Impact of Fe and Ni Addition on the VFAs’ Generation and Process Stability of Anaerobic Fermentation Containing Cd
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Anaerobic Fermentation Experiment
2.3. Measurements
2.4. Data Analysis
3. Results and Discussion
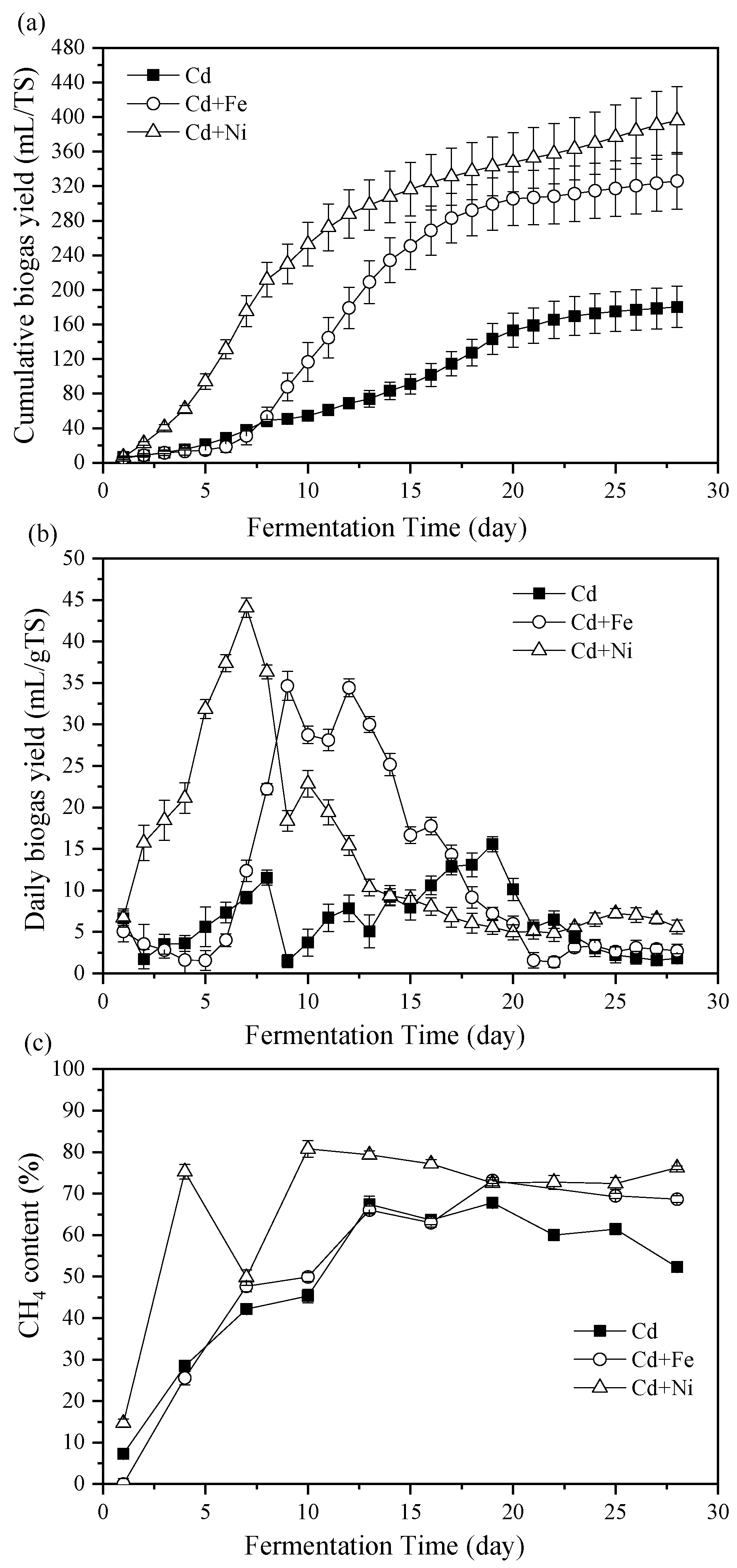
3.1. Biogas Properties
3.1.1. Cumulative and Daily Biogas Yields
3.1.2. CH4 Content
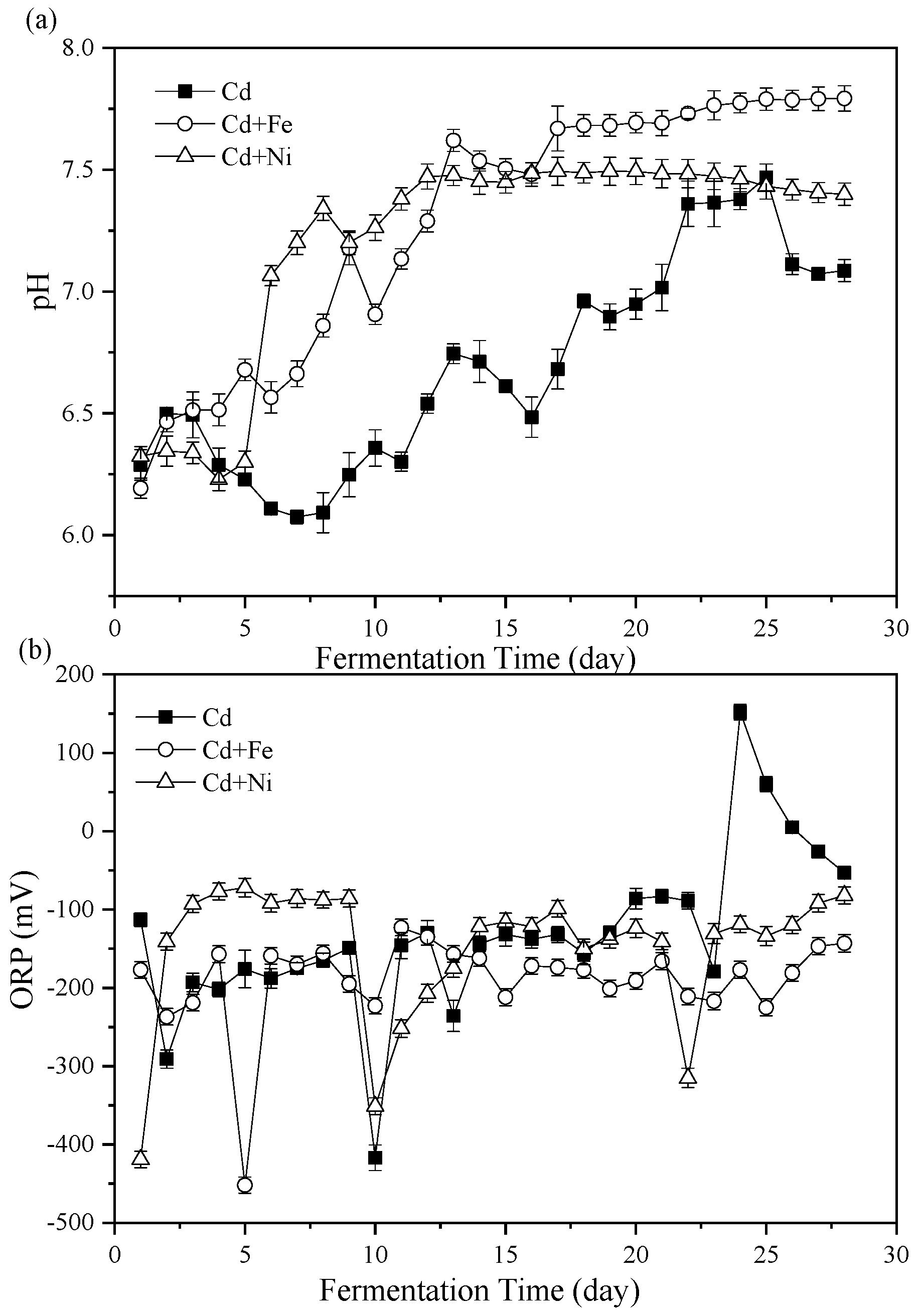
3.2. Process Stability
3.2.1. pH Values
3.2.2. Oxidation Reduction Potential (ORP)
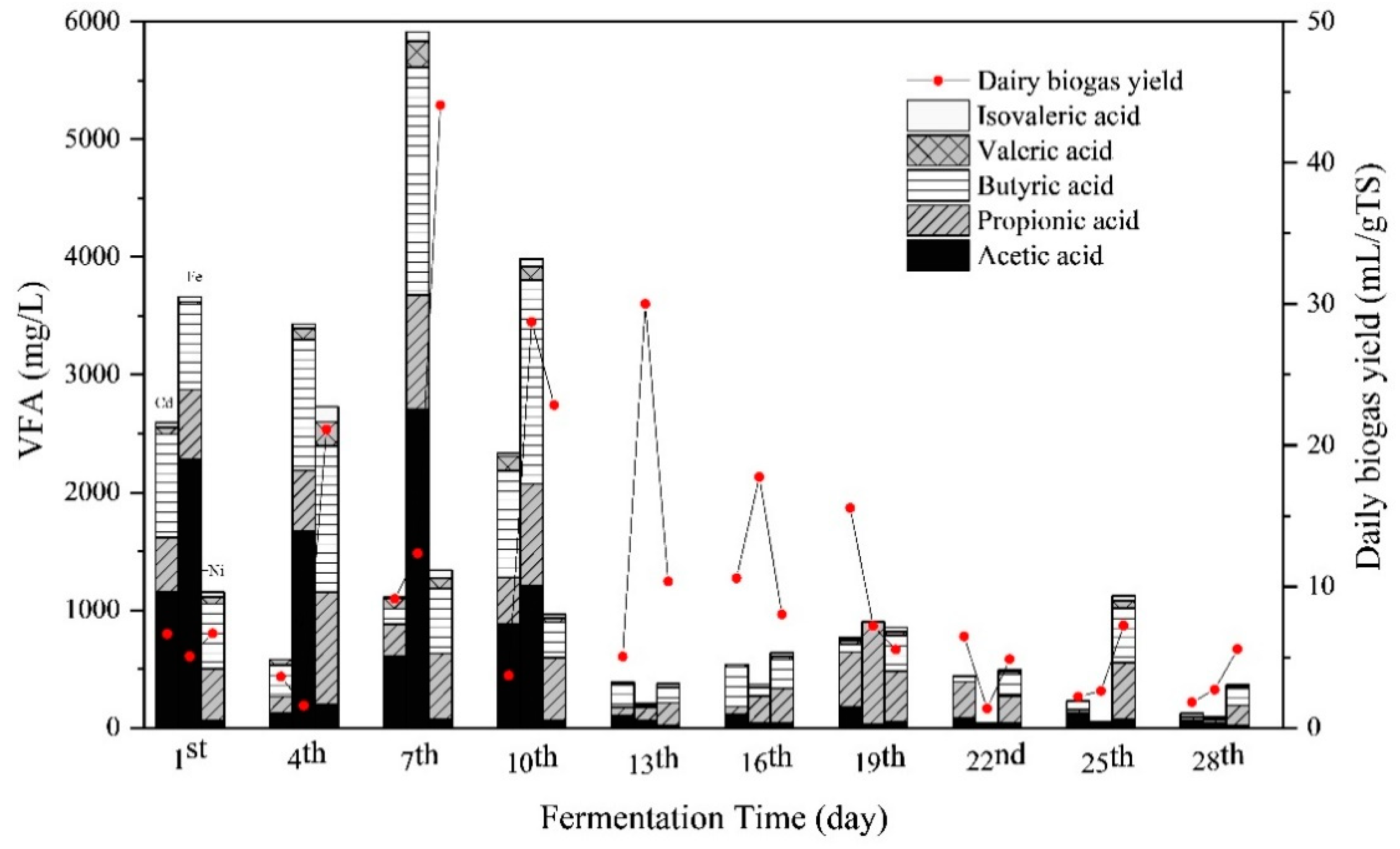
3.3. Substrate Biodegradation
3.3.1. NH4+–N Concentration
3.3.2. Responses of VFA
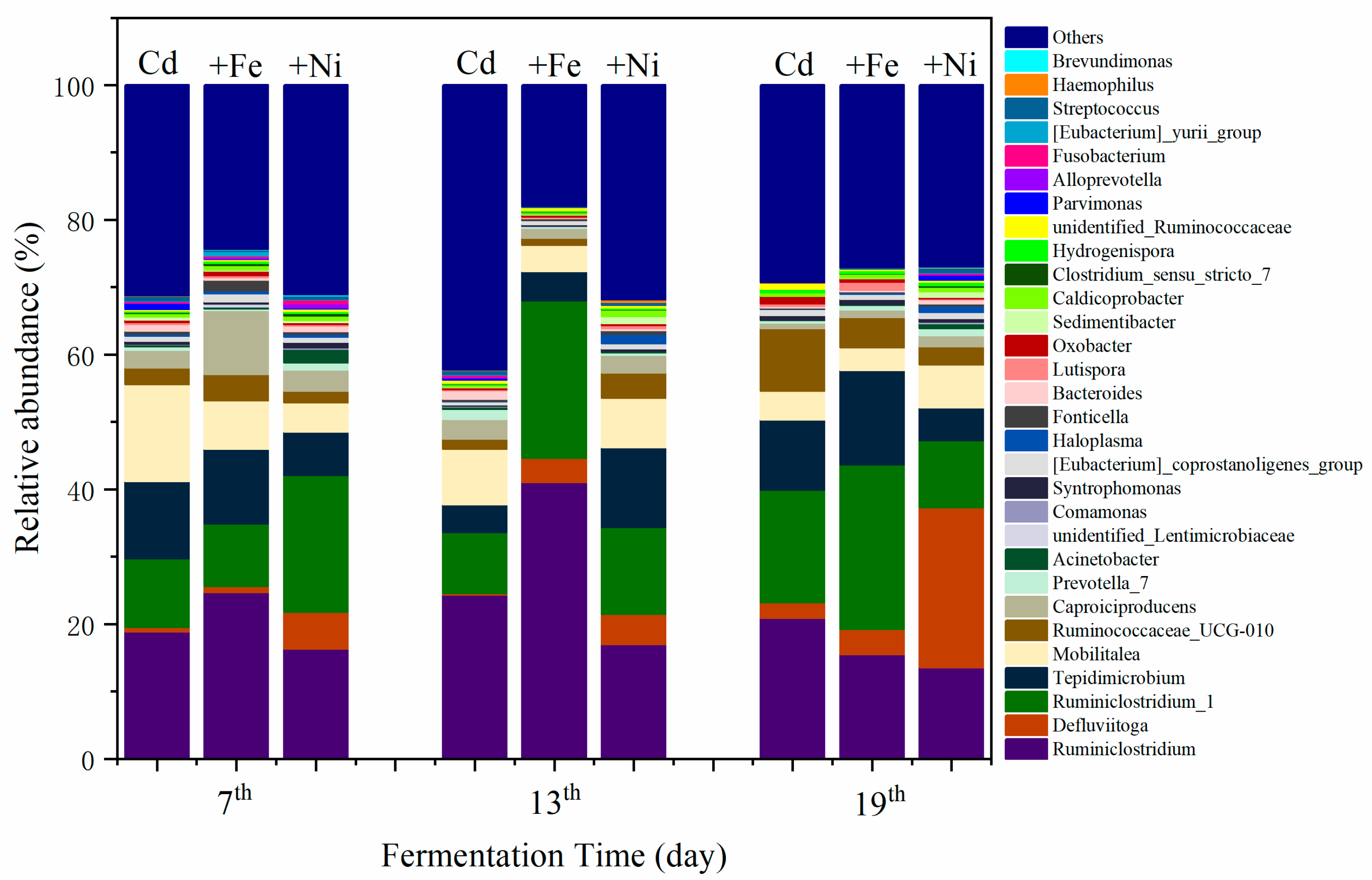
3.4. Microbial Properties
3.4.1. Structure of Bacterial Communities
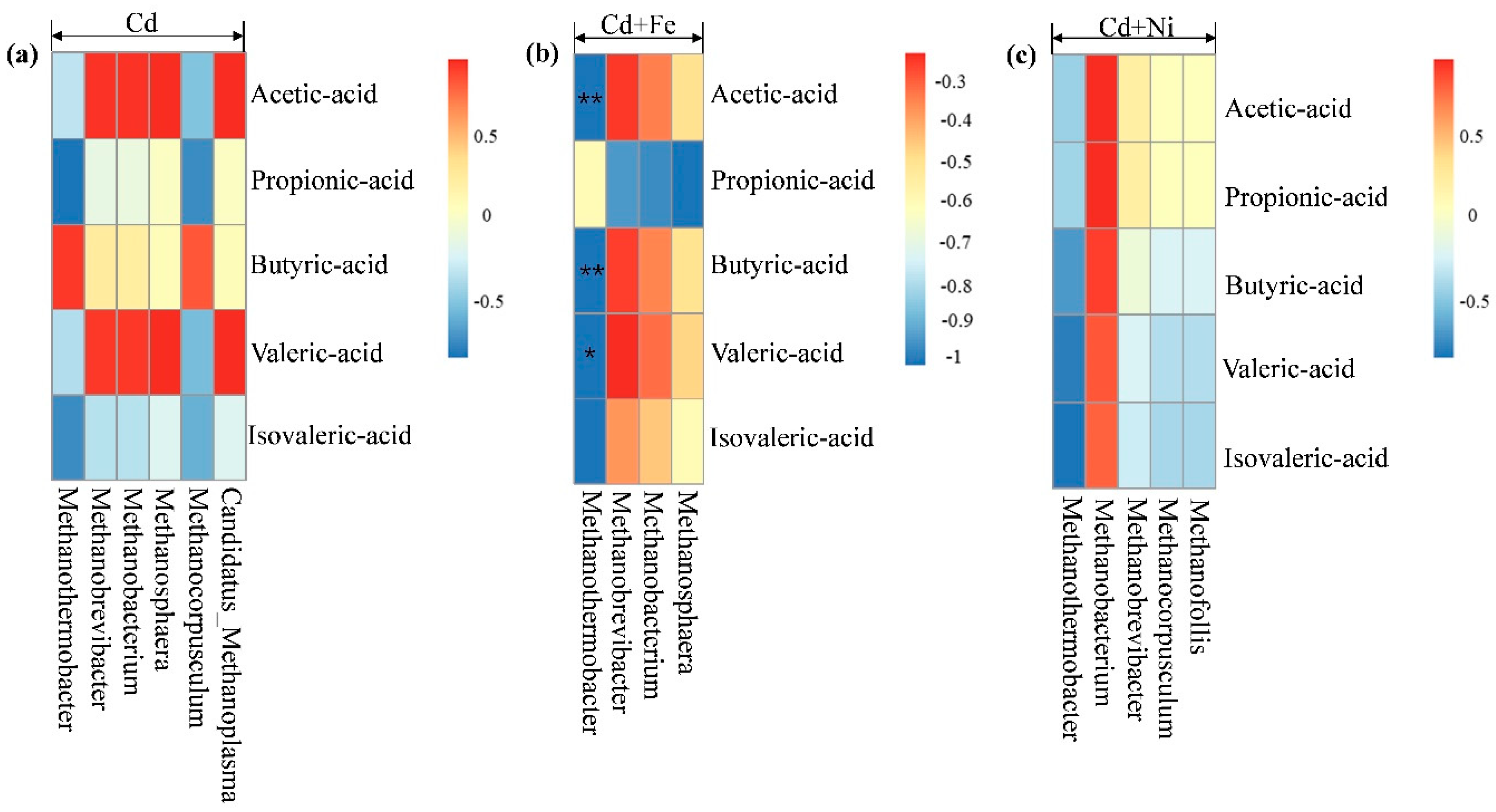
3.4.2. Methanogens and Their Relationships with Fermentation Parameters
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fan, S.Y.; Freedman, B.; Gao, J.X. Potential environmental benefits from increased use of bioenergy in China. Environ. Manag. 2007, 40, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Kibazohi, O.; Sangwan, R. Vegetable oil production potential from Jatropha curcas, Croton megalocarpus, Aleurites moluccana, Moringa oleifera and Pachira glabra: Assessment of renewable energy resources for bio-energy production in Africa. Biomass Bioenergy 2011, 35, 1352–1356. [Google Scholar] [CrossRef]
- Zhuang, D.F.; Jiang, D.; Liu, L. Assessment of bioenergy potential on marginal land in China. Renew. Sustain. Energy Rev. 2011, 15, 1050–1056. [Google Scholar] [CrossRef]
