A Pest to Mental Health? Exploring the Link between Exposure to Agrichemicals in Farmers and Mental Health
Abstract
:1. Introduction
1.1. The Mental Health Burden
1.2. Farmers’ Mental Health
1.3. The Influence of Farming Life and Culture on Mental Health
1.4. Agrichemical Exposure in Farming Populations
1.5. Aims of the Review
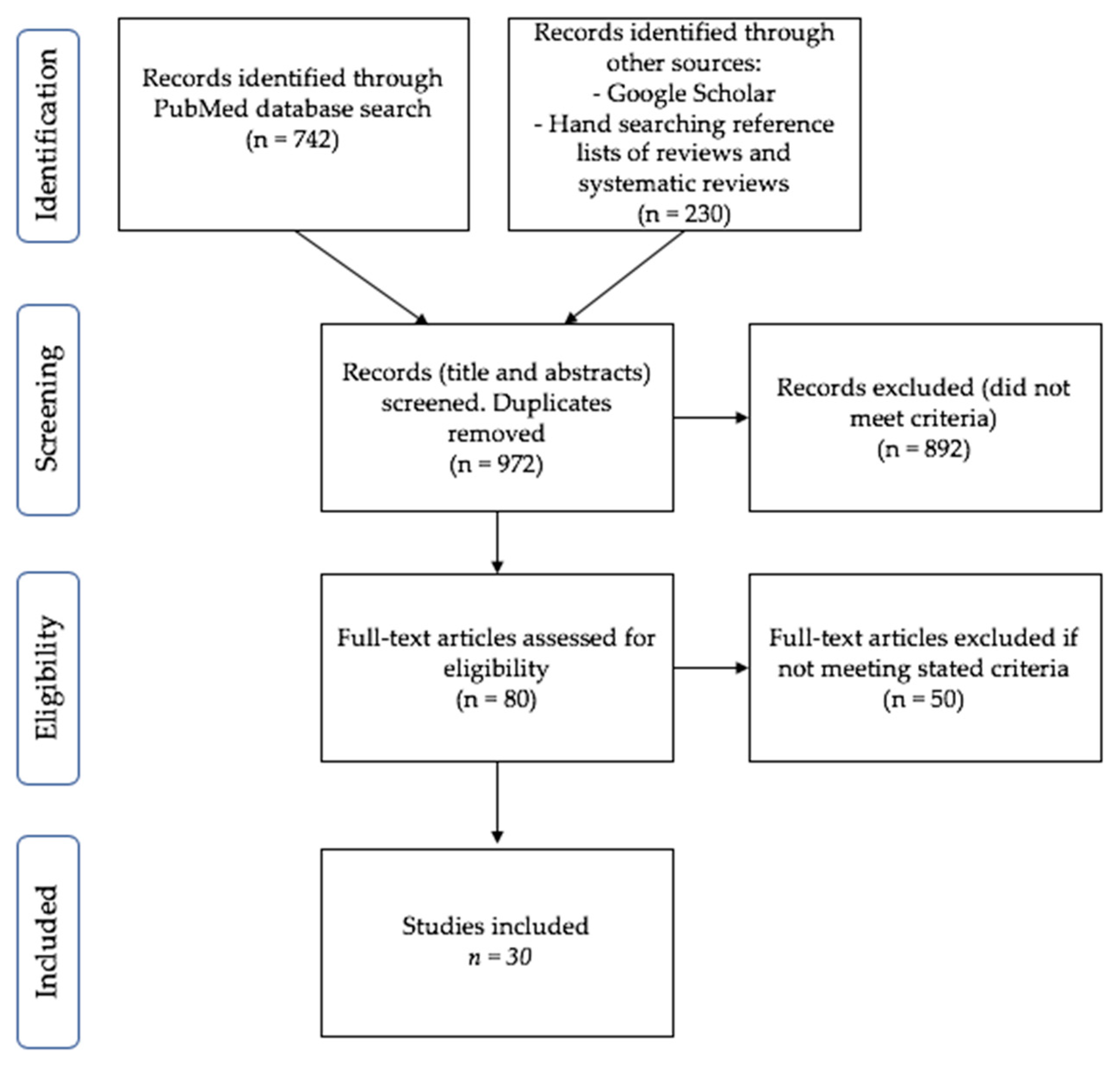
2. Methods
- Research reported on pesticide effect on mental health
- Full text of research article was in English
- Research was peer-reviewed
- Research focus was on a farming/agricultural context
- Research focusing on intentional exposures to pesticide (used for suicide/homicide) were excluded
- Focus on organophosphate-induced health effects
3. Results
3.1. Challenges of Determining Poor Mental Health and Suicide Rates
3.2. The Effect of Organophosphate Exposure on Mental Health
3.2.1. Epidemiological Evidence Displays Inconsistencies but Suggests a Viable Link
3.2.2. Stringent Studies Lay Foundations for the Association between Agrichemical Exposure and Poor Mental Health
3.2.3. Unclear Association between Agrichemical Use and Suicidality
| Study and Year | Study Design | Region | Population Source | Exposure Interest | Outcome Focus | Outcome Measure | Results * |
|---|---|---|---|---|---|---|---|
| Salvi et al., 2003 [59] | Longitudinal | Brazil | Agricultural tobacco farmers 62 participants | Organophosphate exposure | Neuropsychological (extrapyramidal symptoms), psychiatric (depression, anxiety) | MINI – Mini-International Neuropsychiatric Interview (structured questionnaire administered by a psychiatrist) | Three months of organophosphate-free period reduced diagnoses of psychiatric diagnoses |
| Fiedler et al., 1997 [56] | Cross-sectional | USA | Fruit farmers 99 participants | Organophosphate exposure | Neuropsychological | Minnesota Multiphasic Personality Inventory (MMPI-2) | No significant differences in mood |
| Weisskopf et al., 2013 [61] | Cross-sectional | France | Agricultural workers 781 participants | Pesticide exposure | Depression | Single question asking whether they had ever been treated with antidepressants, lithium or sismotherapy, or hospitalised for depression | Elevated depression rate in those using herbicides. Dose–response relationship identified for duration and intensity of use. |
| Mackenzie Ross et al., 2010 [58] | Cross-sectional | England | Sheep dippers 205 participants | Organophosphate exposure | Anxiety, Depression | Hospital Anxiety and Depression Scale | Anxiety and depression higher in exposed group (40% of exposed vs 23% of controls |
| Levin et al., 1976 [57] | Cross-sectional | USA | Commercial pesticide sprayers | Organophosphate exposure | Anxiety | Taylor Manifest Anxiety Scale (derived from the MMPI), Beck Depression Inventory | Sprayers showed higher anxiety levels and lower acetylcholinesterase (AChE) levels compared to controls, no difference in depression scores |
| Savage et al., 1988 [60] | Case-control | USA | Pesticide applicators 100 participants | Organophosphate poisoning | Neurobehavioural (memory, abstraction, reflexes), Mood | MMPI | Those with past poisoning had intellectual function scores consistent with individuals with cerebral damage or dysfunction. |
| Meyer et al., 2010 [82] | Ecological | Brazil (Rio) | Agricultural workers 3517 participants | Use of pesticides | Hospitalisation due to suicide attempts 1998–2007. Suicide deaths in 1981–2005. | Suicide deaths from the Brazilian National Mortality System (using WHO International Classification of Diseases and Related Health Problems (ICD). Hospitalisations due to suicide attempts or mood disorders from the Brazilian Hospital Information System (using ICD) | Suicide: agricultural workers at higher suicide mortality risk compared to three reference populations. Hospitalisation: Higher rates following suicide attempts/mood disorders, also compared to reference populations. |
| Wesseling et al., 2002 [64] | Cross-Sectional | Costa Rica | Banana planation 211 participants | Reduction in pesticide exposure | Neuropsychiatric symptoms | Questionnaire-16 and Brief Symptom Inventory (BSI) | Marked increase in neuropsychiatric symptoms observed in organophosphate-poisoned workers compared to controls |
| Beseler and Stallones, 2008 [62] | Prospective/Longitudinal | USA (Colorado) | Farm residents and spouses: CFFHHS Project 653 participants | Pesticide poisoning at baseline (1993): ever or never | Depressive symptoms: CES-D scale | Center for Epidemiologic Studies-Depression scale | Symptoms of depression were associated with participants that had a history of pesticide poisoning. |
| Parron et al., 2011 [80] | Ecological | Spain (Andalusia) | General population with neurological disorders 1349 participants | High vs Low pesticide exposure areas | Affective psychosis, Suicide attempts. | Hospital records (Andalusian Health Service Minimum Dataset) | Rates and risk of suicide and affective disorders found to be higher in populations exposed to higher levels of pesticides compared to populations exposed to lower levels |
