Evaluation of an Ultraviolet C (UVC) Light-Emitting Device for Disinfection of High Touch Surfaces in Hospital Critical Areas
Abstract
1. Introduction
2. Materials and Methods
2.1. PX-UVC Device
2.2. Study Protocol
2.3. Statistical Analysis
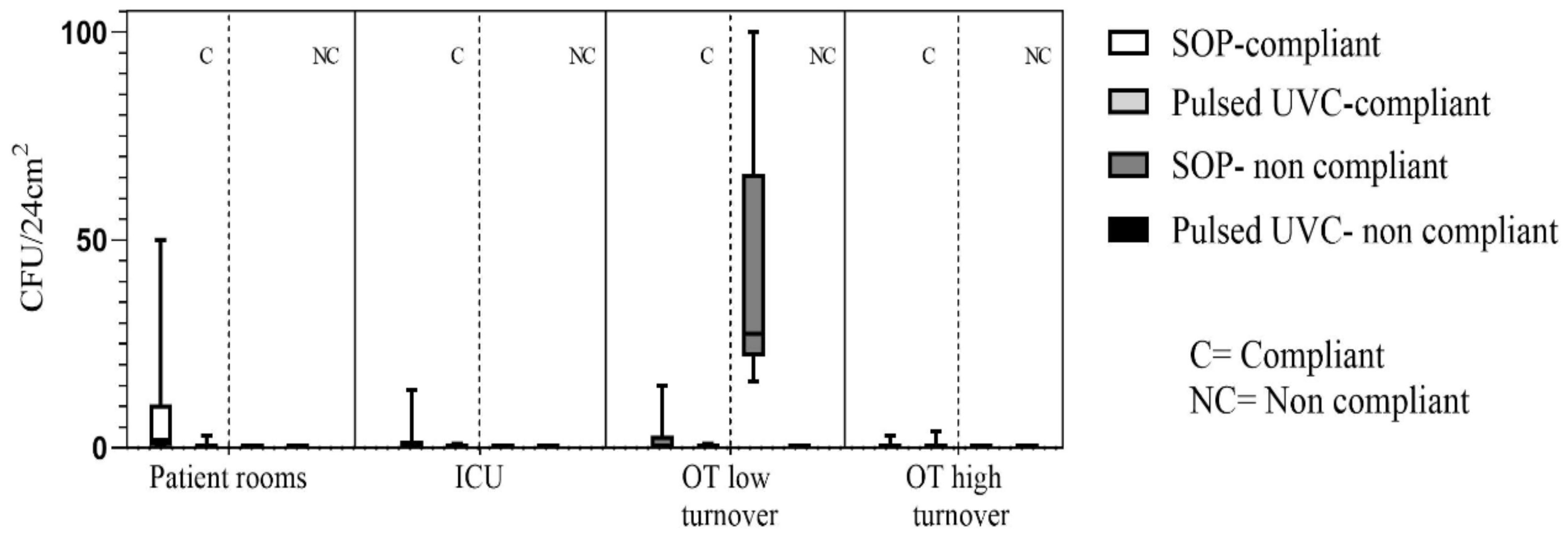
3. Results
High Concern Microorganisms
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weinstein, R.A. Epidemiology and control of nosocomial infections in adult intensive care units. Am. J. Med. 1991, 91 (Suppl. 3B), 179S–184S. [Google Scholar] [CrossRef]
- Adams, C.E.; Smith, J.; Watson, V.; Robertson, C.; Dancer, S.J. Examining the association between surface bioburden and frequently touched sites in intensive care. J. Hosp. Infect. 2017, 95, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Carling, P.C.; Parry, M.M.; Rupp, M.E.; Po, J.L.; Dick, B.; von Beheren, S. Improving cleaning of the environment surrounding patients in 36 acute care hospitals. Infect. Control Hosp. Epidemiol. 2008, 29, 1035–1041. [Google Scholar] [CrossRef] [PubMed]
- Otter, J.A.; Yezli, S.; Salkeld, J.A.; French, G.L. Evidence that contaminated surfaces contribute to the transmission of hospital pathogens and an overview of strategies to address contaminated surfaces in hospital settings. Am. J. Infect. Control 2013, 41, S6–S11. [Google Scholar] [CrossRef] [PubMed]
- Curtis, J.; Donskey, M.D. Does improving surface cleaning and disinfection reduce health care-associated infections? Am. J. Infect. Control 2013, 41, S12–S19. [Google Scholar] [CrossRef]
- Stiefel, U.; Cadnum, J.L.; Eckstein, B.C.; Guerrero, D.M.; Tima, M.A.; Donskey, C.J. Contamination of hands with methicillin-resistant Staphylococcus aureus after contact with environmental surfaces and after contact with the skin of colonized patients. Infect. Control Hosp. Epidemiol. 2011, 32, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Dancer, S.J. Controlling hospital-acquired infection: Focus on the role of the environment and new technologies for decontamination. Clin. Microbiol. Rev. 2014, 27, 665–690. [Google Scholar] [CrossRef]
- White, L.F.; Dancer, S.J.; Robertson, C.; McDonald, J. Are hygiene standards useful in assessing infection risk? Am. J. Infect. Control 2008, 36, 381–384. [Google Scholar] [CrossRef]
- Wilson, A.P.R.; Livermore, D.M.; Otter, J.A.; Warren, R.E.; Jenks, P.; Enoch, D.A.; Newsholme, W.; Oppenheim, B.; Leanord, A.; McNulty, C.; et al. Prevention and control of multi-drug-resistant Gram-negative bacteria: Recommendations from a Joint Working Party. J. Hosp. Infect. 2016, 92, S1–S44. [Google Scholar] [CrossRef]
- Carling, P.C.; Huang, S.S. Improving healthcare environmental cleaning and disinfection. Infect. Control Hosp. Epidemiol 2013, 34, 507–513. [Google Scholar] [CrossRef]
- Vickery, K.; Deva, A.; Jacombs, A.; Allan, J.; Valente, P.; Gosbell, I.B. Presence of biofilm containing viable multiresistant organisms despite terminal cleaning on clinical surfaces in an intensive care unit. J. Hosp. Infect. 2012, 80, 52–55. [Google Scholar] [CrossRef]
