Vitamin D Status and Mortality: A Systematic Review of Observational Studies
Abstract
1. Introduction
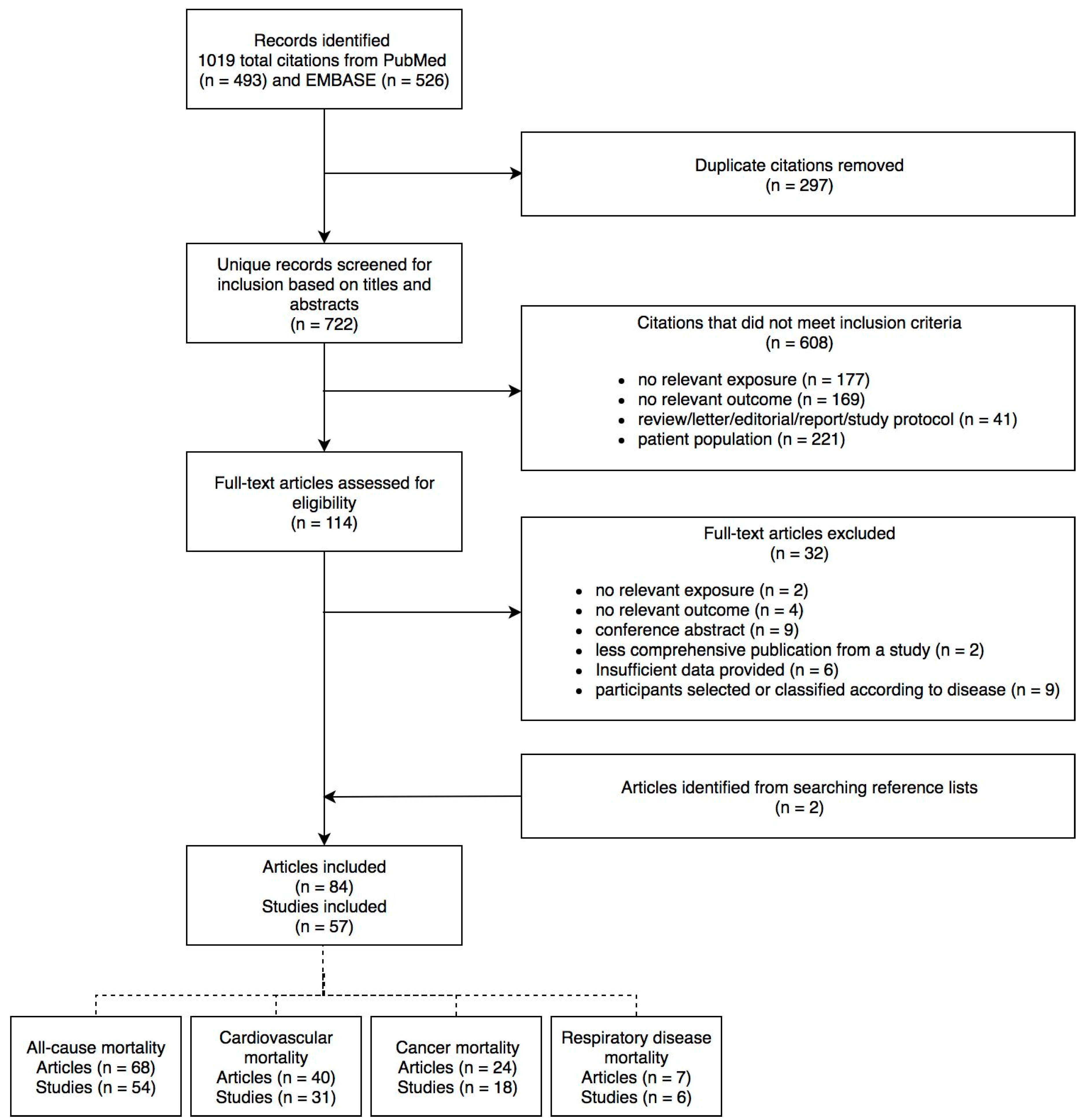
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
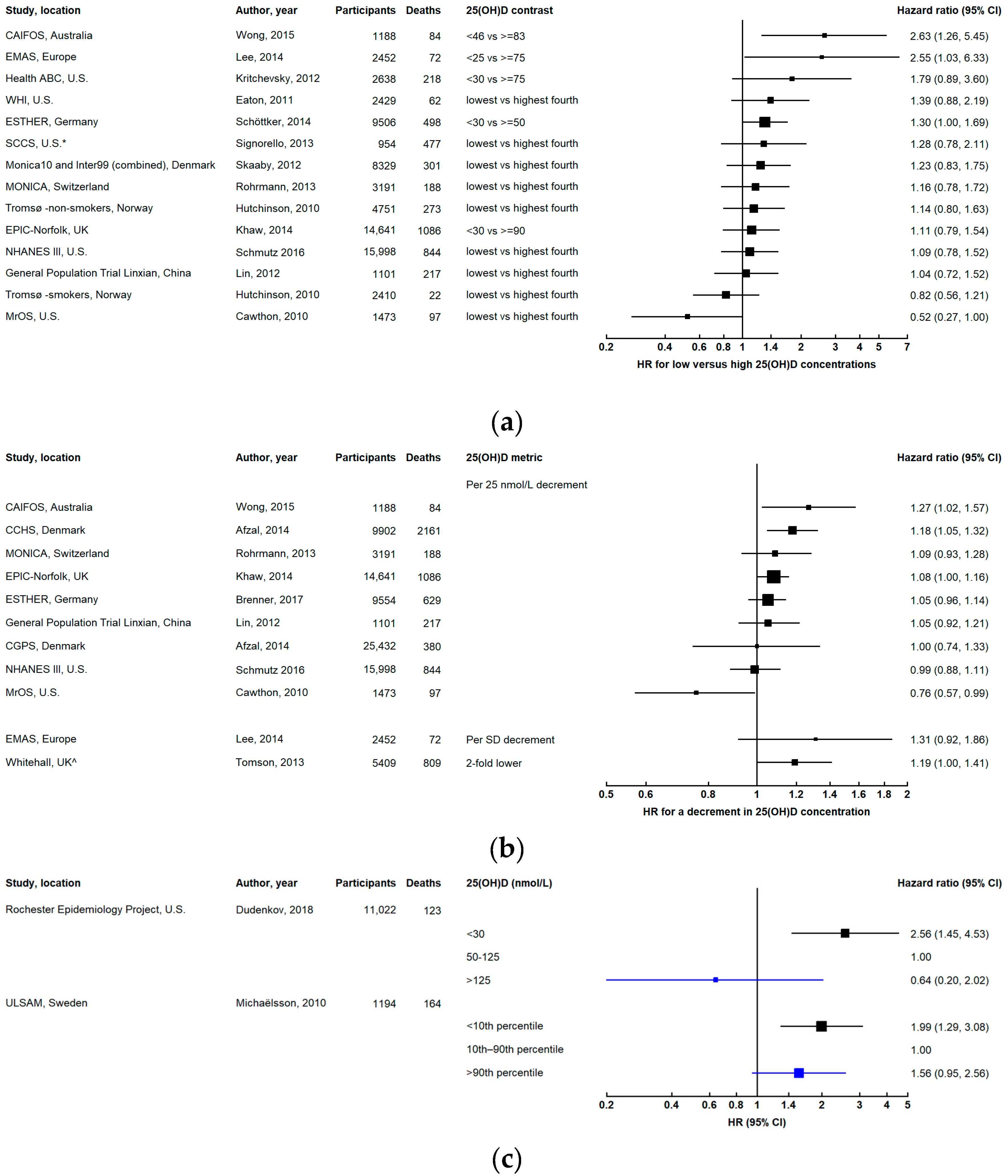
3. Results
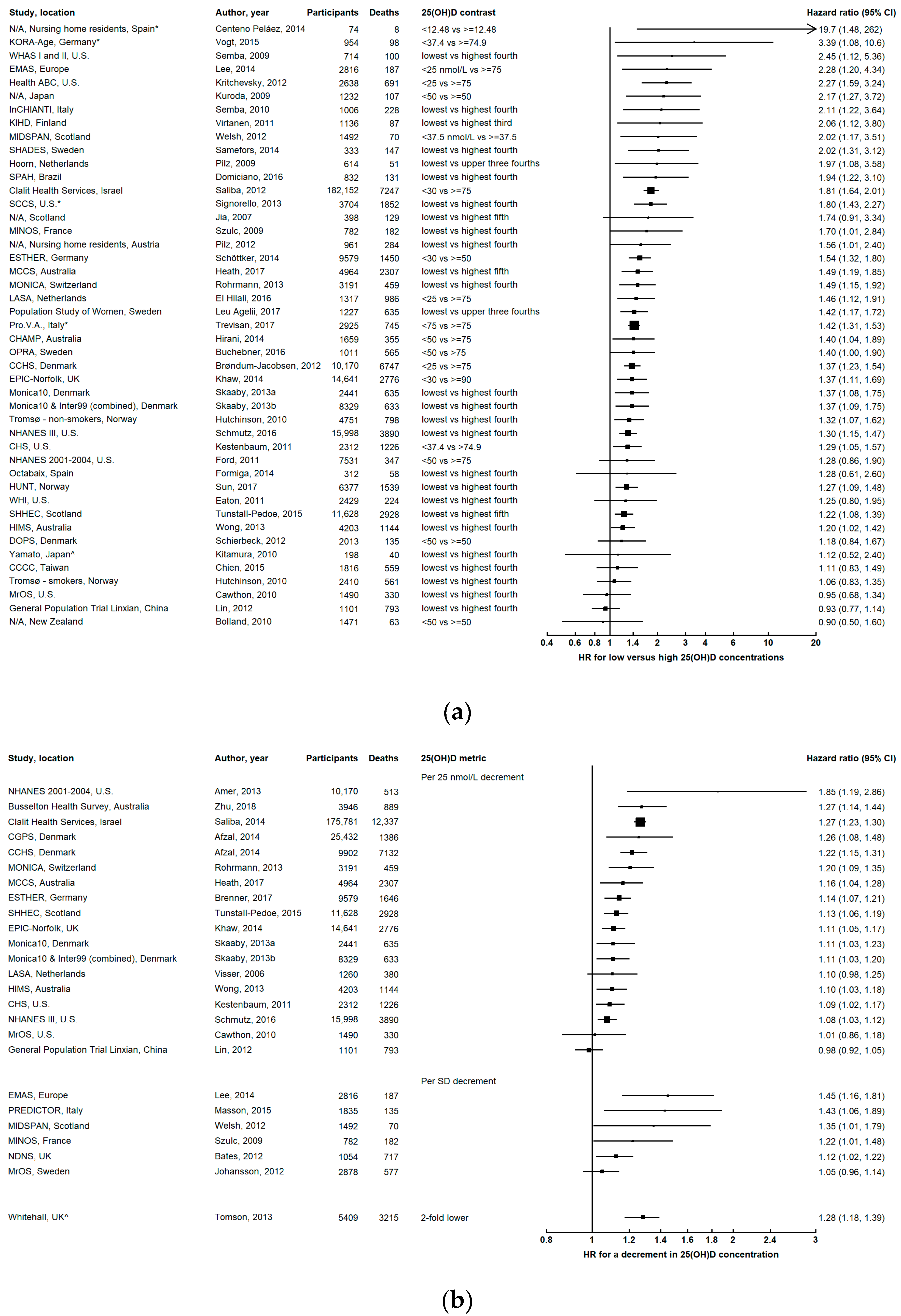
3.1. All-Cause Mortality
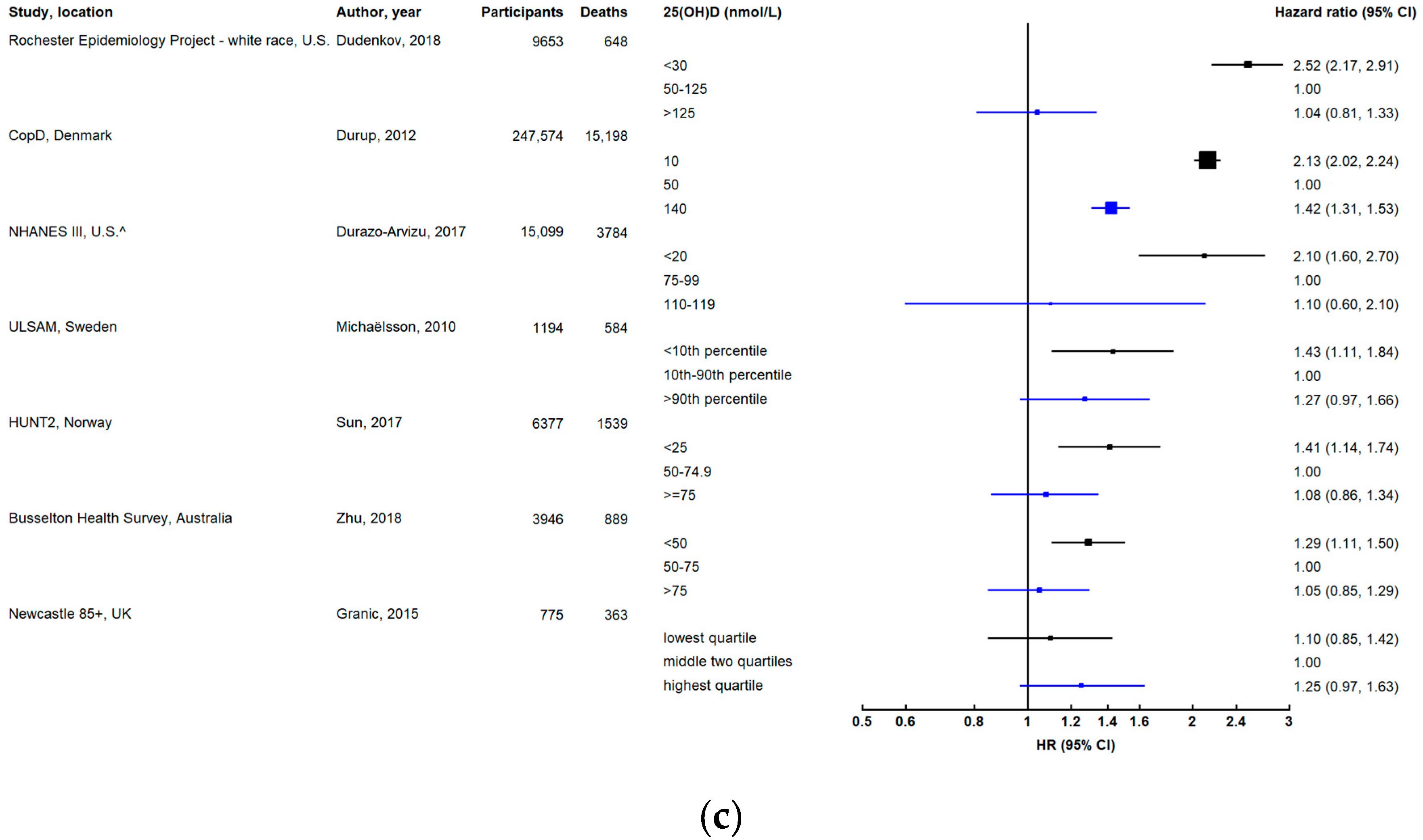
3.1.1. Dose-Response Relationship
3.1.2. Effect of Follow-Up Time and Repeated Measurements of 25(OH)D
3.1.3. Evidence from Other Studies
3.1.4. Vitamin D2 and Vitamin D3
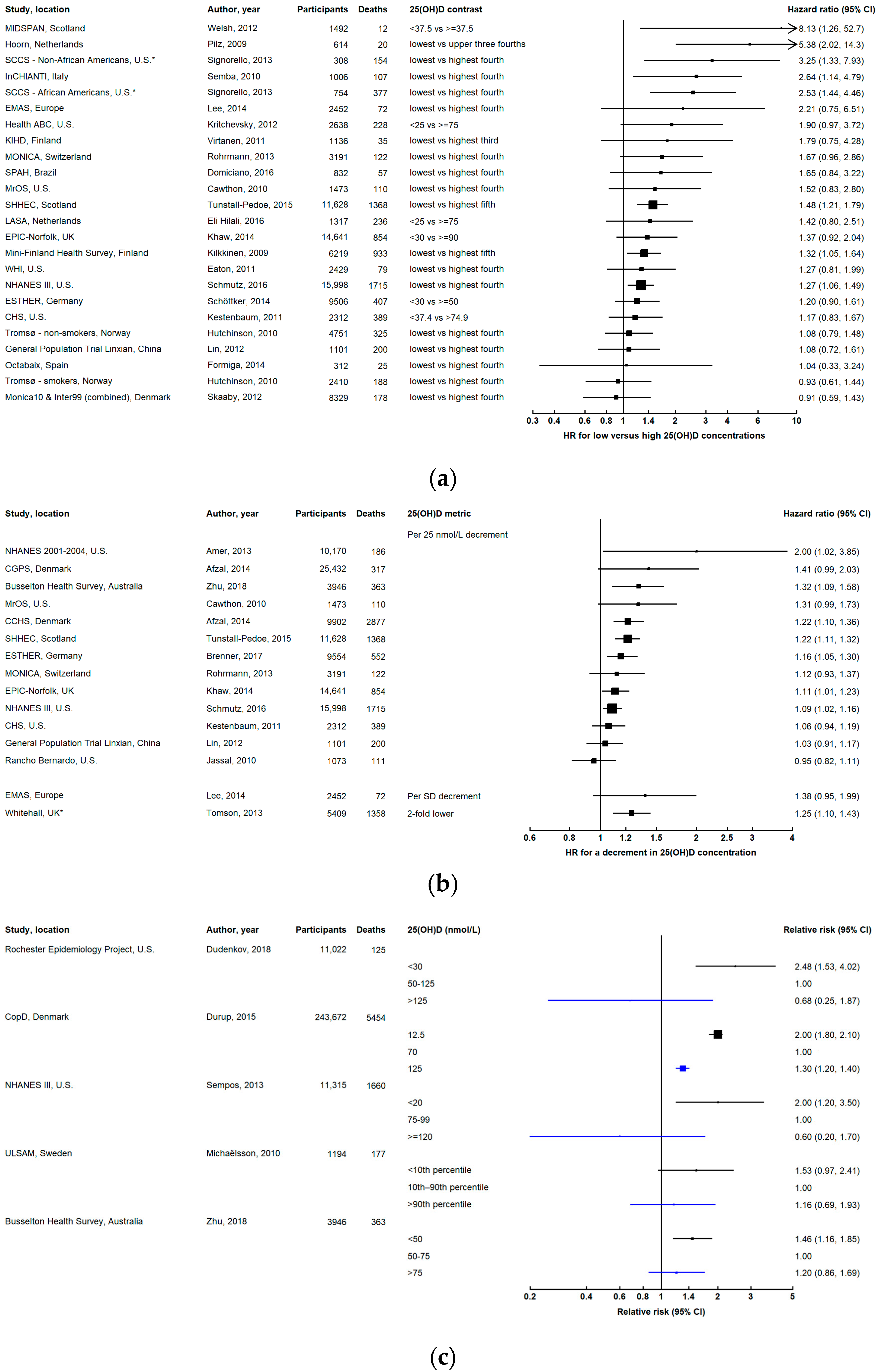
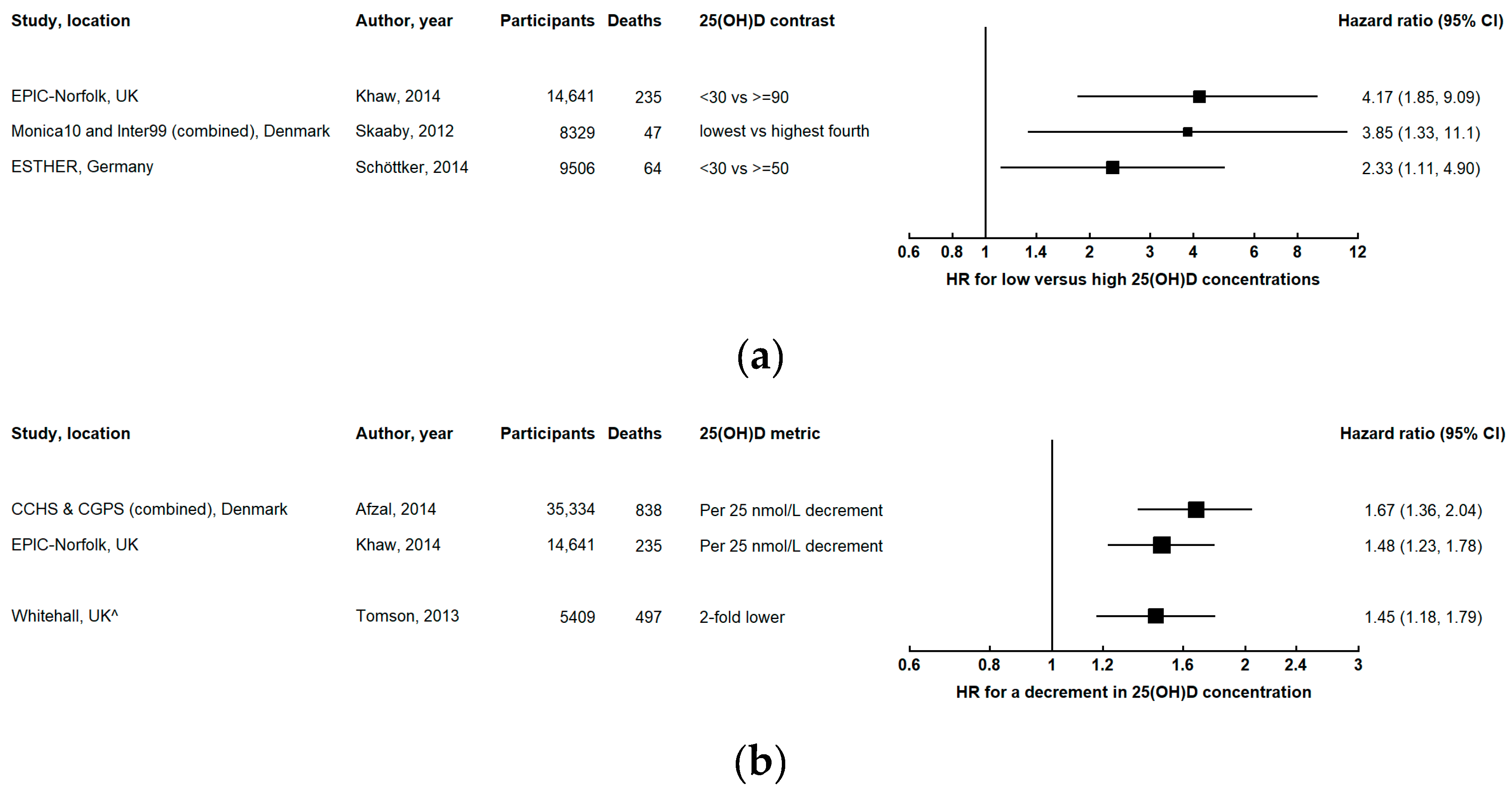
3.2. Cardiovascular Disease Mortality
3.2.1. Subtypes of Cardiovascular Mortality
3.2.2. Evidence from Other Studies
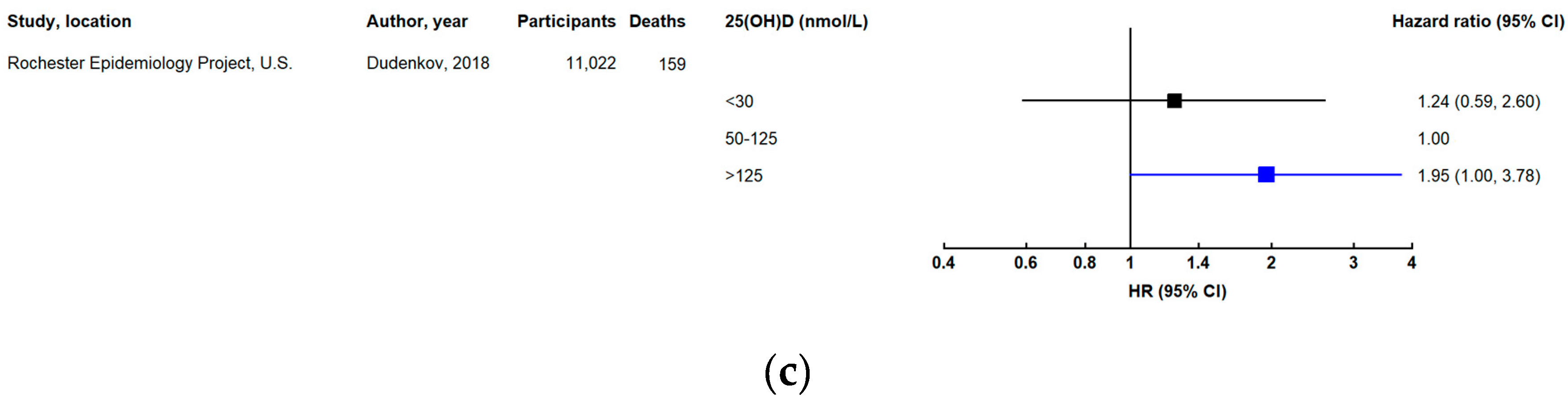
3.3. Cancer Mortality
3.3.1. Site-Specific Cancer Mortality
3.3.2. Evidence from Other Studies
3.4. Respiratory Disease Mortality
3.5. Other Causes of Death
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chowdhury, R.; Kunutsor, S.; Vitezova, A.; Oliver-Williams, C.; Chowdhury, S.; Kiefte-de-Jong, J.C.; Khan, H.; Baena, C.P.; Prabhakaran, D.; Hoshen, M.B.; et al. Vitamin D and risk of cause specific death: Systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ 2014, 348, g1903. [Google Scholar] [CrossRef] [PubMed]
- Garland, C.F.; Kim, J.J.; Mohr, S.B.; Gorham, E.D.; Grant, W.B.; Giovannucci, E.L.; Baggerly, L.; Hofflich, H.; Ramsdell, J.W.; Zeng, K.; et al. Meta-analysis of all-cause mortality according to serum 25-hydroxyvitamin D. Am. J. Public Health 2014, 104, e43–e50. [Google Scholar] [CrossRef] [PubMed]
- Gaksch, M.; Jorde, R.; Grimnes, G.; Joakimsen, R.; Schirmer, H.; Wilsgaard, T.; Mathiesen, E.B.; Njolstad, I.; Lochen, M.L.; Marz, W.; et al. Vitamin D and mortality: Individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS ONE 2017, 12, e0170791. [Google Scholar] [CrossRef] [PubMed]
- Schöttker, B.; Jorde, R.; Peasey, A.; Thorand, B.; Jansen, E.H.; Groot, L.; Streppel, M.; Gardiner, J.; Ordóñez-Mena, J.M.; Perna, L.; et al. Vitamin D and mortality: Meta-analysis of individual participant data from a large consortium of cohort studies from Europe and the United States. BMJ 2014, 348, g3656. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Schöttker, B.; Saum, K.U.; Perna, L.; Ordóñez-Mena, J.M.; Holleczek, B.; Brenner, H. Is vitamin D deficiency a cause of increased morbidity and mortality at older age or simply an indicator of poor health? Eur. J. Epidemiol. 2014, 29, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M.; Burke, F.; Evans, K.N.; Lammas, D.A.; Sansom, D.M.; Liu, P.; Modlin, R.L.; Adams, J.S. Extra-renal 25-hydroxyvitamin D3-1alpha-hydroxylase in human health and disease. J. Steroid Biochem. Mol. Biol. 2007, 103, 316–321. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Hossein-nezhad, A.; Holick, M.F. Vitamin D for health: A global perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef] [PubMed]
- Brøndum-Jacobsen, P.; Benn, M.; Jensen, G.B.; Nordestgaard, B.G. 25-hydroxyvitamin d levels and risk of ischemic heart disease, myocardial infarction, and early death: Population-based study and meta-analyses of 18 and 17 studies. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2794–2802. [Google Scholar] [CrossRef]
- Tomson, J.; Emberson, J.; Hill, M.; Gordon, A.; Armitage, J.; Shipley, M.; Collins, R.; Clarke, R. Vitamin D and risk of death from vascular and non-vascular causes in the Whitehall study and meta-analyses of 12,000 deaths. Eur. Heart J. 2013, 34, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Iodice, S.; Pilz, S.; Grant, W.B.; Bagnardi, V.; Gandini, S. Vitamin D deficiency and mortality risk in the general population: A meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2012, 95, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Song, Y.; Manson, J.E.; Pilz, S.; Marz, W.; Michaëlsson, K.; Lundqvist, A.; Jassal, S.K.; Barrett-Connor, E.; Zhang, C.; et al. Circulating 25-hydroxy-vitamin D and risk of cardiovascular disease: A meta-analysis of prospective studies. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, B.; Gao, X.; Tian, R.; Pan, Y.; Jiang, Y.; Gu, H.; Wang, Y.; Wang, Y.; Liu, G. Serum 25-hydroxyvitamin D and the risk of cardiovascular disease: Dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2017, 105, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Binkley, N.; Dawson-Hughes, B.; Durazo-Arvizu, R.; Thamm, M.; Tian, L.; Merkel, J.M.; Jones, J.C.; Carter, G.D.; Sempos, C.T. Vitamin D measurement standardization: The way out of the chaos. J. Steroid Biochem. Mol. Biol. 2017, 173, 117–121. [Google Scholar] [CrossRef]
- Carter, G.D. Accuracy of 25-hydroxyvitamin D assays: Confronting the issues. Curr. Drug Targets 2011, 12, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Durup, D.; Jorgensen, H.L.; Christensen, J.; Schwarz, P.; Heegaard, A.M.; Lind, B. A reverse J-shaped association of all-cause mortality with serum 25-hydroxyvitamin D in general practice: The CopD study. J. Clin. Endocrinol. Metab. 2012, 97, 2644–2652. [Google Scholar] [CrossRef] [PubMed]
- Durup, D.; Jorgensen, H.L.; Christensen, J.; Tjonneland, A.; Olsen, A.; Halkjaer, J.; Lind, B.; Heegaard, A.M.; Schwarz, P. A reverse J-shaped association between serum 25-hydroxyvitamin D and cardiovascular disease mortality: The CopD study. J. Clin. Endocrinol. Metab. 2015, 100, 2339–2346. [Google Scholar] [CrossRef]
- Granic, A.; Aspray, T.; Hill, T.; Davies, K.; Collerton, J.; Martin-Ruiz, C.; von Zglinicki, T.; Kirkwood, T.B.; Mathers, J.C.; Jagger, C. 25-hydroxyvitamin D and increased all-cause mortality in very old women: The Newcastle 85+ study. J. Intern. Med. 2015, 277, 456–467. [Google Scholar] [CrossRef]
- Michaëlsson, K.; Baron, J.A.; Snellman, G.; Gedeborg, R.; Byberg, L.; Sundstrom, J.; Berglund, L.; Arnlov, J.; Hellman, P.; Blomhoff, R.; et al. Plasma vitamin D and mortality in older men: A community-based prospective cohort study. Am. J. Clin. Nutr. 2010, 92, 841–848. [Google Scholar] [CrossRef]
- Sempos, C.T.; Durazo-Arvizu, R.A.; Dawson-Hughes, B.; Yetley, E.A.; Looker, A.C.; Schleicher, R.L.; Cao, G.; Burt, V.; Kramer, H.; Bailey, R.L.; et al. Is there a reverse J-shaped association between 25-hydroxyvitamin D and all-cause mortality? Results from the U.S. nationally representative NHANES. J. Clin. Endocrinol. Metab. 2013, 98, 3001–3009. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Dobnig, H.; Tomaschitz, A.; Kienreich, K.; Meinitzer, A.; Friedl, C.; Wagner, D.; Piswanger-Solkner, C.; Marz, W.; Fahrleitner-Pammer, A. Low 25-hydroxyvitamin D is associated with increased mortality in female nursing home residents. J. Clin. Endocrinol. Metab. 2012, 97, E653–E657. [Google Scholar] [CrossRef] [PubMed]
- Centeno Peláez, V.; Ausín, L.; Ruiz Mambrilla, M.; Gonzalez-Sagrado, M.; Pérez Castrillón, J.L. Severe vitamin D deficiency, functional impairment and mortality in elderly nursing home residents. J. Aging Res. Clin. Pract. 2014, 3, 218–222. [Google Scholar] [CrossRef]
- Samefors, M.; Ostgren, C.J.; Molstad, S.; Lannering, C.; Midlov, P.; Tengblad, A. Vitamin D deficiency in elderly people in Swedish nursing homes is associated with increased mortality. Eur. J. Endocrinol. 2014, 170, 667–675. [Google Scholar] [CrossRef]
- Formiga, F.; Ferrer, A.; Megido, M.J.; Boix, L.; Contra, A.; Pujol, R. Low serum vitamin D is not associated with an increase in mortality in oldest old subjects: The Octabaix three-year follow-up study. Gerontology 2014, 60, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Domiciano, D.S.; Machado, L.G.; Lopes, J.B.; Figueiredo, C.P.; Caparbo, V.F.; Oliveira, R.M.; Scazufca, M.; McClung, M.R.; Pereira, R.M. Bone mineral density and parathyroid hormone as independent risk factors for mortality in community-dwelling older adults: A population-based prospective cohort study in Brazil. The Sao Paulo Ageing & Health (SPAH) Study. J. Bone Miner. Res. 2016, 31, 1146–1157. [Google Scholar] [CrossRef]
