Nanoscale Zero-Valent Iron and Chitosan Functionalized Eichhornia crassipes Biochar for Efficient Hexavalent Chromium Removal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
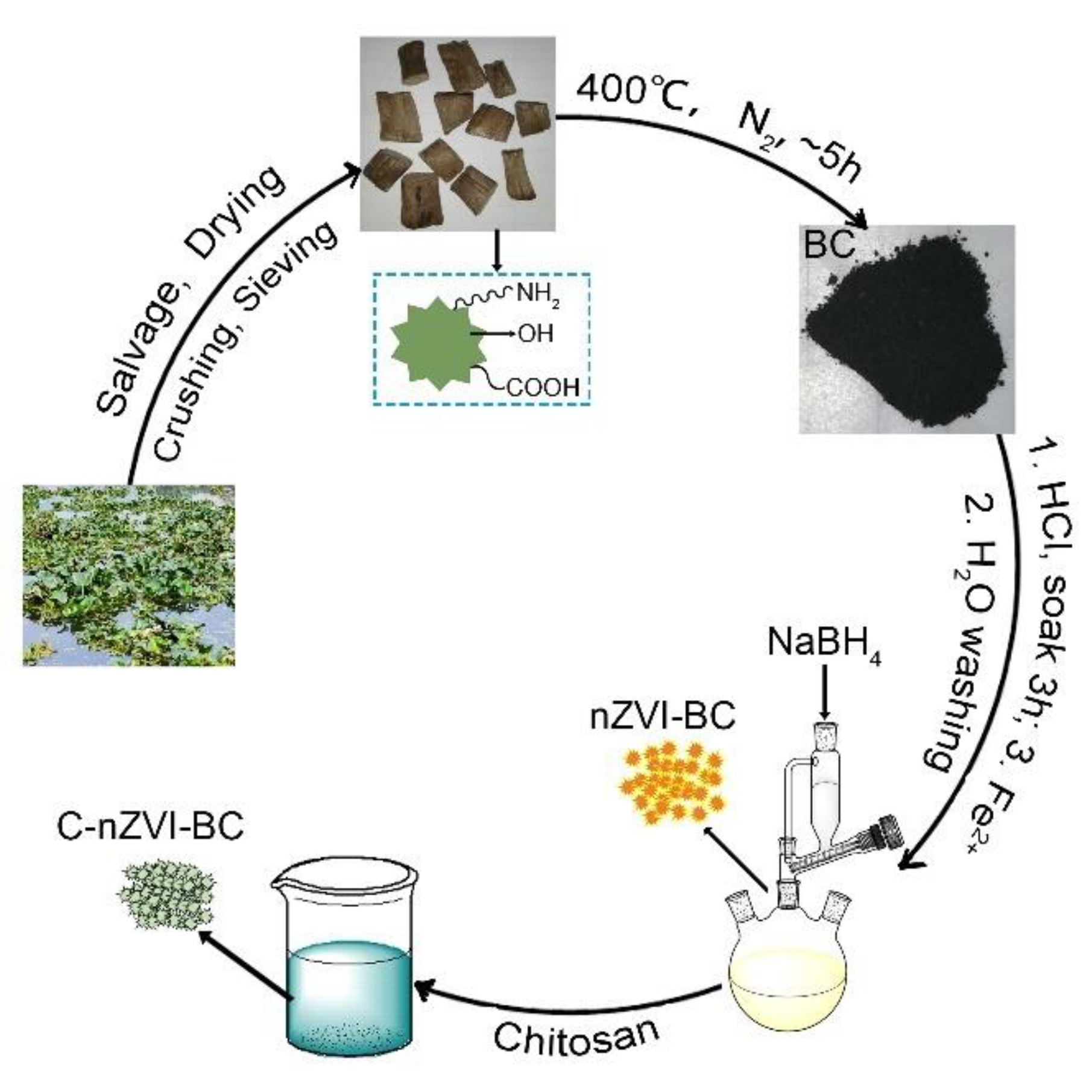
2.2. Preparation of Adsorbents
2.2.1. Preparation of Pristine Biochar (BC)
2.2.2. Preparation of nZVI–BC Composite
2.2.3. Preparation of C–nZVI–BC Composite
2.3. Characterizations
2.4. Sorption Experiments
2.5. Regeneration of C–nZVI–BC Adsorbent
3. Results and Discussion
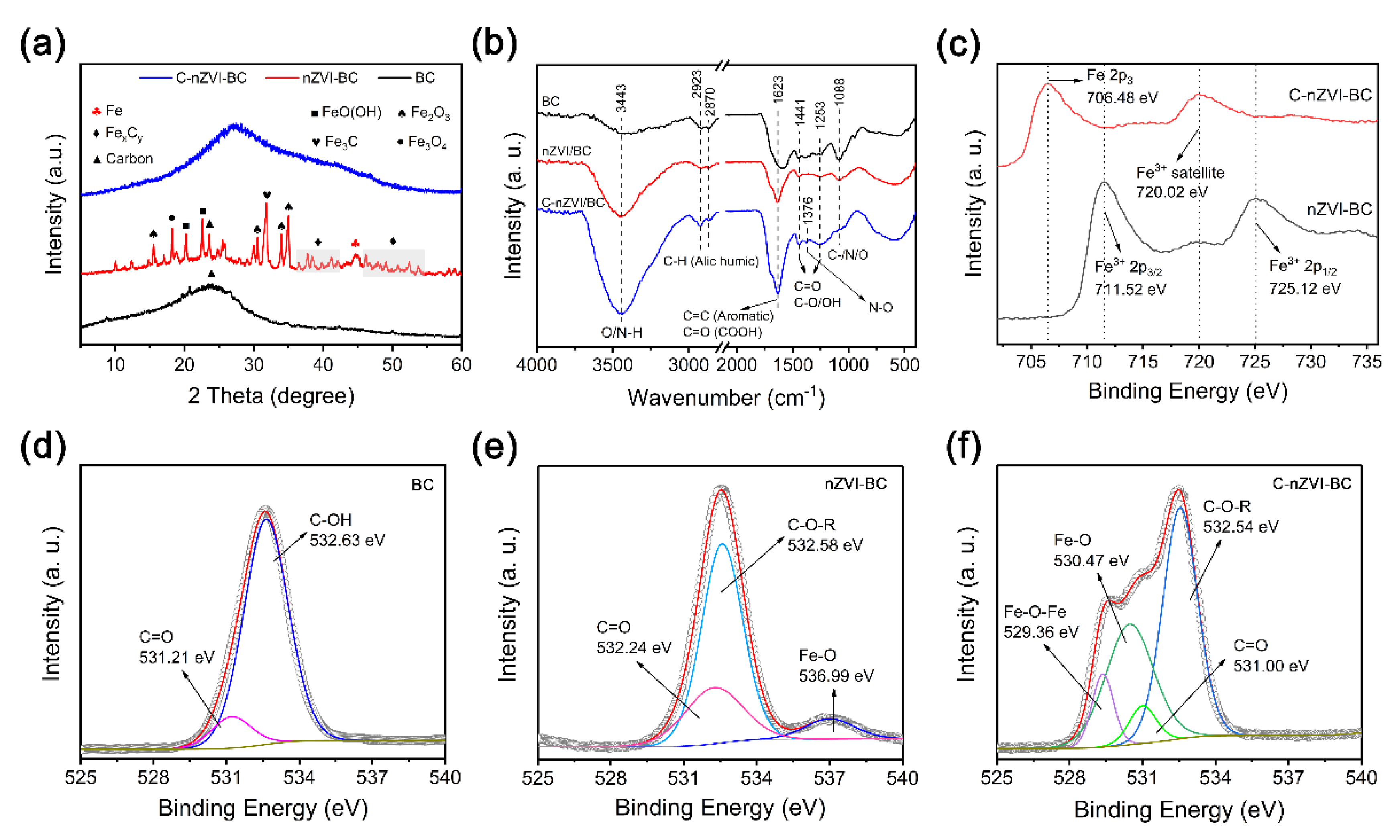
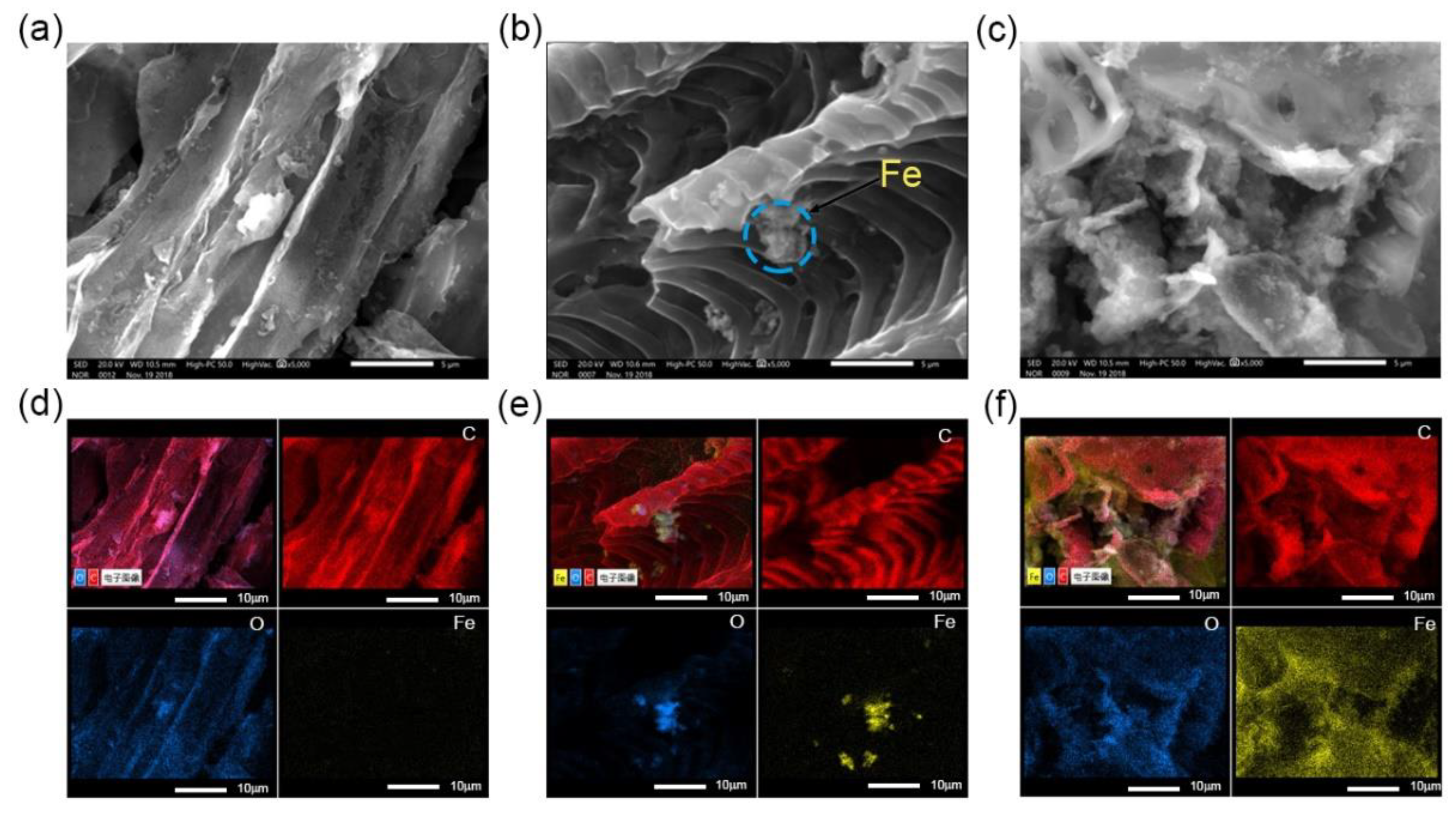
3.1. Characterization of Adsorbents
3.2. Removal of Cr(VI) by Three Biochar Adsorbents
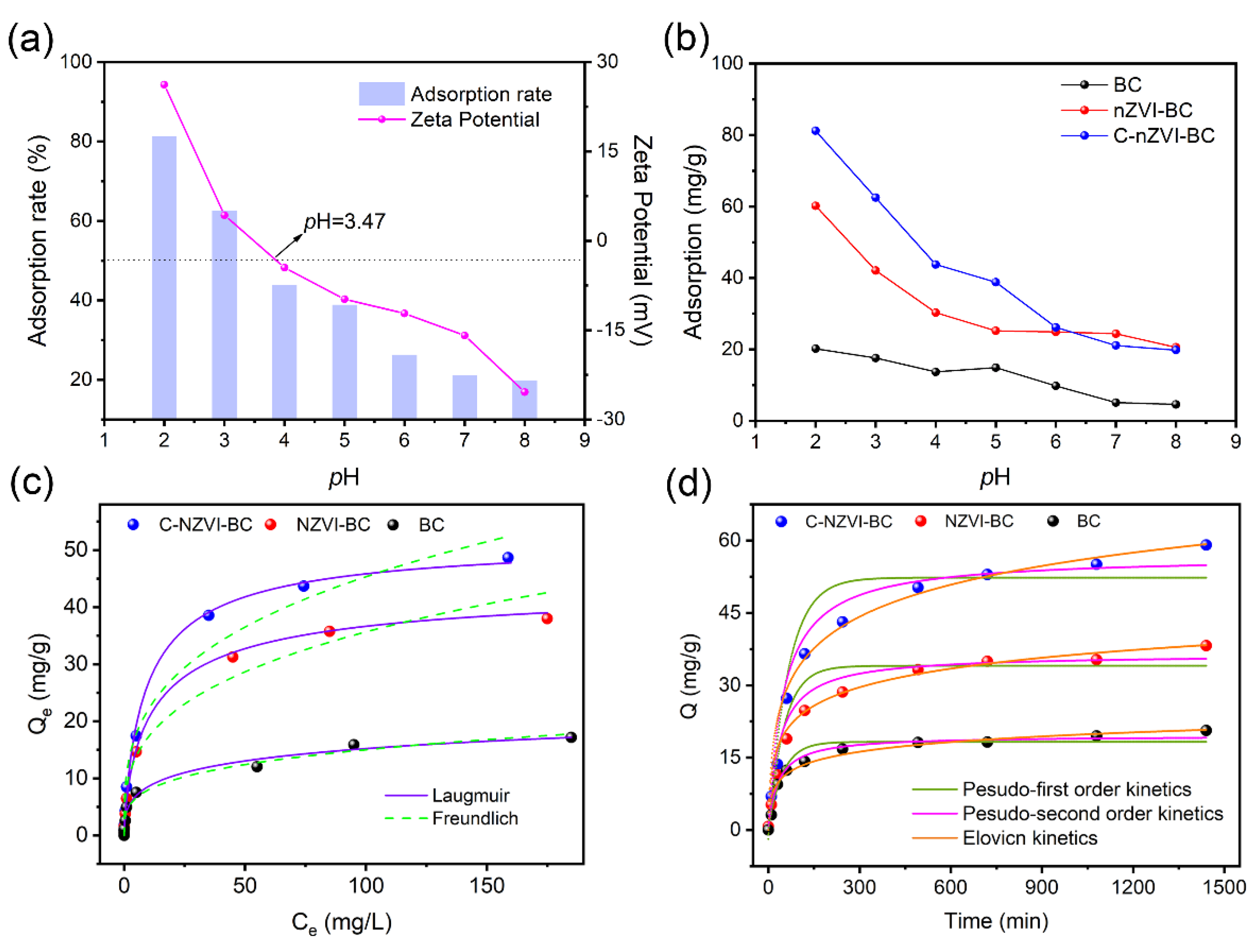
3.2.1. Effect of Solution pH
3.2.2. Effect of Ionic Strength and Temperature
3.3. Sorption Isotherms and Kinetic Study
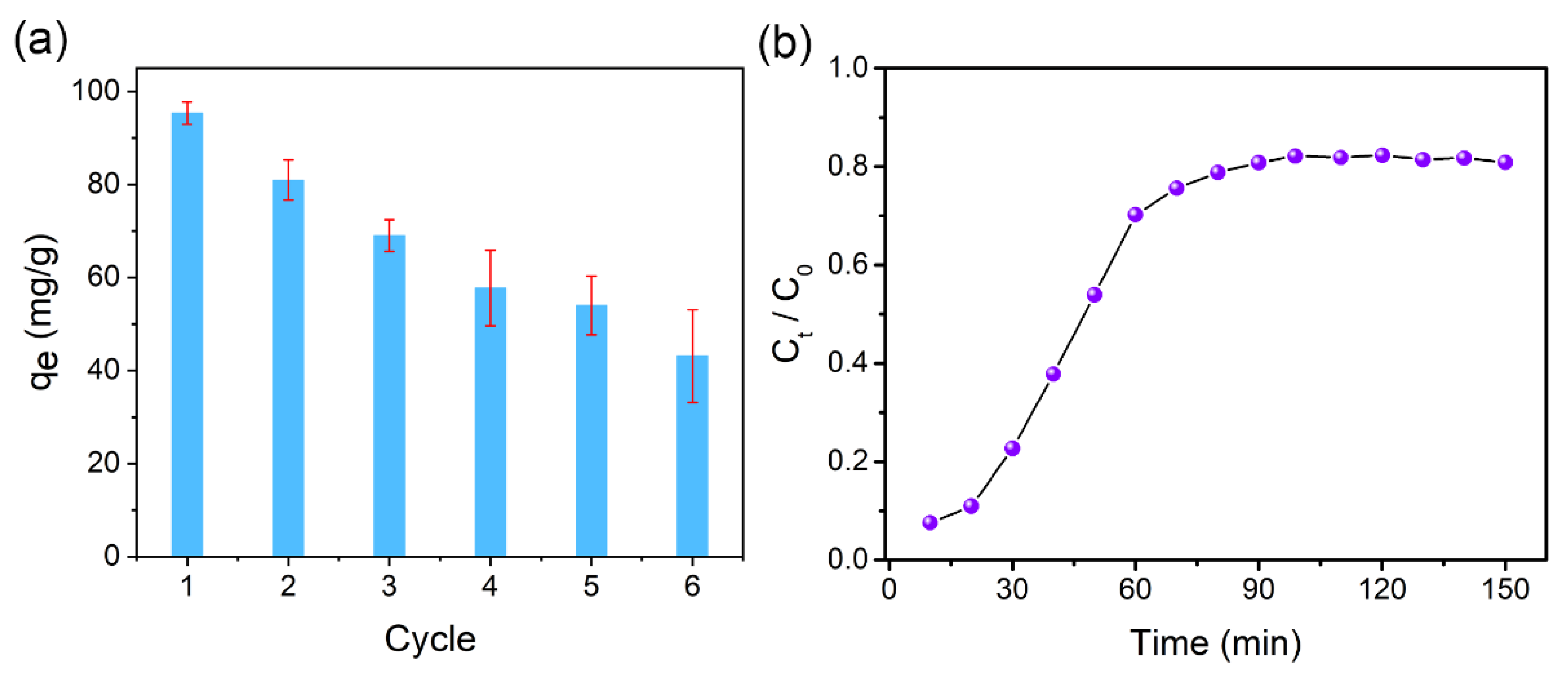
3.4. Reusability of C–nZVI–BC
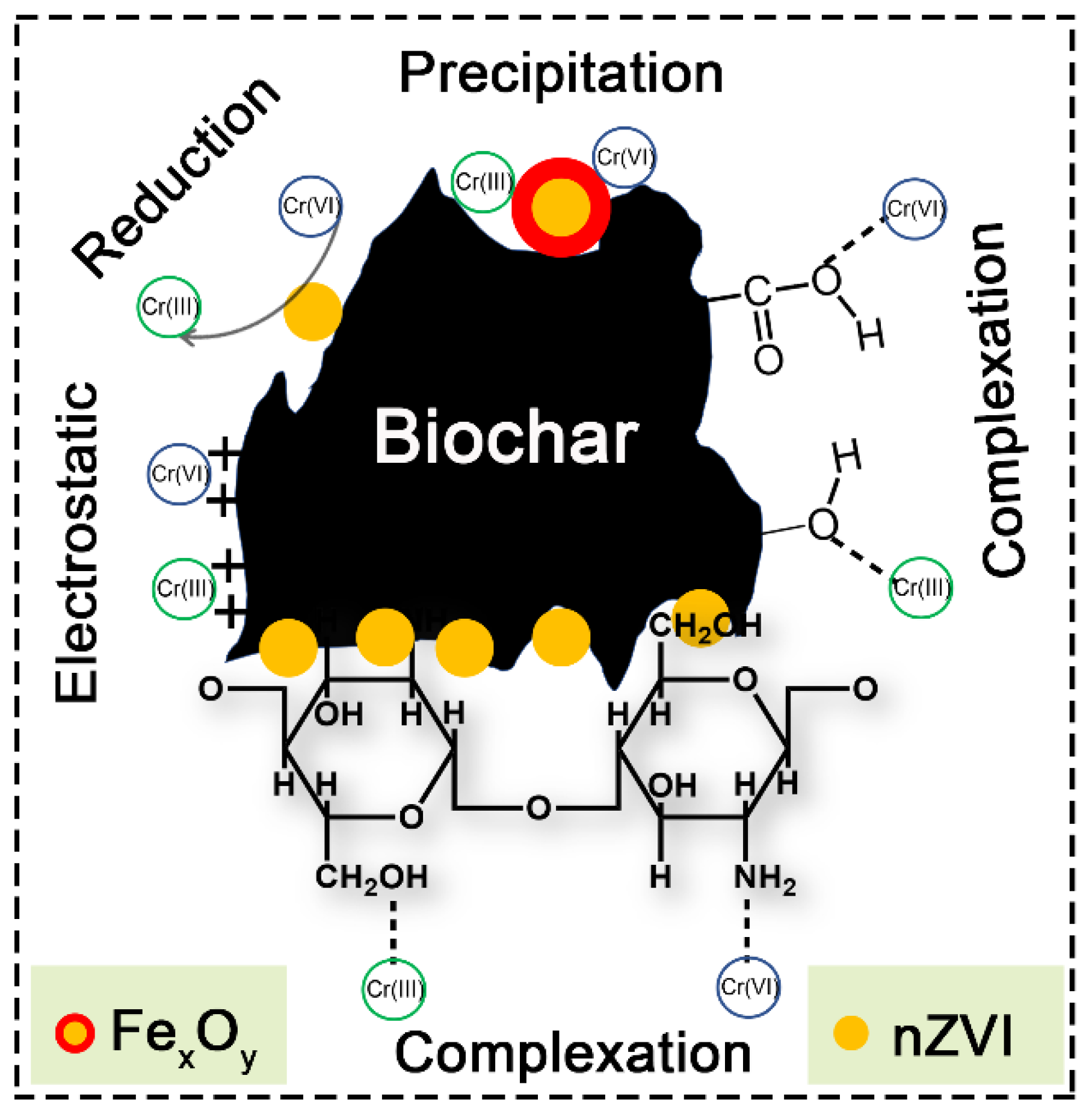
3.5. Sorption Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| E. crassipes | Eichhornia crassipes |
| C | Chitosan |
| nZVI | Nanoscale zero-valent iron |