| Kim et al., 2013 [63] | Cross-sectional | South Korea | Male farmers 1958 participants | Pesticide poisoning | Depressive symptoms | Face-to-face administering of the Korean version of the Geriatric Depression Screening Scale (short form) | Risk of depressive symptoms increased with pesticide poisoning (OR - 1.61, 95% CI, 1.10–2.34). Risk increased with severity of poisoning symptoms. |
| Beard et al., 2014 [66] | Cross-sectional | USA (Iowa and North Carolina) | Agricultural Health Study | Pesticide exposure | Depression | Single written questionnaire question: “Has a DOCTOR ever told you that you had (been diagnosed with) depression requiring medication or shock therapy?” | Positive association between depression and occupational pesticide use among applicators |
| Solomon et al., 2007 [69] | Cross-sectional | UK | Sheep dippers 9844 participants | Pesticide exposure | Anxiety, Depression | Written questionnaire including questions from the Hospital Anxiety and Depression Scale (symptoms experienced in the past 7 days) | Past use of pesticides not associated with anxiety and depression. |
| Delgado et al., 2004 [67] | Prospective | Nicaragua | Hospitalised patients from pesticide poisoning 81 participants | Organophosphate poisoning | Psychiatric symptoms | Modified Spanish version of the Q-16 assessing neuropsychiatric symptoms | Psychiatric symptoms increase with time since the poisoning event. |
| Bazylewicz-Walczak et al., 1999 [65] | Cross-sectional | Poland | Greenhouse workers and unexposed controls 51 participants | Organophosphate exposure | Depression and anxiety questionnaires before and after spraying season | Subclinical neurobehavioural effects using the World Health Organization (WHO) Neurobehavioral Core Test Battery (NCTB) | Increased anxiety, anger, fatigue, depression symptoms. N.B. No significant effects of exposure after a single spraying season. |
| Onwuameze et al., 2013 [68] | Longitudinal | USA (Iowa) | Iowa Certified Safe Farm study | Pesticide exposure | Self-reported depressive symptoms | Single written questionnaire asked quarterly throughout study: “How would you rate your level of depression in the last quarter?” | Pesticide exposure prospectively increased risk of depressive symptoms |
| Beseler et al., 2008 [71] | Nested case-control | USA (Iowa and North Carolina) | Agricultural Health Study 15,585 participants | Cumulative pesticide exposure: <226 days (low), 226–752 days (intermediate), >752 days (high). Diagnosed pesticide poisoning | Self-reported or medically diagnosed depression | Single written questionnaire question: “Has a DOCTOR ever told you that you had (been diagnosed with) depression requiring medication or shock therapy?” | Pesticide poisoning more strongly associated with depression than high cumulative exposure. However, high cumulative exposure in the absence of poisoning significantly associated with depression |
| Beseler et al., 2006 [70] | Nested case-control | USA (Iowa and North Carolina) | Agricultural Health Study 29,704 participants | Pesticide exposure | Self-reported or medically diagnosed depression | Single written questionnaire question: “Has a DOCTOR ever told you that you had (been diagnosed with) depression requiring medication or shock therapy?” | Depression significantly associated with history of pesticide poisoning but not with low or cumulative exposure |
| Harrison et al. 2016 [72] | Cross-sectional | England | Sheep farmers 205 participants | Organophosphate exposure | Anxiety and depression | Hospital Anxiety and Depression Scale, Beck Anxiety and Depression Inventories, Structured Clinical Interview (DSM-IV criteria) | Exposed cohort reported higher rates of depression and anxiety but only held true for anxiety when diagnostic interviews were utilised |
| Study and Year | Study Design | Region | Population Source | Exposure Interest | Outcome Focus | Outcome Measure | Results |
|---|---|---|---|---|---|---|---|
| Stephens et al., 1995 [73] | Cross-sectional | England | Sheep dippers 289 participants | Pesticide exposure | Neurobehavioral | General Health Questionnaire screening tool | Increased susceptibility to psychiatric disorders. |
| Farahat et al., 2003 [75] | Cross-sectional | Egypt | Cotton crop workers 102 participants | Organophosphate exposure | Neurobehavioral | Eysenck Personality Assessment Questionnaire (EPQ) | Statistically significant lower performance in neurobehavioral test for the exposed group. The longer exposure correlated with worsening performance. |
| Green et al., 1991 [81] | Cohort | Canada | Forestry workers 1222 participants | Phenoxy acid herbicides | Suicide/mortality | Canadian Mortality Database using ICD codes | Statistically significant increase in deaths from suicide for exposed cohort. |
