Prevalence of Chlamydia trachomatis Infection, Serovar Distribution and Co-Infections with Seven High-Risk HPV Types among Italian Women with a Recent History of Abnormal Cervical Cytology
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design and Sample Collection
2.2. DNA Extraction and High-Risk HPV Detection
2.3. Chlamydia trachomatis Detection and Typing
2.4. Statistical Analysis
2.5. Ethics Approval and Consent to Participate
3. Results
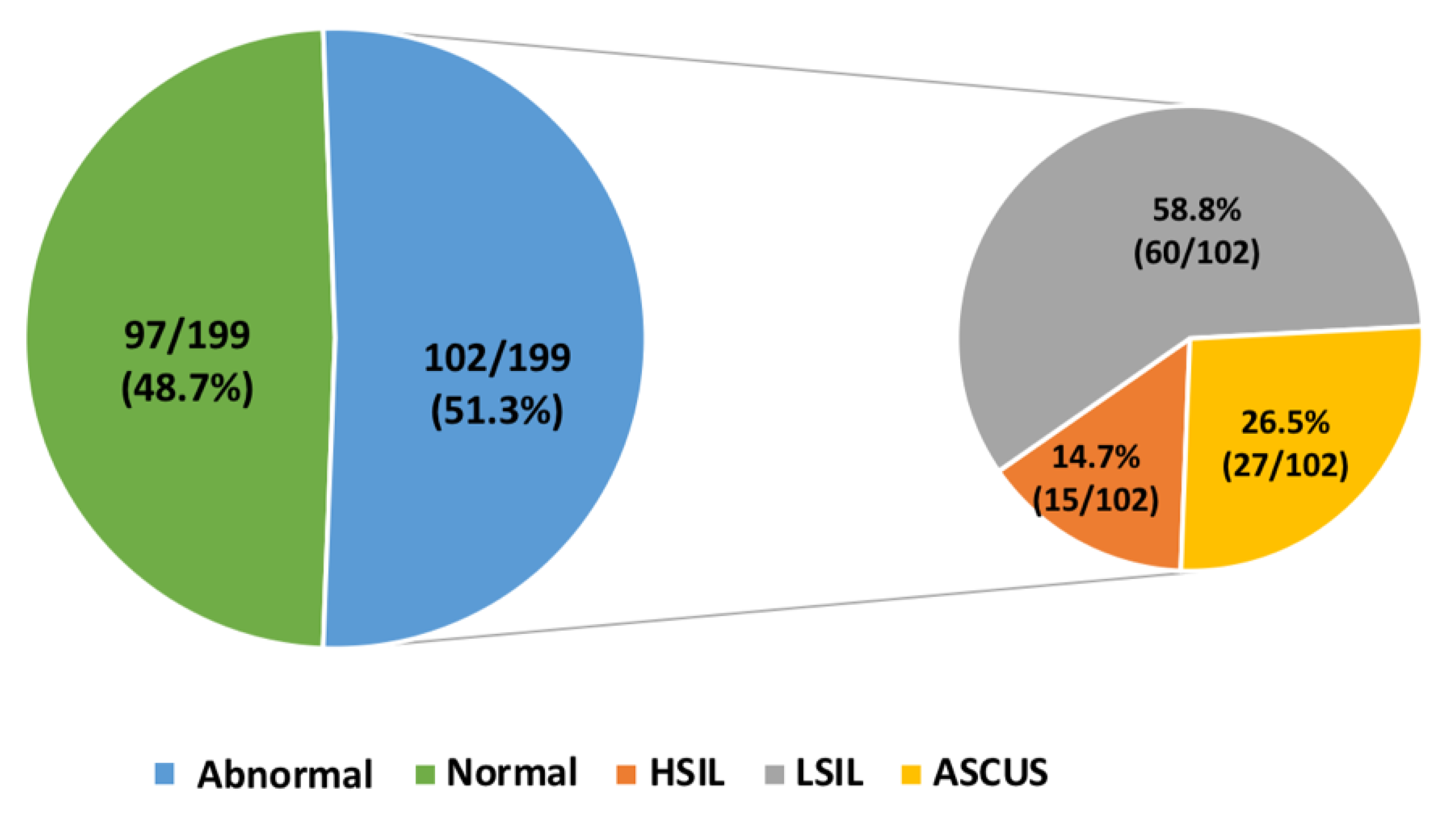
3.1. Cervical Cytology Analysis
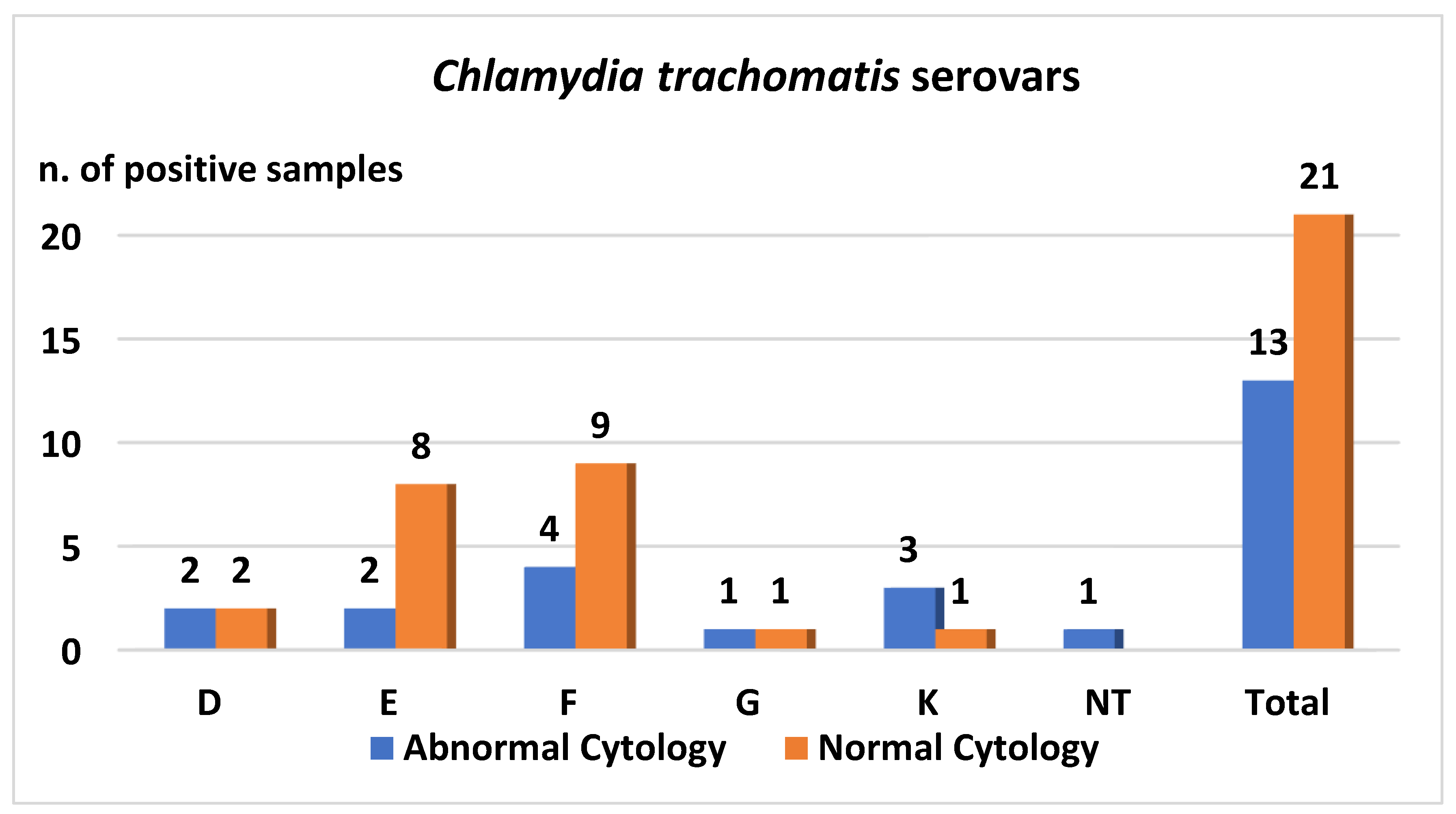
3.2. Chlamydia trachomatis Detection
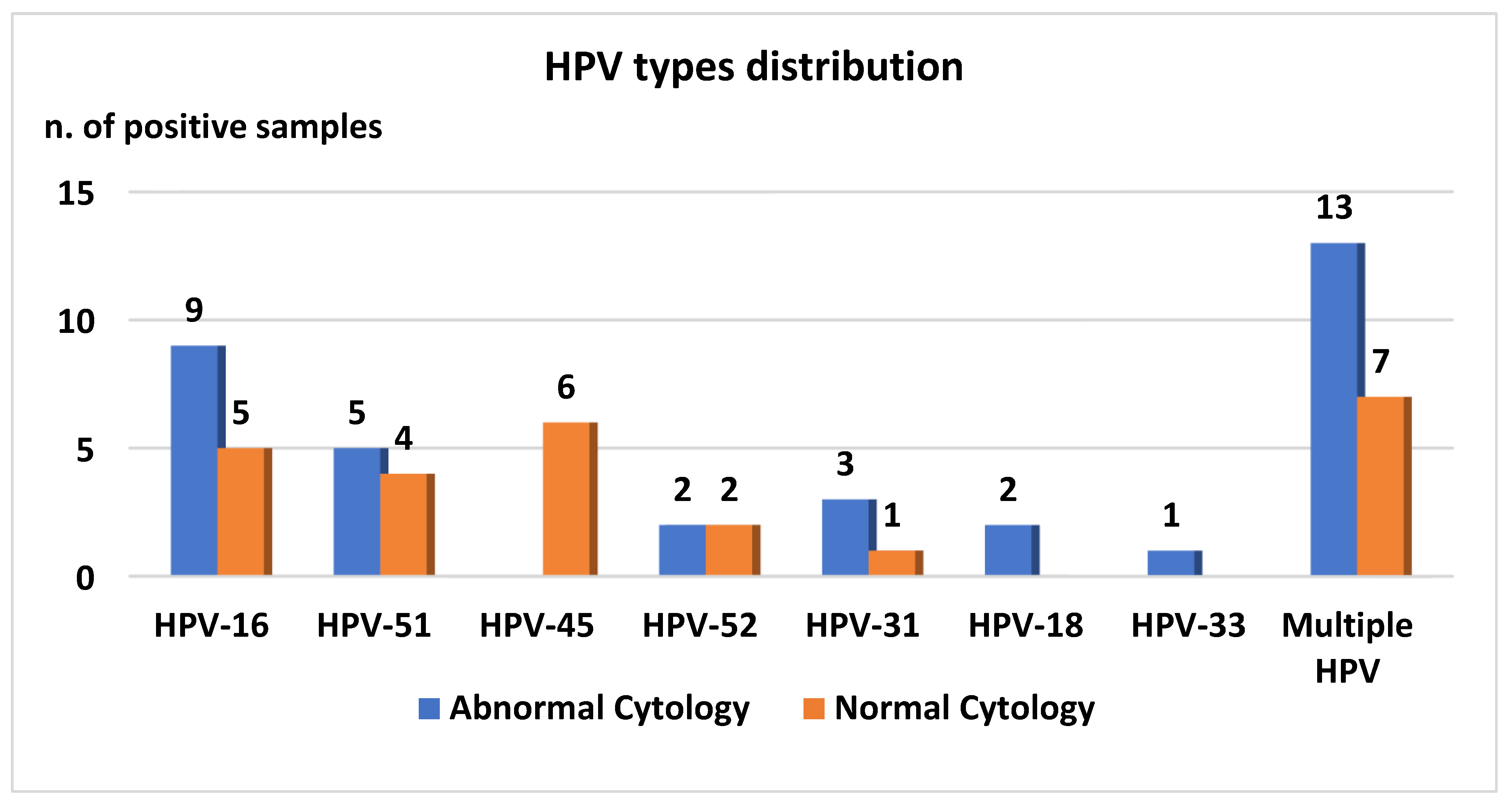
3.3. HPV Detection
3.4. Chlamydia Trachomatis and HPV Co-Infection
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Ct | Chlamydia trachomatis |
| WHO | World Health Organization |
| EU/EEA | European Union/European Economic Area |
| MOMP | major outer membrane protein |
| LGV | lymphogranuloma venereum |
| STDs | sexually transmitted diseases |
| PID | pelvic inflammatory disease |
| HPV | human papillomavirus |
| RFLP | restriction fragment length polymorphism |
| PCR | polymerase chain reaction |
References
- WHO. Guideline for Treatment of Chlamydia Trachomatis Infection; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Surveillance Report, Annual Epidemiological Report for 2016, Chlamydia; eCDC: Solna Municipality, Sweden, 2016.
