019, 10, 30–42. [Google Scholar] [CrossRef]
| Reference | Country | Study Design | N | % Female | Age (Year) | Participants | Weight Status (If Reported) |
|---|---|---|---|---|---|---|---|
| [23] | Japan | Cross-sectional | 1459 | 37.5 | Mean: 19.5 | University students | - |
| [24] | Spain | Cross-sectional | 537 | 52.1% | Range: 21–30 | University students and lecturers | - |
| [25] | German | Cross-sectional | 335 | 58.8 | Mean: 23.15 Range: 17–42 | University students | Mean BMI: 22.93 ± 3.41 kg/m2 |
| [26] | Japan | Cross-sectional | 800 | 100.0 | Mean: 19.26 ± 1.33 Range: 18–29 | Students | - |
| [11] | Japan | Cross-sectional | 3304 | 100.0 | Mean: 18.1 ± 0.3 Range: 18–20 | Dietetics Students | Mean BMI: 20.9 ± 2.8 kg/m2 |
| [27] | Japan | Cross-sectional | 112 | 100.0 | Range: 19–36 | Dietetics students | Mean BMI: 19.9 ±1.8 kg/m2 |
| [28] | Brazil | Cross-sectional | 100 | 77.0 | Mean: 39.5 ± 11.7 Range: 18–65 | Outpatient nutrition clinic | Mean BMI: 26.8 ± 4.02 kg/m2 |
| [13] | Finland | Cross-sectional | 4493 | 67.0 | Mean: 51.9 ± 0.2 Range: 25–74 | Population based study | Mean BMI: 26.8 ± 0.2 kg/m2 |
| [29] | Malaysia | Cross-sectional | 1118 | 56.0 | Mean: 20.06 ± 1.53 Range: 18–27 | University students | - |
| [30] | Germany | Cross-sectional | 66 | Mean: 23.08 ± 2.68 | University students | Mean BMI: 22.22 ± 4.48 kg/m2 | |
| [21] | Spain | Observational Intervention study | 420 | 49.5 | Mean: 42 ± 11 | Outpatient of nutrition clinic | Mean BMI: 31.4 ± 5.4 kg/m2 |
| [31] | USA | Prospective, randomized controlled study | 126 | 77.0 | Range: 18–50 | Population based study | Range BMI: 30–55 kg/m2 |
| [12] | USA | Cross-sectional | 194 | 69.6 | Range: 18–85 | Type 2 diabetes patients | Mean BMI: 35.6 ± 8.3 kg/m2 |
| [20] | USA | Cross-sectional | 194 | 70.0 | Range: 18–85 | Type 2 diabetes patients | Mean BMI: 35.6 ± 8.3 kg/m2 |
| [32] | USA | Prospective study | 137 | 58.0 | Range: 18.25 ± 0.56 | University freshmen | Mean BMI: 21.99 ± 3.24 kg/m2 |
| [10] | Finland | Cross-sectional | 4421 | 54.0 | Range: 25–74 | Population based study | - |
| [33] | Brazil | Cross-sectional | 72 | 72.0 | Mean: 29.2 ± 2.0 | Medical residents | Mean BMI: 22.9 ± 3.4 kg/m2 |
| [34] | Spain | Cross sectional & Interventional longitudinal study | 400,171 finished follow up | Not stated | Range: 30–60 | University staff | Mean BMI:
|
| [8] | USA | Cross-sectional | 439,933 | 56.0 | Mean: 56.5 ± 8.1 Range: 40–69 | Population based study | Mean BMI: 27.4 ± 4.8 kg/m2 |
| [35] | Spain | Prospective cohort study | 252 | 79.0 | Age: 52 ± 11 | Post bariatric surgery patient | Mean BMI: 46.4 ± 6.0 kg/m2 |
| [36] | Brazil | Cross-sectional | 204 | 55.0 | Mean: 21.6 ± 3.9 Range: 18–39 | Undergraduate of Law School | Mean BMI: 22.8 ± 3.2 kg/m2 |
| [37] | Korea | Cross-sectional | 2976 | 51.0 | Mean: 58.02 ± 7.05 Range: 49–79 | Population based study | Mean BMI:
|
| [38] | Japan | Cross-sectional | 218 | 100.0 | Range: 21–63 | Nurses (day and rotating shift) | Mean BMI: 21.7 kg/m2 |
| [39] | Finland | Cross-sectional | 1854 | 54.0% | Range: 25–74 | Population based study | Mean BMI:
|
| [40] | Brazil | Cross-sectional | 721 | 67.7 | Above 18 years old | Undergraduate student | Mean BMI:
|
| [41] | Spain | Cross-sectional | 2126 | 81.0 | Mean: 40 ± 13 | Overweight and obese population | Mean BMI: 31 ± 5 kg/m2 |
| [19] | Thailand | Cross-sectional | 210 | 60.0 | Mean: 58.6 ± 11 | Type 2 diabetes | Mean BMI: 28.4 ± 4.8 kg/m2 |
| [42] | Turkey | Cross-sectional | 142 | 43.0 | Mean: 21.83 ± 2.37 | University student | Mean BMI:
|
| [43] | Japan | Cross-sectional | 2559 | 100.0 | Range: 20–59 | Nurses (day and rotating shift workers) | Mean BMI:
|
| [44] | China | Cross-sectional | 977 | 57.7 | Mean: 20.06 ± 1.25 | University undergraduates | BMI:
