Legionnaires’ Disease Cases at a Large Community Hospital—Common and Underdiagnosed
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.1.1. Inclusion Criteria
- Age ≥ 18 years at enrollment.
- Diagnosis of pneumonia.
- Come to the Emergency department and requiring admission to our hospital via our Internal Medicine (general medical ward) or ICU (Intensive Care Unit) service.
2.1.2. Exclusion Criterion
- Name, date of birth, contact information, home address, work address, occupation, and travel history if applicable.
- Comorbidities as documented on the patient chart.
- Laboratory and imaging information, including final results of Legionella testing.
- Outcome (if known, including death, prolonged hospitalization, discharged [d/c] without complications).
2.2. Sample Collection and Assessment of Legionella Infection
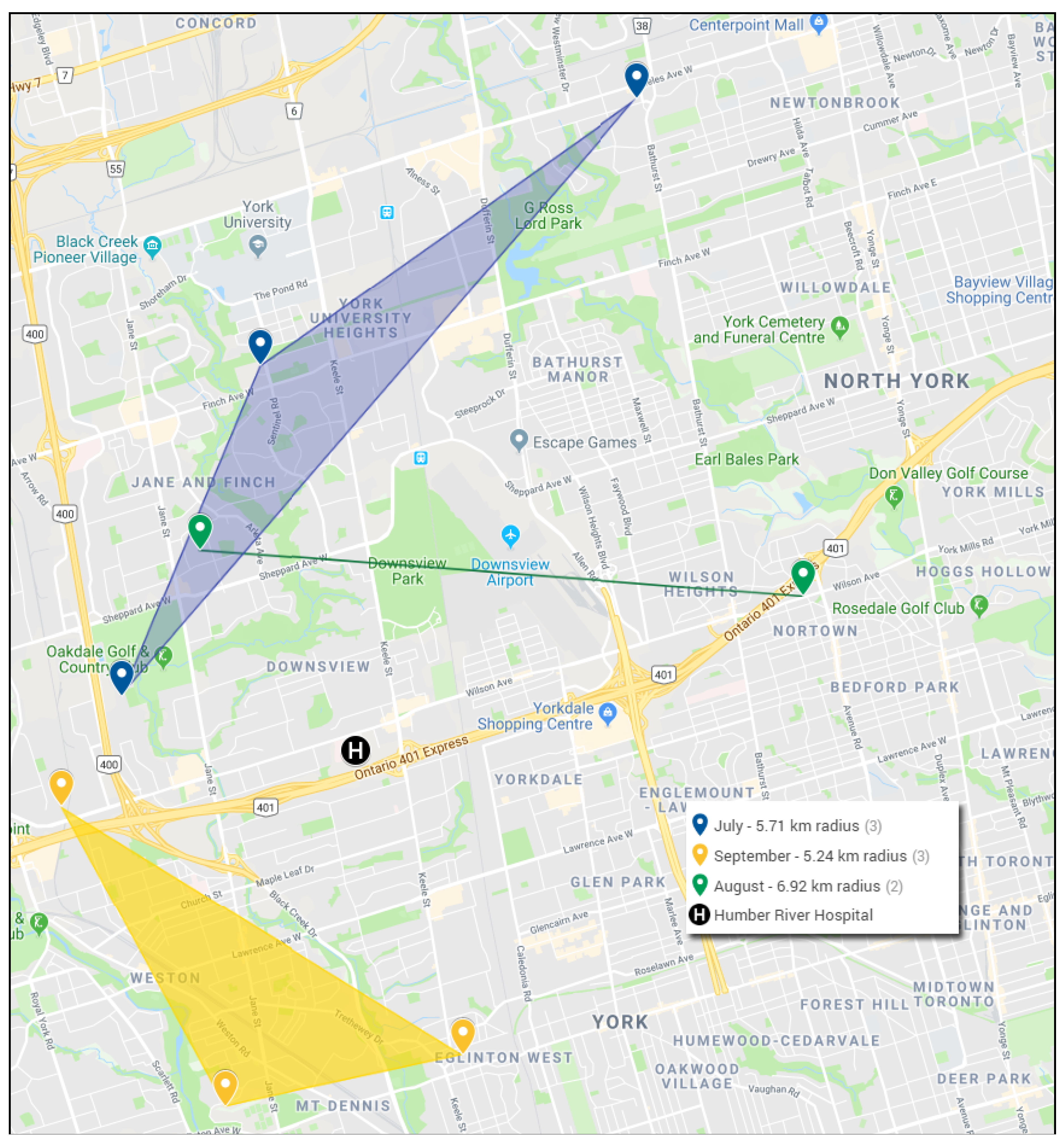
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fraser, D.W.; Tsai, T.R.; Orenstein, W.; Parkin, W.E.; Beecham, H.J.; Sharrar, R.G.; Harris, J.; Mallison, G.F.; Martin, S.M.; McDade, J.E.; et al. Legionnaires’ disease: Description of an epidemic of pneumonia. N. Engl. J. Med. 1977, 297, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Cunha, B.A.; Burillo, A.; Bouza, E. Legionnaires’ disease. Lancet 2016, 387, 376–385. [Google Scholar] [CrossRef]
- Kwong, J.C.; Crowcroft, N.S.; Campitelli, M.A.; Ratnasingham, S.; Daneman, N.; Deeks, S.L.; Manuel, D.G. Ontario Burden of Infections Disease Study (ONBOIDS): An OAHPP/ICES Report; Ontario Agency for Health Protection and Promotion, Institute for Clinical Evaluative Sciences: Toronto, ON, Canada, 2010. [Google Scholar]
- Canadian Institute for Health Information (CIHI). Leading Hospitalization Costs in Acute Inpatient Facilities in 2012–2013; CIHI: Ottawa, ON, Canada, 2014. [Google Scholar]
- Pierre, D.M.; Baron, J.; Yu, V.L.; Stout, J.E. Diagnostic testing for legionnaires’ disease. Ann. Clin. Microbiol. Antimicrob. 2017, 16, 59. [Google Scholar] [CrossRef] [PubMed]
- Mercante, J.W.; Winchell, J.M. Current and emerging Legionella diagnostics for laboratory and outbreak investigations. Clin. Microbiol. Rev. 2015, 28, 95–133. [Google Scholar] [CrossRef] [PubMed]