- Kitamura, K.; Nakamura, K.; Nishiwaki, T.; Ueno, K.; Hasegawa, M. Low body mass index and low serum albumin are predictive factors for short-term mortality in elderly Japanese requiring home care. Tohoku J. Exp. Med. 2010, 221, 29–34. [Google Scholar] [CrossRef]
- Bates, C.J.; Hamer, M.; Mishra, G.D. A study of relationships between bone-related vitamins and minerals, related risk markers, and subsequent mortality in older British people: The National Diet and Nutrition Survey of People Aged 65 Years and Over. Osteoporos Int. 2012, 23, 457–466. [Google Scholar] [CrossRef]
- Afzal, S.; Brondum-Jacobsen, P.; Bojesen, S.E.; Nordestgaard, B.G. Genetically low vitamin D concentrations and increased mortality: Mendelian randomisation analysis in three large cohorts. BMJ 2014, 349, g6330. [Google Scholar] [CrossRef]
- Brenner, H.; Jansen, L.; Saum, K.U.; Holleczek, B.; Schöttker, B. Vitamin D supplementation trials aimed at reducing mortality have much higher power when focusing on people with low serum 25-hydroxyvitamin D concentrations. J. Nutr. 2017, 147, 1325–1333. [Google Scholar] [CrossRef]
- Buchebner, D.; McGuigan, F.; Gerdhem, P.; Ridderstrale, M.; Akesson, K. Association between hypovitaminosis D in elderly women and long- and short-term mortality-results from the Osteoporotic Prospective Risk Assessment cohort. J. Am. Geriatr. Soc. 2016, 64, 990–997. [Google Scholar] [CrossRef] [PubMed]
- El Hilali, J.; de Koning, E.J.; van Ballegooijen, A.J.; Lips, P.; Sohl, E.; van Marwijk, H.W.J.; Visser, M.; van Schoor, N.M. Vitamin D, PTH and the risk of overall and disease-specific mortality: Results of the Longitudinal Aging Study Amsterdam. J. Steroid Biochem. Mol. Biol. 2016, 164, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Heath, A.K.; Williamson, E.J.; Kvaskoff, D.; Hodge, A.M.; Ebeling, P.R.; Baglietto, L.; Neale, R.E.; Giles, G.G.; Eyles, D.W.; English, D.R. 25-Hydroxyvitamin D concentration and all-cause mortality: The Melbourne Collaborative Cohort Study. Public Health Nutr. 2017, 20, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- Hirani, V.; Cumming, R.G.; Naganathan, V.; Blyth, F.; Le Couteur, D.G.; Handelsman, D.J.; Waite, L.M.; Seibel, M.J. Associations between serum 25-hydroxyvitamin D concentrations and multiple health conditions, physical performance measures, disability, and all-cause mortality: The Concord Health and Ageing in Men Project. J. Am. Geriatr. Soc. 2014, 62, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Kestenbaum, B.; Katz, R.; de Boer, I.; Hoofnagle, A.; Sarnak, M.J.; Shlipak, M.G.; Jenny, N.S.; Siscovick, D.S. Vitamin D, parathyroid hormone, and cardiovascular events among older adults. J. Am. Coll. Cardiol. 2011, 58, 1433–1441. [Google Scholar] [CrossRef] [PubMed]
- Khaw, K.T.; Luben, R.; Wareham, N. Serum 25-hydroxyvitamin D, mortality, and incident cardiovascular disease, respiratory disease, cancers, and fractures: A 13-y prospective population study. Am. J. Clin. Nutr. 2014, 100, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Kritchevsky, S.B.; Tooze, J.A.; Neiberg, R.H.; Schwartz, G.G.; Hausman, D.B.; Johnson, M.A.; Bauer, D.C.; Cauley, J.A.; Shea, M.K.; Cawthon, P.M.; et al. 25-Hydroxyvitamin D, parathyroid hormone, and mortality in black and white older adults: The health ABC study. J. Clin. Endocrinol. Metab. 2012, 97, 4156–4165. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, T.; Shiraki, M.; Tanaka, S.; Ohta, H. Contributions of 25-hydroxyvitamin D, co-morbidities and bone mass to mortality in Japanese postmenopausal women. Bone 2009, 44, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.M.; Vanderschueren, D.; Boonen, S.; O’Neill, T.W.; Pendleton, N.; Pye, S.R.; Ravindrarajah, R.; Gielen, E.; Claessens, F.; Bartfai, G.; et al. Association of 25-hydroxyvitamin D, 1,25-dihydroxyvitamin D and parathyroid hormone with mortality among middle-aged and older European men. Age Ageing 2014, 43, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Leu Agelii, M.; Lehtinen-Jacks, S.; Zetterberg, H.; Sundh, V.; Bjorkelund, C.; Lissner, L. Low vitamin D status in relation to cardiovascular disease and mortality in Swedish women - Effect of extended follow-up. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Masson, S.; Agabiti, N.; Vago, T.; Miceli, M.; Mayer, F.; Letizia, T.; Wienhues-Thelen, U.; Mureddu, G.F.; Davoli, M.; Boccanelli, A.; et al. The fibroblast growth factor-23 and Vitamin D emerge as nontraditional risk factors and may affect cardiovascular risk. J. Intern. Med. 2015, 277, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Dobnig, H.; Nijpels, G.; Heine, R.J.; Stehouwer, C.D.A.; Snijder, M.B.; Van Dam, R.M.; Dekker, J.M. Vitamin D and mortality in older men and women. Clin. Endocrinol. 2009, 71, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Rohrmann, S.; Braun, J.; Bopp, M.; Faeh, D. Inverse association between circulating vitamin D and mortality--dependent on sex and cause of death? Nutr. Metab. Cardiovasc. Dis. 2013, 23, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Saliba, W.; Barnett, O.; Rennert, H.S.; Rennert, G. The risk of all-cause mortality is inversely related to serum 25(OH)D levels. J. Clin. Endocrinol. Metab. 2012, 97, 2792–2798. [Google Scholar] [CrossRef] [PubMed]
- Saliba, W.; Barnett-Griness, O.; Rennert, G. Obesity and association of serum 25(OH)D levels with all-cause mortality. Calcif. Tissue Int. 2014, 95, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Houston, D.K.; Bandinelli, S.; Sun, K.; Cherubini, A.; Cappola, A.R.; Guralnik, J.M.; Ferrucci, L. Relationship of 25-hydroxyvitamin D with all-cause and cardiovascular disease mortality in older community-dwelling adults. Eur. J. Clin. Nutr. 2010, 64, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Signorello, L.B.; Han, X.; Cai, Q.; Cohen, S.S.; Cope, E.L.; Zheng, W.; Blot, W.J. A prospective study of serum 25-hydroxyvitamin d levels and mortality among African Americans and non-African Americans. Am. J. Epidemiol. 2013, 177, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Skaaby, T.; Husemoen, L.L.; Linneberg, A. Does liver damage explain the inverse association between vitamin D status and mortality? Ann. Epidemiol. 2013, 23, 812–814. [Google Scholar] [CrossRef]
- Skaaby, T.; Husemoen, L.L.; Pisinger, C.; Jorgensen, T.; Thuesen, B.H.; Fenger, M.; Linneberg, A. Vitamin D status and incident cardiovascular disease and all-cause mortality: A general population study. Endocrine 2013, 43, 618–625. [Google Scholar] [CrossRef]
- Sun, Y.Q.; Langhammer, A.; Skorpen, F.; Chen, Y.; Mai, X.M. Serum 25-hydroxyvitamin D level, chronic diseases and all-cause mortality in a population-based prospective cohort: The HUNT Study, Norway. BMJ Open 2017, 7, e017256. [Google Scholar] [CrossRef]
- Szulc, P.; Claustrat, B.; Delmas, P.D. Serum concentrations of 17beta-E2 and 25- hydroxycholecalciferol (25OHD) in relation to all-cause mortality in older men - The MINOS study. Clin. Endocrinol. 2009, 71, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Tunstall-Pedoe, H.; Woodward, M.; Hughes, M.; Anderson, A.; Kennedy, G.; Belch, J.; Kuulasmaa, K. Prime mover or fellow traveller: 25-hydroxy vitamin D’s seasonal variation, cardiovascular disease and death in the Scottish Heart Health Extended Cohort (SHHEC). Int. J. Epidemiol. 2015, 44, 1602–1612. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, J.K.; Nurmi, T.; Voutilainen, S.; Mursu, J.; Tuomainen, T.P. Association of serum 25-hydroxyvitamin D with the risk of death in a general older population in Finland. Eur. J. Nutr. 2011, 50, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Vogt, S.; Decke, S.; de Las Heras Gala, T.; Linkohr, B.; Koenig, W.; Ladwig, K.H.; Peters, A.; Thorand, B. Prospective association of vitamin D with frailty status and all-cause mortality in older adults: Results from the KORA-Age Study. Prev. Med. 2015, 73, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Welsh, P.; Doolin, O.; McConnachie, A.; Boulton, E.; McNeil, G.; Macdonald, H.; Hardcastle, A.; Hart, C.; Upton, M.; Watt, G.; et al. Circulating 25OHD, dietary vitamin D, PTH, and calcium associations with incident cardiovascular disease and mortality: The MIDSPAN Family Study. J. Clin. Endocrinol. Metab. 2012, 97, 4578–4587. [Google Scholar] [CrossRef]
- Wong, Y.Y.; McCaul, K.A.; Yeap, B.B.; Hankey, G.J.; Flicker, L. Low vitamin D status is an independent predictor of increased frailty and all-cause mortality in older men: The Health in Men Study. J. Clin. Endocrinol. Metab. 2013, 98, 3821–3828. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Knuiman, M.; Divitini, M.; Hung, J.; Lim, E.M.; Cooke, B.R.; Walsh, J.P. Serum 25-hydroxyvitamin D as a predictor of mortality and cardiovascular events: A 20-year study of a community-based cohort. Clin. Endocrinol. 2018, 88, 154–163. [Google Scholar] [CrossRef]
- Schmutz, E.A.; Zimmermann, M.B.; Rohrmann, S. The inverse association between serum 25-hydroxyvitamin D and mortality may be modified by vitamin A status and use of vitamin A supplements. Eur. J. Nutr. 2016, 55, 393–402. [Google Scholar] [CrossRef]
- Trevisan, C.; Veronese, N.; Maggi, S.; Baggio, G.; Toffanello, E.D.; Zambon, S.; Sartori, L.; Musacchio, E.; Perissinotto, E.; Crepaldi, G.; et al. Factors influencing transitions between frailty states in elderly adults: The Progetto Veneto Anziani longitudinal study. J. Am. Geriatr. Soc. 2017, 65, 179–184. [Google Scholar] [CrossRef]
- Semba, R.D.; Houston, D.K.; Ferrucci, L.; Cappola, A.R.; Sun, K.; Guralnik, J.M.; Fried, L.P. Low serum 25-hydroxyvitamin D concentrations are associated with greater all-cause mortality in older community-dwelling women. Nutr. Res. 2009, 29, 525–530. [Google Scholar] [CrossRef]
- Amer, M.; Qayyum, R. Relationship between 25-hydroxyvitamin D and all-cause and cardiovascular disease mortality. Am. J. Med. 2013, 126, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Eaton, C.B.; Young, A.; Allison, M.A.; Robinson, J.; Martin, L.W.; Kuller, L.H.; Johnson, K.C.; Curb, J.D.; Van Horn, L.; McTiernan, A.; et al. Prospective association of vitamin D concentrations with mortality in postmenopausal women: Results from the Women’s Health Initiative (WHI). Am. J. Clin. Nutr. 2011, 94, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Schierbeck, L.L.; Rejnmark, L.; Tofteng, C.L.; Stilgren, L.; Eiken, P.; Mosekilde, L.; Kober, L.; Jensen, J.E. Vitamin D deficiency in postmenopausal, healthy women predicts increased cardiovascular events: A 16-year follow-up study. Eur. J. Endocrinol. 2012, 167, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Aucott, L.S.; McNeill, G. Nutritional status and subsequent all-cause mortality in men and women aged 75 years or over living in the community. Br. J. Nutr. 2007, 98, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Bolland, M.J.; Bacon, C.J.; Horne, A.M.; Mason, B.H.; Ames, R.W.; Wang, T.K.; Grey, A.B.; Gamble, G.D.; Reid, I.R. Vitamin D insufficiency and health outcomes over 5 y in older women. Am. J. Clin. Nutr. 2010, 91, 82–89. [Google Scholar] [CrossRef]
- Cawthon, P.M.; Parimi, N.; Barrett-Connor, E.; Laughlin, G.A.; Ensrud, K.E.; Hoffman, A.R.; Shikany, J.M.; Cauley, J.A.; Lane, N.E.; Bauer, D.C.; et al. Serum 25-hydroxyvitamin D, parathyroid hormone, and mortality in older men. J. Clin. Endocrinol. Metab. 2010, 95, 4625–4634. [Google Scholar] [CrossRef]