| BC | Biochar |
References
- Nriagu, J.O.; Nieboer, E. Chromium in the Natural and Human Environments; John Wiley and Sons: New York, NY, USA, 1988. [Google Scholar]
- Huang, J.; Cao, Y.H.; Qin, B.H.; Zhong, G.; Zhang, J.; Yu, H.; Wang, H.J.; Peng, F. Highly efficient and acid-corrosion resistant nitrogen doped magnetic carbon nanotubes for the hexavalent chromium removal with subsequent reutilization. Chem. Eng. J. 2019, 361, 547–558. [Google Scholar] [CrossRef]
- Zou, Y.; Wang, X.; Khan, A.; Wang, P.; Liu, Y.; Alsaedi, A.; Hayat, T.; Wang, X. Environmental remediation and application of nanoscale zero-valent iron and its composites for the removal of heavy metal ions: A review. Environ. Sci. Technol. 2016, 50, 7290–7304. [Google Scholar] [CrossRef] [PubMed]
- Bhaumik, M.; Maity, A.; Srinivasu, V.V.; Onyango, M.S. Enhanced removal of Cr(VI) from aqueous solution using polypyrrole/Fe3O4 magnetic nanocomposite. J. Hazard. Mater. 2011, 190, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Lyu, H.H.; Tang, J.C.; Huang, Y.; Gai, L.S.; Zeng, E.Y.; Liber, K.; Gong, Y.Y. Removal of hexavalent chromium from aqueous solutions by a novel biochar supported nanoscale iron sulfide composite. Chem. Eng. J. 2017, 322, 516–524. [Google Scholar] [CrossRef]
- Nayak, V.; Jyothi, M.S.; Balakrishna, R.G.; Padaki, M.; Deon, S. Novel modified poly vinyl chloride blend membranes for removal of heavy metals from mixed ion feed sample. J. Hazard. Mater. 2017, 331, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Avila, M.; Burks, T.; Akhtar, F.; Gothelid, M.; Lansaker, P.C.; Toprak, M.S.; Muhammed, M.; Uheida, A. Surface functionalized nanofibers for the removal of chromium(VI) from aqueous solutions. Chem. Eng. J. 2014, 245, 201–209. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. An overview of landfill leachate treatment via activated carbon adsorption process. J. Hazard. Mater. 2009, 171, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.K. A review on the adsorption of heavy metals by clay minerals with special focus on the past decade. Chem. Eng. J. 2017, 308, 438–462. [Google Scholar] [CrossRef]
