Links between Cognitive Status and Trace Element Levels in Hair for an Environmentally Exposed Population: A Case Study in the Surroundings of the Estarreja Industrial Area
Abstract
1. Introduction
2. Methods
2.1. Participants
- (1)
- Sociodemographic and clinical questionnaire: During a personal interview, demographic and clinical data were collected through an extensive sociodemographic questionnaire and an inventory of past habits, current clinical health status, and medical history. The following data were collected: age, marital status, weight, height, nationality, education level, the period of time working in agriculture, pesticide application methods and time of exposure, use of personal protective equipment, home-grown foodstuff consumption, irrigation water source, and drinking water source. Additionally, a full medical record was obtained during this interview, including information on 29 symptoms typically associated with toxic elements exposure or essential elements deficiency [48];
- (2)
- The Mini-Mental State Examination (MMSE) [49,50,51] is a brief screening test for assessment of the global cognitive status. The MMSE score ranges from 0–30, with higher scores indicating better cognitive performance. In this study we considered the following categories: (i) 0–25 points: dementia; (ii) 26–28 points: mild cognitive impairment (MCI); and (iii) 29–30 points: normal cognitive functioning [51]. The MMSE is the most broadly used brief cognitive screening instrument in clinical, epidemiological, and research contexts. Despite the existence of other neuropsychological instruments with greater sensitivity in detecting cognitive decline at earlier stages (e.g., probably due to the lack of MMSE in including executive functioning assessment and the usage of rather simple tasks to assess short-term memory, working memory, attention, and concentration, language and visuospatial skills), the MMSE has been largely validated for different populations, thus representing a common reference in the communication between health professionals, including psychologists, neurologists, and psychiatrists [51];
- (3)
- The Geriatric Depression Scale (GDS) [45,46,47] is a brief instrument to assess depressive symptoms in older adults, composed of 30 dichotomous questions that evaluate emotional and behavioral symptoms. The maximum score is 30 points, with higher scores indicating greater severity of depressive symptomatology. In this study, we considered the following categories: (i) 0–10 points: absence of depressive symptoms; (ii) 11–20 points: mild depressive symptoms; and (iii) 21–30 points: moderate to severe depressive symptoms.
2.2. Study Groups
2.3. Hair Samples and Analysis
2.4. Soil Samples and Analysis
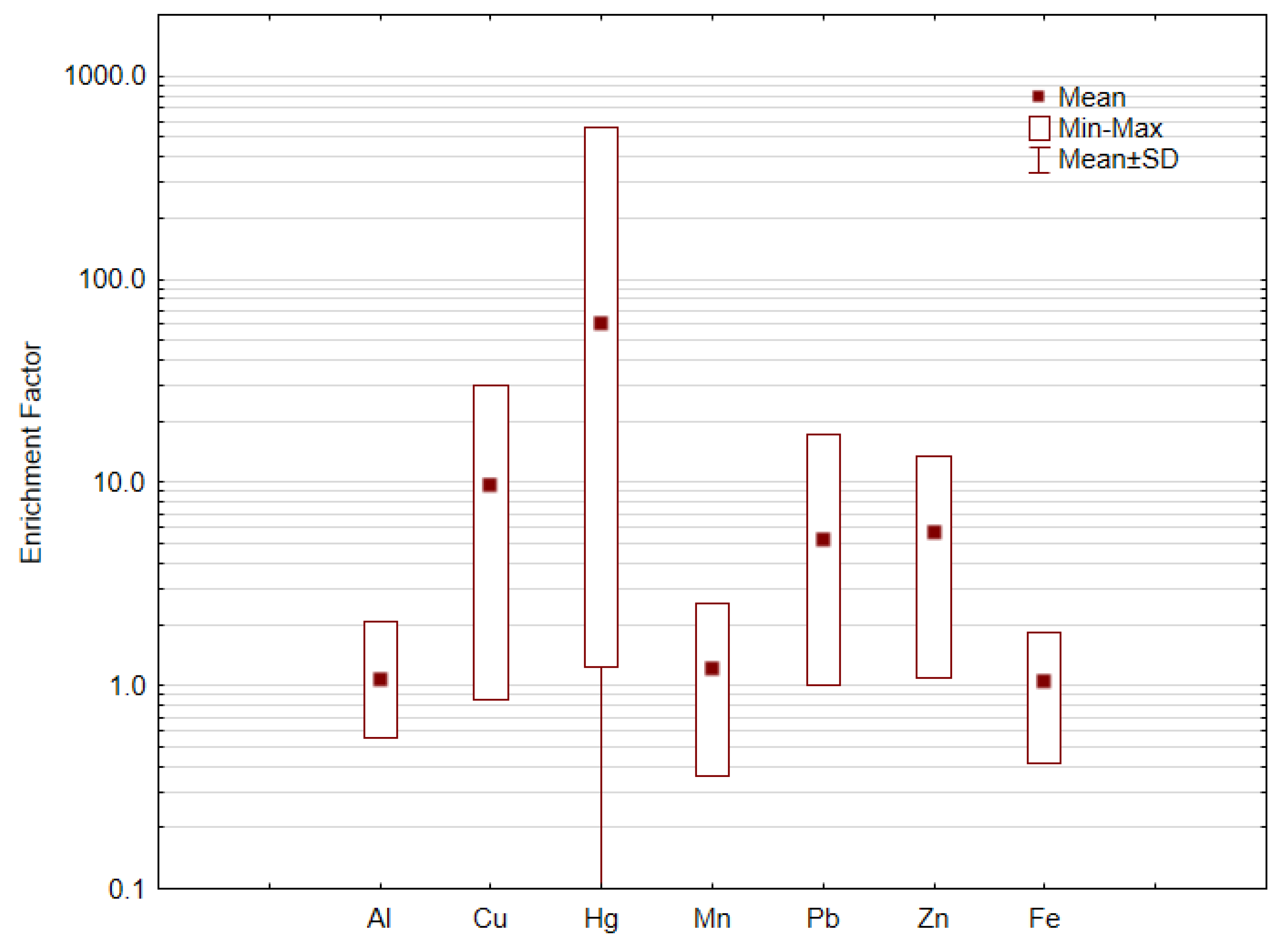
Enrichment Factor
2.5. Statistical Techniques
3. Results and Discussion
3.1. The Study Population Cognitive Status
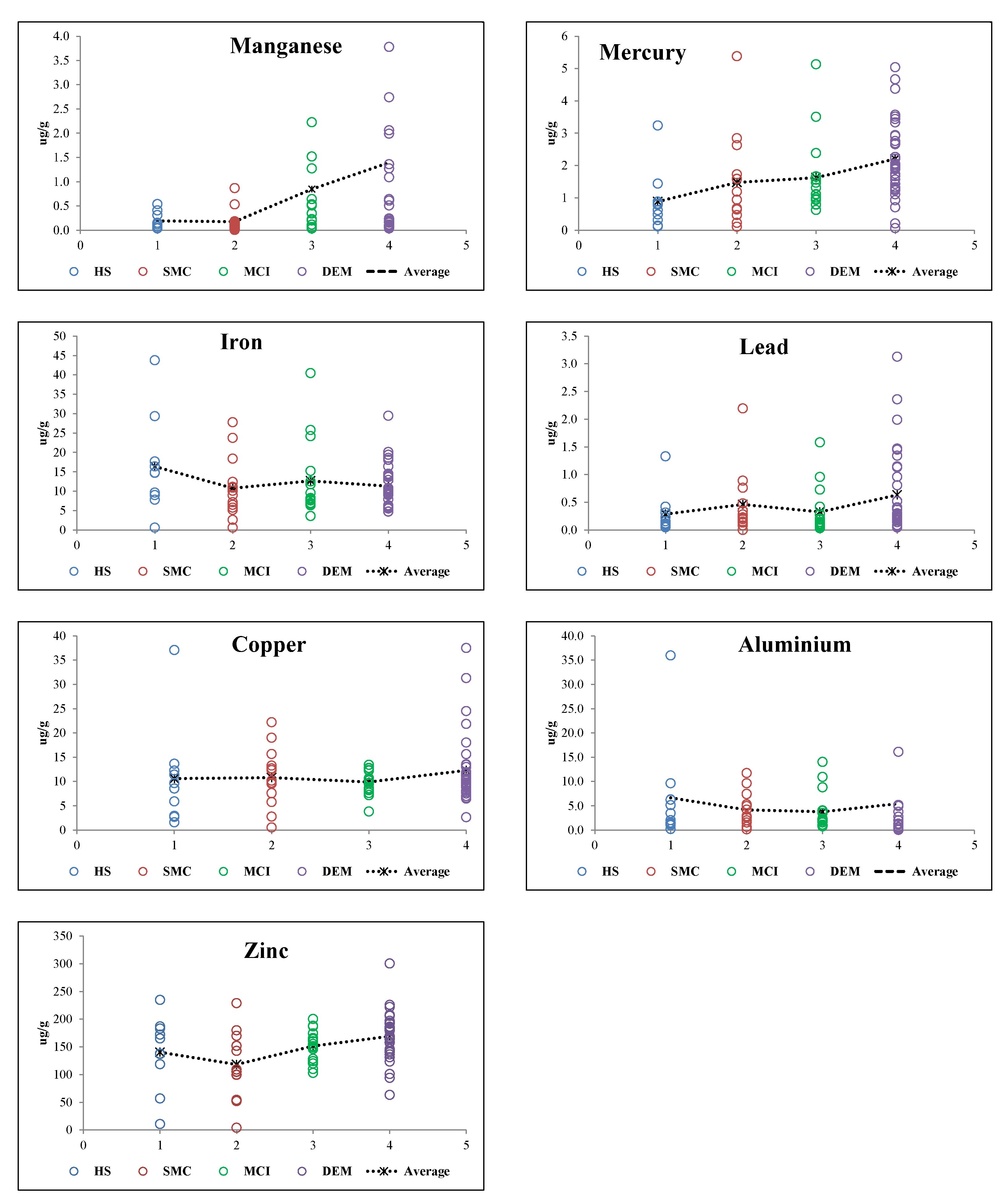
3.2. TE Levels in Hair and Population Cognitive Status Relations
3.3. Trace Elements Hair Versus Risk of AD
3.4. Metal Population Environmental Exposure
4. Conclusions
- -
- Participants whose professional activity was associated with agriculture and fisheries were shown to be the most vulnerable to dementia.
- -
- Participants diagnosed with dementia frequently consume home-grown foodstuffs, some of them probably irrigated with contaminated well water.
- -
- Biomonitoring and the analysis of the cognitive status of the residents surrounding ECC suggest that Hg levels in hair differed significantly between the four cognitive groups (healthy, subjective memory complaint, mild cognitive impairment, and dementia), increasing from healthy to dementia participants.
- -
- Mercury mean levels in soil samples were above Canadian guidelines for soil for agricultural uses and the enrichment factor calculation values highlighted that 26% of the studied soils reached the “extremely contaminated” class for Hg, with 65% of the soils reaching the “significant contamination” class.
- -
