Chlorine Dioxide Degradation Issues on Metal and Plastic Water Pipes Tested in Parallel in a Semi-Closed System
Abstract
1. Introduction
2. Materials and Methods
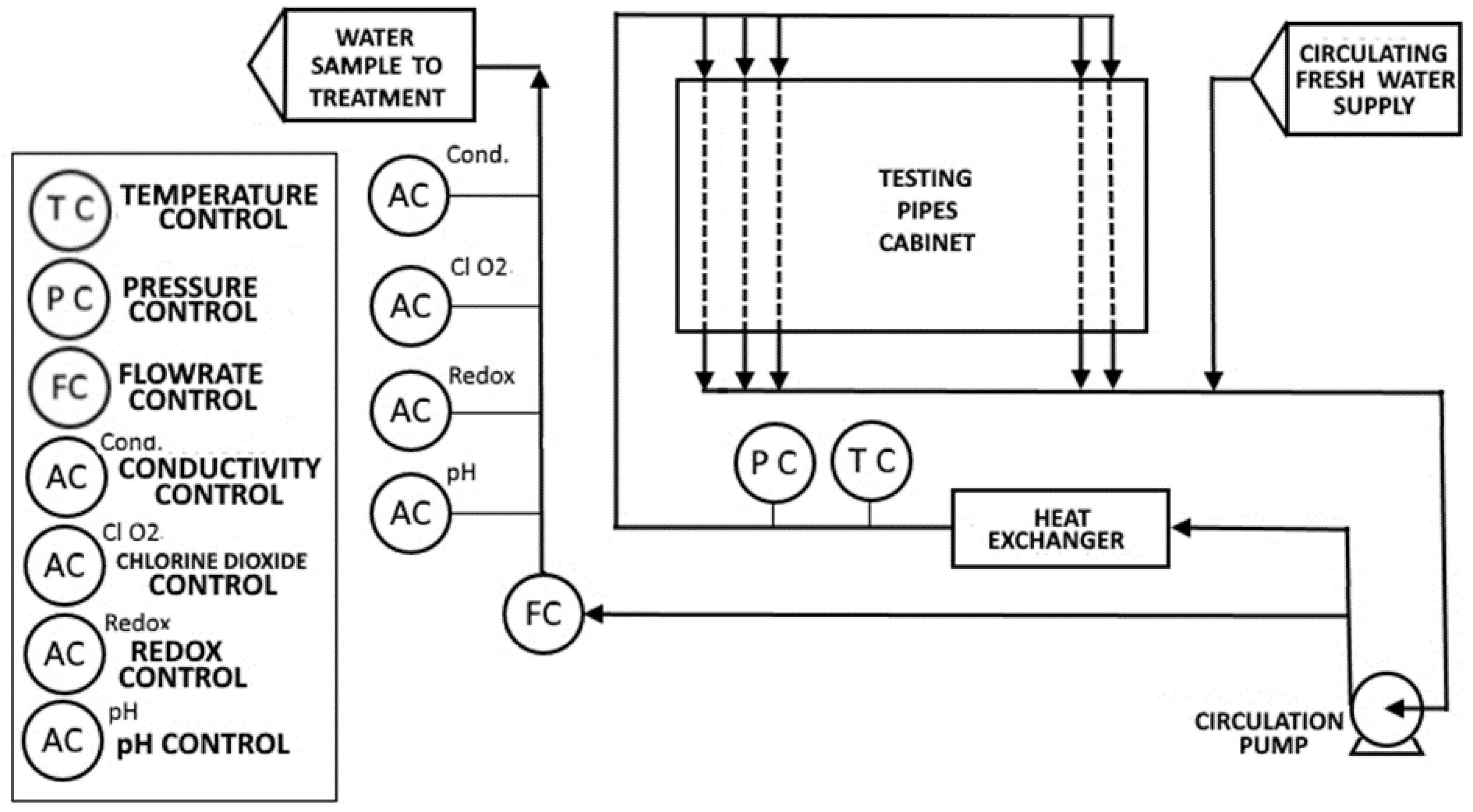
2.1. Experimental Setup
2.2. Characterization
2.2.1. Fourier Transform Infrared Spectroscopy (FT-IR)
2.2.2. Differential Scanning Calorimetry (DSC)
2.2.3. Optical Microscopy
2.2.4. Scanning Electron Microscopy (SEM)
2.2.5. Electrochemical Impedance Spectroscopy