- Herrero-Latorre, C.; Barciela-García, J.; García-Martín, S.; Pena-Crecente, R.M. Graphene and carbon nanotubes as solid phase extraction sorbents for the speciation of chromium: A review. Anal. Chim. Acta 2018, 1002, 1–17. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.X.; Zhao, G.; Chen, C.L.; Chai, Z.F.; Alsaedi, A.; Hayat, T.; Wan, X.K. Metal–organic framework-based materials: Superior adsorbents for the capture of toxic and radioactive metal ions. Chem. Soc. Rev. 2018, 47, 2322. [Google Scholar]
- Inyang, M.I.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.; Mosa, A.; Pullammanappallil, P.; Ok, Y.S.; Cao, X.D. A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Crit. Rev. Environ. Sci. Technol. 2016, 46, 406–433. [Google Scholar] [CrossRef]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotox. Environ. Saf. 2018, 148, 702–712. [Google Scholar] [CrossRef]
- Li, H.B.; Dong, X.L.; da Silva, E.B.; Oliveira, L.M.; Chen, Y.S.; Ma, L.N.Q. Mechanisms of metal sorption by biochars: Biochar characteristics and modifications. Chemosphere 2017, 178, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.Z.; Kumar, A.; Zhang, H.L.; Bellmer, D.; Huhnke, R. Recent advances in utilization of biochar. Renew. Sustain. Energy Rev. 2015, 42, 1055–1064. [Google Scholar] [CrossRef]