- Improved diagnostic results can be obtained using hair TE signatures coupled with MMSE scores. This strategy may prove useful for predictive diagnosis in populations screening of cognitive impairment.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brookmeyer, R.; Johnson, E.; Ziegler-Graham, K.; Arrighi, H.M. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement. 2007, 3, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Zandi, T. Relationship between subjective memory complaints, objective memory performance, and depression among older adults. Am. J. Alzheimers Dis. 2004, 19, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Youn, J.C.; Kim, K.W.; Lee, D.Y.; Jhoo, J.H.; Lee, S.B.; Park, J.H.; Woo, J.I. Development of the subjective memory complaints questionnaire. Dement. Geriatr. Cogn. Disord. 2009, 27, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Benito-León, J.; Mitchell, A.J.; Vega, S.; Bermejo-Pareja, F. A population-based study of cognitive function in older people with subjective memory complaints. J. Alzheimers Dis. 2010, 22, 159–170. [Google Scholar] [CrossRef]
- Genziani, M.; Stewart, R.; Béjot, Y.; Amieva, H.; Artero, S.; Ritchie, K. Subjective memory impairment, objective cognitive functioning and social activity in French older people: Findings from the Three Cities study. Geriatr. Gerontol. Int. 2013, 13, 139–145. [Google Scholar] [CrossRef]
- Vaskivuo, L.; Hokkanen, L.; Hänninen, T.; Antikainen, R.; Bäckman, L.; Laatikainen, T.; Ngandu, T. Associations between Prospective and Retrospective Subjective Memory Complaints and Neuropsychological Performance in Older Adults: The Finger Study. J. Int. Neuropsychol. Soc. 2018, 24, 1099–1109. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Beaumont, H.; Ferguson, D.; Yadegarfar, M.; Stubbs, B. Risk of dementia and mild cognitive impairment in older people with subjective memory complaints: Meta--analysis. Acta Psychiatr. Scand. 2014, 130, 439–451. [Google Scholar] [CrossRef]
- Geerlings, M.I.; Jonker, C.; Bouter, L.M.; Adèr, H.J.; Schmand, B. Association between memory complaints and incident Alzheimer’s disease in elderly people with normal baseline cognition. Am. J. Psychiatry 1999, 156, 531–537. [Google Scholar]
- Jorm, A.F.; Christensen, H.; Korten, A.E.; Jacomb, P.A.; Henderson, A.S. Memory complaints as a precursor of memory impairment in older people: A longitudinal analysis over 7–8 years. Psychol. Med. 2001, 31, 441–449. [Google Scholar] [CrossRef]
- Wang, L.; Van Belle, G.; Crane, P.K.; Kukull, W.A.; Bowen, J.D.; McCormick, W.C.; Larson, E.B. Subjective memory deterioration and future dementia in people aged 65 and older. J. Am. Geriatr. Soc. 2004, 52, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Waldorff, F.B.; Siersma, V.; Vogel, A.; Waldemar, G. Subjective memory complaints in general practice predicts future dementia: A 4-year follow-up study. Int. J. Geriatr. Psychiatry 2012, 27, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Amieva, H.; Letenneur, L.; Dartigues, J.F.; Rouch-Leroyer, I.; Sourgen, C.; D’Alchée-Birée, F.; Fabrigoule, C. Annual rate and predictors of conversion to dementia in subjects presenting mild cognitive impairment criteria defined according to a population-based study. Dement. Geriatr. Cogn. Disord. 2004, 18, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Devier, D.J.; Villemarette-Pittman, N.; Brown, P.; Pelton, G.; Stern, Y.; Sano, M.; Devanand, D.P. Predictive utility of type and duration of symptoms at initial presentation in patients with mild cognitive impairment. Dement. Geriatr. Cogn. Disord. 2010, 30, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.; Guerreiro, M.; Faria, C.; Maroco, J.; Schmand, B.A.; Mendonça, A.D. Significance of subjective memory complaints in the clinical setting. J. Geriatr. Psychiatry Neurol. 2014, 27, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Mol, M.E.; Van Boxtel, M.P.; Willems, D.; Jolles, J. Do subjective memory complaints predict cognitive dysfunction over time? A six-year follow-up of the Maastricht Aging Study. Int. J. Geriatr. Psychiatry J. Child Psychol. Psychiatry 2006, 21, 432–441. [Google Scholar] [CrossRef]
- Reid, L.M.; MacLullich, A.M. Subjective memory complaints and cognitive impairment in older people. Dement. Geriatr. Cogn. Disord. 2006, 22, 471–485. [Google Scholar] [CrossRef]
- Dourlen, P.; Kilinc, D.; Malmanche, N.; Chapuis, J.; Lambert, J.C. The new genetic landscape of Alzheimer’s disease: From amyloid cascade to genetically driven synaptic failure hypothesis? Acta Neuropathol. 2019, 138, 221–226. [Google Scholar] [CrossRef]
- Yegambaram, M.; Manivannan, B.; Beach, G.T.; Halden, U.R. Role of environmental contaminants in the etiology of Alzheimer’s disease: A review. Curr. Alzheimer Res. 2015, 12, 116–146. [Google Scholar] [CrossRef]