|
| [45] | India | Cross-sectional | 203 | 35.5 | Mean: 18.34 | University student (medical) | Mean BMI:
|
| [46] | Turkey | Cross-sectional | 383 | 60.1 | Mean: 21.1 ± 0.1 Range: 17–37 years | University students | Mean BMI: 22.25 ± 3.19 kg/m2 |
| [47] | Finland | Follow-up, 7 years | Baseline: 5024 Follow-up: 1097 | 54.0 | Range: 25–74 | Population based study | - |
| [48] | Brazil | Cross-sectional | 100 | 100 | 27.3 ± 5.7 | Pregnant women | - |
| [49] | Turkey | Cross-sectional | 1323 | 65.8 | Mean: 20.83 ± 1.98 Range: 16–33 | University students | Mean BMI: 21.96 ± 3.03 kg/m2 |
| Reference | Chronotype | |
|---|---|---|
| Assessment | Distribution | |
| [23] | MEQ |
|
| [24] | MEQ |
|
| [25] | CSM |
|
| [26] | ME | Mean ME score: 16.07 (3.53) |
| [11] | Midpoint of sleep |
|
| [27] | MEQ Midpoint of sleep | High MEQ score: 37 (33%) Low MEQ score: 37 (33%) Early midpoint of sleep tertile: 40 (36%) Late midpoint of sleep tertile: 37 (33%) |
| [28] | MEQ | Mean MEQ: 52.4 ± 14.0 |
| [13] | Shortened MEQ | Quintile 5 (extreme M-type): 22% Quintile 1 (extreme E-type): 18% |
| [29] | MEQ |
|
| [30] | MEQ |
|
| [21] | MEQ | Not stated |
| [31] | MEQ |
|
| [12] | MSF | Mean MSF of breakfast skippers: 4:34 (2.18) |
| [20] | MSF | Mean MSF: 3.29 (1.46) |
| [32] | Reduced MEQ |
|
| [10] | MEQ |
|
| [33] | MEQ |
|
| [34] | MEQ |
|
| [8] | Self-report chronotype |
|
| [35] | MEQ |
|
| [36] | MSF | Mean MSF: 5.40 (1.48) |
| [37] | MEQ |
|
| [38] | ME |
|
| [39] | MEQ |
|
| [40] | MEQ |
|
| [41] | MEQ Dichotomous based on median score of population; 53 |
|
| [19] | CSM |
|
| [42] | MEQ |
|
| [43] | MEQ Higher scores and tertiles (T) indicate a tendency towards morningness. | MEQ score in; (a) Day worker:
|
| [44] | MSF | Mean MSF: 4:41(1:06) |
| [45] | MEQ |
|
| [46] | MEQ |
|
| [47] | MEQ |
|
| [48] | MSF |
|
| [49] | MEQ |
|
| Reference | Measure of Dietary Pattern | The Association of Chronotype | ||
|---|---|---|---|---|
| Dietary Behaviour | Nutrient Intake | Other Health Status | ||
| [23] | Life Habits Inventory | E-type significantly had frequent night meal (χ = 65.63, p < 0.001) compared to M-type. 34.8% of E-type skipped breakfast than M-type, 5.5%. No significant different in meal timing; breakfast, lunch and dinner between chronotypes. | - | - |
| [25] | Three-factor eating questionnaire (TFEQ) | Positive significant association between M-type and dietary restraint (r = 0.136, p = 0.013). Negative significant association between M-type with disinhibition (breaking dietary restraint and overeating) (r = −0.151, p = 0.006) and perceived hunger (r = −0.137, p = 0.009). | - | - |
| [26] |
| Higher chronotype score (towards morningness) significantly related to regular breakfast eater (χ2 = 74.55, p < 0.001) and earlier mealtime for breakfast (χ2 = 88.94, p < 0.001). | - | - |
| [21] | Seven-day dietary record | E-type significantly were more late lunch eaters (after 15:00) than M-type (p = 0.032). | - | No significant association between weight loss (%) and MEQ score (p = 0.456). |
| [11] |
| Quintile 5 (towards eveningness) compared to Quintile 1 (towards morningness) significantly:
| Latest midpoint of sleep (towards eveningness) significantly associated with:
| No significant association between BMI and midpoint of sleep (p = 0.30). |
| [29] | Craving of High-calorie foods questionnaire | Not significant relationship between chronotype and high calorie food craving (r = 0.003, p = 0.917) | - | No significant association between BMI and MEQ score (r = 0.043, p = 0.152). |