3. Results and Discussion
3.1. Microscope Analyses
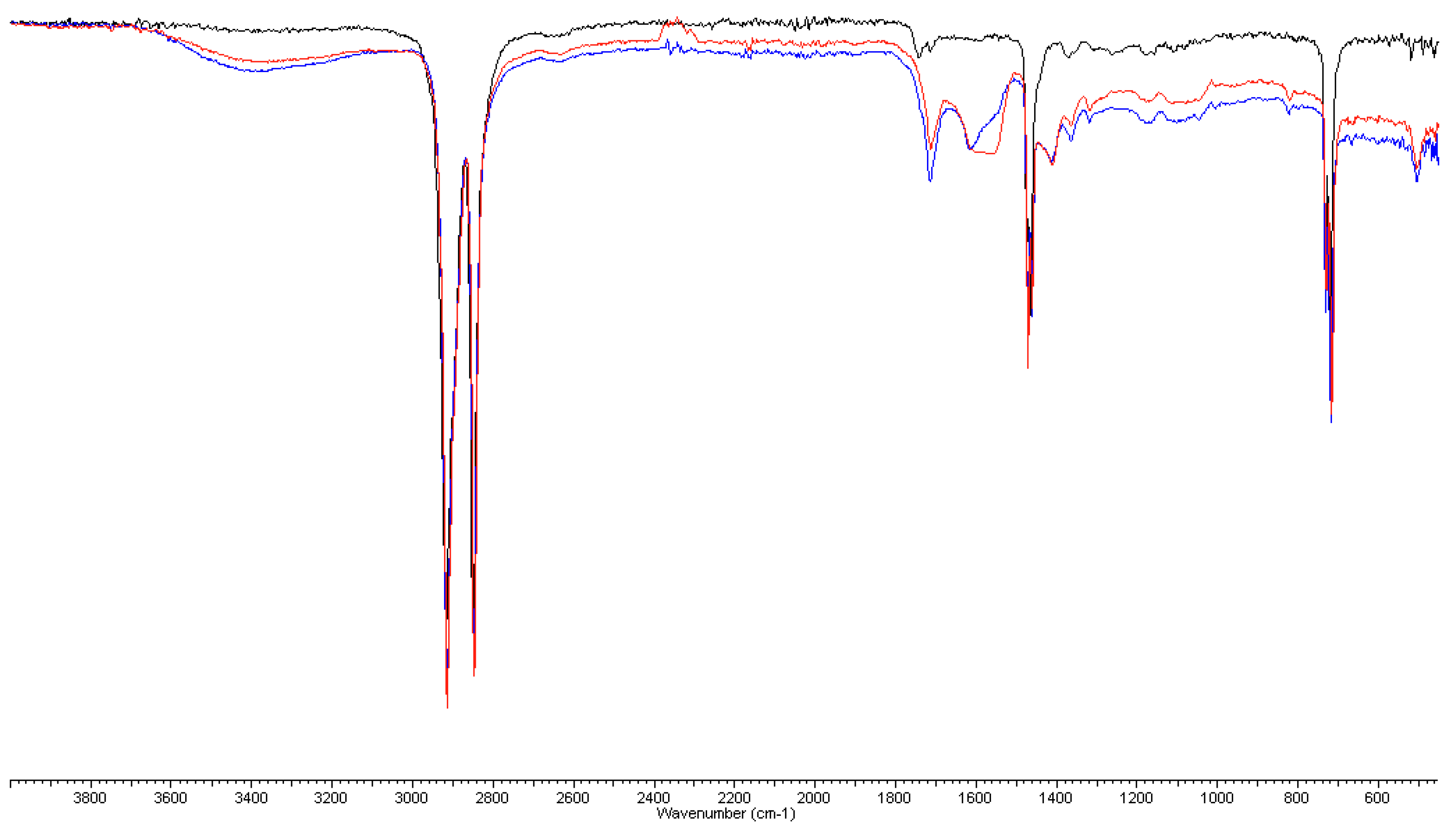
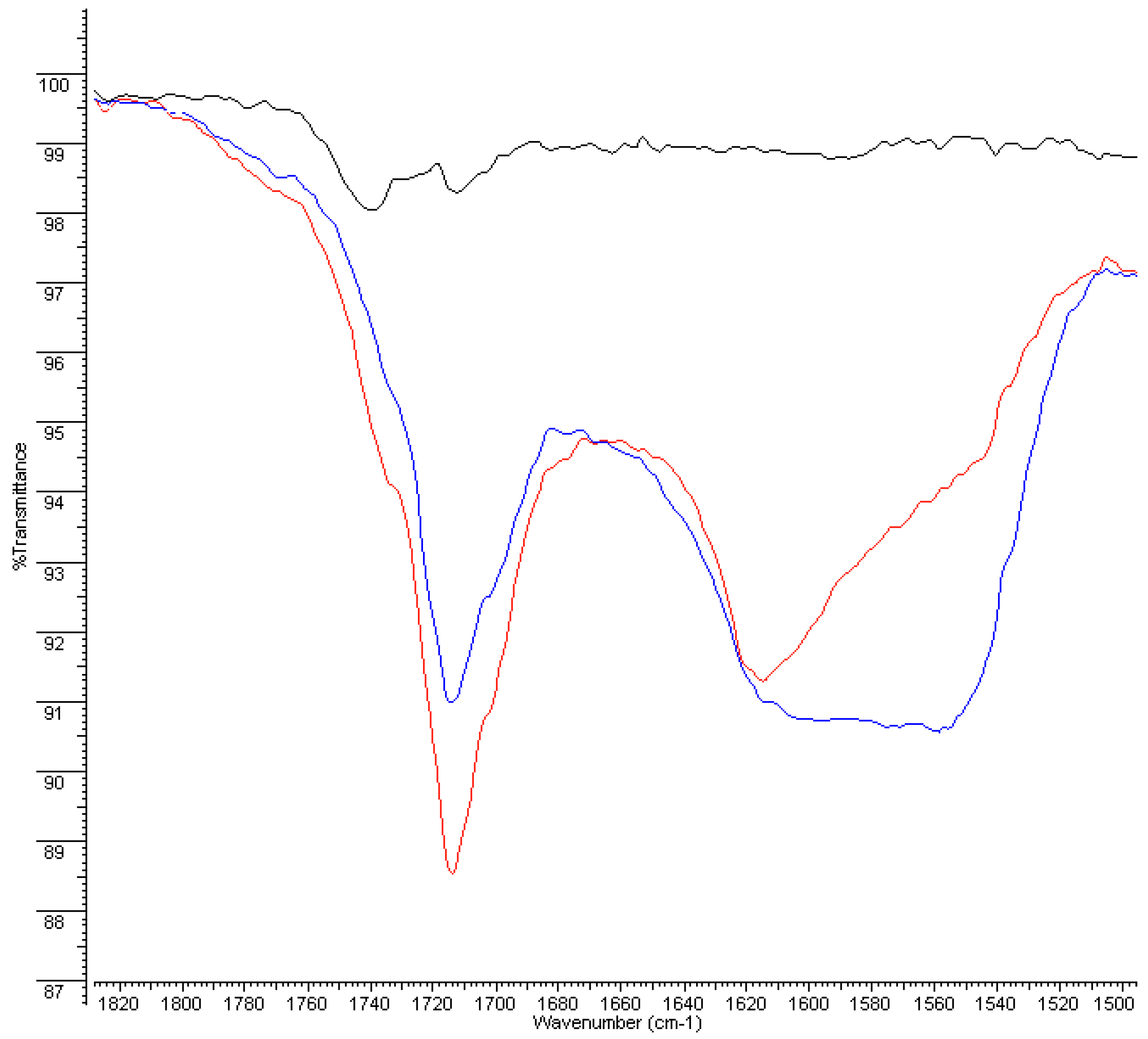
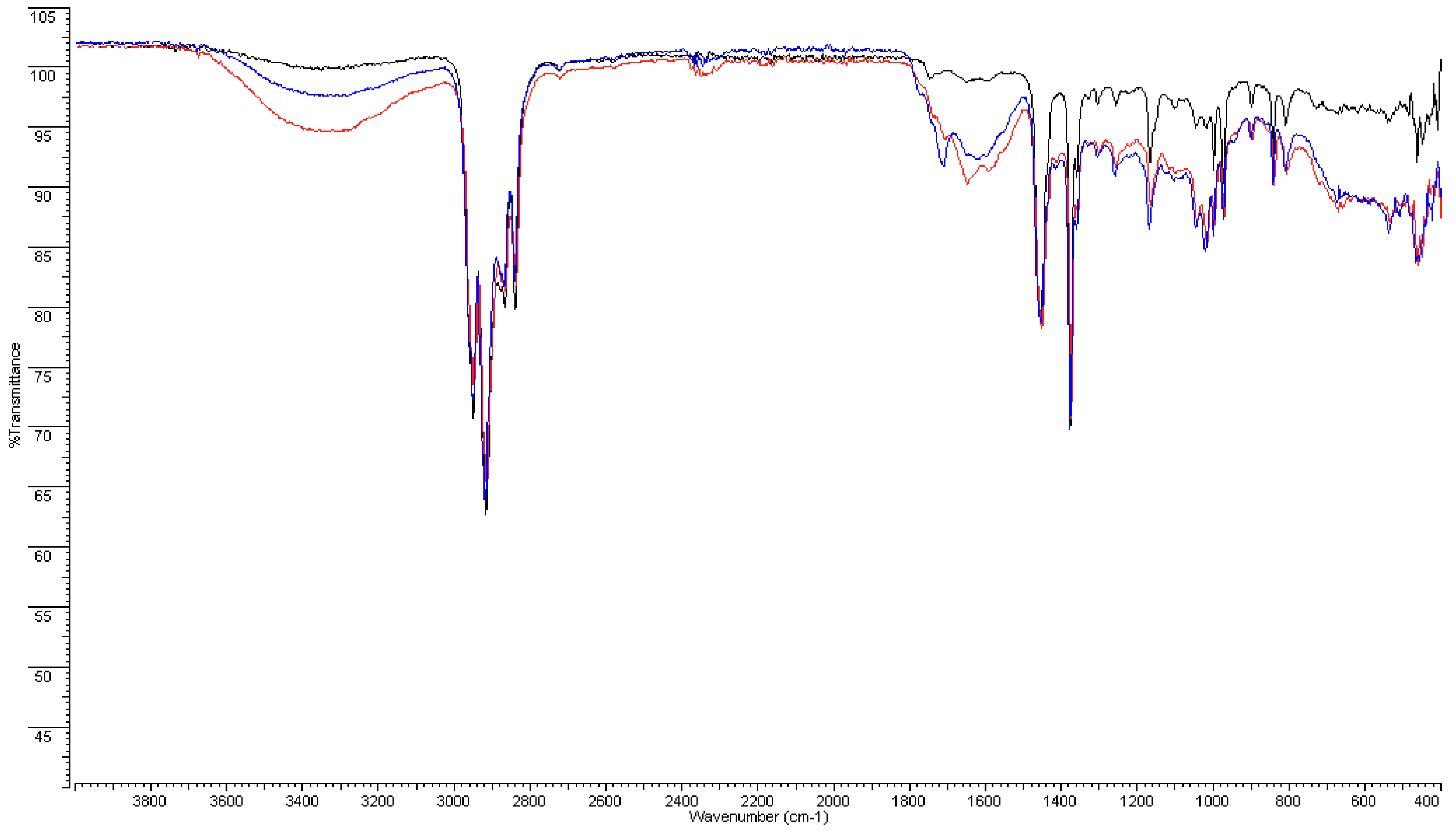
3.2. FT-IR and DSC Analyses on Plastic Pipes
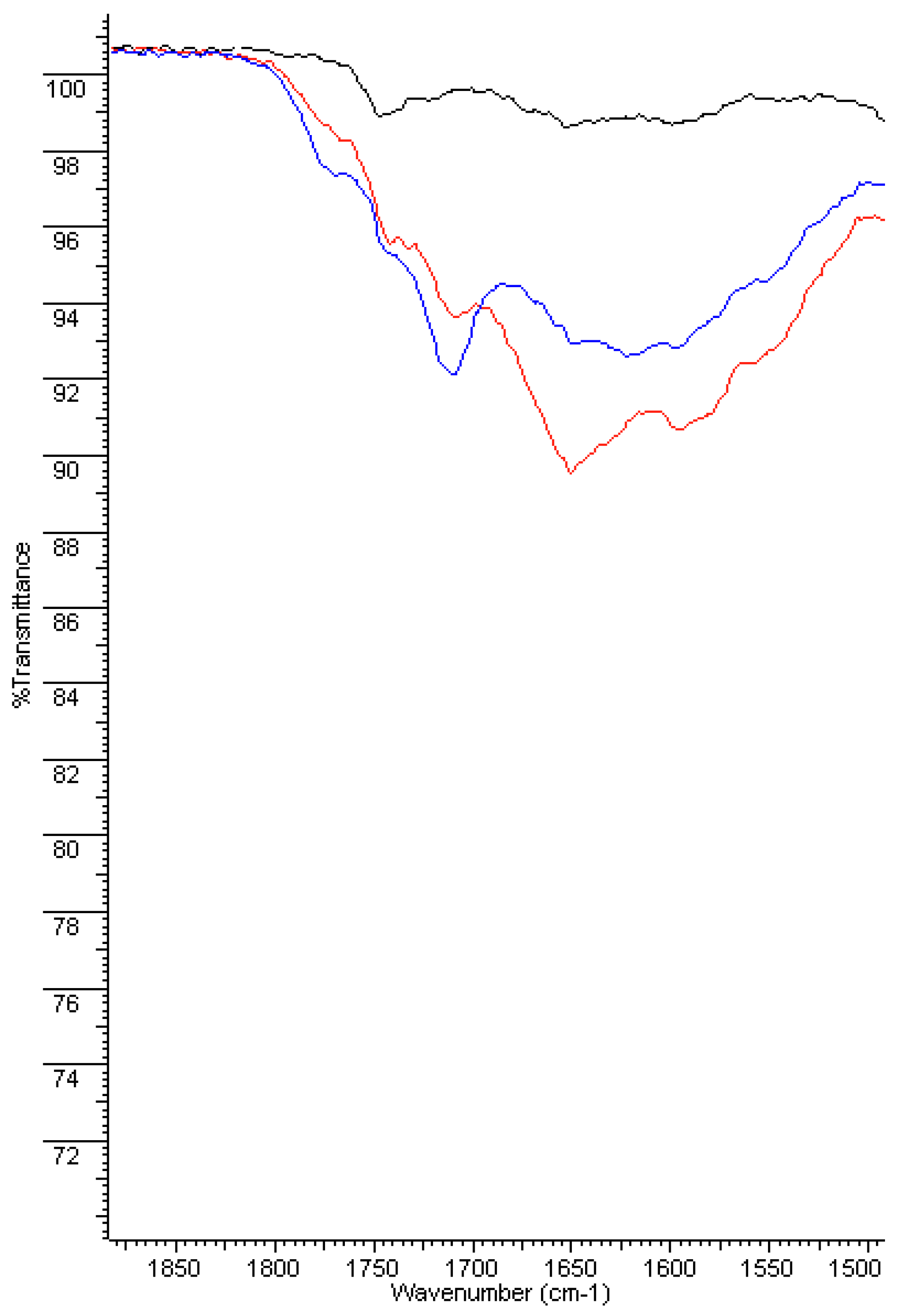
3.2.1. FT-IR Analyses
3.2.2. DSC Analyses
3.3. Impedance Analyses
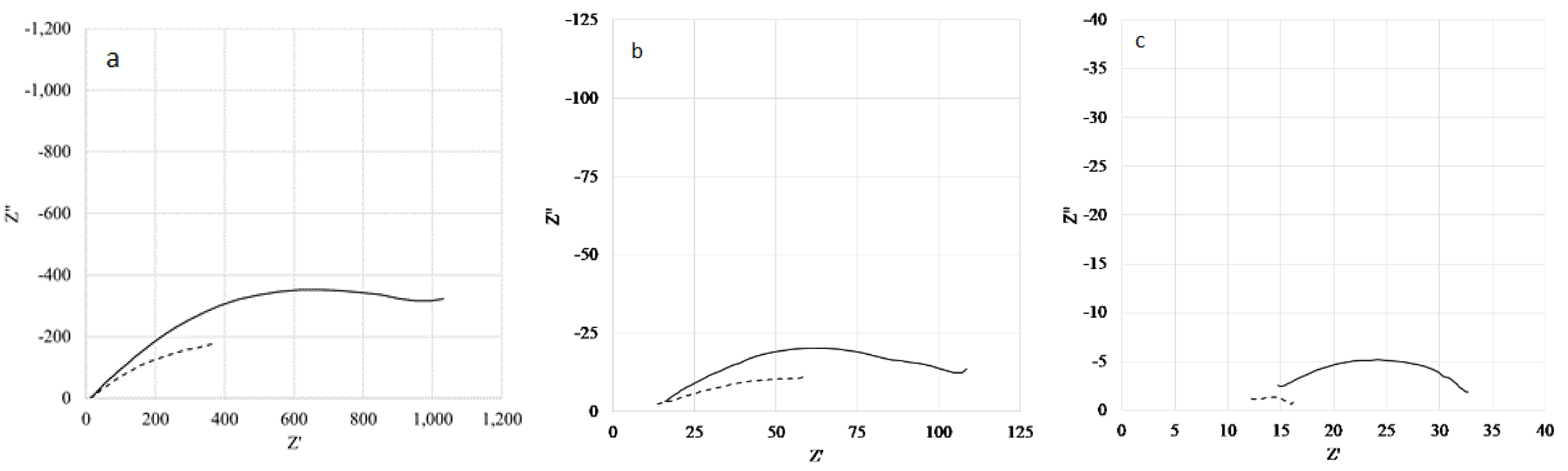
3.3.1. PERT Multilayer Samples
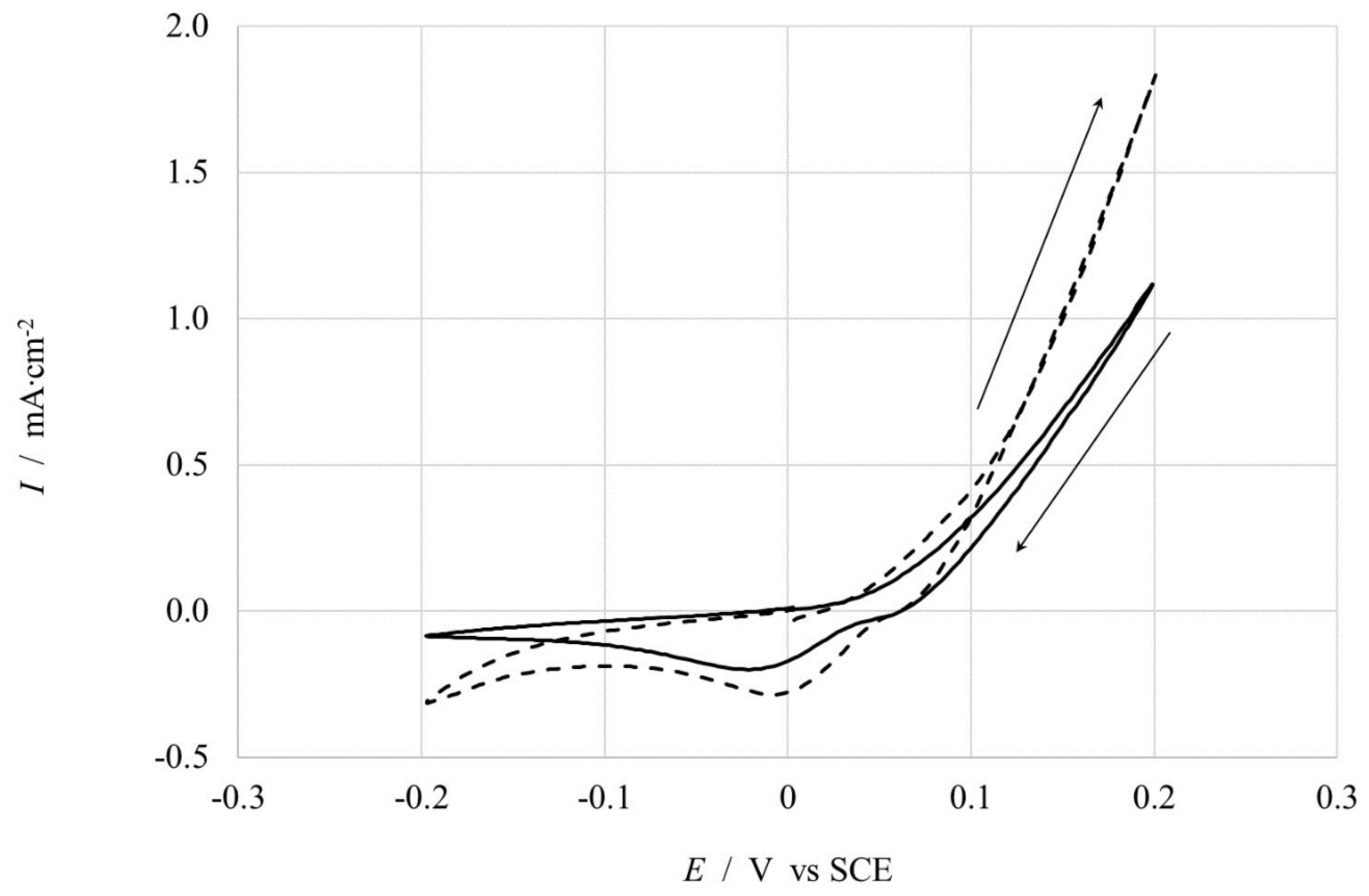
