Shedding Light on Vitamin D Status and Its Complexities during Pregnancy, Infancy and Childhood: An Australian Perspective
Abstract
1. Introduction
1.1. Traditional Practices for Prevention and Treatment of Rickets
1.1.1. Cod Liver Oil
1.1.2. “Sunning”
2. A Resurgence of Rickets in Australia
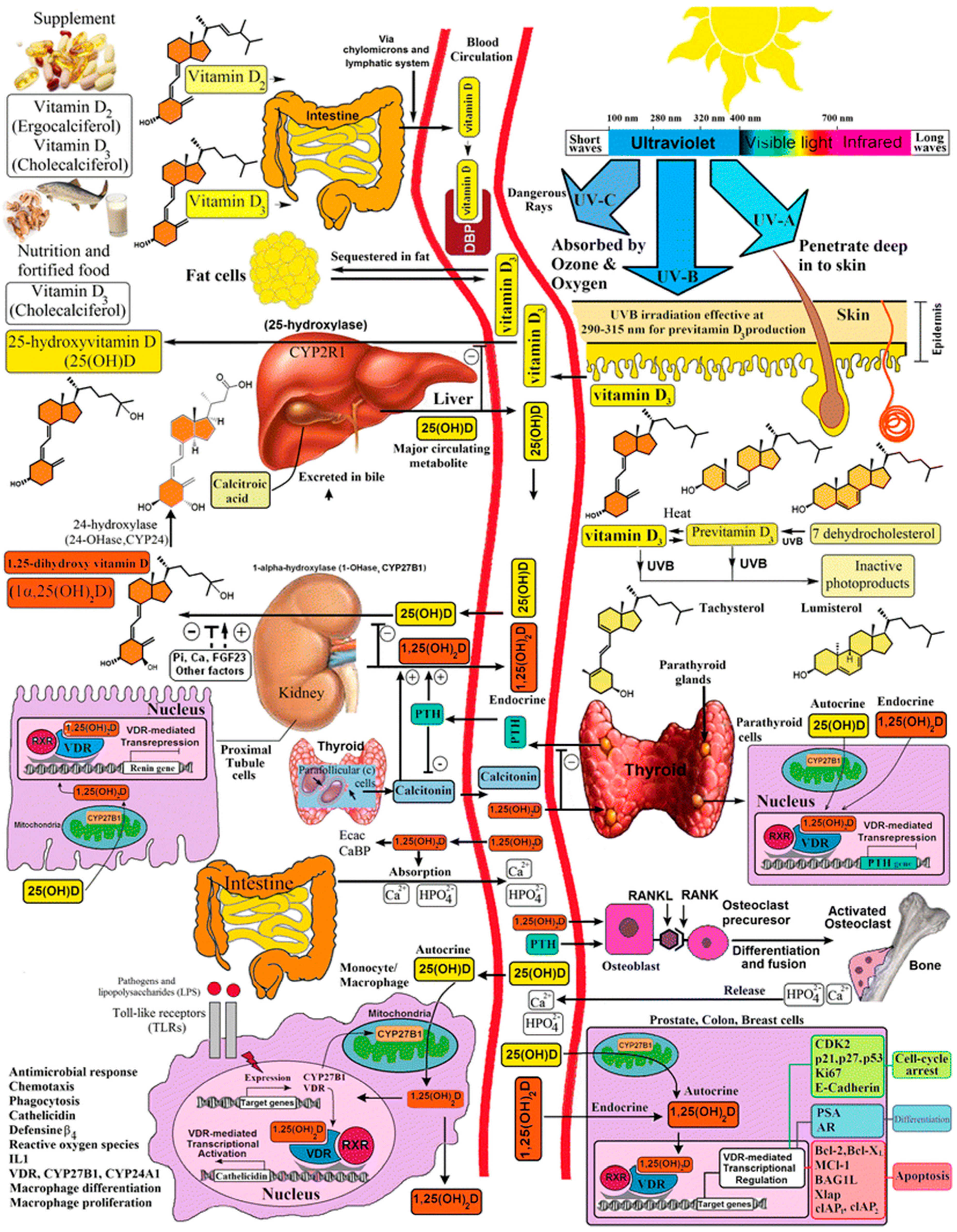
3. Overview of Vitamin D Metabolism during Pregnancy, Infancy and Childhood
4. Vitamin D Assays