- Eid, A.; Mhatre, I.; Richardson, J.R. Gene-environment interactions in Alzheimer’s disease: A potential path to precision medicine. Clin. Pharm. 2019, 199, 173–187. [Google Scholar] [CrossRef]
- Paglia, G.; D’Alessandro, A.; Rolfsson, Ó.; Sigurjónsson, Ó.E.; Bordbar, A.; Palsson, S.; Palsson, B.O. Biomarkers defining the metabolic age of red blood cells during cold storage. Blood 2016, 128, 43–50. [Google Scholar] [CrossRef] [PubMed]
- WHO 2015 Human Biomonitoring: Facts and Figures. Available online: http://www.euro.who.int/__data/assets/pdf_file/0020/276311/Human–biomonitoring-facts-figures-en.pdf (accessed on 1 September 2018).
- Hartmann, S.; Ledur Kist, T.B. A review of biomarkers of Alzheimer’s disease in noninvasive samples. Biomark. Med. 2018, 12, 677–690. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, M.; Hafner, D.; Lombeck, I.; Ohnesorge, F.K. Monitoring of cadmium, copper, lead and zinc status in young-children using toenails—Comparison with scalp hair. Sci. Total Environ. 1991, 103, 199–207. [Google Scholar] [CrossRef]
- Savarino, L.; Granchi, D.; Ciapetti, G.; Cenni, E.; Ravaglia, G.; Forti, P. Serum concentrations of zinc and selenium in elderly people: Results in healthy nonagenarians/ centenarians. Exp. Gerontol. 2001, 36, 327–339. [Google Scholar] [CrossRef]
- Hao, Z.; Li, Y.; Liu, Y.; Li, H.; Wang, W.; Yu, J. Hair elements and healthy aging: A cross-sectional study in Hainan Island, China. Environ. Geochem. Health 2016, 38, 723–735. [Google Scholar] [CrossRef]
- Ahluwalia, N.; Gordon, M.A.; Handte, G.; Mahlon, M.; Li, N.Q.; Beard, J.L. Iron status and stores decline with age in Lewis rats. J. Nutr. 2000, 130, 2378–2383. [Google Scholar] [CrossRef]
- Pike, J.; Chandra, R.K. Effect of vitamin and traceelement supplementation on immune indexes in healthy elderly. Int. J. Vitam. Nutr. Res. 1995, 65, 117–121. [Google Scholar]
- Schmidt, K. Vitamins, minerals, and trace-element in elderly people. Zentralblatt für Hygiene und Umweltmedizin 1991, 191, 327–332. [Google Scholar]
- Selim, A.J.; Fincke, G.; Berlowitz, D.R.; Miller, D.R.; Qian, S.X.; Lee, A. Comprehensive health status assessment of centenarians: Results from the 1999 large health survey of veteran enrollees. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 515–519. [Google Scholar] [CrossRef][Green Version]
- Andersen-Ranberg, K.; Vasegaard, L.; Jeune, B. Dementia is not inevitable: A population-based study of Danish centenarians. J. Gerontol. B Psychol. Sci. Soc. Sci. 2001, 56, 152–159. [Google Scholar] [CrossRef]
- Pereira, M.E.; Lillebø, A.I.; Pato, P.; Válega, M.; Coelho, J.P.; Lopes, C. Mercury pollution in Ria de Aveiro (Portugal): A review of the system assessment. Environ. Monit. Assess. 2009, 155, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Cachada, A.; Pereira, M.E.; Ferreira da Silva, E.; Duarte, A.C. Sources of potentially toxic elements and organic pollutants in an urban area subjected to an industrial impact. Environ. Monit. Assess. 2012, 184, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Inácio, M.; Neves, O.; Pereira, V.; da Silva, E.F. Levels of selected potential harmful elements (PHEs) in soils and vegetables used in diet of the population living in the surroundings of the Estarreja Chemical Complex (Portugal). J. Appl. Geochem. 2014, 44, 38–44. [Google Scholar] [CrossRef]
- Patinha, C.; Reis, A.P.; Dias, A.C.; Abduljelil, A.A.; Noack, Y.; Robert, S.; Cave, M.; da Silva, E.F. The mobility and human oral bioaccessibility of Zn and Pb in urban dusts of Estarreja (N Portugal). Environ. Geochem. Health 2015, 37, 115–131. [Google Scholar] [CrossRef] [PubMed]
- Leitão, T.B.E. Metodologia Para A Reabilitação De Aquíferos Poluídos. Ph.D. Thesis, Faculdade de Ciências da Universidade de Lisboa, Lisboa, Portugal, 1996. [Google Scholar]
- Van der Weijden, C.; Pacheco, F.A.L. Hydrogeochemistry in the Vouga River basin (central Portugal): Pollution and chemical weathering. J. Appl. Geochem. 2006, 21, 580–613. [Google Scholar] [CrossRef]
- Ordens, C.M. Estudo Da Contaminacão Do Aquífero Superior Na Região De Estarreja. Master’s Thesis, Coimbra University, Coimbra, Portugal, 2007. Unpublished. Available online: http://www.lneg.pt/download/3268/ carlos_ordens.pdf (accessed on 11 March 2018).