- Kong, H.; He, J.; Gao, Y.; Wu, H.; Zhu, X. Cosorption of phenanthrene and mercury (II) from aqueous solution by soybean stalk-based biochar. J. Agric. Food Chem. 2011, 59, 12116–12123. [Google Scholar] [CrossRef]
- Lu, K.P.; Yang, X.; Gielen, G.; Bolan, N.; Ok, Y.S.; Niazi, N.K.; Xu, S.; Yuan, G.D.; Chen, X.; Zhang, X.K.; et al. Effect of bamboo and rice straw biochars on the mobility and redistribution of heavy metals (Cd, Cu, Pb and Zn) in contaminated soil. J. Environ. Manag. 2017, 186, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Bashir, S.; Hussain, Q.; Akmal, M.; Riaz, M.; Hu, H.; Ijaz, S.S.; Iqbal, M.; Abro, S.; Mehmood, S.; Ahmad, M. Sugarcane bagasse-derived biochar reduces the cadmium and chromium bioavailability to mash bean and enhances the microbial activity in contaminated soil. J. Soil. Sediment. 2018, 18, 874–886. [Google Scholar] [CrossRef]
- Patel, S. Threats, management and envisaged utilizations of aquatic weed Eichhornia crassipes: An overview. Rev. Environ. Sci. Bio Technol. 2012, 11, 249–259. [Google Scholar] [CrossRef]
- Guna, V.; Ilangovan, M.M.; Anantha Prasad, G.; Reddy, N. Water Hyacinth: A Unique Source for Sustainable Materials and Products. ACS Sustain. Chem. Eng. 2017, 5, 4478–4490. [Google Scholar] [CrossRef]
- Bai, F.; Chisholm, R.; Sang, W.; Dong, M. Spatial risk assessment of alien invasive plants in China. Environ. Sci. Technol. 2013, 47, 7624–7632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Wang, X.; Yin, D.X.; Peng, B.C.; Tan, Y.; Liu, Y.G.; Tan, X.F.; Wu, S.X. Efficiency and mechanisms of Cd removal from aqueous solution by biochar derived from water hyacinth (Eichornia crassipes). J. Environ. Manag. 2015, 153, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Shen, K.X.; Long, X.L.; Wen, J.S.; Xie, X.J.; Zeng, X.Y.; Liang, Y.Y.; Wei, Y.S.; Lin, Z.F.; Huang, W.R.; et al. Preparation and Characterization of Biochars from Eichornia crassipes for Cadmium Removal in Aqueous Solutions. PLoS ONE 2016, 11, e0148132. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, F.; Wei, Y.S.; Li, G.G.; Shen, K.X.; He, H.J. High cadmium adsorption on nanoscale zero-valent iron coated Eichhornia crassipes biochar. Environ. Chem. Lett. 2019, 17, 589–594. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, Y.G.; Liu, S.B.; Tan, X.F.; Zeng, G.M.; Zeng, W.; Ding, Y.; Cao, W.; Zheng, B.H. Enhanced adsorption of methylene blue by citric acid modification of biochar derived from water hyacinth (Eichornia crassipes). Environ. Sci. Pollut. Res. 2016, 23, 23606–23618. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Wang, B.Y.; Xin, J.; Sun, P.; Wu, D. Adsorption behavior and mechanism of Cr(VI) by modified biochar derived from Enteromorpha prolifera. Ecotox. Environ. Saf. 2018, 164, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.M.; Liu, Y.G.; Li, T.T.; Xu, W.H.; Zheng, B.H.; Tan, X.F.; Wang, H.; Guo, Y.M.; Guo, F.Y.; Wang, S.F. Chitosan modification of magnetic biochar produced from Eichhornia crassipes for enhanced sorption of Cr(VI) from aqueous solution. RSC Adv. 2015, 5, 46955–46964. [Google Scholar] [CrossRef]