- Hu, H.; Johani, K.; Gosbell, I.B.; Jacombs, A.S.W.; Almatroudi, A.; Whiteley, G.S.; Deva, A.K.; Jensen, S.; Vickery, K. Intensive care unit environmental surfaces are contaminated by multidrug-resistant bacteria in biofilms: Combined results of conventional culture, pyrosequencing, scanning electron microscopy, and confocal laser microscopy. J. Hosp. Infect. 2015, 91, 35–44. [Google Scholar] [CrossRef]
- Otter, J.A.; Vickery, K.; Walker, J.T.; de Lancey Pulcini, E.; Stoodley, P.; Goldenberg, S.D.; Salkeld, J.A.G.; Chewins, J.; Yezli, S.; Edgeworth, J.D. Surface-attached cells, biofilms and biocide susceptibility: Implications for hospital cleaning and disinfection. J. Hosp. Infect. 2015, 89, 16–27. [Google Scholar] [CrossRef]
- Kowalski, W. UVGI deactivation theory. In Ultraviolet Germicidal Irradiation Handbook; Springer: New York, NY, USA, 2009; pp. 17–50. [Google Scholar]
- Liscynesky, C.; Hines, L.P.; Smyer, J.; Hanrahan, M.; Orellana, R.C.; Mangino, J.E. The Effect of Ultraviolet Light on Clostridium difficile Spore Recovery Versus Bleach Alone. Infect. Control Hosp. Epidemiol. 2017, 38, 1116–1117. [Google Scholar] [CrossRef]
- Wong, T.; Woznow, T.; Petrie, M.; Murzello, E.; Muniak, A.; Kadora, A.; Bryce, E. Post discharge decontamination of MRSA, VRE, and Clostridium difficile isolation rooms using 2 commercially available automated ultraviolet-C–emitting devices. Am. J. Infect. Control 2016, 44, 416–420. [Google Scholar] [CrossRef]
- Ali, S.; Muzslay, M.; Wilson, P. A Novel Quantitative Sampling Technique for Detection and Monitoring of Clostridium difficile Contamination in the Clinical Environment. J. Clin. Microbiol. 2015, 53, 2570–2574. [Google Scholar] [CrossRef][Green Version]
- Hosein, I.; Madeloso, R.; Nagaratnam, W.; Villamaria, F.; Stock, E.; Jinadatha, C. Evaluation of a pulsed xenon ultraviolet light device for isolation room disinfection in a United Kingdom hospital. Am. J. Infect. Control 2016, 44, e157–e161. [Google Scholar] [CrossRef]
- Haddad, L.E.; Ghantoji, S.S.; Stibich, M.; Fleming, J.B.; Segal, C.; Ware, K.M.; Chemaly, R.F. Evaluation of a pulsed xenon ultraviolet disinfection system to decrease bacterial contamination in operating rooms. BMC Infect. Dis. 2017, 17, 672. [Google Scholar] [CrossRef]
- Jinadatha, C.; Quezada, R.; Huber, T.W.; Williams, J.B.; Zeber, J.E.; Copeland, L.A. Evaluation of a pulsed-xenon ultraviolet room disinfection device for impact on contamination levels of methicillin-resistant Staphylococcus aureus. BMC Infect. Dis. 2014, 14, 187. [Google Scholar] [CrossRef]
- Boyce, J.; Donskey, C. Understanding ultraviolet light surface decontamination in hospital rooms: A primer. Infect. Control Hosp. Epidemiol. 2019, 18, 1030–1035. [Google Scholar] [CrossRef]
- Anderson, D.J.; Chen, L.F.; Weber, D.J.; Moehring, R.W.; Lewis, S.S.; Triplett, P.F.; Blocker, M.; Becherer, P.; Schwab, J.C.; Knelson, L.P.; et al. The benefits of enhanced terminal room (BETR) disinfection study: A prospective, cluster randomized, multicenter, crossover study to evaluate the impact of enhanced terminal room disinfection on acquisition and infection caused by multidrug-resistant organisms. Lancet Infect. Dis. 2017, 389, 805–814. [Google Scholar] [CrossRef]