3.3.2. Copper Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Aieta, E.M.; Berg, J.D. A Review of Chlorine Dioxide in Drinking Water Treatment. J. Am. Water Assoc. 1986, 78, 62–72. [Google Scholar] [CrossRef]
- Simpson, G.D.; Miller, R.F.; Laxton, G.D.; Clements, W.R. A Focus on Chlorine Dioxide: The "Ideal" Biocide; Unichem International Inc.: Houston, TX, USA, 1993. [Google Scholar]
- White, C.C. Chlorine Dioxide. In White’s Handbook of Chlorination and Alternative Disinfectants, 5th ed.; Wiley: New York, NY, USA, 2010; pp. 700–757. [Google Scholar]
- Hofmann, R.; Andrews, R.C.; Ye, Q. Impact of Giardia inactivation requirements on ClO2 by-products. Environ. Technol. 1999, 20, 147–158. [Google Scholar] [CrossRef]
- Sadiq, R.; Rodriguez, M.J. Disinfection by-products (DBPs) in drinking water and predictive models for their occurrence: A review. Sci. Total Environ. 2004, 321, 21–46. [Google Scholar] [CrossRef] [PubMed]
- Vincenti, S.; de Waure, C.; Raponi, M.; Teleman, A.A.; Bonint, F.; Bruno, S.; Boccia, S.; Damiani, G.; Laurenti, P. Environmental surveillance of Legionella spp. colonization in the water system of a large academic hospital: Analysis of the four–year results on the effectiveness of the chlorine dioxide disinfection method. Sci. Total Environ. 2019, 657, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Binnie, C.; Kimber, M. Chlorine Dioxide. In Basic Water Treatment, 5th ed.; Royal Society of Chemistry: London, UK, 2013; p. 205. [Google Scholar]
- Stage 1 Disinfection and Disinfection By-Products (D/DBP) Rule; United States Environmental Protection Agency (EPA): Washington DC, USA, 1998.
- UBA Liste der Aufbereitungsstoffe und Desinfectionsverfahren gemäß § 11 TWVO—Trinkwasserverordnung; Umweldbundesamt (UBA): Dessau-Roβlau, Germany, 2001.
- Drinking Water Inspectorate List of Approved Products for Use in Public Water Supply in the United Kingdom; Drinking Water Inspectorate (DWI): London, UK, 2019.