- Li, X.F.; Hou, S.L.; Su, M. Major energy plants and their potential for bioenergy development in China. Environ. Manag. 2010, 46, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Bellarby, J.; Wattenbach, M.; Tuck, G. The potential distribution of bioenergy crops in the UK under present and future climate. Biomass Bioenergy 2010, 34, 1935–1945. [Google Scholar] [CrossRef]
- Čater, M.; Fanedl, L.; Malovrh, Š. Biogas production from brewery spent grain enhanced by bioaugmentation with hydrolytic anaerobic bacteria. Bioresour. Technol. 2015, 186, 261–269. [Google Scholar] [CrossRef]
- Pobeheim, H.; Munk, B.; Lindorfer, H. Impact of nickel and cobalt on biogas production and process stability during semi-continuous anaerobic fermentation of a model substrate for maize silage. Water Res. 2011, 45, 781–787. [Google Scholar] [CrossRef]
- Wei, Y.J.; Liu, Y.S. Effects of sewage sludge compost application on crops and cropland in a 3-year field study. Chemosphere 2005, 59, 1257–1265. [Google Scholar] [CrossRef]
- Mudhoo, A.; Kumar, S. Effects of heavy metals as stress factors on anaerobic digestion processes and biogas production from biomass. Int. J. Environ. Sci. Technol. 2013, 10, 1383–1398. [Google Scholar] [CrossRef]
- Nordell, E.; Nilsson, B.; Påledal, S.N. Co-digestion of manure and industrial waste–The effects of trace element addition. Waste Manag. 2016, 47, 21–27. [Google Scholar] [CrossRef]
- Krishna, R.; Gilbert, W. Toxification and detoxification of heavy metals in anaerobic reactors used in the production of biohydrogen: Future fuel. Int. J. Environ. Eng. Res. 2014, 3, 1–6. [Google Scholar]