| Wesseling et al., 2010 [77] | Cross-sectional | Costa Rica | Banana plantation 208 participants | Previous poisoning with cholinesterase-inhibiting pesticide | Suicidal ideation | Verbal administration of BSI – measuring psychological distress | Higher prevalence of somatisation, depression and anxiety disorders. Odds ratio for suicidal thoughts was 3.72. |
| Zhang et al., 2009 [85] | Cross-sectional | China | Rural residents 9811 participants | Storage of pesticide | Suicidal ideation | Verbal administration of questions to ascertain whether the respondent reported suicidal ideation in the two years before the interview. | Increased rates of suicide for cohort using pesticides. Also, higher incidence of depression and neuropsychiatric disturbances. |
| Parron et al., 1996 [83] | Ecological | Spain (Andalusia) | Agricultural workers 251 participants | Pesticide exposure | Suicide deaths | Suicides determined as cause of death (source of records not stated), psychological autopsy method used to determine reasons for suicide | Increased rates of suicide using pesticides. Higher incidence of depression and neuropsychiatric disturbances. |
| Pires et al., 2005 [84] | Ecological | Brazil | Agricultural workers 640 participants | Insecticides and herbicides | Suicide attempts and death | Suicide attempts determined from records of the Integrated Center for Toxicological Surveillance of the State Health Department of the State of Mato Grosso do Sul. Suicide deaths determined using data from Epidemiology Division of the State Health Secretariat of Mato Grosso do Sul using ICD-10 codes | Increased prevalence of suicide attempts and deaths in regions where higher insecticide use occurred compared with areas of lower use. |
| Ames et al., 1995 [74] | Cross-sectional | USA | Agricultural workers 135 participants | Organophosphate exposure | Mood | Mood scales from the Neurobehavioral Evaluation System (computerized assessment) | No significant abnormality in mood. |
| Jamal et al., 2002 [76] | Case-control | England | Sheep farmers 72 participants | Organophosphate exposure | Neuropathy, Depression and anxiety | Mood and affect assessed using General Health Questionnaire and the Hospital Anxiety and Depression Test | Increasing neuropathy associated with exposure. Neuropathy correlated to increased anxiety and depression scores |
| Amr et al., 1997 [78] | Cross-sectional | Egypt | Urban textile workers 503 participants | Pesticide exposure of formulators and applicators of pesticides | Depressive disorders and mood symptoms (insomnia, anhedonia, anxiety) | In-field screening using General Health Questionnaire, further in-field diagnosis by a psychiatrist according to DSM-III-R criteria | Higher depression in exposed subjects over controls. Particularly, those with longer-term exposure (> 20 years) |
| Roldan-Tapia et al., 2006 [79] | Cross-sectional | Spain | Greenhouse workers 92 participants | Carbamate and organophosphate poisoning | Neuropsychological performance | Taylor Anxiety Scale & Beck Depression Inventory | Exposure linked to increased anxiety. However, exposure for less than 10 years associated with profiles similar to unexposed controls. |
3.3. Lack of Data on Chronic Low-Level Exposure of Agrichemicals
3.4. Variability in Findings IS Linked to Inconsistencies in Study Designs
4. Discussion
4.1. Key Findings and Implications for Policy and Practice
4.2. Limitations of the Existing Literature
4.3. Recommendations for Future Research
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Department of Health. Australian Health Ministers, National Mental Health Plan 2003–2008. Available online: http://www.health.gov.au/internet/publications/publishing.nsf/Content/mental-pubs-n-infopri2-toc~mental-pubs-n-infopri2-exe (accessed on 20 May 2018).
- World Health Organisation. Social Determinants of Mental Health. Available online: http://www.who.int/mental_health/publications/gulbenkian_paper_social_determinants_of_mental_health/en/ (accessed on 20 May 2018).
- Whiteford, H.A.; Degenhardt, L.; Rehm, J.; Baxter, A.J.; Ferrari, A.J.; Erskine, H.E.; Charlson, F.J.; Norman, R.E.; Flaxman, A.D.; Johns, N.; et al. Global burden of disease attributable to mental and substance use disorders: Findings from the Global Burden of Disease Study 2010. Lancet 2013, 382, 1575–1586. [Google Scholar] [CrossRef]
- Kessler, R.C.; Aguilar-Gaxiola, S.; Alonso, J.; Chatterji, S.; Lee, S.; Ormel, J.; Üstün, T.B.; Wang, P.S. The global burden of mental disorders: An update from the WHO World Mental Health (WMH) Surveys. Epidemiol. Psichiatr. Soc. 2009, 18, 23–33. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation: Depression and Other Common Mental Disorders—Global Health Estimates. Available online: https://www.who.int/mental_health/management/depression/prevalence_global_health_estimates/en/ (accessed on 21 March 2019).