- Ifwarson, M.; Aoyama, K. Results and experiences from tests on polyolefin pipes exposed to chlorinated water. In Proceedings of the 10th Plastic Pipes, Plastic Pipes Conference Association, Gothenburg, Sweden, 21–23 September 1998. [Google Scholar]
- Bradley, S.W.; Bradley, W.L. The effect of chlorine on the long-term durability of crosslinked polyethylene pipe. In Proceedings of the 10th International Conference of Deformation, Yield and Fracture of Polymers, Cambridge, UK, 7–10 April 1997; pp. 114–117. [Google Scholar]
- Dear, J.P.; Mason, N.S. The effects of chlorine depletion of antioxidants in PE. Polym. Polym. Compos. 2001, 9, 1–13. [Google Scholar]
- Hassinen, J.; Lundback, M.; Ifwarson, M.; Gedde, U.W. Deterioration of polyethylene pipes exposed to chlorinated water. Polym. Degrad. Stab. 2004, 84, 261–267. [Google Scholar] [CrossRef]
- Yu, W.; Azhdar, B.; Andersson, A.; Reitberger, T.; Hassinen, J.; Hjertberg, T.; Gedde, U.W. Deterioration of polyethylene pipes exposed to water containing chlorine dioxide. Polym. Degrad. Stab. 2011, 96, 790–797. [Google Scholar] [CrossRef]
- Colin, X.; Audouin, L.; Verdu, J.; Rozental-Evesque, M.; Rabaud, B.; Martin, F.; Bourgine, F. Aging of polyethylene pipes transporting drinking water disinfected by chlorine dioxide. I. Chemical aspects. Polym. Eng. Sci. 2009, 49, 1429–1437. [Google Scholar]
- Colin, X.; Audouin, L.; Verdu, J.; Rozental-Evesque, M.; Rabaud, B.; Martin, F.; Bourgine, F. Aging of polyethylene pipes transporting drinking water disinfected by chlorine dioxide. II. Lifetime Prediction. Polym. Eng. Sci. 2009, 49, 1642–1652. [Google Scholar]
- Smith, G.D.; Karlsson, K.; Gedde, U.W. Modeling of antioxidant loss from polyolefins in hot-water applications. I: Model and application to medium density polyethylene pipes. Polym. Eng. Sci. 1992, 32, 658–667. [Google Scholar] [CrossRef]
- Yu, W.; Reitberger, T.; Hjertberg, T.; Oderkerk, J.; Costa, F.R.; Gedde, U.W. Antioxidant consumption in squalane and polyethylene exposed to chlorinated aqueous media. Polym. Degrad. Stab. 2012, 97, 2370–2377. [Google Scholar] [CrossRef]
- Bredéacs, M.; Frank, A.; Bastero, A.; Stolarz, A.; Pinter, G. Accelerated aging of polyethylene pipe grades in aqueous chlorine dioxide at constant concentration. Polym. Degrad. Stab. 2018, 157, 80–89. [Google Scholar] [CrossRef]
- Rabello, M.S.; White, J.R. Crystallization and melting behaviour of photodegraded polypropylene—I. Chemi-crystallization. Polymer 1997, 38, 6379–6387. [Google Scholar] [CrossRef]
- Chung, S.; Oliphant, K.; Vibien, P.; Zhang, J. An Examination of the Relative Impact of Common Potable Water Disinfectants (Chlorine, Chloramines and Chlorine Dioxide) on Plastic Piping System Components. In Proceedings of the Annual Technical Conference—ANTEC, Cincinnati, OH, USA, 6–11 May 2007. [Google Scholar]
- Zhang, Z.; Stout, J.E.; Yu, V.L.; Vidic, R. Effect of pipe corrosion scales on chlorine dioxide consumption in drinking water distribution systems. Water Res. 2008, 42, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; von Gunten, U.; Croué, J.P. Enhanced chlorine dioxide decay in the presence of metal oxides: Relevance to drinking water distribution systems. Environ. Sci. Technol. 2013, 47, 8365–8372. [Google Scholar] [CrossRef] [PubMed]
- Azhdar, B.; Yu, W.; Gedde, U.W. A new method for assessing the efficiency of stabilizers in polyolefins exposed to chlorinated water media. Polym. Test. 2009, 28, 661–667. [Google Scholar] [CrossRef]
- Devilliers, C.; Fayolle, B.; Laiarinandrasana, L.; Oberti, S.; Gaudichet-Maurin, E. Kinetics of chlorine-induced polyethylene degradation in water pipes. Polym. Degrad. Stab. 2011, 96, 1361–1368. [Google Scholar] [CrossRef][Green Version]
- Castillo Montes, J.; Cadoux, D.; Creus, J.; Touzain, S.; Gaudichet-Maurin, E.; Correc, O. Ageing of polyethylene at raised temperature in contact with chlorinated sanitary hot water. Part I—Chemical aspects. Polym. Degrad. Stab. 2012, 97, 149–157. [Google Scholar] [CrossRef]
