The Cost-Effectiveness Analysis of Cervical Cancer Screening Using a Systematic Invitation System in Lithuania
Abstract
1. Introduction
2. Materials and Methods
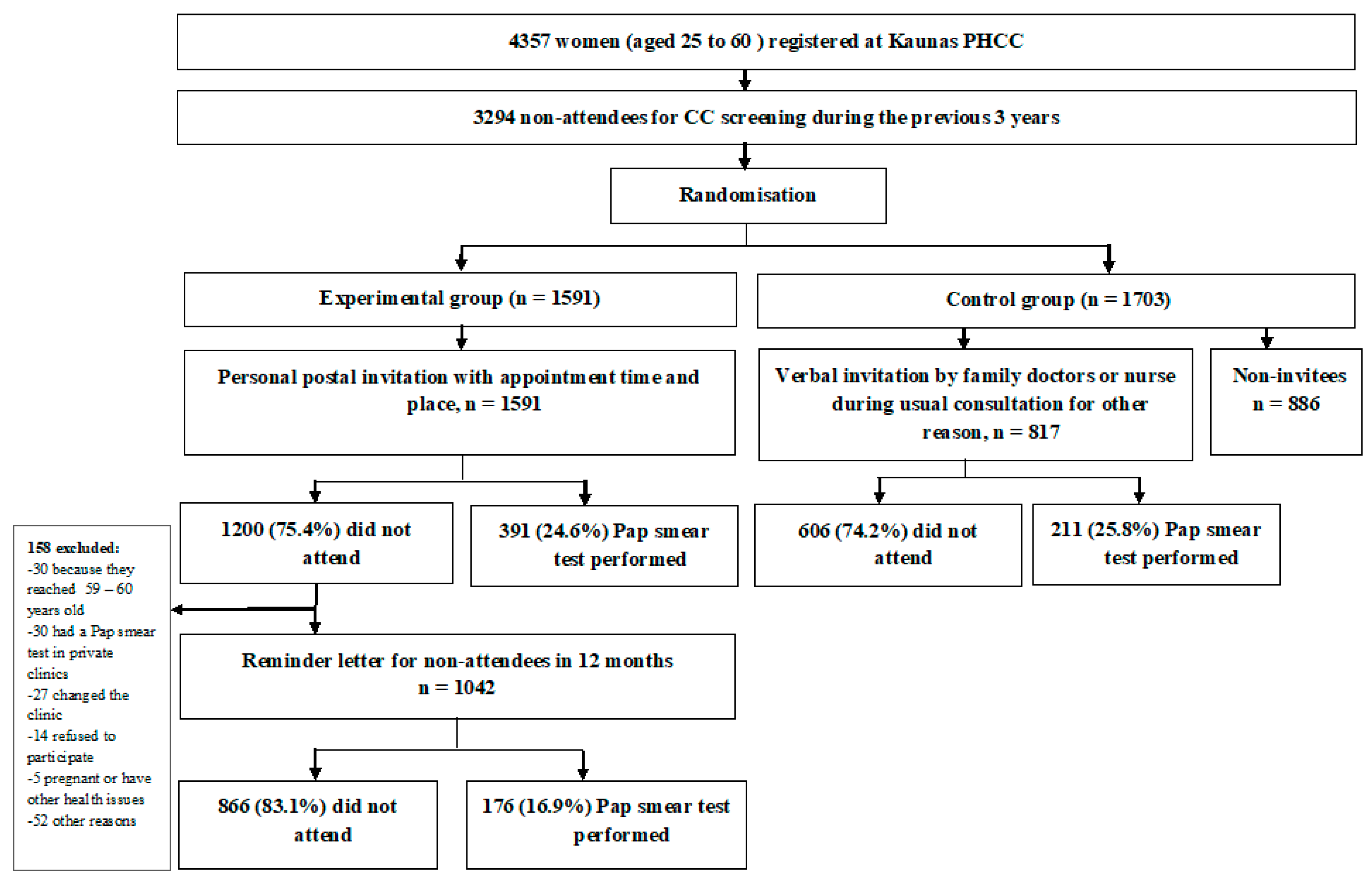
2.1. Data Collection and Study Design
2.2. Invitation Strategies and Costs
2.3. The Cost of Healthcare Services
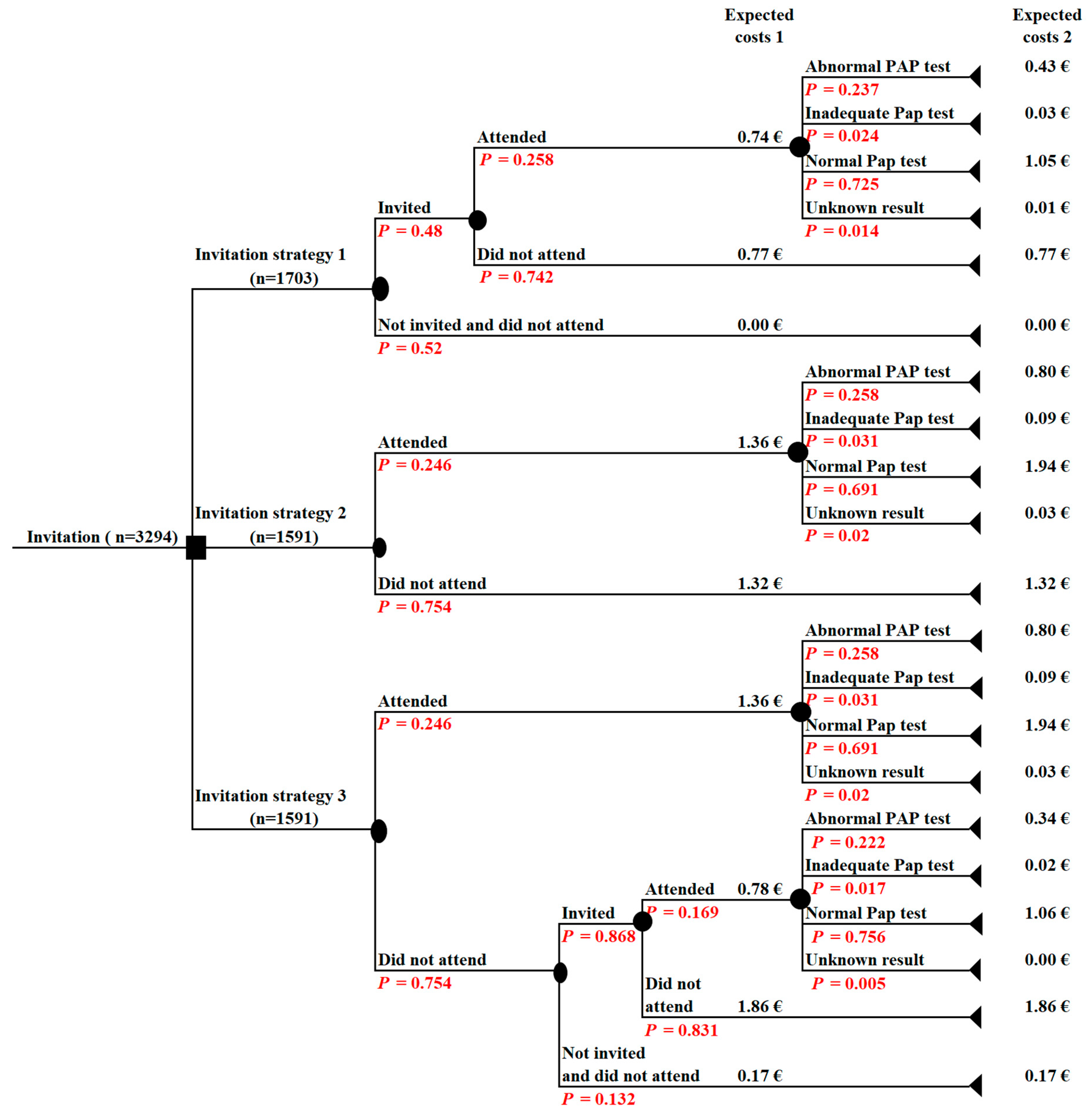
2.4. Economic Analysis
3. Results
3.1. The Participation in CC Screening and Pap Smear Test Results
3.2. The Cost-Effectiveness Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Albero, G.; Serrano, B.; Mena, M.; Gómez, D.; Muñoz, J.; Bosch, F.X.; de Sanjosé, S. ICO/IARCInformation Centre on HPV and Cancer (HPV Information Centre). Human Papillomavirus and RelatedDiseases in Lithuania. Summary Report 17 June 2019. Available online: https://hpvcentre.net/statistics/reports/LTU.pdf (accessed on 15 November 2019).
- Council Recommendation of 2 December 2003 on Cancer Screening (2003/878/EC). OJ L 327: 34–38. Available online: https://ec.europa.eu/jrc/sites/jrcsh/files/2_December_2003%20cancer%20screening.pdf (accessed on 13 November 2019).