- Reis, A.P.; Costa, S.; Santos, I.; Patinha, C.; Noack, Y.; Wragg, J. Investigating relationships between biomarkers of exposure and environmental copper and manganese levels in house dusts from a Portuguese industrial city. Environ. Geochem. Health 2015, 37, 725–744. [Google Scholar] [CrossRef]
- Plumejeaud, S.; Reis, A.P.; Tassistro, V.; Patinha, C.; Noack, Y.; Orsière, T. Potentially harmful elements in house dust from Estarreja, Portugal: Characterization and genotoxicity of the bioaccessible fraction. Environ. Geochem. Health 2018, 40, 127–144. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; Marinho-Reis, A.P.; Almeida, A.; Ordens, C.M.; Silva, M.M.; Freitas, S.; Simões, M.; Dinis, P.; Moreira, P.; de Melo, T.C.; et al. Human predisposition to cognitive impairment and its relation with environmental exposure to potentially toxic elements. Environ. Geochem. Health 2018, 40, 1767–1784. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; Marinho-Reis, A.P.; Almeida, A.; Freitas, S.; Simões, M.R.; Diniz, M.L.; da Silva, E.F.; Moreira, P.I. Fingernail trace element content in environmentally exposed individuals and its influence on their cognitive status in ageing. Expo. Health 2018, 11, 181–194. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; Ordens, C.M.; de Melo, M.T.C.; Inácio, M.; Almeida, A.; Pinto, E.; da Silva, E.A.F. An Inter-disciplinary Approach to Evaluate Human Health Risks Due to Long-Term Exposure to Contaminated Groundwater Near a Chemical Complex. Expo. Health 2019, 1–16. [Google Scholar] [CrossRef]
- Cabral-Pinto, M.M.S.; Inácio, M.; Neves, O.; Almeida, A.A.; Pinto, E.; Oliveiros, B.; Ferreira da Silva, E.A.F. Human Health Risk Assessment Due to Agricultural Activities and Crop Consumption in the Surroundings of an Industrial Area. Expo. Health 2019, 1–12. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1983, 17, 37–49. [Google Scholar] [CrossRef]
- Pocinho, M.T.S.; Farate, C.; Dias, C.A.; Lee, T.T.; Yesavage, J.A. Clinical and psychometric validation of the Geriatric Depression Scale (GDS) for Portuguese Elders. Clin. Gerontol. 2009, 32, 223–236. [Google Scholar] [CrossRef]
- Simões, M.R.; Prieto, G.; Pinho, M.S.; Firmino, H. Geriatric Depression Scale (GDS-30). In Escalas e Testes na Demência (3a. edição) (Scales and tests in dementia), 3rd ed.; Simões, M.R., Isabel Santana e Grupo de Estudos de Envelhecimento Cerebral e Demência, Eds.; Novartis: Lisboa, Portugal, 2015; pp. 128–133. [Google Scholar]
- Kuiper, N.; Rowell, C.; Nriagu, J.; Shomar, B. What do the trace metal contents of urine and toenail samples from Qatar’s farm workers bioindicate? Environ. Res. 2014, 131, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.; Folstein, S.; McHugh, P. Mini-mental state: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Freitas, S.; Simões, M.R.; Alves, L.; Santana, I. Montreal Cognitive Assessment (MoCA): Validation study for mild cognitive impairment and Alzheimer’s disease. Alzheimer Dis. Assoc. Disord. 2013, 27, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Freitas, S.; Simões, M.R.; Alves, L.; Santana, I. The relevance of sociodemographic and health variables on MMSE normative data. Appl. Neuropsychol. Adult 2015, 22, 311–319. [Google Scholar] [CrossRef]
- Kaup, A.R.; Nettiksimmons, J.; LeBlanc, E.S.; Yaffe, K. Memory complaints and risk of cognitive impairment after nearly 2 decades among older women. Neurology 2015, 85, 1852–1858. [Google Scholar] [CrossRef]
- O’Bryant, S.E.; Humphreys, J.D.; Smith, G.E.; Ivnik, R.J.; GraffRadford, N.R.; Petersen, R.C.; Lucas, J.A. Detecting dementia with the Mini-Mental State Examination (MMSE) in highly educated individuals. Arch. Neurol. 2008, 65, 963–967. [Google Scholar] [CrossRef]
- Bass, D.A.; Hickok, D.; Quig, D.; Urek, K. Trace element analysis in hair: Factors determining accuracy, precision, and reliability. Altern. Med. Rev. 2001, 6, 472–481. [Google Scholar]
- Buat-Menard, P.; Chesselet, R. Variable influence of the atmospheric flux on the trace metal chemistry of oceanic suspended matter. Earth Planet. Sci. Lett. 1979, 42, 399–411. [Google Scholar] [CrossRef]
- Islam, S.; Ahmed, K.; Masunaga, S. Potential ecological risk of hazardous elements in different land-use urban soils of Bangladesh. Sci. Total Environ. 2015, 512, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.N.; Park, J.H. Immobilization and reduction of bioavailability of lead in shooting range soil through hydrothermal treatment. J. Environ. Manag. 2017, 191, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Cabral Pinto, M.M.S.; Silva, M.M.; da Silva, E.A.F.; Dinis, P.A.; Rocha, F. Transfer processes of potentially toxic elements (PTE) from rocks to soils and the origin of PTE in soils: A case study on the island of Santiago (Cape Verde). J. Geochem. Explor. 2017, 183, 140–151. [Google Scholar] [CrossRef]
- Sutherland, R.A. Depth variation in copper, lead and zinc concentrations and mass enrichment ratios in soils of an urban watershed. J. Environ. Qual. 2000, 29, 1414–1422. [Google Scholar] [CrossRef]
- Anderson, T.M.; Sachdev, P.S.; Brodaty, H.; Trollor, J.; Andrews, G. Effects of sociodemographic and health variables on MiniMental State Exam scores in older Australians. Am. J. Geriatr. Psychiatry 2007, 15, 467–476. [Google Scholar] [CrossRef]
- Bravo, G.; Hébert, R. Age and education specific reference values for the Mini-Mental and Modified Mini-Mental State Examination derived from a non-demented elderly population. Int. J. Geriatr. Psychiatry 1997, 12, 1008–1018. [Google Scholar] [CrossRef]