5. Vitamin D Physiology during Pregnancy and in the Foetus and Infant
5.1. Pregnancy
5.2. Foetus
5.3. Newborn
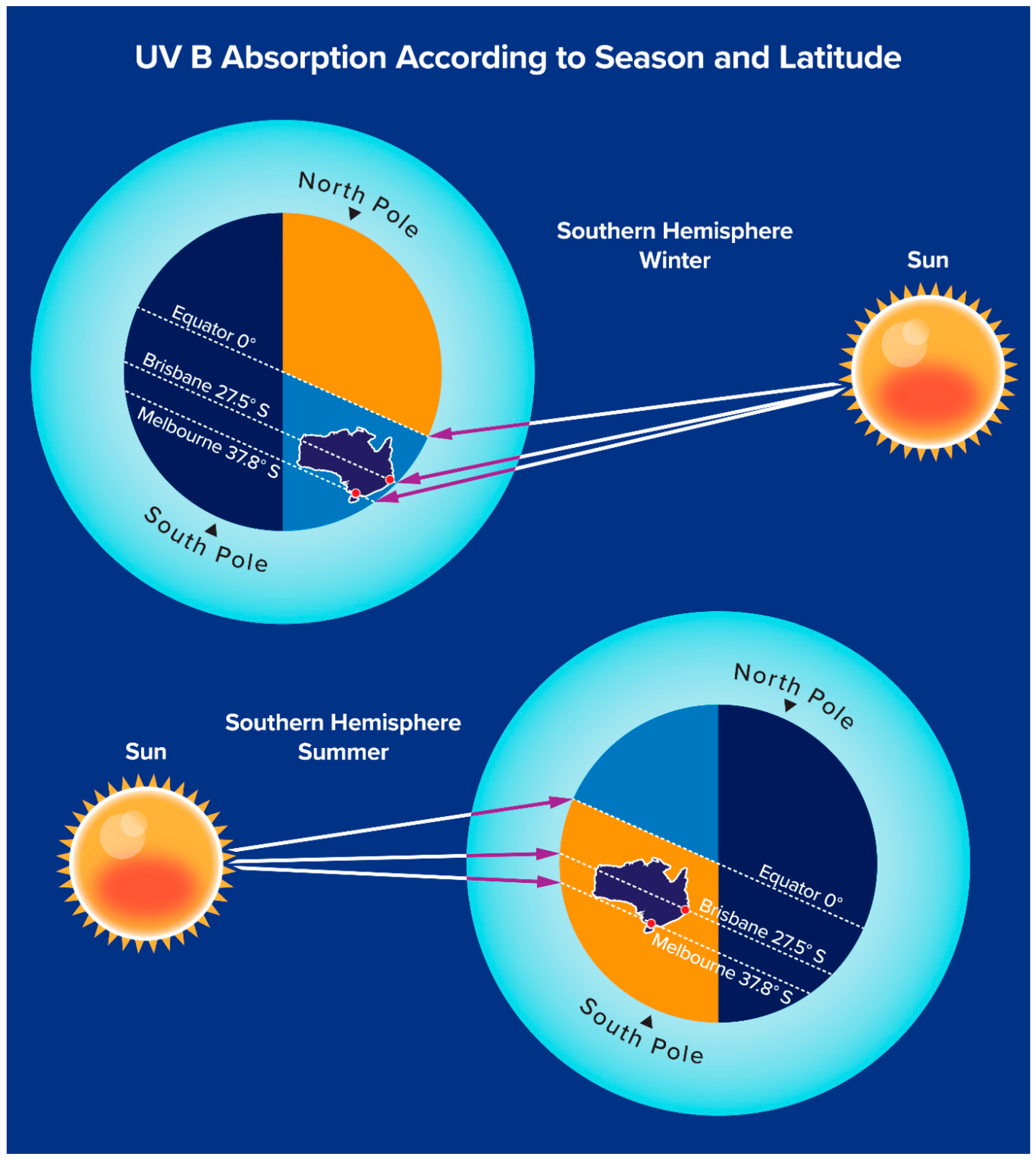
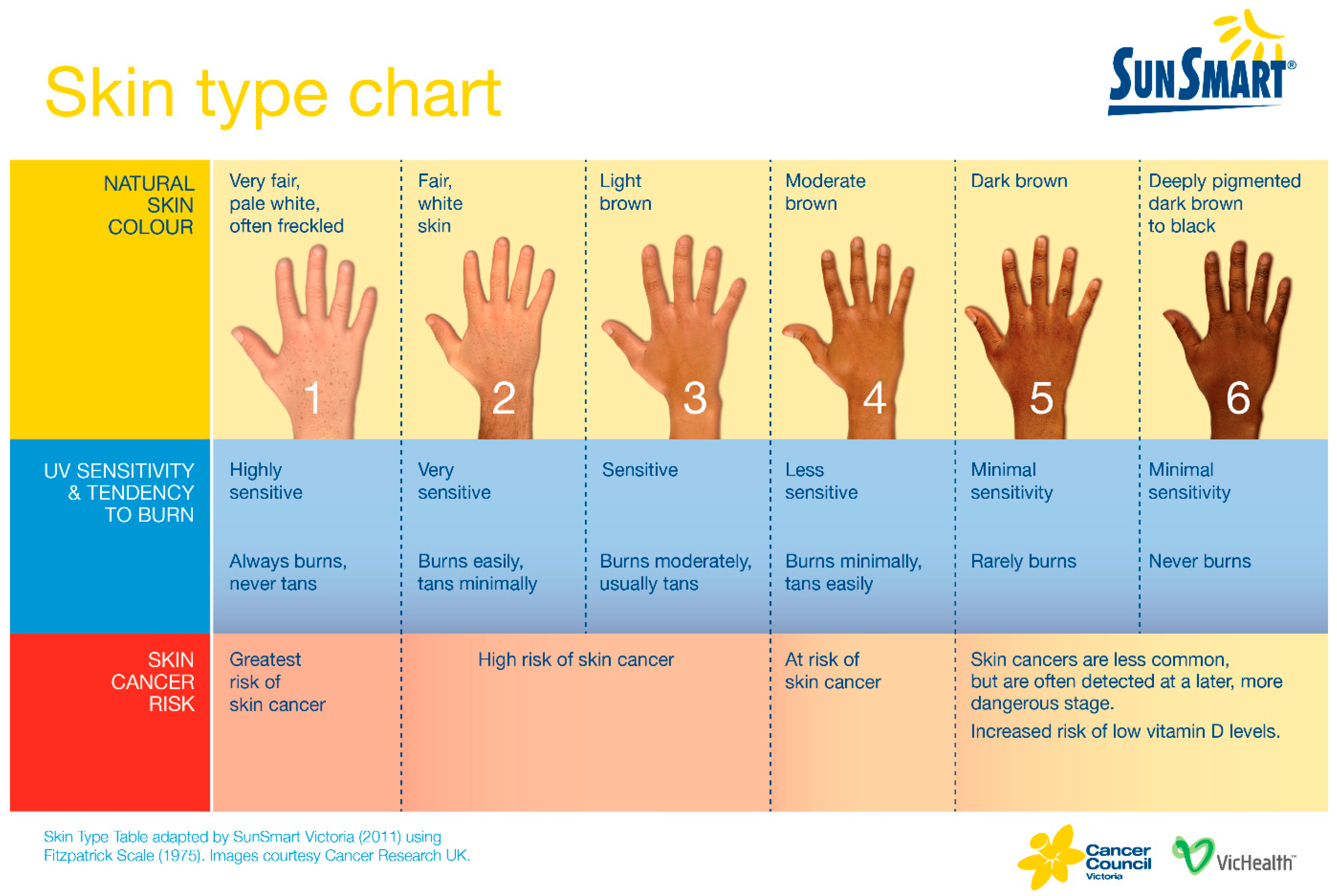
6. Public Health Aspects of Vitamin D Deficiency: Vitamin D Adequacy and “Sun Smart” Behaviours
7. Clinical Features of Rickets
7.1. Normal Bone Physiology
7.2. Rickets
8. Congenital (Genetic) Forms of Rickets
9. Maintaining Vitamin D Adequacy during Pregnancy and Breast Feeding