- Anderson, D.J.; Moehring, R.W.; Weber, D.J.; Lewis, S.S.; Chen, L.F.; Schwab, J.C.; Becherer, P.; Blocker, M.; Triplett, P.F.; Knelson, L.P.; et al. for the CDC Prevention Epicenters Program Effectiveness of targeted enhanced terminal room disinfection on hospital-wide acquisition and infection with multidrug-resistant organisms and Clostridium difficile: A secondary analysis of a multicentre cluster randomised controlled trial with crossover design (BETR Disinfection). Lancet Infect. Dis. 2018, 18, 845–853. [Google Scholar] [CrossRef]
- Italian Workers Compensation Authority, INAIL (ex ISPESL, Istituto Superiore per la Prevenzione E la Sicurezza del Lavoro), LINEE GUIDA SUGLI STANDARD DI SICUREZZA E DI IGIENE NELREPARTO OPERATORIO. Available online: https://www.inail.it/cs/internet/docs/linee-guida-igiene-reparto-operatorio.pdf?section=attivita (accessed on 7 March 2018).
- Dancer, S.J. How do we assess hospital cleaning? A proposal for microbiological standards for surface hygiene in hospitals. J. Hosp. Infect. 2004, 56, 10–15. [Google Scholar] [CrossRef]
- Boyce, J.M. Modern technologies for improving cleaning and disinfection of environmental surfaces in hospitals. Antimicrob. Resist. Infect. Control 2016, 5, 10. [Google Scholar] [CrossRef]
- Shams, A.M.; Rose, L.J.; Edwards, J.R.; Cali, S.; Harris, A.D.; Jacob, J.T.; LaFae, A.; Pineles, L.L.; Thom, K.A.; McDonald, L.C.; et al. Assessment of the overall and multidrug-resistant organism bioburden on environmental surfaces in healthcare facilities. Infect. Control Hosp. Epidemiol 2016, 37, 1426–1432. [Google Scholar] [CrossRef]
- Weber, D.J.; Rutala, W.A. Understanding and Preventing Transmission of Healthcare-Associated Pathogens Due to the Contaminated Hospital Environment. Infect. Control Hosp. Epidemiol. 2013, 34, 449–452. [Google Scholar] [CrossRef]
- Dancer, S.J. Importance of the environment in meticillin-resistant Staphylococcus aureus acquisition: The case for hospital cleaning. Lancet Infect. Dis. 2008, 8, 101–113. [Google Scholar] [CrossRef]
- Rutala, W.A.; Weber, D.J. The benefits of surface disinfection. Am. J. Infect. Control 2004, 32, 226–231. [Google Scholar] [CrossRef]
- Sharma, G.; Malik, D.J. The uses and abuses of rapid bioluminescence-based ATP assays. Inter. J. Hyg. Environ. Health 2012, 216, 115–125. [Google Scholar] [CrossRef]
- Marra, R.; Schweizer, A.L.; Michael, M.E. No-Touch Disinfection Methods to Decrease Multidrug-Resistant Organism Infections: A Systematic Review and Meta-analysis. Infect. Control Hosp. Epidemiol. 2017, 39, 1–12. [Google Scholar] [CrossRef]
- Toffolutti, V.; Reeves, A.; McKee, M.; Stuckler, D. Outsourcing cleaning services increases MRSA incidence: Evidence from 126 english acute trusts. Soc. Sci. Med. 2017, 174, 64–69. [Google Scholar] [CrossRef]
- Sattar, S.A.; Bradley, C.; Kibbee, R.; Wesgate, R.; Wilkinson MA, C.; Sharpe, T.; Maillard, J.Y. Disinfectant wipes are appropriate to control microbial bioburden from surfaces: Use of a new ASTM standard test protocol to demonstrate efficacy. J. Hosp. Infect. 2015, 91, 319–325. [Google Scholar] [CrossRef]