- Li, C.L.; Fang, H. Inhibition of heavy metals on fermentative hydrogen production by granular sludge. Chemosphere 2007, 67, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Zerkle, A.L.; House, C.H.; Brantley, S.L. Biogeochemical signatures through time as inferred from whole microbial genomes. Am. J. Sci. 2005, 305, 467–502. [Google Scholar] [CrossRef]
- Glass, J.; Orphan, V.J. Trace metal requirements for microbial enzymes involved in the production and consumption of methane and nitrous oxide. Front. Microbiol. 2012, 3, 61. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.L.; Zhang, H.Y.; Chai, Y. Biogas properties and enzymatic analysis during anaerobic fermentation of Phragmites australis straw and cow dung: Influence of nickel chloride supplement. Biodegradation 2017, 28, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Tian, Y.L.; Wang, L.J. Effect of ferrous chloride on biogas production and enzymatic activities during anaerobic fermentation of cow dung and Phragmites straw. Biodegradation 2016, 27, 69–82. [Google Scholar] [CrossRef]
- Choong, Y.Y.; Norli, I.; Abdullah, A.Z. Impacts of trace element supplementation on the performance of anaerobic digestion process: A critical review. Bioresour. Technol. 2016, 209, 369–379. [Google Scholar] [CrossRef]
- Ashley, N.; Davies, M.; Hurst, T.J. The effect of increased nickel ion concentrations on microbial populations in the anaerobic digestion of sewage sludge. Water Res. 1982, 16, 963–971. [Google Scholar] [CrossRef]
- Takashima, M.; Speece, R.; Parkin, G.F. Mineral requirements for methane fermentation. Crit. Rev. Environ. Sci. Technol. 1990, 19, 465–479. [Google Scholar] [CrossRef]