- Somboonna, N.; Mead, S.; Liu, J.; Dean, D. Discovering and differentiating new and emerging clonal populations of Chlamydia trachomatis with a novel shotgun cell culture harvest assay. Emerg. Infect. Dis. 2008, 14, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Spaargaren, J.; Fennema, H.S.; Morré, S.A.; de Vries, H.J.; Coutinho, R.A. New lymphogranuloma venereum Chlamydia trachomatis variant, Amsterdam. Emerg. Infect. Dis. 2005, 11, 1090–1092. [Google Scholar] [PubMed]
- Carlson, J.H.; Porcella, S.F.; McClarty, G.; Caldwell, H.D. Comparative genomic analysis of Chlamydia trachomatis oculotropic and genitotropic strains. Infect. Immun. 2005, 73, 6407–6418. [Google Scholar] [CrossRef] [PubMed]
- Del Prete, R.; Ronga, L.; Lestingi, M.; Addati, G.; Angelotti, U.F.; Di Carlo, D.; Miragliotta, G. Simultaneous detection and identification of STI pathogens by multiplex Real-Time PCR in genital tract specimens in a selected area of Apulia, a region of Southern Italy. Infection 2017, 45, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Foschi, C.; Nardini, P.; Banzola, N.; D’Antuono, A.; Compri, M.; Cevenini, R.; Marangoni, A. Chlamydia trachomatis infection prevalence and serovar distribution in a high-density urban area in the north of Italy. J. Med. Microbiol. 2016, 65, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Salfa, M.C.; Suligoi, B.; Italian STI Laboratory-based Surveillance Working Group. Prevalence of Chlamydia trachomatis, Trichomonas vaginalis and Neisseria gonorrhoeae based on Data Collected by a Network of Clinical Microbiology Laboratories, in Italy. Adv. Exp. Med. Biol. 2016, 901, 47–57. [Google Scholar] [PubMed]
- Bianchi, S.; Boveri, S.; Igidbashian, S.; Amendola, A.; Urbinati, A.M.; Frati, E.R.; Bottari, F.; Colzani, D.; Landoni, F.; Tanzi, E.; et al. Chlamydia trachomatis infection and HPV/Chlamydia trachomatis co-infection among HPV-vaccinated young women at the beginning of their sexual activity. Arch. Gynecol. Obstet. 2016, 294, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Frati, E.R.; Fasoli, E.; Martinelli, M.; Colzani, D.; Bianchi, S.; Carnelli, L.; Amendola, A.; Olivani, P.; Tanzi, E. Sexually Transmitted Infections: A Novel Screening Strategy for Improving Women’s Health in Vulnerable Populations. Int. J. Mol. Sci. 2017, 18, 1311. [Google Scholar] [CrossRef]
- Silva, J.; Cerqueira, F.; Medeiros, R. Chlamydia trachomatis infection: Implications for HPV status and cervical cancer. Arch. Gynecol. Obstet. 2014, 289, 715–723. [Google Scholar] [CrossRef]
- Smith, J.H. Bethesda 2001—Review. Cytopathology 2002, 13, 4–10. [Google Scholar] [CrossRef]
- Broccolo, F.; Cocuzza, C.E. Automated extraction and quantitation of oncogenic HPV genotypes from cervical samples by a real-time PCR-based system. J. Virol. Methods 2008, 148, 48–57. [Google Scholar] [CrossRef]
- Broccolo, F.; Chiari, S.; Piana, A.; Castiglia, P.; Dell’Anna, T.; Garcia-Parra, R.; Maneo, A.; Villa, A.; Leone, E.B.; Perego, P.; et al. Prevalence and viral load of oncogenic human papillomavirus types associated with cervical carcinoma in a population of North Italy. J. Med. Virol. 2009, 81, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Lindh, M.; Görander, S.; Andersson, E.; Horal, P.; Mattsby-Balzer, I.; Ryd, W. Real-time Taqman PCR targeting 14 human papilloma virus types. J. Clin. Virol. 2007, 40, 321–324. [Google Scholar] [CrossRef]