- Walser, S.M.; Gerstner, D.G.; Brenner, B.; Holler, C.; Liebl, B.; Herr, C.E. Assessing the environmental health relevance of cooling towers—A systematic review of legionellosis outbreaks. Int. J. Hyg. Environ. Health 2014, 217, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Marrie, T.J.; De Carolis, E.; Yu, V.L.; Stout, J. Canadian Community-Acquired Pneumonia, I. Legionnaires’ disease—Results of a multicentre Canadian study. Can. J. Infect. Dis. 2003, 14, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Mandell, L.A.; Marrie, T.J.; Grossman, R.F.; Chow, A.W.; Hyland, R.H. Canadian guidelines for the initial management of community-acquired pneumonia: An evidence-based update by the Canadian infectious diseases society and the Canadian thoracic society. The Canadian community-acquired pneumonia working group. Clin. Infect. Dis. 2000, 31, 383–421. [Google Scholar] [CrossRef] [PubMed]
- Yu, V.L.; Stout, J.E. Community-acquired legionnaires disease: Implications for underdiagnosis and laboratory testing. Clin. Infect. Dis. 2008, 46, 1365–1367. [Google Scholar] [CrossRef] [PubMed]
- Prussin, A.J., 2nd; Schwake, D.O.; Marr, L.C. Ten questions concerning the aerosolization and transmission of legionella in the built environment. Build. Environ. 2017, 123, 684–695. [Google Scholar] [CrossRef] [PubMed]
- Stout, J.E.; Muder, R.R.; Mietzner, S.; Wagener, M.M.; Perri, M.B.; DeRoos, K.; Goodrich, D.; Arnold, W.; Williamson, T.; Ruark, O.; et al. Role of environmental surveillance in determining the risk of hospital-acquired legionellosis: A national surveillance study with clinical correlations. Infect. Control Hosp. Epidemiol. 2007, 28, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Merault, N.; Rusniok, C.; Jarraud, S.; Gomez-Valero, L.; Cazalet, C.; Marin, M.; Brachet, E.; Aegerter, P.; Gaillard, J.L.; Etienne, J.; et al. Specific real-time PCR for simultaneous detection and identification of Legionella pneumophila serogroup 1 in water and clinical samples. Appl. Environ. Microbiol. 2011, 77, 1708–1717. [Google Scholar] [CrossRef] [PubMed]
- Llewellyn, A.C.; Lucas, C.E.; Roberts, S.E.; Brown, E.W.; Nayak, B.S.; Raphael, B.H.; Winchell, J.M. Distribution of Legionella and bacterial community composition among regionally diverse US cooling towers. PLoS ONE 2017, 12, e0189937. [Google Scholar] [CrossRef] [PubMed]
- French Ministry of the Environment. The Appearance of Legionella in the Cooling System of a Sugar Refinery; Bureau for Analysis of Industrial Risks and Pollutions: Lyon, France, 2000.