- Gallacher, J.E.; Elwood, P.C.; Hopkinson, C.; Rabbitt, P.M.; Stollery, B.T.; Sweetnam, P.M.; Huppert, F.A. Cognitive function in the Caerphilly study: Associations with age, social class, education and mood. Eur. J. Epidemiol. 1999, 15, 161–169. [Google Scholar] [CrossRef]
- Matallana, D.; Santacruz, C.; Cano, C.; Reyes, P.; Samper-Ternent, R.; Markides, K.S.; Reyes-Ortiz, C.A. The relationship between educational level and Mini-Mental State Examination domains among older Mexican Americans. J. Geriatr. Psychiatry Neurol. 2011, 24, 9–18. [Google Scholar] [CrossRef]
- Moraes, C.; Pinto, J.A.; Lopes, M.A.; Litvoc, J.; Bottino, C.M. Impact of sociodemographic and health variables on Mini-Mental State Examination in a community-based sample of older people. Eur. Arch. Psychiatry Clin. Neurosci. 2010, 260, 535–542. [Google Scholar] [CrossRef]
- Han, C.; Jo, S.A.; Jo, I.; Kim, E.; Park, M.H.; Kang, Y. An adaptation of the Korean Mini-Mental State Examination (K-MMSE) in elderly Koreans: Demographic influence and population-based norms (the AGE Study). Arch Gerontol. Geriatr. 2008, 47, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Mathuranath, P.S.; Cherian, J.P.; Mathew, R.; George, A.; Alexander, A.; Sarma, S.P. Mini Mental State Examination and the Addenbrooke’s Cognitive Examination: Effect of education and norms for a multicultural population. Neurol. India 2007, 55, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Measso, G.; Cavarzeran, F.; Zappalà, G.; Lebowitz, B.D.; Crook, T.H.; Pirozzollo, F.J.; Grigoletto, F. The Mini-Mental State Examination: Normative study of an Italian random sample. Dev. Neuropsychol. 1993, 9, 77–85. [Google Scholar] [CrossRef]
- Thiele, S.; Mensink, G.B.; Beitz, R. Determinants of diet quality. Public Health Nutr. 2004, 7, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Powdthavee, N. Does more education lead to better health habits? Evidence from the school reforms in Australia. Soc. Sci. Med. 2015, 127, 83–91. [Google Scholar] [PubMed]
- Mías, C.D.; Sassi, M.; Masih, M.E.; Querejeta, A.; Krawchik, R. Deterioro cognitivo leve: Estúdio de prevalência y factores sociodemográficos en la ciudad de Córdoba, Argentina [Mild cognitive impairment: A prevalence and sociodemographic factors study in the city of Córdoba, Argentina]. Rev. Neurol. 2007, 44, 733–738. [Google Scholar] [PubMed]
- Ribeiro, P.C.; Oliveira, B.H.; Cupertino, A.P.; Neri, A.L.; Yassuda, M.S. Desempenho de idosos na bateria cognitiva CERAD: Relações com variáveis sociodemográficas e saúde percebida [Performance of the elderly in the CERAD Cognitive Battery: Relations with socio-demographic variables and perceived health]. Psicologia, Reflexão e Crítica 2010, 23, 102–109. [Google Scholar] [CrossRef]
- Scazufca, M.; Almeida, O.P.; Vallada, H.P.; Tasse, W.A.; Menezes, P.R. Limitations of the Mini-Mental State Examination for screening dementia in a community with low socioeconomic status. Eur. Arch. Psychiatry Clin. Neurosci. 2009, 259, 8–15. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, W.; Ye, T.; Lin, X.; Zhang, J. Alzheimer’s Disease Neuroimaging Initiative. Sex Difference in the Association of APOE4 with Memory Decline in Mild Cognitive Impairment. J. Alzheimer’s Dis. 2019, 69, 1161–1169. [Google Scholar] [CrossRef]
- Iwata, A.; Iwatsubo, T.; Ihara, R.; Suzuki, K.; Matsuyama, Y.; Tomita, N.; Ikeuchi, T. Effects of sex, educational background, and chronic kidney disease grading on longitudinal cognitive and functional decline in patients in the Japanese Alzheimer’s Disease Neuroimaging Initiative study. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2018, 4, 765–774. [Google Scholar] [CrossRef]
- Gorelick, P.B.; Scuteri, A.; Black, S.E.; DeCarli, C.; Greenberg, S.M.; Iadecola, C.; Petersen, R.C. Vascular contributions to cognitive impairment and dementia: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011, 42, 2672–2713. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Mercury Study Report to Congress; Office of Air Quality Planning and Standards and Office of Research and Development: Washington, DC, USA; USEPA: Washington, DC, USA, 1997. [Google Scholar]
- Skalny, A.V.; Skalnaya, M.G.; Tinkov, A.A.; Serebryansky, E.P.; Demidov, V.A.; Lobanova, Y.N.; Skalnaya, O.A. Hair concentration of essential trace elements in adult non-exposed Russian population. Environ. Monit. Assess. 2015, 187, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Mikulewicz, M.; Chojnacka, K.; Gedrange, T.; Górecki, H. Reference values of elements in human hair: A systematic review. Environ. Toxicol. Pharmacol. 2013, 36, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Rodushkin, I.; Axelsson, M.D. Application of double focusing sector field ICP-MS for multielemental characterization of human hair and nails. Part II. A study of the inhabitants of northern Sweden. Sci. Total Environ. 2000, 262, 21–36. [Google Scholar] [CrossRef]
- Kazi, T.G.; Memon, A.R.; Afridi, H.I.; Jamali, M.K.; Arain, M.B.; Jalbani, N.; Sarfraz, R.A. Determination of cadmium in whole blood and scalp hair samples of Pakistani male lung cancer patients by electrothermal atomic absorption spectrometer. Sci. Total Environ. 2008, 389, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Brulle, R.J.; Pellow, D.N. Environmental justice: Human health and environmental inequalities. Annu. Rev. Publ. Health. 2006, 27, 103–124. [Google Scholar] [CrossRef] [PubMed]