- Lin, C.Y. Effect of heavy metals on acidogenesis in anaerobic digestion. Water Res. 1993, 27, 147–152. [Google Scholar] [CrossRef]
- Ahring, B.K. Westermann Peter. Sensitivity of thermophilic methanogenic bacteria to heavy metals. Curr. Microbiol. 1985, 12, 273–276. [Google Scholar] [CrossRef]
- Tsai, Y.P.; You, S.J.; Pai, T.Y. Effect of cadmium on composition and diversity of bacterial communities in activated sludges. Int. Biodeterior. Biodegrad. 2005, 55, 285–291. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Tian, Y.L.; Wang, L.J. Ecophysiological characteristics and biogas production of cadmium-contaminated crops. Bioresour. Technol. 2013, 146, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Zhang, H.; Zheng, L. Effect of Zn addition on the Cd-Containing anaerobic fermentation process: Biodegradation and microbial Communities. Int. J. Environ. Res. Public Health 2019, 16, 2998. [Google Scholar] [CrossRef]
- Tian, Y.L.; Zhang, H.Y. Producing biogas from agricultural residues generated during phytoremediation process: Possibility, threshold, and challenges. Int. J. Green Energy 2016, 13, 1556–1563. [Google Scholar] [CrossRef]
- Sun, W.; Qu, Y.; Yu, Q. Adsorption of organic pollutants from coking and papermaking wastewaters by bottom ash. J. Hazard. Mater. 2008, 154, 595–601. [Google Scholar] [CrossRef]
- Wei, Y.S.; Houten, R.T.V.; Brger, A.R. Minimization of excess sludge production for biological wastewater treatment. Water Res. 2003, 37, 4453–4467. [Google Scholar] [CrossRef]
- Allen, G.C.; Floresvergara, M.A.; Krasynanski, S. A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat. Protoc. 2006, 1, 2320–2325. [Google Scholar] [CrossRef]