- Arbyn, M.; Anttila, A.; Jordan, J.; Ronco, G.; Schenck, U.; Segnan, N.; Wiener, H.; Herbert, A.; von Karsa, L. European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition—Summary document. Ann. Oncol. 2010, 21, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Arbyn, M.; Raifu, A.O.; Weiderpass, E.; Bray, F.; Anttila, A. Trends of cervical cancer mortality in the member states of the European Union. Eur. J. Cancer 2009, 45, 2640–2648. [Google Scholar] [CrossRef] [PubMed]
- Serraino, D.; Gini, A.; Taborelli, M.; Ronco, G.; Giorgi-Rossi, P.; Zappa, M.; Crocetti, E.; Franzo, A.; Falcini, F.; Visioli, C.B.; et al. Changes in cervical cancer incidence following the introduction of organized screening in Italy. Prev. Med. 2015, 75, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Altobelli, E.; Lattanzi, A. Cervical carcinoma in the European Union: An update on disease burden, screening program state of activation, and coverage as of March 2014. Int. J. Gynecol. Cancer 2015, 25, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Salo, H.; Nieminen, P.; Kilpi, T.; Auranen, K.; Leino, T.; Vänskä, S.; Tiihonen, P.; Lehtinen, M.; Anttila, A. Divergent coverage, frequency and costs of organised and opportunistic Pap testing in Finland. Int. J. Cancer 2014, 135, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Camilloni, L.; Ferroni, E.; Cendales, B.J.; Pezzarossi, A.; Furnari, G.; Borgia, P.; Guasticchi, G.; Giorgi Rossi, P. Methods to increase participation in organised screening programs: A systematic review. BMC Public Health 2013, 13, 464. [Google Scholar] [CrossRef] [PubMed]
- The order of the Lithuanian Minister of Health No V-482. Valstybes zinios, 3 July 2004, No. 104-3856. Available online: https://www.e-tar.lt/portal/en/legalActEditions/TAR.8FD6BCF64FD4 (accessed on 13 November 2019).
- Solomon, D.; Davey, D.; Kurman, R.; Moriarty, A.; O’Connor, D.; Prey, M.; Raab, S.; Sherman, M.; Wilbur, D.; Wright, T., Jr.; et al. The 2001 Bethesda System: Terminology for reporting results of cervical cytology. JAMA 2002, 287, 2114–2119. [Google Scholar] [CrossRef]
- Virtanen, A.; Anttila, A.; Luostarinen, T.; Malila, N.; Nieminen, P. Improving cervical cancer screening attendance in Finland. Int. J. Cancer 2015, 136, E677–E684. [Google Scholar] [CrossRef]
- Elfström, K.M.; Arnheim-Dahlström, L.; von Karsa, L.; Dillner, J. Cervical cancer screening in Europe: Quality assurance and organisation of programmes. Eur. J. Cancer 2015, 51, 950–968. [Google Scholar] [CrossRef]
- Vale, D.B.; Anttila, A.; Ponti, A.; Senore, C.; Sankaranaryanan, R.; Ronco, G.; Segnan, N.; Tomatis, M.; Žakelj, M.P.; Elfström, K.M.; et al. Invitation strategies and coverage in the population-based cancer screening programmes in the European Union. Eur. J. Cancer Prev. 2019, 28, 131–140. [Google Scholar] [CrossRef]
- Everatt, R.; Intaite, B. Trends in cervical cancer mortality rates in Lithuania, 1987–2016. Cancer Epidemiol. 2018, 57, 85–89. [Google Scholar] [CrossRef]
- Denny, L.; Prendiville, W. Cancer of the cervix: Early detection and cost-effective solutions. Int. J. Gynaecol. Obstet. 2015, 131, S28–S32. [Google Scholar] [CrossRef]
- Pedersen, K.; Sørbye, S.W.; Burger, E.A.; Lönnberg, S.; Kristiansen, I.S. Using Decision-Analytic Modeling to isolate interventions that are feasible, efficient and optimal: An application from the Norwegian Cervical Cancer Screening Program. Value Health 2015, 18, 1088–1097. [Google Scholar] [CrossRef]
- Rashid, R.M.; Ramli, S.; John, J.; Dahlui, M. Cost effective analysis of recall methods for cervical cancer screening in Selangor--results from a prospective randomized controlled trial. Asian Pac. J. Prev. 2014, 15, 5143–5147. [Google Scholar] [CrossRef]