- Chung, S.; Li, T.; Oliphant, K.; Vibien, P. The mechanisms of chlorine dioxide oxidation of plastic piping systems. In Proceedings of the Plastic Pipes XIV, Plastic Pipes Conference Association, Budapest, Hungary, 22–24 September 2008. [Google Scholar]
- Osawa, Z.; Tsurumi, K. Evaluation of Some Commercial Copper Deactivators by Chemiluminescence. Polym. Degrad. Stab. 1989, 26, 151–160. [Google Scholar] [CrossRef]
- Hansen, R.H.; Russell, C.A.; De Benedictis, T.; Martin, W.M.; Pascale, J.V. Inhibition of the Copper-Catalyzed Oxidation of Polypropylene. J. Polym. Sci. Part A 1964, 2, 587–609. [Google Scholar] [CrossRef]
- Stevens, A.A.; Seeger, D.R. Ozone/Chlorine Dioxide Oxidation Products of Organic Materials; Rice, R.G., Cotruvo, J.A., Eds.; Ozone Press International: Cleveland, OH, USA, 1978; pp. 383–399. [Google Scholar]
- Vand der Stok, E.J.W.; Jacobson, K.; Jansma, S.O.; Lukes, D. Literature review: Effect of ClO2 on ageing of polymer materials and related test methods. In Proceedings of the 19th Plastic Pipes Conference, Las Vegas, NV, USA, 24–26 September 2018. [Google Scholar]


| Material | Elements—New Pipe (w/w) | Elements—Pipe after 8 Weeks (w/w) |
|---|---|---|
| Copper | Cu (100%) | Cu, O |
| Galvanized Steel | Zn (97.7%) Fe (2.3%) | Zn (60.3%), O (21.9%), Fe (13.6%), Cu (2.4%), Cl (0.8%), Al (0.7%), K (0.3%) |
| PPR | C, O (trace) | O (71.3%), C (26.2%), Ti (0.9%), Fe (0.4%), Si (0.3%), Cu (0.3%), Cl (0.1%) |
| PE-RT | C, O (trace) | O (72.1%), C (27.0%), Cu (0.5%), Cl (0.1%), Si (0.1%) |
| Sample | Aging (Weeks) | Position | Tm | ΔHm | Tc | ΔHc | χc |
|---|---|---|---|---|---|---|---|
| °C | J/g | °C | J/g | % | |||
| PERT | 0 | 127.9 | −133.4 | 115.8 | 155.0 | 46.5 | |
| 4 | Surface | 126.8 | −143.8 | 116.6 | 164.5 | 50.1 | |
| Bulk | 127.7 | −155.6 | 116.3 | 159.7 | 54.3 | ||
| 8 | Surface | 127.1 | −155.1 | 116.9 | 168.0 | 54.1 | |
| Bulk | 127.2 | −159.0 | 116.4 | 168.1 | 55.4 | ||
| PPR | 0 | 141.5 | −50.9 | 111.2 | 60.7 | 24.6 | |
| 4 | Surface | 143.6 | −58.5 | 111.6 | 62.8 | 28.3 | |
| Bulk | 143.4 | −54.2 | 111.0 | 64.2 | 26.2 | ||
| 8 | Surface | 145.9 | −45.0 | 111.0 | 63.4 | 21.7 | |
| Bulk | 141.7 | −62.7 | 111.8 | 63.7 | 30.3 |
| Sample | Cdl, F |
|---|---|
| PERT new pipe | (3.00 ± 0.10) 10−11 |
| PERT aged 8 weeks | (4.40 ± 0.04) 10−11 |
| Cu | Rct, Ω | CPE, F | α |
|---|---|---|---|
| New pipe 0 V | (1486 ± 19) | (4.35 ± 0.05) 10−4 | (0.578 ± 0.002) |
| Aged 8 weeks 0 V | (1279 ± 127) | (2.30 ± 0.06) 10−3 | (0.453 ± 0.006) |
| New pipe 0.05 V | (108.9 ± 0.8) | (1.37 ± 0.03) 10−3 | (0.450 ± 0.002) |
| Aged 8 weeks 0.05 V | (121 ± 6) | (1.18 ± 0.03) 10−2 | (0.227 ± 0.005) |
| New pipe 0.1 V | (22.6 ± 0.3) | (6.6 ± 0.3) 10−4 | (0.524 ± 0.008) |
| Aged 8 weeks 0.1 V | (6.7 ± 0.5) | (3.6 ± 0.6) 10−3 | (0.423 ± 0.003) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vertova, A.; Miani, A.; Lesma, G.; Rondinini, S.; Minguzzi, A.; Falciola, L.; Ortenzi, M.A. Chlorine Dioxide Degradation Issues on Metal and Plastic Water Pipes Tested in Parallel in a Semi-Closed System. Int. J. Environ. Res. Public Health 2019, 16, 4582. https://doi.org/10.3390/ijerph16224582
Vertova A, Miani A, Lesma G, Rondinini S, Minguzzi A, Falciola L, Ortenzi MA. Chlorine Dioxide Degradation Issues on Metal and Plastic Water Pipes Tested in Parallel in a Semi-Closed System. International Journal of Environmental Research and Public Health. 2019; 16(22):4582. https://doi.org/10.3390/ijerph16224582
Chicago/Turabian StyleVertova, Alberto, Alessandro Miani, Giordano Lesma, Sandra Rondinini, Alessandro Minguzzi, Luigi Falciola, and Marco Aldo Ortenzi. 2019. "Chlorine Dioxide Degradation Issues on Metal and Plastic Water Pipes Tested in Parallel in a Semi-Closed System" International Journal of Environmental Research and Public Health 16, no. 22: 4582. https://doi.org/10.3390/ijerph16224582
APA StyleVertova, A., Miani, A., Lesma, G., Rondinini, S., Minguzzi, A., Falciola, L., & Ortenzi, M. A. (2019). Chlorine Dioxide Degradation Issues on Metal and Plastic Water Pipes Tested in Parallel in a Semi-Closed System. International Journal of Environmental Research and Public Health, 16(22), 4582. https://doi.org/10.3390/ijerph16224582