- Kupfer, D.J.; Regier, D.A.; Narrow, W.E.; Blazer, D.G.; Burke, J.D.; Carpenter, W.T.; Castellanos, F.X.; Compton, W.M.; Dimsdale, J.E.; Escobar, J.I.; et al. American Psychiatric Association 2013: Diagnostic and Statistical Manual of Mental Disorders: Diagnostic and Statistical Manual of Mental Disorders DSM-5, 5th ed.; Americal Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- Australian Bureau of Statistics. National Survey of Mental Health and Wellbeing: Summary of Results. 2007. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/mf/4326.0 (accessed on 20 May 2018).
- Australian Bureau of Statistics. Gender Indicators, Australia. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/4125.0main+features3150Jan%202013 (accessed on 20 May 2018).
- Engel, G.L. The need for a new medical model: A challenge for biomedicine. Science 1977, 196, 129–136. [Google Scholar] [CrossRef]
- Borrell-Carrió, F.; Suchman, A.L.; Epstein, R.M. The biopsychosocial model 25 years later: Principles, practice, and scientific inquiry. Ann. Fam. Med. 2004, 2, 576–582. [Google Scholar] [CrossRef]
- Roberts, R.E.; Lee, E.S. Occupation and the prevalence of major depression, alcohol, and drug abuse in the United States. Environ. Res. 1993, 61, 266–278. [Google Scholar] [CrossRef]
- Sanne, B.; Mykletun, A.; Dahl, A.A.; Moen, B.E.; Tell, G.S. Occupational differences in levels of anxiety and depression: The Hordaland Health Study. J. Occup. Environ. Med. 2003, 45, 628–638. [Google Scholar] [CrossRef]
- Klingelschmidt, J.; Milner, A.; Khireddine-Medouni, I.; Witt, K.; Alexopoulos, E.C.; Toivanen, S.; LaMontagne, A.D.; Chastang, J.F.; Niedhammer, I. Suicide among agricultural, forestry, and fishery workers: A systematic literature review and meta-analysis. Scand. J. Work. Health 2018, 44, 3–15. [Google Scholar] [CrossRef]
- Judd, F.; Jackson, H.; Fraser, C.; Murray, G.; Robins, G.; Komiti, A. Understanding suicide in Australian farmers. Soc. Psychiatry Psychiatr. Epidemiol. 2006, 41, 1–10. [Google Scholar] [CrossRef]
- Kunde, L.; Kõlves, K.; Kelly, B.; Reddy, P.; De Leo, D. Pathways to Suicide in Australian Farmers: A Life Chart Analysis. Int. J. Environ. Res. Public Health 2017, 14, 352. [Google Scholar] [CrossRef] [PubMed]
- Fragar, L.; Stain, H.J.; Perkins, D.; Kelly, B.; Fuller, J.; Coleman, C.; Lewin, T.J.; Wilson, J.M. Distress among rural residents: Does employment and occupation make a difference? Aust. J. Rural Health 2010, 18, 25–31. [Google Scholar] [CrossRef]
- Thomas, H.V.; Lewis, G.; Thomas, D.R.; Salmon, R.L.; Chalmers, R.M.; Coleman, T.J.; Kench, S.M.; Morgan-Capner, P.; Meadows, D.; Sillis, M.; et al. Mental health of British farmers. Occup. Environ. Med. 2003, 60, 181–185, discussion 185–186. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.; Hawgood, J.; Klieve, H.; Kolves, K.; De Leo, D. Suicide in selected occupations in Queensland: Evidence from the State suicide register. Aust. N. Z. J. Psychiatry 2010, 44, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Malmberg, A.; Simkin, S.; Hawton, K. Suicide in farmers. Br. J. Psychiatry J. Ment. Sci. 1999, 175, 103–105. [Google Scholar] [CrossRef]
- Kelly, S.; Bunting, J. Trends in suicide in England and Wales, 1982–1996. Popul. Trends 1998, 92, 29–41. [Google Scholar]
- Page, A.N.; Fragar, L.J. Suicide in Australian farming, 1988–1997. Aust. N. Z. J. Psychiatry 2002, 36, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Arnautovska, U.; McPhedran, S.; Kelly, B.; Reddy, P.; De Leo, D. Geographic variation in suicide rates in Australian farmers: Why is the problem more frequent in Queensland than in New South Wales? Death Stud. 2016, 40, 367–372. [Google Scholar] [CrossRef]
- Arnautovska, U.; McPhedran, S.; De Leo, D. A regional approach to understanding farmer suicide rates in Queensland. Soc. Psychiatry Psychiatr. Epidemiol. 2014, 49, 593–599. [Google Scholar] [CrossRef]
- Caldwell, T.M.; Jorm, A.F.; Dear, K.B. Suicide and mental health in rural, remote and metropolitan areas in Australia. Med. J. Aust. 2004, 181, S10–S14. [Google Scholar]
- Freire, C.; Koifman, S. Pesticides, depression and suicide: A systematic review of the epidemiological evidence. Int. J. Hyg. Environ. Health 2013, 216, 445–460. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. 4102.0—Australian Social Trends. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/Lookup/4102.0Main+Features10Dec+2012 (accessed on 20 October 2018).
- Australian Bureau of Statistics—2011 Census QuickStats. Available online: http://quickstats.censusdata.abs.gov.au/census_services/getproduct/census/2011/quickstat/0 (accessed on 20 October 2018).
- Australian Bureau of Statistics. 4627.0—Land Management and Farming in Australia, 2016–2017. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/mf/4627.0 (accessed on 20 October 2018).