- Flores-Munguia, R.; Siegel, E.; Klimecki, W.T.; Giuliano, A.R. Performance assessment of eight high-throughput PCR assays for viral load quantitation of oncogenic HPV types. J. Mol. Diagn. 2004, 6, 115–124. [Google Scholar] [CrossRef]
- Mahony, J.B.; Luinstra, K.E.; Sellors, J.W.; Chernesky, M.A. Comparison of plasmid- and chromosome-based polymerase chain reaction assays for detecting Chlamydia trachomatis nucleic acids. J. Clin. Microbiol. 1993, 31, 1753–1758. [Google Scholar]
- Lan, J.; Walboomers, J.M.; Roosendaal, R.; van Doornum, G.J.; MacLaren, D.M.; Meijer, C.J.; van den Brule, A.J. Direct detection and genotyping of Chlamydia trachomatis in cervical scrapes by using polymerase chain reaction and restriction fragment length polymorphism analysis. J. Clin. Microbiol. 1993, 31, 1060–1065. [Google Scholar] [PubMed]
- Seraceni, S.; De Seta, F.; Colli, C.; Del Savio, R.; Pesel, G.; Zanin, V.; D’Agaro, P.; Contini, C.; Comar, M. High prevalence of hpv multiple genotypes in women with persistent chlamydia trachomatis infection. Infect. Agents Cancer 2014, 9, 30. [Google Scholar] [CrossRef]
- Mancini, F.; Vescio, F.; Mochi, S.; Accardi, L.; di Bonito, P.; Ciervo, A. HPV and Chlamydia trachomatis coinfection in women with Pap smear abnormality: Baseline data of the HPV Pathogen ISS study. Infez. Med. 2018, 26, 139–144. [Google Scholar]
- Bellaminutti, S.; Seraceni, S.; De Seta, F.; Gheit, T.; Tommasino, M.; Comar, M. HPV and Chlamydia trachomatis co-detection in young asymptomatic women from high incidence area for cervical cancer. J. Med. Virol. 2014, 86, 1920–1925. [Google Scholar] [CrossRef]
- Marangoni, A.; Foschi, C.; Nardini, P.; D’Antuono, A.; Banzola, N.; Di Francesco, A.; Ostanello, F.; Russo, I.; Donati, M.; Cevenini, R. Chlamydia trachomatis serovar distribution and other sexually transmitted coinfections in subjects attending an STD outpatients clinic in Italy. New Microbiol. 2012, 35, 215–219. [Google Scholar] [CrossRef]
- Wikström, E.; Surcel, H.M.; Merikukka, M.; Ohman, H.; Namujju, P.B.; Tasanen, K.; Tiitinen, A.; Paavonen, J.; Lehtinen, M. Changes over time in the Chlamydia trachomatis serotype distribution in Finnish women. Scand. J. Infect. Dis. 2014, 46, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Cocuzza, C.E.; Martinelli, M.; Sina, F.; Piana, A.; Sotgiu, G.; Dell’Anna, T.; Musumeci, R. Human papillomavirus DNA detection in plasma and cervical samples of women with a recent history of low grade or precancerous cervical dysplasia. PLoS ONE 2017, 12, e0188592. [Google Scholar] [CrossRef]
- Wohlmeister, D.; Vianna, D.R.; Helfer, V.E.; Gimenes, F.; Consolaro, M.E.; Barcellos, R.B.; Rossetti, M.L.; Calil, L.N.; Buffon, A.; Pilger, D.A. Association of human papillomavirus and Chlamydia trachomatis with intraepithelial alterations in cervix samples. Mem. Inst. Oswaldo Cruz. 2016, 111, 106–113. [Google Scholar] [CrossRef]