| [27] | Brief diet history questionnaire (BDHQ) | - | A lower chronotype score (towards eveningness) was significantly associated with less energy from protein and intake of calcium, magnesium, zinc, vitamin D, riboflavin, vitamin B6 and folate. No association between chronotype score with total energy intake/day, carbohydrate, fats and other micronutrients. Latest midpoint of sleep (towards eveningness) significantly associated with lower energy from protein and intake of cholesterol, potassium, calcium, magnesium, zinc, vitamin D, riboflavin, vitamin B6 and vitamin B12. No association between midpoint of sleep with total energy intake/day, carbohydrate, fats and other micronutrients. | No significant association between BMI and midpoint of sleep (p = 0.67) and MEQ score (p = 0.78). |
| [12] | 24 h diet recall | Later MSF (towards eveningness) significantly were breakfast skippers (p = 0.002). | - | - |
| [30] | Food craving questionnaires | Significantly more M-type; 91.2% had breakfast compared to E-type; 46.4% (p < 0.001). There is no significant difference in hunger between morning and evening type. | - | No significant association between BMI and MEQ score (F = 0.52, p > 0.05). |
| [28] | Binge eating score (BES), eating attitudes test (EAT) and night eating syndrome (NES) | Lower chronotype score (towards eveningness) was significantly associated with higher binge eating (r = −0.33, p = 0.001) and night eating syndrome score (r = −0.24, p = 0.015). In multivariate regression (r2 = 0.12, F = 6.8, p = 0.002), only binge eating remained significantly associated with chronotype score (β = −0.25, p = 0.028) No correlation was found between EAT and chronotype. | - | No significant association between BMI and MEQ score (r = −0.101, p = 0.319). |
| [31] | Three-day food record | E-type delay in breakfast time during working (8:38 ± 1:52 vs. 7:17 ± 1:31, p < 0.001) and non-working days (9:52 ± 2:32 vs. 8:56 ± 2:30, p = 0.075) compared to M-type. Lower chronotype score (towards eveningness) related to consumptions of more calories after 20:00 (β = 0.459, p < 0.001). No significant differences in portion size and number of eating occasion between chronotypes. | No significant differences in total energy intake between chronotype. Meals consumed after 20:00 contained less carbohydrate (49 ± 16% vs. 53 ± 10%, p = 0.021) and protein (12 ± 7% vs. 14 ± 4%, p = 0.006) and more fat (34 ± 14% vs. 32 ± 7%, p = 0.069). However, there was no significant difference between chronotype in these macronutrients. | No significant difference in weight between M and E-type. However, chronotype score (moving from morningness to eveningness score) was associate with increase in BMI (r2 = 0.057, p = 0.048), larger neck circumference (r2 = 0.488, p = 0.028) and lower HDL-C levels (r2 = 0.095, p = 0.026). |
| [37] | Sleep interfering behaviour scale | E-type significantly ate heavy meal before bedtime compared to I-type and M-type (p < 0.001). | - | No significant difference in BMI between the chronotypes. |
| [13] | Food frequency questionnaire | - | Quintile 1 significantly consume more energy from alcohol (2.5 vs. 1.8 E%, p < 0.001), fat (31.3 vs. 30.8 E%, p < 0.001), saturated fatty acid (11.6 vs. 11.5 E%, p = 0.002) and sucrose (9.8 vs. 10.0, p = 0.001) compared to Quintile 5. Quintile 5 significantly consume more energy from carbohydrates (49.5 vs. 48.6 E%, p < 0.001), protein (17.9 vs. 17.6 E%, p = 0.16) and intake of more fibre (32 vs. 30 g, p < 0.001), folic acid (426 vs. 412 µg, p < 0.001), vitamin D (9.9 vs. 9.5 µg, p < 0.001) and sodium (3.9 vs. 3.8 g, p < 0.001) compared to Quintile 1. No significant difference between chronotype quintiles in total energy intake, vitamin C, and calcium intake. | No significant association between BMI and chronotype score (p = 0.35). |