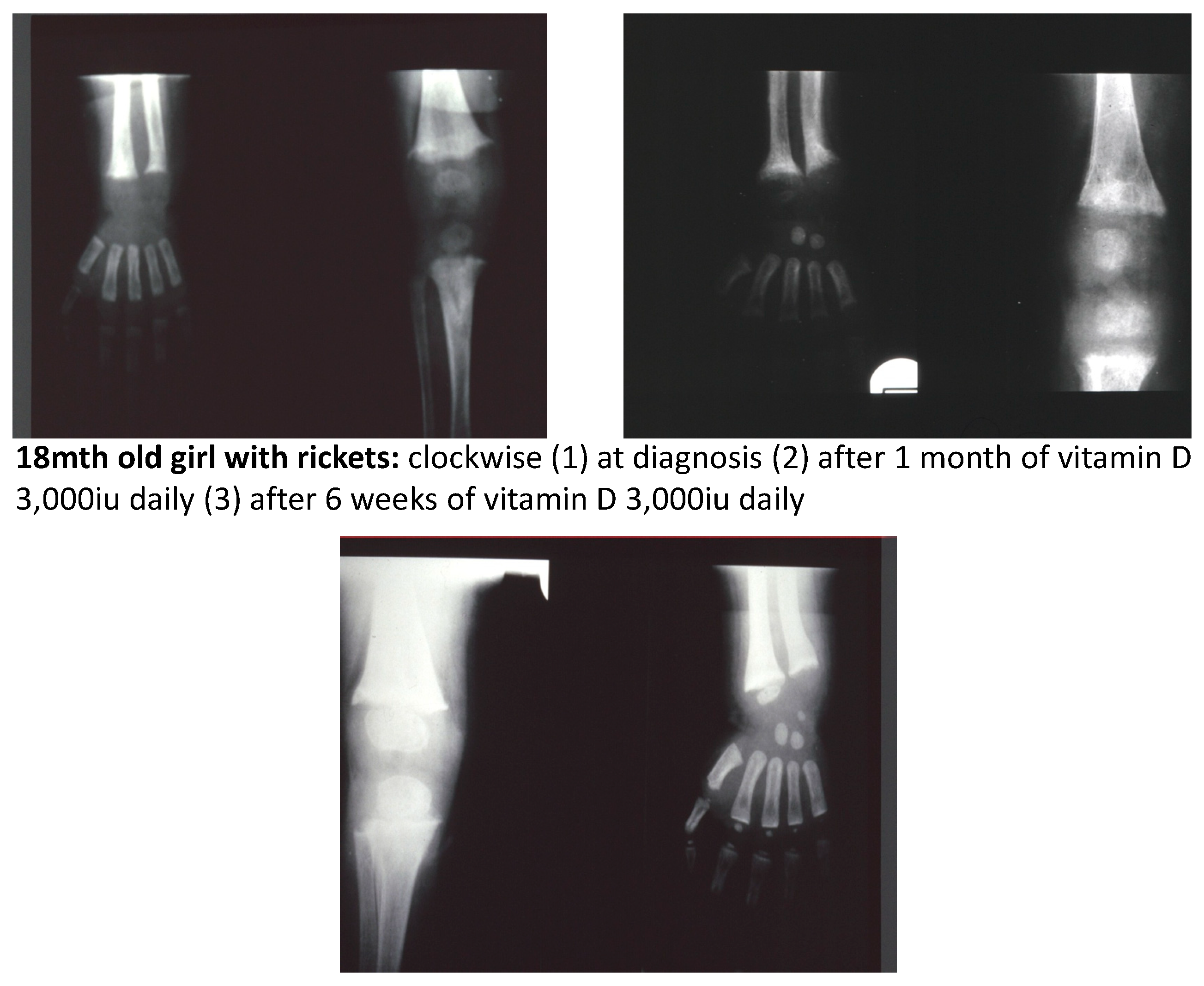
10. Treatment of Vitamin D Deficiency Rickets
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Clarke, E. Whistler and Glisson on Rickets. Bull. Hist. Med. 1962, 36, 45–61. [Google Scholar] [PubMed]
- Holick, M.F. Sunlight, Ultraviolet radiation, Vitamin D and skin cancer. How much sunlight do we need? Adv. Exp. Med. Biol. 2014, 810, 1–16. [Google Scholar] [PubMed]
- Rajakumar, K. Vitamin D, cod-liver oil, sunlight, and rickets: A historical perspective. Paediatrics 2003, 112, e132–e135. [Google Scholar] [CrossRef]
- Guy, R.A. The history of cod liver oil as a remedy. Am. J. Dis. Child. 1923, 26, 112–116. [Google Scholar] [CrossRef]
- Get Ready for FoodData Central, a New USDA Food and Nutrient Data System. Available online: https://ndb.nal.usda.gov/ (accessed on 12 February 2019).