- Thelin, A.; Holmberg, S. Farmers and retirement: A longitudinal cohort study. J. Agromed. 2010, 15, 38–46. [Google Scholar] [CrossRef]
- Brew, B.; Inder, K.; Allen, J.; Thomas, M.; Kelly, B. The health and wellbeing of Australian farmers: A longitudinal cohort study. BMC Public Health 2016, 16, 988. [Google Scholar] [CrossRef] [PubMed]
- Fraser, C.E.; Smith, K.B.; Judd, F.; Humphreys, J.S.; Fragar, L.J.; Henderson, A. Farming and mental health problems and mental illness. Int. J. Soc. Psychiatry 2005, 51, 340–349. [Google Scholar] [CrossRef]
- Bouwmeester, O.; Kok, T. Moral or Dirty Leadership: A Qualitative Study on How Juniors Are Managed in Dutch Consultancies. Int. J. Environ. Res. Public Health 2018, 15, 2506. [Google Scholar] [CrossRef]
- Del Pozo-Antúnez, J.; Ariza-Montes, A.; Fernández-Navarro, F.; Molina-Sánchez, H. Effect of a Job Demand-Control-Social Support Model on Accounting Professionals’ Health Perception. Int. J. Environ. Res. Public Health 2018, 15, 2437. [Google Scholar] [CrossRef]
- Peters, S.; Grant, M.; Rodgers, J.; Manjourides, J.; Okechukwu, C.; Dennerlein, J. A Cluster Randomized Controlled Trial of a Total Worker Health® Intervention on Commercial Construction Sites. Int. J. Environ. Res. Public Health 2018, 15, 2354. [Google Scholar] [CrossRef]
- Thompson, J.; Schwatka, N.; Tenney, L.; Newman, L. Total Worker Health: A Small Business Leader Perspective. Int. J. Environ. Res. Public Health 2018, 15, 2416. [Google Scholar] [CrossRef] [PubMed]
- Yoo, G.; Lee, S. It Doesn’t End There: Workplace Bullying, Work-to-Family Conflict, and Employee Well-Being in Korea. Int. J. Environ. Res. Public Health 2018, 15, 1548. [Google Scholar] [CrossRef]
- Chari, R.; Chang, C.C.; Sauter, S.L.; Petrun Sayers, E.L.; Cerully, J.L.; Schulte, P.; Schill, A.L.; Uscher-Pines, L. Expanding the Paradigm of Occupational Safety and Health: A New Framework for Worker Well-Being. J. Occup. Environ. Med. 2018, 60, 589–593. [Google Scholar] [CrossRef]
- Brumby, S.; Willder, S.J.; Martin, J. Milking their health for all its worth? Improving the health of farming families through facilitated learning. Ext. Farming Syst. J. 2010, 6, 1. [Google Scholar]
- Brumby, S.A.; Willder, S.J.; Martin, J. The sustainable farm families project: Changing attitudes to health. Rural Remote Health 2009, 9, 1012. [Google Scholar] [CrossRef]
- Brumby, S.; Hatherell, T.; Jensen, J.; Nelson, L. 1412 Sustainable farm families across the globe—The most important part of any farm is a healthy farm family. Occup. Environ. Med. 2018, 75, A466. [Google Scholar] [CrossRef]
- Muñoz-Quezada, M.T.; Lucero, B.A.; Iglesias, V.P.; Muñoz, M.P.; Cornejo, C.A.; Achu, E.; Baumert, B.; Hanchey, A.; Concha, C.; Brito, A.M.; et al. Chronic exposure to organophosphate (OP) pesticides and neuropsychological functioning in farm workers: A review. Int. J. Occup. Environ. Health 2016, 22, 68–79. [Google Scholar] [CrossRef]
- Aktar, M.W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdiscip. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef]
- Cotton, J.; Lewandowski, P.; Brumby, S. Cholinesterase Research Outreach Project (CROP): Measuring cholinesterase activity and pesticide use in an agricultural community. BMC Public Health 2015, 15, 748. [Google Scholar] [CrossRef]
- De Silva, H.J.; Samarawickrema, N.A.; Wickremasinghe, A.R. Toxicity due to organophosphorus compounds: What about chronic exposure? Trans. R. Soc. Trop. Med. Hyg. 2006, 100, 803–806. [Google Scholar] [CrossRef]
- Ross, S.M.; McManus, I.C.; Harrison, V.; Mason, O. Neurobehavioral problems following low-level exposure to organophosphate pesticides: A systematic and meta-analytic review. Crit. Rev. Toxicol. 2013, 43, 21–44. [Google Scholar] [CrossRef]
- Nalwanga, E.; Ssempebwa, J.C. Knowledge and practices of in-home pesticide use: A community survey in Uganda. J. Environ. Public Health 2011, 2011, 230894. [Google Scholar] [CrossRef]
- Ibitayo, O.O. Egyptian farmers’ attitudes and behaviors regarding agricultural pesticides: Implications for pesticide risk communication. Risk Anal.: Off. Publ. Soc. Risk Anal. 2006, 26, 989–995. [Google Scholar] [CrossRef]
- Sivayoganathan, C.; Gnanachandran, S.; Lewis, J.; Fernando, M. Protective measure use and symptoms among agropesticide applicators in Sri Lanka. Soc. Sci. Med. (1982) 1995, 40, 431–436. [Google Scholar] [CrossRef]
- Mekonnen, Y.; Agonafir, T. Pesticide sprayers’ knowledge, attitude and practice of pesticide use on agricultural farms of Ethiopia. Occup. Med. (Oxf. Engl.) 2002, 52, 311–315. [Google Scholar]
- Gesesew, H.A.; Woldemichael, K.; Massa, D.; Mwanri, L. Farmers Knowledge, Attitudes, Practices and Health Problems Associated with Pesticide Use in Rural Irrigation Villages, Southwest Ethiopia. PLoS ONE 2016, 11, e0162527. [Google Scholar] [CrossRef]
- Lekei, E.E.; Ngowi, A.V.; London, L. Farmers’ knowledge, practices and injuries associated with pesticide exposure in rural farming villages in Tanzania. BMC Public Health 2014, 14, 389. [Google Scholar] [CrossRef]
- Kennedy, A.J.; Maple, M.J.; McKay, K.; Brumby, S.A. Suicide and accidental death in Australia’s rural farming communities: A review of the literature. Rural Remote Health 2014, 14, 2517. [Google Scholar]
- Research Compendium—The NIOSH Total Worker Health (TM) Program: Seminal Research Papers 2012. Available online: https://www.cdc.gov/niosh/docs/2012-146/pdfs/2012-146.pdf?id=10.26616/NIOSHPUB2012146 (accessed on 14 November 2018).