- Marshall, A.G.; Bellucci, E.C. Legionella pneumophila: A continuing threat. Hosp. Rev. 1986, 4, 6–16. [Google Scholar]
- Marston, B.J.; Lipman, H.B.; Breiman, R.F. Surveillance for legionnaires’ disease. Risk factors for morbidity and mortality. Arch. Intern. Med. 1994, 154, 2417–2422. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Legionnaires’ Disease in Europe, 2011; ECDC: Solna, Sweden, 2013. [Google Scholar]
| Date Collected (Year-Month-Day) | Sex | Age | Home Structure | Occupation/Industry | Smoking History | Co-Morbidities * | Legionella Status | Outcome | |
|---|---|---|---|---|---|---|---|---|---|
| 2018-07-09 | M | 57 | Detached-house | Driver | Current, 1 pk/d since 1974 | HTN | + | ICU admission | |
| 2018-07-02 | F | 87 | High-rise building | Retired | Never | CAD, PVD, CKD | + | Prolonged admission | |
| 2018-07-06 | F | 63 | High-rise building | Personal support worker | Never | HTN, Asthma | + | D/C without complication | |
| 2018-08-21 | F | 86 | High-rise building | Retired Nurse | Never | HTN, RA, AF | + | D/C without complication | |
| 2018-08-22 | M | 71 | Semi-detached house | Trucking Industry | Never | HTN, DM | + | D/C without complication | |
| 2018-09-03 | M | 54 | Detached house | Automotive Industry | Current, ½ pk/d since 1985 | - | + | D/C without complication | |
| 2018-09-06 | M | 65 | Detached house | Welder/bus driver | Former, 1 pk/d for 20 y | - | + | ICU admission | |
| 2018-09-02 | M | 69 | Semi-detached house | Heating industry | Never | HTN, CAD, Gout | + | ICU admission | |
| 2018-09-09 | M | 64 | High-rise building | Insurance | Never | Alcohol abuse | + | ICU admission | |
| 2018-08-27 | F | 36 | Low-rise building | Cashier | Current, 1 pk/d since 1998 | Asthma, DM | - | D/C without complication | |
| 2018-06-27 | F | 61 | Semi-detached house | On disability | Never | Multiple myeloma | - | Death | |
| Not performed | F | 86 | Detached house | Housewife | Never | HTN | NA | Prolonged Admission | |
| 2018-06-06 | F | 79 | Detached house | retired | Never | HTN, CKD, Gout | - | D/C without complication | |
| 2018-07-18 | F | 40 | High-rise building | Interpreter | Never | Crohn’s | - | D/C without complication | |
| 2018-07-26 | M | 84 | Detached house | Construction worker | Former, 40 y until 1998 | COPD, HTN, DM | - | D/C without complication | |
| 2018-07-25 | F | 47 | Low-rise building | Housewife | Never | CAD, Grave’s | - | D/C without complication | |
| Not performed | M | 88 | Semi-detached-house | Business owner | Former, 40 y until 1995 | HTN, lung cancer, DM | NA | D/C without complication | |
| 2018-07-25 | M | 74 | High-rise building | Retired, government | Former, 50 y until 2013 | Lung cancer, DM | - | D/C without complication | |
| 2018-08-08 | M | 87 | Detached house | Retired Librarian | Former, 56 y until 2003 | HTN, COPD, CKD | - | D/C without complication | |
| 2018-08-05 | F | 90 | High-rise building | Housewife | Current, ½ pk/d since 1958 | CAD, AF, CKD | - | D/C without complication | |