- Zhang, X.L.; Tian, X.Q.; Ma, L.Y. Biodiversity of the symbiotic bacteria associated with toxic marine Dinoflagellate Alexandrium tamarense. J. Biosci. Med. 2015, 3, 23–28. [Google Scholar] [CrossRef]
- Zhang, W.; Lei, Z.; Li, A. Enhanced anaerobic digestion of food waste by trace metal elements supplementation and reduced metals dosage by green chelating agent [S, S]-EDDS via improving metals bioavailability. Water Res. 2015, 84, 266–277. [Google Scholar] [CrossRef]
- Tang, M.; Xian, P.; Ying, X.U. Effects of Fe2+, Co2+ and Ni2+ on anaerobic process for landfill leachate treatment. Acta Sci. Circumstantiae 2014, 34, 2573–2579. [Google Scholar]
- Fermoso, F.G.; Bartacek, J.; Jansen, S. Metal supplementation to UASB bioreactors: From cell-metal interactions to full-scale application. Sci. Total Environ. 2009, 407, 3652–3667. [Google Scholar] [CrossRef] [PubMed]
- Guang, Y.; Guang, Z.; Pan, Z. Enhancement of sludge anaerobic digestion by adding trace element Fe and Ni. J. Environ. Eng.-China 2017, 11, 4971–4977. [Google Scholar]
- Wang, K.; Yin, J.; Shen, D. Anaerobic digestion of food waste for volatile fatty acids (VFAs) production with different types of inoculum: Effect of pH. Bioresour. Technol. 2014, 161, 395–401. [Google Scholar] [CrossRef]
- Lo, H.; Chiang, C.; Tsao, H. Effects of spiked metals on the MSW anaerobic digestion. Waste Manag. Res. 2012, 30, 32–48. [Google Scholar] [CrossRef]
- Zhang, X.; Qiu, W.; Chen, H. Enhancing the hydrolysis and acidification of steam-exploded cornstalks by intermittent pH adjustment with an enriched microbial community. Bioresour. Technol. 2012, 123, 30–35. [Google Scholar] [CrossRef]