- Chien, K.L.; Hsu, H.C.; Chen, P.C.; Lin, H.J.; Su, T.C.; Chen, M.F.; Lee, Y.T. Total 25-hydroxyvitamin D concentration as a predictor for all-cause death and cardiovascular event risk among ethnic Chinese adults: A cohort study in a Taiwan community. PLoS ONE 2015, 10, e0123097. [Google Scholar] [CrossRef]
- Johansson, H.; Oden, A.; Kanis, J.; McCloskey, E.; Lorentzon, M.; Ljunggren, O.; Karlsson, M.K.; Thorsby, P.M.; Tivesten, A.; Barrett-Connor, E.; et al. Low serum vitamin D is associated with increased mortality in elderly men: MrOS Sweden. Osteoporos Int. 2012, 23, 991–999. [Google Scholar] [CrossRef]
- Lin, S.W.; Chen, W.; Fan, J.H.; Dawsey, S.M.; Taylor, P.R.; Qiao, Y.L.; Abnet, C.C. Prospective study of serum 25-hydroxyvitamin D concentration and mortality in a Chinese population. Am. J. Epidemiol. 2012, 176, 1043–1050. [Google Scholar] [CrossRef]
- Hutchinson, M.S.; Grimnes, G.; Joakimsen, R.M.; Figenschau, Y.; Jorde, R. Low serum 25-hydroxyvitamin D levels are associated with increased all-cause mortality risk in a general population: The Tromsø study. Eur. J. Endocrinol. 2010, 162, 935–942. [Google Scholar] [CrossRef]
- Grant, W.B.; Karras, S.N.; Bischoff-Ferrari, H.A.; Annweiler, C.; Boucher, B.J.; Juzeniene, A.; Garland, C.F.; Holick, M.F. Do studies reporting ‘U’-shaped serum 25-hydroxyvitamin D-health outcome relationships reflect adverse effects? Dermatoendocrinol 2016, 8, e1187349. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A. Cautious interpretation of the reverse J-shaped association between circulating 25-hydroxyvitamin D and total mortality is necessary. J. Clin. Endocrinol. Metab. 2012. [E-Letter to the article by Durup et al. 2012;97:2644–2652], Published online 14 June. [Google Scholar]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldei, J.; Boehm, B.O.; Weihrauch, G.; Maerz, W. Independent association of low serum 25-hydroxyvitamin d and 1,25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch. Intern. Med. 2008, 168, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Schleithoff, S.S.; Frisch, S.; Gotting, C.; Kuhn, J.; Koertke, H.; Kleesiek, K.; Tenderich, G.; Koerfer, R. Circulating calcitriol concentrations and total mortality. Clin. Chem. 2009, 55, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Durazo-Arvizu, R.A.; Dawson-Hughes, B.; Kramer, H.; Cao, G.; Merkel, J.; Coates, P.M.; Sempos, C.T. The reverse J-shaped association between serum total 25-hydroxyvitamin D concentration and all-cause mortality: The impact of assay standardization. Am. J. Epidemiol. 2017, 185, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Dudenkov, D.V.; Mara, K.C.; Petterson, T.M.; Maxson, J.A.; Thacher, T.D. Serum 25-hydroxyvitamin D values and risk of all-cause and cause-specific mortality: A population-based cohort study. Mayo Clin. Proc. 2018, 93, 721–730. [Google Scholar] [CrossRef]
- Scragg, R. Emerging evidence of thresholds for beneficial effects from vitamin D supplementation. Nutrients 2018, 10, 561. [Google Scholar] [CrossRef]
- Grant, W.B. Effect of follow-up time on the relation between prediagnostic serum 25-hydroxyvitamin D and all-cause mortality rate. Dermatoendocrinol 2012, 4, 198–202. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D supplements and prevention of cancer and cardiovascular disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Whitfield, K.; Wetterslev, J.; Simonetti, R.G.; Bjelakovic, M.; Gluud, C. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst. Rev. 2014, 1, CD007470. [Google Scholar] [CrossRef]
- Kilkkinen, A.; Knekt, P.; Aro, A.; Rissanen, H.; Marniemi, J.; Heliovaara, M.; Impivaara, O.; Reunanen, A. Vitamin D status and the risk of cardiovascular disease death. Am. J. Epidemiol. 2009, 170, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Skaaby, T.; Husemoen, L.L.; Pisinger, C.; Jorgensen, T.; Thuesen, B.H.; Fenger, M.; Linneberg, A. Vitamin D status and cause-specific mortality: A general population study. PLoS ONE 2012, 7, e52423. [Google Scholar] [CrossRef] [PubMed]