| Test cancelled | M | 46 | Townhouse | Construction | Current, ½ pk/d since 1998 | CHF | NA | D/C without complication | |
| 2018-08-02 | M | 53 | Semi-detached house | Construction worker | Current, 5 cig/wk since 1985 | CAD | - | D/C without complication | |
| 2018-08-12 | M | 81 | Detached house | Machinery Inspector | Never | CAD, DM | - | D/C without complication | |
| 2018-08-10 | M | 60 | High-rise building | Factory worker | Former, 30 y until 2014 | CHF, DM, HTN | - | D/C without complication | |
| 2018-08-20 | M | 74 | Low-rise building | Rental car/Airport | Former, 46 y until 2000 | HTN | - | D/C without complication | |
| 2018-08-19 | F | 93 | Detached house | Retired | Never | HTN | - | D/C without complication | |
| 2018-08-30 | M | 77 | Semi-detached house | Stone Mason | Former, 5 cig/d until August 2018 | AF, HTN, gout | - | D/C without complication | |
| 2018-08-28 | M | 66 | High-rise building | Truck driver | Never | - | - | D/C without complication | |
| 2018-08-30 | M | 88 | Detached house | Retired | Never | CAD, Prostate cancer | - | Prolonged Admission | |
| 2018-09-05 | M | 58 | Detached house | Maintenance | Current, 1 pk/d since 1978 | COPD | - | Prolonged Admission | |
| 2018-09-04 | M | 71 | Detached house | Car industry | Former, 20 y, 2–3 pk/d until 1999 | CAD, PVD, CKD, DM | - | Prolonged Admission | |
| 2018-09-02 | M | 57 | Detached house | Accounting | Former, 5 cig/d until 1984 | HTN | - | D/C without complication | |
| 2018-09-11 | M | 75 | Detached house | Housewife | Never | CHF, AF | - | Prolonged Admission | |
| 2018-09-19 | M | 85 | Detached house | Business/machine shop | Never | AF, CAD | - | ICU admission | |
| 2018-09-17 | M | 32 | Detached house | Security guard | Never | Asthma, Obese | - | D/C without complication | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spiegelman, J.; Pedutem, T.; Francisco, M.J. Legionnaires’ Disease Cases at a Large Community Hospital—Common and Underdiagnosed. Int. J. Environ. Res. Public Health 2020, 17, 332. https://doi.org/10.3390/ijerph17010332
Spiegelman J, Pedutem T, Francisco MJ. Legionnaires’ Disease Cases at a Large Community Hospital—Common and Underdiagnosed. International Journal of Environmental Research and Public Health. 2020; 17(1):332. https://doi.org/10.3390/ijerph17010332
Chicago/Turabian StyleSpiegelman, Jamie, Theresa Pedutem, and Mary June Francisco. 2020. "Legionnaires’ Disease Cases at a Large Community Hospital—Common and Underdiagnosed" International Journal of Environmental Research and Public Health 17, no. 1: 332. https://doi.org/10.3390/ijerph17010332
APA StyleSpiegelman, J., Pedutem, T., & Francisco, M. J. (2020). Legionnaires’ Disease Cases at a Large Community Hospital—Common and Underdiagnosed. International Journal of Environmental Research and Public Health, 17(1), 332. https://doi.org/10.3390/ijerph17010332