- Nutrients. Available online: https://www.nrv.gov.au/nutrients (accessed on 12 February 2019).
- Aladag, N.; Filiz, T.M.; Topsever, P.; Gorpelioglu, S. Parents’ knowledge and behaviour concerning sunning their babies; a cross-sectional, descriptive study. BMC Pediatr. 2006, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.L.; Buettner, P.G.; MacLennan, R. Why do mothers still sun their infants? J. Paediatr. Child Health 1999, 35, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Saggese, G.; Vierucci, F.; Boot, A.M.; Czech-Kowalska, J.; Weber, G.; Camargo, C.A.; Mallet, E.; Fanos, M.; Shaw, N.J.; Holick, M.F. Vitamin D in childhood and adolescence: An expert position statement. Eur. J. Pediatr. 2015, 174, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Stanton, W.R.; Chakma, B.; O’Riordan, D.L.; Eyeson-Annan, M. Sun exposure and primary prevention of skin cancer for infants and young children during autumn/winter. Aust. N. Z. J. Public Health 2000, 24, 178–184. [Google Scholar] [CrossRef]
- Mayne, V.; McCredie, D. Rickets in Melbourne. Med. J. Aust. 1972, 2, 873–875. [Google Scholar]
- Fraser, W.D.; Milan, A.M. Vitamin D Assays: Past and present debates, difficulties and developments. Calcif. Tissue Int. 2013, 92, 118–127. [Google Scholar] [CrossRef]
- Campbell, K. Australian Dictionary of Biography; National Centre of Biography, (MUP) Australian National University: Canberra, Australia, 1988; Volume 11. [Google Scholar]
- Delvin, E.E.; Glorieux, F.H.; Salle, B.L. Control of vitamin D metabolism in preterm infants: Fetomaternal relationships. Arch. Dis. Child. 1982, 57, 754–757. [Google Scholar] [CrossRef] [PubMed]
- Salle, B.L.; Glorieux, F.H.; Bishop, N. Perinatal Vitamin D metabolism. Semin. Neonatol. 1998, 3, 143–147. [Google Scholar] [CrossRef]
- Pillow, J.J.; Forrest, P.J.; Rodda, C.P. Vitamin D deficiency in infants and young children born to migrant parents. J. Paediatr. Child Health 1995, 31, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Nozza, J.M.; Rodda, C.P. Vitamin D deficiency in mothers of infants with rickets. Med. J. Aust. 2001, 175, 253–255. [Google Scholar] [PubMed]
- Munns, C.F.; Simm, P.J.; Rodda, C.P.; Garnett, S.P.; Zacharin, M.R.; Ward, L.M.; Geddes, J.; Cherian, S.; Zurynski, Y.; Cowell, C.T. Incidence of vitamin D deficiency rickets among Australian children: An Australian Paediatric Surveillance Unit Study. Med. J. Aust. 2012, 196, 466–468. [Google Scholar]
- Grover, S.R.; Morley, R. Vitamin D deficiency in veiled or dark-skinned pregnant women. Med. J. Aust. 2001, 175, 251–252. [Google Scholar]
- Vitamin D and UV Radiation. Available online: http://www.sunsmart.com.au/downloads/resources/info-sheets/vitamin-d-uv-radiation-info-sheet.pdf (accessed on 12 February 2019).