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Boell, S.K.; Cecez-Kecmanovic, D. A Hermeneutic Approach for Conducting Literature Reviews and Literature Searches. Commun. Assoc. Inf. Syst. 2014, 34, 257–286. [Google Scholar] [CrossRef]
- Fiedler, N.; Kipen, H.; Kelly-McNeil, K.; Fenske, R. Long-term use of organophosphates and neuropsychological performance. Am. J. Ind. Med. 1997, 32, 487–496. [Google Scholar] [CrossRef]
- Levin, H.S.; Rodnitzky, R.L.; Mick, D.L. Anxiety associated with exposure to organophosphate compounds. Arch. Gen. Psychiatry 1976, 33, 225–228. [Google Scholar] [CrossRef]
- Mackenzie Ross, S.J.; Brewin, C.R.; Curran, H.V.; Furlong, C.E.; Abraham-Smith, K.M.; Harrison, V. Neuropsychological and psychiatric functioning in sheep farmers exposed to low levels of organophosphate pesticides. Neurotoxicol. Teratol. 2010, 32, 452–459. [Google Scholar] [CrossRef]
- Salvi, R.M.; Lara, D.R.; Ghisolfi, E.S.; Portela, L.V.; Dias, R.D.; Souza, D.O. Neuropsychiatric evaluation in subjects chronically exposed to organophosphate pesticides. Toxicol. Sci. Off. J. Soc. Toxicol. 2003, 72, 267–271. [Google Scholar] [CrossRef]
- Savage, E.P.; Keefe, T.J.; Mounce, L.M.; Heaton, R.K.; Lewis, J.A.; Burcar, P.J. Chronic neurological sequelae of acute organophosphate pesticide poisoning. Arch. Environ. Health 1988, 43, 38–45. [Google Scholar] [CrossRef]
- Weisskopf, M.G.; Moisan, F.; Tzourio, C.; Rathouz, P.J.; Elbaz, A. Pesticide exposure and depression among agricultural workers in France. Am. J. Epidemiol. 2013, 178, 1051–1058. [Google Scholar] [CrossRef]
- Beseler, C.L.; Stallones, L. A cohort study of pesticide poisoning and depression in Colorado farm residents. Ann. Epidemiol. 2008, 18, 768–774. [Google Scholar] [CrossRef]
- Kim, J.; Ko, Y.; Lee, W.J. Depressive symptoms and severity of acute occupational pesticide poisoning among male farmers. Occup. Environ. Med. 2013, 70, 303–309. [Google Scholar] [CrossRef]
- Wesseling, C.; Keifer, M.; Ahlbom, A.; McConnell, R.; Moon, J.-D.; Rosenstock, L.; Hogstedt, C. Long-Term Neurobehavioral Effects of Mild Poisonings with Organophosphate and n-Methyl Carbamate Pesticides among Banana Workers. Int. J. Occup. Environ. Health 2002, 8, 27–34. [Google Scholar] [CrossRef]
- Bazylewicz-Walczak, B.; Majczakowa, W.; Szymczak, M. Behavioral effects of occupational exposure to organophosphorous pesticides in female greenhouse planting workers. Neurotoxicology 1999, 20, 819–826. [Google Scholar]
- Beard, J.D.; Umbach, D.M.; Hoppin, J.A.; Richards, M.; Alavanja, M.C.; Blair, A.; Sandler, D.P.; Kamel, F. Pesticide exposure and depression among male private pesticide applicators in the agricultural health study. Environ. Health Perspect. 2014, 122, 984–991. [Google Scholar] [CrossRef]
- Delgado, E.; McConnell, R.; Miranda, J.; Keifer, M.; Lundberg, I.; Partanen, T.; Wesseling, C. Central nervous system effects of acute organophosphate poisoning in a two-year follow-up. Scand. J. Work. Health 2004, 30, 362–370. [Google Scholar] [CrossRef]
- Onwuameze, O.E.; Paradiso, S.; Peek-Asa, C.; Donham, K.J.; Rautiainen, R.H. Modifiable risk factors for depressed mood among farmers. Ann. Clin. Psychiatry Off. J. Am. Acad. Clin. Psychiatr. 2013, 25, 83–90. [Google Scholar]
- Solomon, C.; Poole, J.; Palmer, K.T.; Peveler, R.; Coggon, D. Neuropsychiatric symptoms in past users of sheep dip and other pesticides. Occup. Environ. Med. 2007, 64, 259–266. [Google Scholar] [CrossRef]
- Beseler, C.; Stallones, L.; Hoppin, J.A.; Alavanja, M.C.; Blair, A.; Keefe, T.; Kamel, F. Depression and pesticide exposures in female spouses of licensed pesticide applicators in the agricultural health study cohort. J. Occup. Environ. Med. 2006, 48, 1005–1013. [Google Scholar] [CrossRef]
- Beseler, C.L.; Stallones, L.; Hoppin, J.A.; Alavanja, M.C.; Blair, A.; Keefe, T.; Kamel, F. Depression and pesticide exposures among private pesticide applicators enrolled in the Agricultural Health Study. Environ. Health Perspect. 2008, 116, 1713–1719. [Google Scholar] [CrossRef]
- Harrison, V.; Mackenzie Ross, S. Anxiety and depression following cumulative low-level exposure to organophosphate pesticides. Environ. Res. 2016, 151, 528–536. [Google Scholar] [CrossRef]
- Stephens, R.; Spurgeon, A.; Calvert, I.A.; Beach, J.; Levy, L.S.; Berry, H.; Harrington, J.M. Neuropsychological effects of long-term exposure to organophosphates in sheep dip. Lancet 1995, 345, 1135–1139. [Google Scholar] [CrossRef]
- Ames, R.G.; Steenland, K.; Jenkins, B.; Chrislip, D.; Russo, J. Chronic neurologic sequelae to cholinesterase inhibition among agricultural pesticide applicators. Arch. Environ. Health 1995, 50, 440–444. [Google Scholar] [CrossRef]