- Mandelblatt, J.S.; Lawrence, W.F.; Gaffikin, L.; Limpahayom, K.K.; Lumbiganon, P.; Warakamin, S.; King, J.; Yi, B.; Ringers, P.; Blumenthal, P.D. Costs and benefits of different strategies to screen for cervical cancer in less-developed countries. J. Nat. Cancer Inst. 2002, 94, 1469–1483. [Google Scholar] [CrossRef]
- Trapero-Bertran, M.; Acera Pérez, A.; de Sanjosé, S.; Manresa Domínguez, J.M.; Rodríguez Capriles, D.; Rodriguez Martinez, A.; Bonet Simó, J.M.; Sanchez Sanchez, N.; Hidalgo Valls, P.; Díaz Sanchis, M. Cost-effectiveness of strategies to increase screening coverage for cervical cancer in Spain: The CRIVERVA study. BMC Public Health 2017, 17, 194. [Google Scholar] [CrossRef]
- Heranney, D.; Fender, M.; Velten, M.; Baldauf, J.J. A prospective randomized study of two reminding strategies: Telephone versus mail in the screening of cervical cancer in women who did not initially respond. Acta Cytol. 2011, 55, 334–340. [Google Scholar] [CrossRef]
- Broberg, G.; Jonasson, J.M.; Ellis, J.; Gyrd-Hansen, D.; Anjemark, B.; Glantz, A.; Söderberg, L.; Ryd, M.L.; Holtenman, M.; Milsom, I.; et al. Increasing participation in cervical cancer screening: Telephone contact with long-term non-attendees in Sweden. Results from RACOMIP, a randomized controlled trial. Int. J. Cancer 2013, 133, 164–171. [Google Scholar] [CrossRef]
- Haguenoer, K.; Sengchanh, S.; Gaudy-Graffin, C.; Boyard, J.; Fontenay, R.; Marret, H.; Goudeau, A.; Pigneaux de Laroche, N.; Rusch, E.; Giraudeau, B. Vaginal self-sampling is a cost-effective way to increase participation in a cervical cancer screening programme: A randomised trial. Br. J. Cancer 2014, 111, 2187–2196. [Google Scholar] [CrossRef]
- Nahvijou, A.; Hadji, M.; Marnani, A.B.; Tourang, F.; Bayat, N.; Weiderpass, E.; Daroudi, R.; Sari, A.A.; Zendehdel, K. A systematic review of economic aspects of cervical cancer screening strategies worldwide: Discrepancy between economic analysis and policymaking. Asian Pac. J. Cancer Prev. 2014, 15, 8229–8237. [Google Scholar] [CrossRef]
- Vassilakos, P.; Poncet, A.; Catarino, R.; Viviano, M.; Petignat, P.; Combescure, C. Cost-effectiveness evaluation of HPV self-testing offered to non-attendees in cervical cancer screening in Switzerland. Gynecol. Oncol. 2019, 153, 92–99. [Google Scholar] [CrossRef]
| Cost Category | Description | Unit Cost (€) |
|---|---|---|
| A verbal invitation from a family doctor | Reimbursed cost of the invitation | 1.45 |
| Cost of time spent for a verbal invitation by a family doctor (calculated using an average of hourly wage rate). | 0.70 | |
| A personal invitation letter | Reimbursed cost of the invitation | 1.45 |
| Cost of time of the screening department staff for identification of unscreened women, preparation and sending out of invitation letters (calculated using hourly wage rate). | 0.30 | |
| A personal reminder letter | Cost of a personal invitation letter discounted by 5%. | 1.67 |
| Pap smear test-taking | Reimbursed cost | 3.79 |
| Conventional Pap smear test assessment | Reimbursed cost | 5.79 |
| Information about the Pap smear test result | Cost of time spent (calculated using hourly wage rate): | |
| a family doctor | 2.80 | |
| screening department staff | 0.08 | |
| a nurse | 1.28 |
| Pap Smear Test Results | Experimental Group | Control Group | |
|---|---|---|---|
| The First Invitation Letter (n = 391) | The Reminder Letter (n = 176) | Invitation by a Family Doctor (n = 211) | |
| n (%) | n (%) | n (%) | |
| Unknown a | 8 (2.0) | 1 (0.6) | 3 (1.4) |
| Inadequate b | 12 (3.1) | 3 (1.7) | 5 (2.4) |
| Normal | 270 (69.1) | 133 (75.6) | 153 (72.5) |
| Abnormal c | 101 (25.8) | 39 (22.2) | 50 (23.7) |
| ASC-US | 42 (10.7) | 14 (7.9) | 17 (8.1) |
| ASC-H | - | 1 (0.6) | 1 (0.5) |
| LSIL | 59 (15.1) | 24 (13.6) | 30 (14.2) |
| HSIL | - | - | 2 (0.9) |
| Cost Category | Unit Cost (€) | Invitation Strategy 1 (n = 1703) | Invitation Strategy 2 (n = 1591) | Invitation Strategy 3 (n = 1591) | |||