- Holick, M.F.; Hossein-Nezhad, A. The D-lemma: Narrow band UV type B radiation vs vitamin D supplementation vs sunlight for cardiovascular and immune health. Am. J. Clin. Nutr. 2017, 105, 1031–1032. [Google Scholar] [CrossRef]
- Bikle, D.; Adams, J.S.; Christakos, S. Vitamin D: Production, metabolism, mechanism of action and clinical requirements. In Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism, 8th ed.; Rosen, C.J., Ed.; Wiley-Blackwell: Singapore, 2013; Chapter 29; pp. 235–247. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Mason, R.S.; Reichrath, J. Sunlight, vitamin D and skin cancer. Anticancer Agents Med. Chem. 2013, 13, 83–97. [Google Scholar] [CrossRef]
- Van Der Pols, J.C.; Russell, A.; Bauer, U.; Neale, R.E.; Kimlin, M.G.; Green, A.C. Vitamin D status and skin cancer risk independent of time outdoors: 11 year prospective study in an Australian Community. J. Investig. Dermatol. 2013, 133, 637–641. [Google Scholar] [CrossRef]
- Holick, M.F.; MacLaughlin, J.A.; Clark, M.B. Photosynthesis of preVitamin D3 in human skin and the physiological consequences. Science 1980, 210, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.T.; Moore, C.G.; Hall, C.; Olabopo, F.; Rozario, N.L.; Holick, M.F.; Greenspan, S.L.; Rajakumar, K. Utility of sun-reactive skin typing and melanin index for discerning Vitamin D deficiency. Paediatr. Res. 2017, 82, 444–451. [Google Scholar] [CrossRef]
- Kovacs, C.S. Bone Development and mineral homeostasis in the fetus and neonate: Roles of calciotropic and phosphotropic hormones. Physiol. Rev. 2014, 94, 1143–1218. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, C.S. Maternal mineral and bone metabolism during pregnancy, lactation and post-weaning recovery. Physiol. Rev. 2016, 96, 449–547. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D Supplementation During Pregnancy: Double-Blind, Randomized Clinical Trial of Safety and Effectiveness. J. Bone Miner. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef] [PubMed]
- Rodda, C.P.; Kubota, M.; Heath, J.A.; Ebeling, P.R.; Moseley, J.M.; Care, A.D.; Caple, I.W.; Martin, T.J. Evidence for a novel parathyroid hormone-related protein in fetal lamb parathyroid glands and sheep placenta: Comparisons with a similar protein implicated in humoral hypercalcaemia of malignancy. J. Endocrinol. 1988, 117, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and Human Health: Lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, R.; Abrahamsen, B.; Bauerek, M.; Holst, C.; Jensen, C.B.; Knop, J.; Raymond, K.; Rasmussen, L.B.; Stougaard, M.; Sørensen, T.I.; et al. The influence of early exposure to Vitamin D for development of diseases later in life. BMC Public Health 2013, 13, 515. [Google Scholar] [CrossRef] [PubMed]
- Hart, P.H.; Lucas, R.M.; Walsh, J.P.; Zosky, G.R.; Whitehouse, A.J.; Zhu, K.; Allen, K.L.; Kusel, M.M.; Anderson, D.; Mountain, J.A. Vitamin D in fetal development: Findings from a birth cohort study. Pediatrics 2015, 135, e167–e173. [Google Scholar] [CrossRef]
- Wei, S.-Q.; Qi, H.-P.; Luo, Z.-C.; Fraser, W.D. Maternal vitamin D status and adverse pregnancy outcomes: A systematic review and meta-analysis. J. Matern.-Fetal Med. 2013, 26, 889–899. [Google Scholar] [CrossRef]
- Holland, P.C.; Wilkinson, A.R.; Diez, J.; Lindsell, D.R.M. Prenatal deficiency of phosphate, phosphate supplementation, and rickets in very-low-birthweight infants. Lancet 1990, 335, 697–701. [Google Scholar] [CrossRef]
- Takeuchi, A.; Okano, T.; Tsugawa, N.; Tasaka, Y.; Kobayashi, T.; Kodama, S.; Matsuo, T. Effects of ergocalciferol supplementation on the concentration of vitamin D and its metabolites in human milk. J. Nutr. 1989, 119, 1639–1646. [Google Scholar] [CrossRef] [PubMed]
- Saadi, H.F.; Dawodu, A.; Afandi, B.O.; Zayed, R.; Benedict, S.; Nagelkerke, N. Efficacy of daily and monthly high-dose calciferol in vitamin D-deficient nulliparous and lactating women. Am. J. Clin. Nutr. 2007, 85, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L.; Howard, C.R.; Ebeling, M.; Shary, J.R.; Smith, P.G.; Taylor, S.N.; Morella, K.; Lawrence, R.A.; Hulsey, T.C. Maternal versus infant Vitamin D supplementation during lactation: A randomised controlled trial. Paediatrics 2015, 136, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, B.J.; Taylor, B.J.; Herbison, P.; Haszard, J.J.; Mikhail, A.; Jones, S.; Harper, M.J.; Houghton, L.A. High dose monthly maternal cholecalciferol supplementation during breastfeeding affects maternal and infant vitamin D status at 5 months postpartum: A randomised controlled trial. J. Nutr. 2016, 146, 1999–2006. [Google Scholar] [CrossRef] [PubMed]
- Skin Cancer. Available online: http://www.sunsmart.com.au/skin-cancer (accessed on 12 February 2019).