- Mao, C.; Feng, Y.; Wang, X. Review on research achievements of biogas from anaerobic digestion. Renew. Sustain. Energy Rev. 2015, 45, 540–555. [Google Scholar] [CrossRef]
- Lee, S.J. Relationship between Oxidation Reduction Potential (ORP) and Volatile Fatty Acid (VFA) Production in the Acid-Phase Anaerobic Digestion Process. Master’s Thesis, University of Canterbury, Christchurch, New Zealand, 2008. [Google Scholar]
- Liu, R.; Wang, Y.; Sun, C. Experimental study on biogas production from vegetable waste by anaerobic fermentation. Trans. CSAE 2008, 24, 209–213. [Google Scholar]
- Zhang, Y.; Cañas, M.Z.; Zhu, Z. Robustness of archaeal populations in anaerobic co-digestion of dairy and poultry wastes. Bioresour. Technol. 2011, 102, 779–785. [Google Scholar] [CrossRef]
- Sawayama, S.; Tada, C.; Tsukahara, K. Effect of ammonium addition on methanogenic community in a fluidized bed anaerobic digestion. J. Biosci. Bioeng. 2004, 97, 65–70. [Google Scholar] [CrossRef]
- Boe, K.; Batstone, D.J.; Steyer, J.P. State indicators for monitoring the anaerobic digestion process. Water Res. 2010, 44, 5973–5980. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Dang, Y.; Li, C. Inhibitory effect of high NH4+-N concentration on anaerobic biotreatment of fresh leachate from a municipal solid waste incineration plant. Waste Manag. 2015, 43, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Angsana, R.; Warinthom, S.; Annop, N. Combination effect of pH and acetate on enzymatic cellulose hydrolysis. J. Environ. Sci.-China 2009, 21, 965–970. [Google Scholar]