- Ginter, E.; Simko, V. Women live longer than men. Bratisl. Lek. Listy 2013, 114, 45–49. [Google Scholar] [CrossRef]
- Luy, M.; Gast, K. Do women live longer or do men die earlier? Reflections on the causes of sex differences in life expectancy. Gerontology 2014, 60, 143–153. [Google Scholar] [CrossRef]
- Serdar, M.A.; Akin, B.S.; Razi, C.; Akin, O.; Tokgoz, S.; Kenar, L.; Aykut, O. The correlation between smoking status of family members and concentrations of toxic trace elements in the hair of children. Biol. Trace Elem. Res. 2012, 148, 11–17. [Google Scholar] [CrossRef]
- Zatta, P.; Lucchini, R.; Van Rensburg, S.J.; Taylor, A. The role of metals in neurodegenerative processes: Aluminum, manganese, and zinc. Brain Res. Bull. 2003, 62, 15–28. [Google Scholar] [CrossRef]
- Loef, M.; Von Stillfried, N.; Walach, H. Zinc diet and Alzheimer’s disease: A systematic review. Nutr. Neurosci. 2012, 15, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Prodan, C.I.; Holland, N.R. CNS demyelination from zinc toxicity? Neurology 2000, 54, 1705–1706. [Google Scholar] [CrossRef] [PubMed]
- Genuis, S.J.; Kelln, K.L. Toxicant exposure and bioaccumulation: A common and potentially reversible cause of cognitive dysfunction and dementia. Behav. Neurol. 2015, 2015, 620143. [Google Scholar] [CrossRef] [PubMed]
- Zahir, F.; Rizwi, S.J.; Haq, S.K.; Khan, R.H. Low dose mercury toxicity and human health. Environ. Toxicol. Chem. 2005, 20, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Waldron, I.; Johnston, S. Why do women live longer than men? J. Hum. Stresss 1976, 2, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, T.; Ohmori, H.; Hashimoto, K.; Tsuruta, K.; Ekino, S. Expansion of methylmercury poisoning outside minamata: An epidemiological study on chronic methylmercury poisoninig outside of Minamata. Environ. Res. 1995, 70, 47–50. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, W.; Liu, X.; Zhang, C.; Wang, P.; Zhao, X. Circulatory Levels of Toxic Metals (Aluminum, Cadmium, Mercury, Lead) in Patients with Alzheimer’s Disease: A Quantitative Meta-Analysis and Systematic Review. J. Alzheimers Dis. 2018, 62, 361–372. [Google Scholar] [CrossRef]
- Meleleo, D.; Notarachille, G.; Mangini, V.; Arnesano, F. Concentration-dependent effects of mercury and lead on Aβ42: Possible implications for Alzheimer’s disease. Eur. Biophys. J. 2019, 48, 173–187. [Google Scholar] [CrossRef]
- Food and Agriculture Organization World Health of the United Nations. In Proceedings of the Joint FAO/WHO Expert Committee on Food Additives Sixty-Third Meeting, Geneva, Switzerland, 8–17 June 2004. Available online: http://www.fao.org/3/a-at878e.pdf (accessed on 1 April 2019).
- JECFA. Evaluation of Certain Contaminants in Food. In The Seventy-Second Report of Joint FAO/WHO Expert Committee on Food Additives; WHO technical report series; JECFA: Geneva, Switzerland, 2011; Volume 959, pp. 1–115. [Google Scholar]
- WHO. Environmental Health Criteria 101—Methylmercury; World Health Organization: Geneva, Switzerland, 1990. [Google Scholar]
- Agusa, T.; Kunito, T.; Sudaryanto, A.; Monirith, I.; Kan-Atireklap, S.; Iwata, H.; Tanabe, S. Exposure assessment for trace elements from consumption of marine fish in Southeast Asia. Environ. Pollut. 2007, 145, 766–777. [Google Scholar] [CrossRef]
- Freire, C.; Ramos, R.; Lopez-Espinosa, M.J.; Díez, S.; Vioque, J.; Ballester, F.; Fernández, M.F. Hair mercury levels, fish consumption, and cognitive development in preschool children from Granada, Spain. Environ. Res. 2010, 110, 96–104. [Google Scholar] [CrossRef]
- Díez, S.; Esbrí, J.M.; Tobias, A.; Higueras, P.; Martínez-Coronado, A. Determinants of exposure to mercury in hair from inhabitants of the largest mercury mine in the world. Chemosphere 2011, 84, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Dolbec, J.; Mergler, D.; Larribe, F.; Roulet, M.; Lebel, J.; Lucotte, M. Sequential analysis of hair mercury levels in relation to fish diet of an Amazonian population, Brazil. Sci. Total Environ. 2001, 271, 87–97. [Google Scholar] [CrossRef]
- Koseoglu, E.; Koseoglu, R.; Kendirci, M.; Saraymen, R.; Saraymen, B. Trace metal concentrations in hair and nails from Alzheimer’s disease patients: Relations with clinical severity. J. Trace Elem. Med. Biol. 2017, 39, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Vance, D.E.; Ehmann, W.D.; Markesbery, W.R. Trace element imbalances in hair and nails of Alzheimer’s disease patients. Neurotoxicology 1998, 9, 197–208. [Google Scholar]
- Koc, E.R.; Ilhan, A.; Ayturk, Z.; Acar, B.; Gürler, M.; Altuntaş, A.; Karapirli, M.; Bodur, A.S. A comparison of hair and serum trace elements in patients with Alzheimer disease and healthy. Turk. J. Med. Sci. 2015, 45, 1034–1039. [Google Scholar] [CrossRef]
- Paglia, G.; Miedico, O.; Cristofano, A.; Vitale, M.; Angiolillo, A.; Chiaravalle, A.E.; DiCostanzo, A. Distinctive pattern of serum elements during the progression of Alzheimer’s disease. Sci. Rep. 2016, 6, 22679. [Google Scholar] [CrossRef]
- Li, Y.H.; Zou, X.Y.; Lv, J.M.; Yang, L.S.; Li, H.R.; Wang, W.Y. Trace elements in fingernails of healthy Chinese centenarians. Biol. Trace Elem. Res. 2012, 145, 158–165. [Google Scholar] [CrossRef]
- Inácio, M.M.S. Dados Geoquímicos De Base De Solos De Portugal Continental, Utilizando Amostragem De Baixa Densidade. Ph.D. Thesis, University of Aveiro, Aveiro, Portugal, 2004. Available online: http://hdl.handle.net/10773/18827 (accessed on 20 February 2019).