| [10] | Food frequency questionnaire | - | In women, a lower chronotype score (towards eveningness) was associated with more % energy intake from fat (p < 0.018), adjusted to age. No significant association among men. No significant differences in total daily energy intake between chronotype in both men and women. | In men, there was a positive association between chronotype and BMI (B = 0.048, p = 0.041). Chronotype modified the association between the healthy diet and body fat % and waist circumference. |
| [20] | 24 h diet recall | MSF Quartile 5 (towards eveningness) compared to MSF Quartile 1 (towards morningness) significantly:
| No significant differences in total daily energy intake between chronotype. | Later MSF was associated with higher BMI (p = 0.03). Later MSF was significantly associated with higher HbA1c (B = 0.025, p = 0.001); 1 h delay of MSF was associated with an increase in HbA1c of 2.5% from the original level. |
| [33] | 3 days food diary | - | Lower chronotype score (towards eveningness) were significantly negatively associated with consumption of more calories (β = −0.28, p = 0.02), carbohydrate (β = −0.26, p = 0.03), protein (β = −0.23, p = 0.04) and cholesterol (β = −0.24, p = 0.04). | Chronontype scores not associated with BMI (B = −0.01, p = 0.98), WC (B = 0.09, p = 0.41) and weight gain (B = −0.1, p = 0.48) after the beginning of residency. |
| [36] | Food frequency questionnaire | Breakfast skippers (12.2%) significantly had later MSF (towards eveningness) (6:19 vs. 5:28, p = 0.02) than those who had breakfast. Later MSF significantly positively associated with delay meal timing during breakfast (r = 0.24, p < 0.001) and lunch (r = 0.19, p < 0.01) | Later MSF (towards eveningness) was significantly positively associated with greater servings/day of meat (β = 0.21, p = 0.003), adjusted for age and BMI. | - |
| [41] |
| A lower chronotype score (towards eveningness) was significantly associated with delayed meal timing during breakfast (p < 0.001), lunch (p = 0.002) and dinner (p = 0.007). E-type significantly had higher eating behaviour score (larger portion sizes, second rounds and energy rich foods) and emotional eating score compared to M-type (p < 0.001). | E-type consume significantly less carbohydrate (193.78 ± 3.18 vs. 204.59 ± 3.07 g, p = 0.017) than M-type. There was no significant difference between chronotype in total energy, protein, and fats intake. | Lower chronotype score (towards eveningness) was significantly associated with higher BMI (p = 0.032). and triglyceride (p = 0.006). and lower HDL-cholesterol (p = 0.001). |
| [35] | 4 day food records | E-type significantly delayed mealtime during lunch (14:19 vs. 14:04, p = 0.017) and dinner (21:31 vs. 21:06, p < 0.001) compared to M-type. | No significant differences in total daily energy intake and macronutrients (carbohydrate, protein and fat) during baseline and follow up between chronotype. | E-type had more body weight (126.0 ± 22.3 vs. 119.8 ± 15.9 kg, p = 0.020) during pre-bariatric surgery and loss less excess weight loss (EWL) (77.9 ± 23.3 vs. 82.9 ± 22.6%, p = 0.041) post-bariatric surgery than M-type. In CLOCK 3111 carrier of risk allele C, E-type significantly were more obese during baseline than M-type (p = 0.012). |
| [45] | Proforma (questionnaire) | There were significantly more E-type (75.34%) had delay in dinner timing (later than 21:00) that I- (41.38%) and M-type (34.88%), p < 0.001. | - | E-type had significantly greater BMI than I and M-type (p = 0.029). |
| [34] | Food frequency questionnaire | Among normal weight participants, compared to M-type, E-type significantly consume:
| No significant differences in total daily energy intake, carbohydrate, protein and fat between chronotype among overweight participants. Among normal weight participants, evening type significantly consumed more carbohydrate (p = 0.024) and protein (p = 0.003) than M-type. No significant difference in total energy and fat intake. | M-type group loses more body weight (−0.75 ± 0.54 vs. −0.60 ± 0.46 kg/week, p = 0.153), BMI (-3.30 ± 0.53 vs. −2.63 ± 0.49, p = 0.133) and body fat (−5.41 ± 1.98 vs. −5.37 ± 2.3 %, p = 0.912) than E-type. |
| [38] | Eating behaviour questionnaire | Lower chronotype scores significantly negatively associated to higher meal contents (β = −0.172, p = 0.041) and temporal meal timing (β = −0.338, p < 0.001). A higher meal contents score represents greater preference towards high-fat diet and sweets. A higher temporal eating score represent greater irregular meal timing and total of meals consumed and delay meal timing. | - | - |
| [19] | 24 h dietary recall | E-type significantly delay meal timing during breakfast (7:30–9:00 vs. 7:00–8:30, p < 0.001), lunch (12:00–13:23 vs. 12:00–13:00, p = 0.032), dinner (18:00–19:00 vs. 17:30–19:00, p = 0.031) and last meal (18:00–19:38 vs. 17:53–19:00, p = 0.03) time than M-type. M-type was significantly negatively associated with breakfast time (β = −0.614, p < 0.001). | There was no significant difference between total energy intake between chronotype. | Greater preference towards eveningness was associated with greater BMI (B = −0.141, p = 0.019). Mediation analysis shown, M-type was associated with earlier breakfast time and thus lower BMI by 0.37 kg/m2 (B = −0.365, 95%CI: −0.877, −0.066). |
| [49] | Yale food addiction scale | Chronotype score (towards eveningness) was significantly negative associated with food addiction (r = −0.10, p < 0.01). | - | No significant difference in BMI between the chronotypes. |
| [39] | 48 h dietary recalls 3 days food records | - | During weekdays and weekends, E-type significantly consumed:
On weekdays, lower chronotype score (towards eveningness) was significantly associated with:
| No significant difference in BMI between the chronotype. |
| [40] | 24 h diet recall | E-type significantly had delay meal timing during breakfast (8:00 ± 1.2 vs. 7:20 ± 1.1, p < 0.001) and lunch (12:38 ± 1.00 vs. 12:13 ± 0.48, p = 0.02) than M-type. E-type significantly more breakfast skippers (21.8% vs. 10.1%, p = 0.02) than M-type. E-type was 1.7 times more likely to skip breakfast than M-type (CI 95%: 1.1–2.9, p = 0.02). Among breakfast skippers, the chronotype scores were negatively associated with dinner time (β = −0.17, p = 0.04). | - | No significant difference in BMI, waist circumference and abdominal fat between the chronotypes. |
| [42] |
| All E-type participants skipped a meal, while 11% of M-type and 14% of I-type skipped a meal. All chronotypes skipped breakfast the most; 60% E-type, 33% M-type and 44% I-type. | Among men, E-type compared to M-type significantly had:
| No significant difference in BMI between the chronotypes. |
| [43] | Semi-quantitative food frequency questionnaire | - | There was no association between chronotype scores with total energy and macronutrient intake (carbohydrate, protein and fat). | - |
| [46] |
| E-type had significantly higher NEQ (night eating symptoms) and EAT score (higher score, higher severity of disordered eating) than other chronotypes (p < 0.001). Chronotype score (towards eveningness) was significantly negative associated with NEQ (r = −0.29, p < 0.01). | - | No significant association between BMI and chronotypes. |
| [47] | 48 h diet recall | - | There was no significant difference between M (7709 kJ ± 97) and E-type (7679 kJ ± 215) in total daily energy intake. E-type compared to M-type significantly had:
| E-type (+1.4 kg ± 0.5) gained the most weight after seven years, but not significantly different from M- (+0.6 kg ± 0.2) and I-type (+0.8 kg ± 0.3). |