|---|---|---|---|---|---|---|---|
| n | Cost (€) | n | Cost (€) | n | Cost (€) | ||
| A verbal invitation from a family doctor | 2.15 | 817 | 1756.55 | ||||
| A personal invitation letter | 1.75 | 1591 | 2784.25 | 1591 | 2784.25 | ||
| A personal reminder letter | 1.67 | 1042 | 1740.14 | ||||
| Pap smear test-taking | 3.79 | 211 | 799.69 | 391 | 1481.89 | 567 | 2117.25 |
| Conventional Pap smear test assessment | 5.79 | 204 | 1181.16 | 383 | 2217.57 | 558 | 3181.82 |
| Information about the Pap smear test result: | |||||||
| family doctor | 2.80 | 70 | 196 | ||||
| screening department staff | 0.08 | 290 | 23.2 | 427 | 34.16 | ||
| nurse | 1.28 | 101 | 129.28 | 140 | 176.86 | ||
| Cost of screened women: invitation + performed Pap smear test | 2556.24 | 4266.14 | 6641.64 | ||||
| Total cost | 3933.40 | 6636.19 | 10034.48 | ||||
| Expected cost per one screened woman | 1.51 | 2.68 | 4.17 | ||||
| Expected cost per one abnormal Pap smear test detected | 2.29 | 4.18 | 6.31 | ||||
| Probability of a woman being screened * | 0.124 | 0.246 | 0.357 | ||||
| Probability of abnormal Pap smear test detected * | 0.029 | 0.063 | 0.088 | ||||
| ICER per one additionally screened woman | 9.67 | 13.47 | |||||
| ICER per one additional abnormal Pap smear test detected | 55.21 | 86.88 | |||||
| Scenario | Invitation Strategy | ICER (1) | ICER (2) |
|---|---|---|---|
| Base case | Strategy 2 (invitation letter) versus strategy 1 (regular practice) | €9.67 | €55.21 |
| Strategy 3 (invitation letter + reminder letter) versus strategy 2 (invitation letter) | €13.47 | €86.88 | |
| Without the discounting of a reminder letter | Strategy 2 (invitation letter) versus strategy 1 (regular practice) | €9.67 | €55.21 |
| Strategy 3 (invitation letter + reminder letter) versus strategy 2 (invitation letter) | €14.15 | €91.23 | |
| Without salary expenses | Strategy 2 (invitation letter) versus strategy 1 (regular practice) | €9.96 | €55.86 |
| Strategy 3 (invitation letter + reminder letter) versus strategy 2 (invitation letter) | €11.78 | €77.78 | |
| Expenses of invitation services decrease by 20% | Strategy 2 (invitation letter) versus strategy 1 (regular practice) | €8.43 | €50.79 |
| Strategy 3 (invitation letter + reminder letter) versus strategy 2 (invitation letter) | €11.89 | €79.62 | |
| Expenses of invitation services increase by 20% | Strategy 2 (invitation letter) versus strategy 1 (regular practice) | €10.90 | €59.63 |
| Strategy 3 (invitation letter + reminder letter) versus strategy 2 (invitation letter) | €15.12 | €94.33 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paulauskiene, J.; Stelemekas, M.; Ivanauskiene, R.; Petkeviciene, J. The Cost-Effectiveness Analysis of Cervical Cancer Screening Using a Systematic Invitation System in Lithuania. Int. J. Environ. Res. Public Health 2019, 16, 5035. https://doi.org/10.3390/ijerph16245035
Paulauskiene J, Stelemekas M, Ivanauskiene R, Petkeviciene J. The Cost-Effectiveness Analysis of Cervical Cancer Screening Using a Systematic Invitation System in Lithuania. International Journal of Environmental Research and Public Health. 2019; 16(24):5035. https://doi.org/10.3390/ijerph16245035
Chicago/Turabian StylePaulauskiene, Justina, Mindaugas Stelemekas, Rugile Ivanauskiene, and Janina Petkeviciene. 2019. "The Cost-Effectiveness Analysis of Cervical Cancer Screening Using a Systematic Invitation System in Lithuania" International Journal of Environmental Research and Public Health 16, no. 24: 5035. https://doi.org/10.3390/ijerph16245035
APA StylePaulauskiene, J., Stelemekas, M., Ivanauskiene, R., & Petkeviciene, J. (2019). The Cost-Effectiveness Analysis of Cervical Cancer Screening Using a Systematic Invitation System in Lithuania. International Journal of Environmental Research and Public Health, 16(24), 5035. https://doi.org/10.3390/ijerph16245035