- Liu, A.; Xu, S.; Lu, C. Anaerobic fermentation by aquatic product wastes and other auxiliary materials. Clean Technol. Environ. 2014, 16, 415–421. [Google Scholar] [CrossRef]
- Fosses, A.; Maté, M.; Franche, N. A seven-gene cluster in Ruminiclostridium cellulolyticum is essential for signalization, uptake and catabolism of the degradation products of cellulose hydrolysis. Biotechnol. Biofuels 2017, 10, 250. [Google Scholar] [CrossRef]
- Podosokorskaya, O.; Bonch-Osmolovskaya, E.; Beskorovaynyy, A. Mobilitalea sibirica gen. nov., sp. nov., a halotolerant polysaccharide-degrading bacterium. Int. J. Syst. Evol. Microbiol. 2014, 64, 2657–2661. [Google Scholar] [CrossRef]
- Niu, L.L.; Song, L.; Liu, X.L. Tepidimicrobium xylanilyticum sp. nov., an anaerobic xylanolytic bacterium, and emended description of the genus Tepidimicrobium. Int. J. Syst. Evol. Microbiol. 2009, 59, 2698–2701. [Google Scholar] [CrossRef]
- Kim, B.C.; Jeon, B.S.; Kim, S. Caproiciproducens galactitolivorans gen. nov., sp. nov., a bacterium capable of producing caproic acid from galactitol, isolated from a wastewater treatment plant. Int. J. Syst. Evol. Microbiol. 2015, 65, 4902–4908. [Google Scholar] [CrossRef]
- Maus, I.; Cibis, K.G.; Wibberg, D. Complete genome sequence of the strain Defluviitoga tunisiensis L3, isolated from a thermophilic, production-scale biogas plant. J. Biotechnol. 2015, 203, 17–18. [Google Scholar] [CrossRef] [PubMed]
- Leahy, S.; Kelly, W.; Ronimus, R. Genome sequencing of rumen bacteria and archaea and its application to methane mitigation strategies. Animal 2013, 2, 235–243. [Google Scholar] [CrossRef]