- Canadian Guideline: Minister of the Environment (Canada). Soil, Ground Water and Sediment Standards for Use under Part XV.1 of the Environmental Protection Act. Available online: http://www.mah.gov.on.ca/Asset Facto ry.aspx?did=8993 (accessed on 15 March 2018).
- Thiombane, M.; Di Bonito, M.; Albanese, S.; Zuzolo, D.; Lima, A.; De Vivo, B. Geogenic versus anthropogenic behaviour and geochemical footprint of Al, Na, K and P in the Campania region (Southern Italy) soils through compositional data analysis and enrichment factor. Geoderma 2019, 335, 12–26. [Google Scholar] [CrossRef]
- Reimann, C.; Garrett, R.G. Geochemical background—concept and reality. Sci. Total Environ. 2005, 350, 12–27. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; da Silva, E.F.; Silva, M.; Melo-Gonçalves, P.; Candeias, C. Environmental risk assessment based on high-resolution spatial maps of potentially toxic elements sampled on stream sediments of Santiago, Cape Verde. Geosciences 2014, 4, 297–315. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; da Silva, E.F.; Silva, M.; Melo-Gonçalves, P. Heavy metals of Santiago Island (Cape Verde) top soils: Estimated background value maps and environmental risk assessment. J. Afr. Earth Sci. 2015, 101, 162–176. [Google Scholar] [CrossRef]
- Reis, A.T. Impact of Mercury on Human Health: Aveiro, A Study Case. Master’s Thesis, University of Aveiro, Aveiro, Portugal, 2008; p. 112. [Google Scholar]
- Dutch Soil Remediation Circular. Esdat Environmental Database Managment Software. 2009. Available online: ww.esdat.net (accessed on 10 October 2019).
- Ministry of Health of the People’s Republic of China. The maximum levels of contaminants in foods (GB 2762-2005). Maximum Levels of Contaminants in Foods_Beijing_China—Peoples Republic of_12-11-2014.pdf. 2005. Available online: https://apps.fas.usda.gov/newgainapi/api/report/downloadreportbyfilename?filename=Maximum%20Levels%20of%20Contaminants%20in%20Foods%20_Beijing_China%20-%20Peoples%20Republic%20of_12-11-2014.pdf (accessed on 11 April 2019).
- Jornal Oficial das Comunidades Europeias, L77/1 de 16/3/2001. p. 1. Available online: https://www.europarl.europa.eu/RegData/PDF/r1049_pt.pdf (accessed on 6 March 2019).
| HS | SMC | MCI | DEM | ||
|---|---|---|---|---|---|
| n = 10 | n = 14 | n = 16 | n = 39 | ||
| Age (mean ± SD) | 74.0 ± 9.6 | 73.3 ± 7.2 | 78.4 ± 7.8 | 81.7 ± 9.0 | |
| Gender (n; %) | Male | 2; 20% | 2; 14% | 6; 37% | 3; 8% |
| Female | 8; 80% | 12; 86% | 10; 63% | 36; 92% | |
| Level of Education (mean ± SD) | 7.40 ± 5.10 ** | 4.43 ± 2.34 ** | 2.88 ± 1.54 | 1.91 ± 3.36 ** | |
| Time of residence (mean ± SD) | 53.20 ± 17.76 * | 58.36 ± 27.80 | 58.69 ± 26.64 | 70.82 ± 26.08 * | |
| Profession | Housewife | 2; 20% | 2; 14% | 4; 25% | 8; 21% |
| Agriculture/Fishery | 2; 20% | 3; 21 % | 4; 25% | 16; 41% | |
| Industry/Construction | - | 5; 36% | 4; 25% | 5; 13% | |
| Commerce/Services | 6; 60% | 4; 29 % | 4; 25% | 10; 26% | |
| Medical History (n, %) | |||||
| Diabetes | - | - | 2; 13% | 6; 15% | |
| Dyslipidemia | 1; 10% | 3; 21% | - | 1; 3% | |
| Cardiovascular | 3; 30% | 5; 36% | 8; 50% | 9; 25% | |
| Respiratory | 3; 30% | 1; 7% | 1; 6% | 2; 5% | |
| Other | 2; 20% | 3; 21% | 2; 13% | 5; 13% | |
| Lifestyle factors (n; %) | |||||
| Supplements | - | 5; 36% | 3; 19% | 7; 18% | |
| Homegrown food | 7; 70% | 11; 79% | 13; 81% | 26; 67% | |
| Drinking Water | Bottled | 6; 60% | 11; 79% | 12; 75% | 22; 56% |
| Bottled & tap water | 2; 20% | 1; 7% | - | 1; 3% | |
| Tap water | 2; 20% | 1; 7% | 4; 25% | 11; 28% | |
| Well or borehole | - | 1; 7% | - | - |
| Element | HS | SMC | MCI | DEM | Kruskal-Wallis | Pairwise Comparisons (a) (p-Value) | Hair Reference Values * | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| µg/g | n = 10 | n = 14 | n = 16 | n = 39 | H(3) | p-Value | DEM vs. HS | DEM vs. SMC | DEM vs. MCI | HS vs. SMC | HS vs. MCI | SMC vs. MCI | |
| Al | 6.67 ± 10.69 | 4.18 ± 3.36 | 3.80 ± 3.95 | 6.26 ± 8.76 | 1.222 | 0.748 | N/A | N/A | N/A | N/A | N/A | N/A | 2.91–11.63 |
| (0.23–35.96) | (0.16–11.70) | (0.78–14.02) | (0.51–49.08) | ||||||||||
| Mn | 0.19 ± 0.17 | 0.18 ± 0.24 | 0.85±1.38 | 1.39 ± 2.83 | 7.089 | 0.069 | N/A | N/A | N/A | N/A | N/A | N/A | 0.002–0.91 |
| (0.04–0.54) | (0.05–0.87) | (0.03–5.46) | (0.03–16.10) | ||||||||||