- Kockott, D.; Herzog, B.; Reichrath, J.; Keane, K.; Holick, M.F. New approach to develop optimised sunscreens that enable cutaneous vitamin D formation with minimal erythematous risk. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Glorieux, F.H. Rickets, the continuing challenge. N. Engl. J. Med. 1991, 325, 1875–1877. [Google Scholar] [CrossRef] [PubMed]
- Rauch, F. The dynamics of bone structure development during pubertal growth. J. Musculoskelet. Neuronal Interact. 2012, 12, 1–6. [Google Scholar] [PubMed]
- Paxton, G.A.; Teale, G.R.; Nowson, C.A.; Mason, R.S.; McGrath, J.J.; Thompson, M.J.; Siafarikas, A.; Rodda, C.P.; Munns, C.F. Vitamin D and health in pregnancy, infants, children and adolescents in Australia and New Zealand: A position statement. Med. J. Aust. 2013, 198, 142–143. [Google Scholar] [CrossRef] [PubMed]
- Pai, B.; Shaw, N. Understanding rickets. Paediatr. Child Health 2011, 21, 315–321. [Google Scholar] [CrossRef]
- Munns, C.F.; Shaw, N.; Kiely, M.; Specker, B.L.; Thacher, T.D.; Ozono, K.; Michigami, T.; Tiosano, D.; Mughal, M.Z.; Mäkitie, O.; et al. Global Consensus Recommendations on Prevention and Management of Nutritional Rickets. J. Clin. Endocrinol. Metab. 2016, 101, 394–415. [Google Scholar] [CrossRef] [PubMed]
- Thacher, T.D.; Fischer, P.R.; Pettifor, J.M.; Lawson, J.O.; Isichei, C.O.; Reading, J.C.; Chan, G.M. A comparison of calcium, vitamin D or both for nutritional rickets in Nigerian children. N. Engl. J. Med. 1999, 341, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Elder, C.J.; Bishop, N.J. Rickets. Lancet 2014, 383, 1665–1676. [Google Scholar] [CrossRef]
- Whyte, M.P.; Thakker, R.V. Rickets and Osteomalacia. Medicine 2013, 41, 594–599. [Google Scholar] [CrossRef]
- Thacher, T.D.; Levine, M.A. CYP2R1 mutations causing vitamin D-deficiency rickets. J. Steroid Biochem. Mol. Biol. 2017, 173, 333–336. [Google Scholar] [CrossRef]
- Roizen, J.D.; Li, D.; O’Lear, L.; Javaid, M.K.; Shaw, N.J.; Ebeling, P.R.; Nguyen, H.H.; Rodda, C.P.; Thummel, K.E.; Thacher, T.D.; et al. CYP3A4 mutation causes Vitamin D-dependent rickets type 3. J. Clin. Investig. 2018, 128, 1913–1918. [Google Scholar] [CrossRef]
- Huynh, J.; Lu, T.; Liew, D.; Doery, J.C.G.; Tudball, R.; Jona, M.; Bhamjee, R.; Rodda, C.P. Vitamin D in newborns: A random controlled trial comparing daily and single oral bolus Vitamin D in infants. Paediatr. Child Health 2017, 53, 163–169. [Google Scholar] [CrossRef]
- McBride, L.; Crosthwaite, A.; Houlihan, C.; Rodda, C. Instructive case: A rare cause of maternal and neonatal hypercalcaemia. J. Paediatr. Child Health 2019, 55, 232–235. [Google Scholar] [CrossRef]
- Heaney, R.P.; Recker, R.R.; Grote, J.; Horst, R.L.; Armas, L.A. Vitamin D(3) is more potent than vitamin D(2) in humans. J. Clin. Endocrinol. Metab. 2011, 96, E447–E452. [Google Scholar] [CrossRef]
- Nordblad, M.I.; Graham, F.; Mughal, M.Z.; Padidela, R. Rapid assessment of dietary calcium assessment. Arch. Dis. Child 2016, 101, 634–636. [Google Scholar] [CrossRef]
- Gernand, A.D.; Simhan, H.N.; Baca, K.M.; Caritis, S.; Bodnar, L.M. Vitamin D, pre-eclampsia, and preterm birth among pregnancies at high risk for pre-eclampsia: An analysis of data from a low-dose aspirin trial. BJOG 2017, 124, 1874–1882. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, J.; Wan, Y.; Xia, X.; Pan, J.; Gu, W.; Li, M. Serum vitamin D deficiency in children and adolescents is associated with type 1 diabetes mellitus. Endocr. Connect. 2018. [Google Scholar] [CrossRef] [PubMed]