- Casini, B.; Righi, A.; De Feo, N.; Totaro, M.; Giorgi, S.; Zezza, L.; Valentini, P.; Tagliaferri, E.; Costa, A.L.; Barnini, S.; et al. Improving Cleaning and Disinfection of High-Touch Surfaces in Intensive Care during Carbapenem-Resistant Acinetobacter baumannii Endemo-Epidemic Situations. Int J. Environ. Res. Public Health 2018, 15, 2305. [Google Scholar] [CrossRef]
- Smith, D.L.; Gillanders, S.; Holah, J.T.; Gush, C. Assessing the efficacy of different microfibre cloths at removing surface micro-organisms associated with healthcare-associated infections. J. Hosp. Infect. 2011, 78, 182–186. [Google Scholar] [CrossRef]
- Smith, D.L.; Gillanders, S.; Holah, J.T.; Gush, C. Limitations of the Efficacy of Surface Disinfection in the Healthcare Setting. Infect. Control Epidemiol. 2009, 30, 570–573. [Google Scholar] [CrossRef]
| Setting | Timing of Sampling | n (Samles) | Median | Lower | Higher | IQR |
|---|---|---|---|---|---|---|
| Patient rooms | Before C&D | 25 | 43 | 0 | 180 | 93 |
| After SOP | 25 | 2 | 0 | 50 | 7 | |
| After SOP + Pulsed-UVC | 25 | 0 | 0 | 3 | 1 | |
| ICU | Before C&D | 10 | 23 | 1 | 50 | 45 |
| After SOP | 10 | 1 | 0 | 14 | 2 | |
| After SOP + Pulsed-UVC | 10 | 0 | 0 | 1 | 0 | |
| OT low turnover | Before C&D | 60 | 1 | 0 | 100 | 4 |
| After SOP | 80 | 1 | 0 | 100 | 6 | |
| After SOP + Pulsed-UVC | 30 | 0 | 0 | 1 | 0 | |
| OT high turnover | Before C&D | 40 | 7 | 0 | 38 | 25 |
| After SOP | 10 | 0 | 0 | 3 | 1 | |
| After Pulsed-UVC | 20 | 0 | 0 | 4 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casini, B.; Tuvo, B.; Cristina, M.L.; Spagnolo, A.M.; Totaro, M.; Baggiani, A.; Privitera, G.P. Evaluation of an Ultraviolet C (UVC) Light-Emitting Device for Disinfection of High Touch Surfaces in Hospital Critical Areas. Int. J. Environ. Res. Public Health 2019, 16, 3572. https://doi.org/10.3390/ijerph16193572
Casini B, Tuvo B, Cristina ML, Spagnolo AM, Totaro M, Baggiani A, Privitera GP. Evaluation of an Ultraviolet C (UVC) Light-Emitting Device for Disinfection of High Touch Surfaces in Hospital Critical Areas. International Journal of Environmental Research and Public Health. 2019; 16(19):3572. https://doi.org/10.3390/ijerph16193572
Chicago/Turabian StyleCasini, Beatrice, Benedetta Tuvo, Maria Luisa Cristina, Anna Maria Spagnolo, Michele Totaro, Angelo Baggiani, and Gaetano Pierpaolo Privitera. 2019. "Evaluation of an Ultraviolet C (UVC) Light-Emitting Device for Disinfection of High Touch Surfaces in Hospital Critical Areas" International Journal of Environmental Research and Public Health 16, no. 19: 3572. https://doi.org/10.3390/ijerph16193572
APA StyleCasini, B., Tuvo, B., Cristina, M. L., Spagnolo, A. M., Totaro, M., Baggiani, A., & Privitera, G. P. (2019). Evaluation of an Ultraviolet C (UVC) Light-Emitting Device for Disinfection of High Touch Surfaces in Hospital Critical Areas. International Journal of Environmental Research and Public Health, 16(19), 3572. https://doi.org/10.3390/ijerph16193572