| Fe | 16.37 ± 12.24 | 10.79 ± 7.75 | 12.67 ± 9.65 | 18.47 ± 40.24 | 2.266 | 0.519 | N/A | N/A | N/A | N/A | N/A | N/A | 3.66–36.8 |
| (0.59–43.77) | (0.59–27.78) | (3.60–40.47) | (3.41–259.26) | ||||||||||
| Cu | 10.57 ± 10.24 | 10.77 ± 5.82 | 9.85 ± 2.54 | 17.99 ± 36.71 | 1.581 | 0.664 | N/A | N/A | N/A | N/A | N/A | N/A | 7.2–82.7 |
| (1.62–37.10) | (0.56–22.20) | (3.83–13.39) | (2.66–237.40) | ||||||||||
| Zn | 140.29 ± 65.66 | 118.17 ± 58.33 | 151.69 ± 28.64 | 258.10 ± 519.33 | 11.723 | 0.008 ** | n.s. | 0.006 ** | n.s. | n.s. | n.s. | n.s. | 30–327 |
| (10.70–234.86) | (3.99–229.00) | (103.00–200.38) | (63.25–3396.26) | ||||||||||
| Hg | 0.88 ± 0.92 | 1.48 ± 1.40 | 1.63 ± 1.18 | 4.43 ± 13.86 | 17.772 | <0.001 ** | 0.001** | n.s. | n.s. | n.s. | n.s. | n.s. | 0.17–1.19 |
| (0.12–3.24) | (0.11–5.38) | (0.63–5.13) | (0.06–88.46) | ||||||||||
| Pb | 0.29 ± 0.39 | 0.46 ± 0.56 | 0.33 ± 0.42 | 1.02 ± 2.11 | 8.839 | 0.077 | N/A | N/A | N/A | N/A | N/A | N/A | 0.19–1.39 |
| (0.048–1.33) | (0.001–2.20) | (0.03–1.58) | (0.03–12.75) | ||||||||||
| Element | ANCOVA a | Pairwise Comparisons b (p-Value) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| F(3, 68) | p-Value | partial η2 | DEMvsHS | DEMvsSMC | DEMvsMCI | HSvsSMC | HSvsMCI | SMCvsMCI | |
| Al | 0.597 | 0.620 | 0.03 | N/A | N/A | N/A | N/A | N/A | N/A |
| Mn | 1.13 | 0.344 | 0.05 | N/A | N/A | N/A | N/A | N/A | N/A |
| Fe | 0.982 | 0.407 | 0.05 | N/A | N/A | N/A | N/A | N/A | N/A |
| Cu | 0.455 | 0.715 | 0.22 | N/A | N/A | N/A | N/A | N/A | N/A |
| Zn | 2.477 | 0.07 | 0.11 | N/A | N/A | N/A | N/A | N/A | N/A |
| Hg | 4.411 | 0.007 * | 0.18 | 0.005 * | n.s. | n.s. | n.s. | n.s. | n.s. |
| Pb | 2.757 | 0.500 | 0.02 | N/A | N/A | N/A | N/A | N/A | N/A |
| Al | Cu | Hg | Mn | Fe | Pb | Zn | |
|---|---|---|---|---|---|---|---|
| Minimum | 0.31 | 3 | 0.03 | 36 | 0.26 | 6 | 16 |
| Mean | 0.60 | 33 | 1.5 | 120 | 0.66 | 33 | 84 |
| Median | 0.56 | 23 | 0.15 | 110 | 0.62 | 23 | 67 |
| Maximum | 1.15 | 103 | 14 | 255 | 1.15 | 109 | 199 |
| SD | 0.21 | 27 | 4 | 56 | 0.2 | 23 | 44 |
| Cambisoil | 2.14 | 2 | 0.05 | 251 | 2.39 | 20 | 55 |
| Podzol | 0.13 | 12 | 0.05 | 154 | 0.17 | 6 | 4 |
| BG | 1.14 | 7 | 0.05 | 203 | 1.28 | 13 | 30 |
| CEQG | NA | 62 | 0.16 | NA | NA | 45 | 290 |
| % > BG | 0 | 96 | 100 | 12 | 0 | 84 | 96 |
| % > CEQG | NA | 15 | 35 | NA | NA | 23 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabral Pinto, M.M.S.; Marinho-Reis, P.; Almeida, A.; Pinto, E.; Neves, O.; Inácio, M.; Gerardo, B.; Freitas, S.; Simões, M.R.; Dinis, P.A.; et al. Links between Cognitive Status and Trace Element Levels in Hair for an Environmentally Exposed Population: A Case Study in the Surroundings of the Estarreja Industrial Area. Int. J. Environ. Res. Public Health 2019, 16, 4560. https://doi.org/10.3390/ijerph16224560
Cabral Pinto MMS, Marinho-Reis P, Almeida A, Pinto E, Neves O, Inácio M, Gerardo B, Freitas S, Simões MR, Dinis PA, et al. Links between Cognitive Status and Trace Element Levels in Hair for an Environmentally Exposed Population: A Case Study in the Surroundings of the Estarreja Industrial Area. International Journal of Environmental Research and Public Health. 2019; 16(22):4560. https://doi.org/10.3390/ijerph16224560
Chicago/Turabian StyleCabral Pinto, Marina M. S., Paula Marinho-Reis, Agostinho Almeida, Edgar Pinto, Orquídia Neves, Manuela Inácio, Bianca Gerardo, Sandra Freitas, Mário R. Simões, Pedro A. Dinis, and et al. 2019. "Links between Cognitive Status and Trace Element Levels in Hair for an Environmentally Exposed Population: A Case Study in the Surroundings of the Estarreja Industrial Area" International Journal of Environmental Research and Public Health 16, no. 22: 4560. https://doi.org/10.3390/ijerph16224560
APA StyleCabral Pinto, M. M. S., Marinho-Reis, P., Almeida, A., Pinto, E., Neves, O., Inácio, M., Gerardo, B., Freitas, S., Simões, M. R., Dinis, P. A., Diniz, L., Ferreira da Silva, E., & Moreira, P. I. (2019). Links between Cognitive Status and Trace Element Levels in Hair for an Environmentally Exposed Population: A Case Study in the Surroundings of the Estarreja Industrial Area. International Journal of Environmental Research and Public Health, 16(22), 4560. https://doi.org/10.3390/ijerph16224560