- Robial, R.; Longatto-Filho, A.; Roteli-Martins, C.M.; Silveira, M.F.; Stauffert, D.; Ribeiro, G.G.; Linhares, I.M.; Tacla, M.; Zonta, M.A.; Baracat, E.C. Frequency of Chlamydia trachomatis infection in cervical intraepithelial lesions and the status of cytological p16/Ki-67 dual-staining. Infect Agents Cancer 2017, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, S.; Frati, E.R.; Panatto, D.; Martinelli, M.; Amicizia, D.; Zotti, C.M.; Martinese, M.; Bonanni, P.; Boccalini, S.; Coppola, R.C.; et al. Detection and genotyping of human papillomavirus in urine samples from unvaccinated male and female adolescents in Italy. PLoS ONE 2013, 8, e79719. [Google Scholar] [CrossRef]
- Piana, A.; Sotgiu, G.; Castiglia, P.; Pischedda, S.; Cocuzza, C.; Capobianco, G.; Marras, V.; Dessole, S.; Muresu, E. Prevalence and type distribution of human papillomavirus infection in women from North Sardinia, Italy. BMC Public Health 2011, 11, 785. [Google Scholar] [CrossRef]
- Piana, A.; Sotgiu, G.; Cocuzza, C.; Musumeci, R.; Marras, V.; Pischedda, S.; Deidda, S.; Muresu, E.; Castiglia, P. High HPV-51 prevalence in invasive cervical cancers: Results of a pre-immunization survey in North Sardinia, Italy. PLoS ONE 2013, 8, e63395. [Google Scholar] [CrossRef]
- Orlando, G.; Fasolo, M.; Mazza, F.; Ricci, E.; Esposito, S.; Frati, E.; Zuccotti, G.V.; Cetin, I.; Gramegna, M.; Rizzardini, G.; et al. Valhidate Study Group. Risk of cervical HPV infection and prevalence of vaccine-type and other high-risk HPV types among sexually active teens and young women (13–26 years) enrolled in the VALHIDATE study. Hum. Vaccines Immunother. 2014, 10, 986–994. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinelli, M.; Musumeci, R.; Rizzo, A.; Muresu, N.; Piana, A.; Sotgiu, G.; Landoni, F.; Cocuzza, C. Prevalence of Chlamydia trachomatis Infection, Serovar Distribution and Co-Infections with Seven High-Risk HPV Types among Italian Women with a Recent History of Abnormal Cervical Cytology. Int. J. Environ. Res. Public Health 2019, 16, 3354. https://doi.org/10.3390/ijerph16183354
Martinelli M, Musumeci R, Rizzo A, Muresu N, Piana A, Sotgiu G, Landoni F, Cocuzza C. Prevalence of Chlamydia trachomatis Infection, Serovar Distribution and Co-Infections with Seven High-Risk HPV Types among Italian Women with a Recent History of Abnormal Cervical Cytology. International Journal of Environmental Research and Public Health. 2019; 16(18):3354. https://doi.org/10.3390/ijerph16183354
Chicago/Turabian StyleMartinelli, Marianna, Rosario Musumeci, Alberto Rizzo, Narcisa Muresu, Andrea Piana, Giovanni Sotgiu, Fabio Landoni, and Clementina Cocuzza. 2019. "Prevalence of Chlamydia trachomatis Infection, Serovar Distribution and Co-Infections with Seven High-Risk HPV Types among Italian Women with a Recent History of Abnormal Cervical Cytology" International Journal of Environmental Research and Public Health 16, no. 18: 3354. https://doi.org/10.3390/ijerph16183354
APA StyleMartinelli, M., Musumeci, R., Rizzo, A., Muresu, N., Piana, A., Sotgiu, G., Landoni, F., & Cocuzza, C. (2019). Prevalence of Chlamydia trachomatis Infection, Serovar Distribution and Co-Infections with Seven High-Risk HPV Types among Italian Women with a Recent History of Abnormal Cervical Cytology. International Journal of Environmental Research and Public Health, 16(18), 3354. https://doi.org/10.3390/ijerph16183354