- Danielsson, R.; Dicksved, J.; Sun, L. Methane production in dairy cows correlates with rumen methanogenic and bacterial community structure. Front. Microbiol. 2017, 8, 226. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Zhang, Y.L.; Wang, J.B. Effects of volatile fatty acid concentrations on methane yield and methanogenic bacteria. Biomass Bioenergy 2009, 33, 848–853. [Google Scholar] [CrossRef]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Xu, Y.; Tian, Y.; Zheng, L.; Hao, H.; Huang, H. Impact of Fe and Ni Addition on the VFAs’ Generation and Process Stability of Anaerobic Fermentation Containing Cd. Int. J. Environ. Res. Public Health 2019, 16, 4066. https://doi.org/10.3390/ijerph16214066
Zhang H, Xu Y, Tian Y, Zheng L, Hao H, Huang H. Impact of Fe and Ni Addition on the VFAs’ Generation and Process Stability of Anaerobic Fermentation Containing Cd. International Journal of Environmental Research and Public Health. 2019; 16(21):4066. https://doi.org/10.3390/ijerph16214066
Chicago/Turabian StyleZhang, Huayong, Yanli Xu, Yonglan Tian, Lei Zheng, He Hao, and Hai Huang. 2019. "Impact of Fe and Ni Addition on the VFAs’ Generation and Process Stability of Anaerobic Fermentation Containing Cd" International Journal of Environmental Research and Public Health 16, no. 21: 4066. https://doi.org/10.3390/ijerph16214066
APA StyleZhang, H., Xu, Y., Tian, Y., Zheng, L., Hao, H., & Huang, H. (2019). Impact of Fe and Ni Addition on the VFAs’ Generation and Process Stability of Anaerobic Fermentation Containing Cd. International Journal of Environmental Research and Public Health, 16(21), 4066. https://doi.org/10.3390/ijerph16214066