- Farahat, T.M.; Abdelrasoul, G.M.; Amr, M.M.; Shebl, M.M.; Farahat, F.M.; Anger, W.K. Neurobehavioural effects among workers occupationally exposed to organophosphorous pesticides. Occup. Environ. Med. 2003, 60, 279–286. [Google Scholar] [CrossRef]
- Jamal, G.A.; Hansen, S.; Pilkington, A.; Buchanan, D.; Gillham, R.A.; Abdel-Azis, M.; Julu, P.O.; Al-Rawas, S.F.; Hurley, F.; Ballantyne, J.P. A clinical neurological, neurophysiological, and neuropsychological study of sheep farmers and dippers exposed to organophosphate pesticides. Occup. Environ. Med. 2002, 59, 434–441. [Google Scholar] [CrossRef]
- Wesseling, C.; van Wendel de Joode, B.; Keifer, M.; London, L.; Mergler, D.; Stallones, L. Symptoms of psychological distress and suicidal ideation among banana workers with a history of poisoning by organophosphate or n-methyl carbamate pesticides. Occup. Environ. Med. 2010, 67, 778–784. [Google Scholar] [CrossRef]
- Amr, M.M.; Halim, Z.S.; Moussa, S.S. Psychiatric disorders among Egyptian pesticide applicators and formulators. Environ. Res. 1997, 73, 193–199. [Google Scholar] [CrossRef]
- Roldan-Tapia, L.; Nieto-Escamez, F.A.; del Aguila, E.M.; Laynez, F.; Parron, T.; Sanchez-Santed, F. Neuropsychological sequelae from acute poisoning and long-term exposure to carbamate and organophosphate pesticides. Neurotoxicol. Teratol. 2006, 28, 694–703. [Google Scholar] [CrossRef]
- Parron, T.; Requena, M.; Hernandez, A.F.; Alarcon, R. Association between environmental exposure to pesticides and neurodegenerative diseases. Toxicol. Appl. Pharmacol. 2011, 256, 379–385. [Google Scholar] [CrossRef]
- Green, L.M. A cohort mortality study of forestry workers exposed to phenoxy acid herbicides. Br. J. Ind. Med. 1991, 48, 234–238. [Google Scholar] [CrossRef]
- Meyer, A.; Koifman, S.; Koifman, R.J.; Moreira, J.C.; de Rezende Chrisman, J.; Abreu-Villaca, Y. Mood disorders hospitalizations, suicide attempts, and suicide mortality among agricultural workers and residents in an area with intensive use of pesticides in Brazil. J. Toxicol. Environ. Health. Part A 2010, 73, 866–877. [Google Scholar] [CrossRef]
- Parron, T.; Hernandez, A.F.; Villanueva, E. Increased risk of suicide with exposure to pesticides in an intensive agricultural area. A 12-year retrospective study. Forensic Sci. Int. 1996, 79, 53–63. [Google Scholar] [CrossRef]
- Pires, D.X.; Caldas, E.D.; Recena, M.C. Pesticide use and suicide in the State of Mato Grosso do Sul, Brazil. Cad. De Saude Publica 2005, 21, 598–605. [Google Scholar] [CrossRef]
- Zhang, J.; Stewart, R.; Phillips, M.; Shi, Q.; Prince, M. Pesticide exposure and suicidal ideation in rural communities in Zhejiang province, China. Bull. World Health Organ. 2009, 87, 745–753. [Google Scholar]
- O’Malley, M. Clinical evaluation of pesticide exposure and poisonings. Lancet 1997, 349, 1161–1166. [Google Scholar] [CrossRef]
- Rohlman, D.S.; Anger, W.K.; Lein, P.J. Correlating neurobehavioral performance with biomarkers of organophosphorous pesticide exposure. Neurotoxicology 2011, 32, 268–276. [Google Scholar] [CrossRef]
- Banks, C.N.; Lein, P.J. A review of experimental evidence linking neurotoxic organophosphorus compounds and inflammation. Neurotoxicology 2012, 33, 575–584. [Google Scholar] [CrossRef]
- Durham, W.F.; Wolfe, H.R.; Quinby, G.E. Organophosphorus Insecticides and Mental Alertness. Arch. Environ. Health: Int. J. 1965, 10, 55–66. [Google Scholar] [CrossRef]
- Gershon, S.; Shaw, F.H. Psychiatric sequelae of chronic exposure to organophosphorus insecticides. Lancet 1961, 1, 1371–1374. [Google Scholar] [CrossRef]
- Grob, D.; Lilienthal, J.L., Jr.; Harvey, A.M.; Jones, B.F. The administration of di-isopropyl fluorophosphate (DFP) to man; effect on plasma and erythrocyte cholinesterase; general systemic effects; use in study of hepatic function and erythropoiesis; and some properties of plasma cholinesterase. Bull. Johns Hopkins Hosp. 1947, 81, 217–244. [Google Scholar]
- Metcalf, D.R.; Holmes, J.H., VII. Toxicology and physiology. EEG, psychological, and neurological alterations in humans with organophosphorus exposure. Ann. N. Y. Acad. Sci. 1969, 160, 357–365. [Google Scholar]
- Dille, J.R.; Smith, P.W. Central nervous system effects of chronic exposure to organophosphate insecticides. Aerosp. Med. 1964, 35, 474–478. [Google Scholar]
- Wesseling, C.; van Wendel de Joode, B.; Monge, P. Pesticide-related illness and injuries among banana workers in Costa Rica: A comparison between 1993 and 1996. Int. J. Occup. Environ. Health 2001, 7, 90–97. [Google Scholar] [CrossRef]
- Lundberg, I.; Hogberg, M.; Michelsen, H.; Nise, G.; Hogstedt, C. Evaluation of the Q16 questionnaire on neurotoxic symptoms and a review of its use. Occup. Environ. Med. 1997, 54, 343–350. [Google Scholar] [CrossRef]