| [48] | Three 24 h diet recall | - | There was no association between chronotype and calorie intake. | - |
| Association of Chronotype and Food Group | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| References | Grains | Legumes | Meat | Fish | Dairy Product | Fruits | Vegetables | Sweets | FAT & Oil | Caffeine | Alcohol |
| [23] | NA | NA | NA | NA | NA | NA | NA | NA | NA | +a | + a |
| [24] | NA | NA | NA | NA | NA | NA | NA | NA | NA | + a,b | + a |
| [26] | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA | + b |
| [11] | − b | − b | + b | = | − b | = | − b | + b | + b | NA | + b |
| [27] | = | NA | = | = | = | = | − b | = | NA | NA | NA |
| [13] | − b | NA | = | − b | = | = | − b | + b | = | NA | + b |
| [32] | Pre | ||||||||||
| NA | NA | NA | NA | NA | NA | NA | NA | NA | = | = | |
| Post | |||||||||||
| NA | NA | NA | NA | NA | NA | NA | NA | NA | = | + a | |
| [10] | Men | ||||||||||
| − b | NA | = | − b | = | = | = | NA | NA | NA | + b | |
| Women | |||||||||||
| − b | NA | = | = | = | = | = | NA | NA | NA | + b | |
| [33] | = | = | = | NA | = | = | + b | + b | − b | NA | NA |
| [34] | = | NA | NA | NA | = | − a | = | NA | + a | NA | + a |
| [8] | NA | NA | NA | NA | NA | − a | − a | NA | NA | NA | NA |
| [36] | = | NA | + b | NA | = | = | = | = | = | NA | NA |
| [37] | NA | NA | NA | NA | NA | NA | NA | NA | NA | + a | = |
| [43] | NA | NA | NA | NA | NA | − a | − a | NA | NA | + a | NA |
| [44] | NA | NA | NA | NA | NA | NA | NA | + b | NA | = | + b |
| [45] | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA | = |
| [48] | + b | = | = | = | = | − b | = | NA | = | NA | NA |
| Morning Chronotype | Evening Chronotype | |
|---|---|---|
| Dietary Behaviour | ||
| Delay meal timing | − | + |
| Regular breakfast eater | + | − |
| Breakfast skipper | − | + |
| Excessive calorie during night | − | + |
| Food addiction | Limited | Limited |
| Feeling hunger | Limited | Limited |
| Longer eating duration | Limited | Limited |
| Watching TV during meal | Limited | Limited |
| Binge eating | Limited | Limited |
| Portion size | Limited | Limited |
| Skipped meal | Limited | Limited |
| Nutrient Intake | ||
| Energy | = | = |
| Carbohydrate | = | = |
| Protein | + | − |
| Fat | = | = |
| Cholesterol | = | = |
| Fibre | = | = |
| Sucrose | - | + |
| Vitamins | Limited | Limited |
| Minerals | Limited | Limited |
| Food Group Intake | ||
| Grains | Not enough evidence | Not enough evidence |
| Legumes | = | = |
| Meat | = | = |
| Fish | = | = |
| Dairy products | = | = |
| Fruits | Not enough evidence | Not enough evidence |
| Vegetables | + | − |
| Sweets | − | + |
| Fats & oil | Not enough evidence | Not enough evidence |
| Caffeine | − | + |
| Alcohol | − | + |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazri, F.H.; Manaf, Z.A.; Shahar, S.; Mat Ludin, A.F. The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. Int. J. Environ. Res. Public Health 2020, 17, 68. https://doi.org/10.3390/ijerph17010068
Mazri FH, Manaf ZA, Shahar S, Mat Ludin AF. The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. International Journal of Environmental Research and Public Health. 2020; 17(1):68. https://doi.org/10.3390/ijerph17010068
Chicago/Turabian StyleMazri, Fatin Hanani, Zahara Abdul Manaf, Suzana Shahar, and Arimi Fitri Mat Ludin. 2020. "The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review" International Journal of Environmental Research and Public Health 17, no. 1: 68. https://doi.org/10.3390/ijerph17010068
APA StyleMazri, F. H., Manaf, Z. A., Shahar, S., & Mat Ludin, A. F. (2020). The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. International Journal of Environmental Research and Public Health, 17(1), 68. https://doi.org/10.3390/ijerph17010068