- Jassal, S.K.; Chonchol, M.; Von Mhlen, D.; Smits, G.; Barrett-Connor, E. Vitamin D, parathyroid hormone, and cardiovascular mortality in older adults: The Rancho Bernardo study. Am. J. Med. 2010, 123, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Schöttker, B.; Haug, U.; Schomburg, L.; Köhrle, J.; Perna, L.; Müller, H.; Holleczek, B.; Brenner, H. Strong associations of 25-hydroxyvitamin D concentrations with all-cause, cardiovascular, cancer, and respiratory disease mortality in a large cohort study. Am. J. Clin. Nutr. 2013, 97, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Daraghmeh, A.H.; Bertoia, M.L.; Al-Qadi, M.O.; Abdulbaki, A.M.; Roberts, M.B.; Eaton, C.B. Evidence for the vitamin D hypothesis: The NHANES III extended mortality follow-up. Atherosclerosis 2016, 255, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Perna, L.; Schöttker, B.; Holleczek, B.; Brenner, H. Serum 25-hydroxyvitamin D and incidence of fatal and nonfatal cardiovascular events: A prospective study with repeated measurements. J. Clin. Endocrinol. Metab. 2013, 98, 4908–4915. [Google Scholar] [CrossRef] [PubMed]
- Michos, E.D.; Reis, J.P.; Post, W.S.; Lutsey, P.L.; Gottesman, R.F.; Mosley, T.H.; Sharrett, A.R.; Melamed, M.L. 25-Hydroxyvitamin D deficiency is associated with fatal stroke among whites but not blacks: The NHANES-III linked mortality files. Nutrition 2012, 28, 367–371. [Google Scholar] [CrossRef]
- Wong, G.; Lim, W.H.; Lewis, J.; Craig, J.C.; Turner, R.; Zhu, K.; Lim, E.M.; Prince, R. Vitamin D and cancer mortality in elderly women. BMC Cancer 2015, 15, 106. [Google Scholar] [CrossRef]
- Liu, L.; Chen, M.; Hankins, S.R.; Nunez, A.E.; Watson, R.A.; Weinstock, P.J.; Newschaffer, C.J.; Eisen, H.J. Serum 25-hydroxyvitamin D concentration and mortality from heart failure and cardiovascular disease, and premature mortality from all-cause in United States adults. Am. J. Cardiol. 2012, 110, 834–839. [Google Scholar] [CrossRef]
- Freedman, D.M.; Looker, A.C.; Abnet, C.C.; Linet, M.S.; Graubard, B.I. Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III study (1988-2006). Cancer Res. 2010, 70, 8587–8597. [Google Scholar] [CrossRef]
- Ong, J.S.; Gharahkhani, P.; An, J.Y.; Law, M.H.; Whiteman, D.C.; Neale, R.E.; MacGregor, S. Vitamin D and overall cancer risk and cancer mortality: A Mendelian randomization study. Hum. Mol. Genet. 2018, 27, 4315–4322. [Google Scholar] [CrossRef] [PubMed]
- Scragg, R.; Khaw, K.T.; Toop, L.; Sluyter, J.; Lawes, C.M.M.; Waayer, D.; Giovannucci, E.; Camargo, C.A., Jr. Monthly high-dose vitamin D supplementation and cancer risk: A post hoc analysis of the Vitamin D Assessment randomized clinical trial. JAMA Oncol. 2018, 4, e182178. [Google Scholar] [CrossRef] [PubMed]
- Keum, N.; Giovannucci, E. Vitamin D supplements and cancer incidence and mortality: A meta-analysis. Br. J. Cancer 2014, 111, 976–980. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B. Letter to the Editor: The J-shaped 25-hydroxyvitamin D concentration-cardiovascular disease mortality relation is very likely due to starting vitamin D supplementation late in life. J. Clin. Endocrinol. Metab. 2015, 100, L49–L50. [Google Scholar] [CrossRef] [PubMed]
- Mondul, A.M.; Weinstein, S.J.; Layne, T.M.; Albanes, D. Vitamin D and cancer risk and mortality: State of the science, gaps, and challenges. Epidemiol. Rev. 2017, 39, 28–48. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Whitfield, K.; Krstic, G.; Wetterslev, J.; Gluud, C. Vitamin D supplementation for prevention of cancer in adults. Cochrane Database Syst. Rev. 2014, 6, CD007469. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, E.T.; Kohler, L.N.; Kunihiro, A.G.; Jurutka, P.W. Vitamin D and colorectal, breast, and prostate cancers: A review of the epidemiological evidence. J. Cancer 2016, 7, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Maalmi, H.; Walter, V.; Jansen, L.; Boakye, D.; Schöttker, B.; Hoffmeister, M.; Brenner, H. Association between blood 25-hydroxyvitamin D levels and survival in colorectal cancer patients: An updated systematic review and meta-analysis. Nutrients 2018, 10, 896. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.Y.; Yao, Q.; Zhuo, Z.; Ma, Z.; Chen, G. Circulating vitamin D level and mortality in prostate cancer patients: A dose-response meta-analysis. Endocr. Connect. 2018, 7, R294–R303. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.J.; Callen, D.F.; Li, J.Y.; Zheng, H. Circulating vitamin D and overall survival in breast cancer patients: A dose-response meta-analysis of cohort studies. Integr. Cancer Ther. 2018, 17, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Maalmi, H.; Ordóñez-Mena, J.M.; Schöttker, B.; Brenner, H. Serum 25-hydroxyvitamin D levels and survival in colorectal and breast cancer patients: Systematic review and meta-analysis of prospective cohort studies. Eur. J. Cancer 2014, 50, 1510–1521. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Huang, X.Z.; Chen, W.J.; Wu, J.; Chen, Y.; Wu, C.C.; Wang, Z.N. Plasma 25-hydroxyvitamin D levels, vitamin D intake, and pancreatic cancer risk or mortality: A meta-analysis. Oncotarget 2017, 8, 64395–64406. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, G.; He, X.; Gao, J.; Wang, R.; Wang, Y.; Zhao, W. Serum 25-hydroxyvitamin D levels and prognosis in hematological malignancies: A systematic review and meta-analysis. Cell. Physiol. Biochem. 2015, 35, 1999–2005. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.; Newman, C.; Tomson, J.; Hin, H.; Kurien, R.; Cox, J.; Lay, M.; Sayer, J.; Hill, M.; Emberson, J.; et al. Estimation of the optimum dose of vitamin D for disease prevention in older people: Rationale, design and baseline characteristics of the BEST-D trial. Maturitas 2015, 80, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Neale, R.E.; Armstrong, B.K.; Baxter, C.; Duarte Romero, B.; Ebeling, P.; English, D.R.; Kimlin, M.G.; McLeod, D.S.; RL, O.C.; van der Pols, J.C.; et al. The D-Health Trial: A randomized trial of vitamin D for prevention of mortality and cancer. Contemp. Clin. Trials 2016, 48, 83–90. [Google Scholar] [CrossRef]
- Pradhan, A.D.; Manson, J.E. Update on the Vitamin D and OmegA-3 trial (VITAL). J. Steroid Biochem. Mol. Biol. 2016, 155, 252–256. [Google Scholar] [CrossRef]
- Scragg, R.; Waayer, D.; Stewart, A.W.; Lawes, C.M.M.; Toop, L.; Murphy, J.; Khaw, K.T.; Camargo, C.A., Jr. The Vitamin D Assessment (ViDA) Study: Design of a randomized controlled trial of vitamin D supplementation for the prevention of cardiovascular disease, acute respiratory infection, falls and non-vertebral fractures. J. Steroid Biochem. Mol. Biol. 2016, 164, 318–325. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heath, A.K.; Kim, I.Y.; Hodge, A.M.; English, D.R.; Muller, D.C. Vitamin D Status and Mortality: A Systematic Review of Observational Studies. Int. J. Environ. Res. Public Health 2019, 16, 383. https://doi.org/10.3390/ijerph16030383
Heath AK, Kim IY, Hodge AM, English DR, Muller DC. Vitamin D Status and Mortality: A Systematic Review of Observational Studies. International Journal of Environmental Research and Public Health. 2019; 16(3):383. https://doi.org/10.3390/ijerph16030383
Chicago/Turabian StyleHeath, Alicia K, Iris Y Kim, Allison M Hodge, Dallas R English, and David C Muller. 2019. "Vitamin D Status and Mortality: A Systematic Review of Observational Studies" International Journal of Environmental Research and Public Health 16, no. 3: 383. https://doi.org/10.3390/ijerph16030383
APA StyleHeath, A. K., Kim, I. Y., Hodge, A. M., English, D. R., & Muller, D. C. (2019). Vitamin D Status and Mortality: A Systematic Review of Observational Studies. International Journal of Environmental Research and Public Health, 16(3), 383. https://doi.org/10.3390/ijerph16030383