- Stallones, L.; Beseler, C.L. Assessing the connection between organophosphate pesticide poisoning and mental health: A comparison of neuropsychological symptoms from clinical observations, animal models and epidemiological studies. Cortex J. Devoted Study Nerv. Syst. Behav. 2016, 74, 405–416. [Google Scholar] [CrossRef]
- Genuis, S.J. Our genes are not our destiny: Incorporating molecular medicine into clinical practice. J. Eval. Clin. Pract. 2008, 14, 94–102. [Google Scholar] [CrossRef]
- Rodnitzky, R.L. Occupational exposure to organophosphate pesticides: A neurobehavioral study. Arch. Environ. Health 1975, 30, 98–103. [Google Scholar] [CrossRef]
- Orozco, F.A.; Cole, D.C.; Ibrahim, S.; Wanigaratne, S. Health promotion outcomes associated with a community-based program to reduce pesticide-related risks among small farm households. Health Promot. Int. 2011, 26, 432–446. [Google Scholar] [CrossRef]
- Salvatore, A.L.; Chevrier, J.; Bradman, A.; Camacho, J.; Lopez, J.; Kavanagh-Baird, G.; Minkler, M.; Eskenazi, B. A community-based participatory worksite intervention to reduce pesticide exposures to farmworkers and their families. Am. J. Public Health 2009, 99 (Suppl. 3), S578–S581. [Google Scholar] [CrossRef]
- Lehtola, M.M.; Rautiainen, R.H.; Day, L.M.; Schonstein, E.; Suutarinen, J.; Salminen, S.; Verbeek, J.H. Effectiveness of interventions in preventing injuries in agriculture—A systematic review and meta-analysis. Scand. J. Work. Health 2008, 34, 327–336. [Google Scholar] [CrossRef]
- Brumby, S.; Martin, J.; Willder, S. Sustainable Farm Families: Future Directions; Rural Industries Research and Development Corporation (RIRDC): Barton, Australia, 2013. [Google Scholar]
- Pearson, D. Victorian Auditor-General’s Report: Sustainable Farm Families Program (ISBN 978 1 921650 50 5). Available online: http://download.audit.vic.gov.au/files/20100109_SFF_full_report.pdf (accessed on 27 February 2019).
- Brumby, S. 1659i Agrichemical exposure in australian farmers—Monitoring, measuring and making a difference. Occup. Environ. Med. 2018, 75, A471. [Google Scholar] [CrossRef]
- Brumby, S.A.; Ruldolphi, J.; Rohlman, D.; Donham, K.J. Translating agricultural health and medicine education across the Pacific: A United States and Australian comparison study. Rural Remote Health 2017, 17, 3931. [Google Scholar] [CrossRef]
- Alavanja, M.C.R. Pesticides Use and Exposure Extensive Worldwide. Rev. Environ. Health 2009, 24, 303–309. [Google Scholar] [CrossRef]
- Alavanja, M.C.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef]
- Blair, A.; Hayes, R.B.; Stewart, P.A.; Zahm, S.H. Occupational epidemiologic study design and application. Occup. Med. (Phila. PA) 1996, 11, 403–419. [Google Scholar]
- Arcury, T.A.; Quandt, S.A. Chronic agricultural chemical exposure among migrant and seasonal farmworkers. Soc. Nat. Resour. 1998, 11, 829–843. [Google Scholar] [CrossRef]
- Colosio, C.; Tiramani, M.; Maroni, M. Neurobehavioral effects of pesticides: State of the art. Neurotoxicology 2003, 24, 577–591. [Google Scholar] [CrossRef]
- Beseler, C.L.; Stallones, L. Safety knowledge, safety behaviors, depression, and injuries in Colorado farm residents. Am. J. Ind. Med. 2010, 53, 47–54. [Google Scholar] [CrossRef]
- Cherry, N.; Mackness, M.; Durrington, P.; Povey, A.; Dippnall, M.; Smith, T.; Mackness, B. Paraoxonase (PON1) polymorphisms in farmers attributing ill health to sheep dip. Lancet 2002, 359, 763–764. [Google Scholar] [CrossRef]
- Mackness, B.; Durrington, P.; Povey, A.; Thomson, S.; Dippnall, M.; Mackness, M.; Smith, T.; Cherry, N. Paraoxonase and susceptibility to organophosphorus poisoning in farmers dipping sheep. Pharmacogenetics 2003, 13, 81–88. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, N.; Kennedy, A.; Cotton, J.; Brumby, S. A Pest to Mental Health? Exploring the Link between Exposure to Agrichemicals in Farmers and Mental Health. Int. J. Environ. Res. Public Health 2019, 16, 1327. https://doi.org/10.3390/ijerph16081327
Khan N, Kennedy A, Cotton J, Brumby S. A Pest to Mental Health? Exploring the Link between Exposure to Agrichemicals in Farmers and Mental Health. International Journal of Environmental Research and Public Health. 2019; 16(8):1327. https://doi.org/10.3390/ijerph16081327
Chicago/Turabian StyleKhan, Nufail, Alison Kennedy, Jacqueline Cotton, and Susan Brumby. 2019. "A Pest to Mental Health? Exploring the Link between Exposure to Agrichemicals in Farmers and Mental Health" International Journal of Environmental Research and Public Health 16, no. 8: 1327. https://doi.org/10.3390/ijerph16081327
APA StyleKhan, N., Kennedy, A., Cotton, J., & Brumby, S. (2019). A Pest to Mental Health? Exploring the Link between Exposure to Agrichemicals in Farmers and Mental Health. International Journal of Environmental Research and Public Health, 16(8), 1327. https://doi.org/10.3390/ijerph16081327