- Baggerly, C.A.; Cuomo, R.E.; French, C.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Heaney, R.P.; Holick, M.F.; Hollis, B.W.; McDonnell, S.L.; et al. Sunlight and Vitamin D: Necessary for Public Health. J. Am. Coll. Nutr. 2015, 34, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; McCloskey, E.V.; Johansson, H.; Strom, O.; Borgstrom, F.; Oden, A.; Compston, J.E.; Cooper, A.L.; Cooper, C.; Francis, R.; et al. Case finding for the management of osteoporosis with FRAX—Assessment and intervention thresholds for the UK. Osteoporos. Int. 2008, 19, 1395–1408. [Google Scholar] [CrossRef] [PubMed]
- Australian Type 2 Diabetes Risk Assessment Tool (AUSDRISK). Available online: http://www.health.gov.au/preventionoftype2diabetes (accessed on 12 February 2019).
- Annweiler, C.; Kabeshova, A.; Callens, A.; Paty, M.L.; Duval, G.T.; Holick, M.F. Self administered Vitamin D status predictor: Older adults are able to use a self questionnaire for evaluation of their vitamin D status. PLoS ONE 2017, 12, e0186578. [Google Scholar] [CrossRef]
| Source | Vitamin D Content |
|---|---|
| Natural sources | |
| Fish | |
| Salmon (canned: 100 g) | Approximately 30–600 iu (7.5–15 µg) vit D3 |
| Sardines (canned: 100 g) | Approximately 300 iu (7.5 µg) vit D3 |
| Mackeral (canned: 100 g) | Approximately 250 iu (6.25 µg) vit D3 |
| Tuna (canned: 100 g) | Approximately 230 iu (5.75 µg) vit D3 |
| Cod liver oil (1 tsp/5 mL) | Approximately 400–1000 iu (10–25 µg) vit D3 |
| Shitake mushrooms | |
| Fresh (100 g) | Approximately 100 iu (2.5 µg) vit D2 |
| Sundried (100 g) | Approximately 1600 iu (40 µg) vit D2 |
| Egg yoke | Approximately 20 iu (0.5 µg) vit D2 or D3 |
| UV B sun exposure (0.5 minimal erythematous dose: equivalent to approximately 5–10 min or more of direct sun exposure to arms and legs depending on skin colour [Figure 3], latitude, season and time of day) | Approximately 3000 iu (75 µg) vit D3 |
| Infant feeding formulas | 400iu (10µg)/L vitamin D3 |
| Supplements | |
| For example: Ostelin © | 1000iu (25µg) vitamin D3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Marco, N.; Kaufman, J.; Rodda, C.P. Shedding Light on Vitamin D Status and Its Complexities during Pregnancy, Infancy and Childhood: An Australian Perspective. Int. J. Environ. Res. Public Health 2019, 16, 538. https://doi.org/10.3390/ijerph16040538
Di Marco N, Kaufman J, Rodda CP. Shedding Light on Vitamin D Status and Its Complexities during Pregnancy, Infancy and Childhood: An Australian Perspective. International Journal of Environmental Research and Public Health. 2019; 16(4):538. https://doi.org/10.3390/ijerph16040538
Chicago/Turabian StyleDi Marco, Nelfio, Jonathan Kaufman, and Christine P. Rodda. 2019. "Shedding Light on Vitamin D Status and Its Complexities during Pregnancy, Infancy and Childhood: An Australian Perspective" International Journal of Environmental Research and Public Health 16, no. 4: 538. https://doi.org/10.3390/ijerph16040538
APA StyleDi Marco, N., Kaufman, J., & Rodda, C. P. (2019). Shedding Light on Vitamin D Status and Its Complexities during Pregnancy, Infancy and Childhood: An Australian Perspective. International Journal of Environmental Research and Public Health, 16(4), 538. https://doi.org/10.3390/ijerph16040538