- Sun, Q.Z.; Saratale, R.G.; Saratale, G.D.; Kim, D.S. Pristine and modified radix Angelicae dahuricae (Baizhi) residue for the adsorption of methylene blue from aqueous solution: A comparative study. J. Mol. Liq. 2018, 265, 36–45. [Google Scholar] [CrossRef]
- Zhang, S.; Lyu, H.H.; Tang, J.C.; Song, B.; Zhen, M.; Liu, X.M. A novel biochar supported CMC stabilized nano zero-valent iron composite for hexavalent chromium removal from water. Chemosphere 2019, 217, 686–694. [Google Scholar] [CrossRef]
- Gao, J.; Yang, L.Z.; Liu, Y.Y.; Shao, F.L.; Liao, Q.; Shang, J. Scavenging of Cr(VI) from aqueous solutions by sulfide-modified nanoscale zero-valent iron supported by biochar. J. Taiwan Inst. Chem. Eng. 2018, 91, 449–456. [Google Scholar] [CrossRef]
- Toli, A.; Varouxaki, A.; Mystrioti, C.; Xenidis, A.; Papassiopi, N. Green Synthesis of Resin Supported Nanoiron and Evaluation of Efficiency for the Remediation of Cr(VI) Contaminated Groundwater by Batch Tests. Bull. Environ. Contam. Toxicol. 2018, 101, 711–717. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnçrzak, R.; Leahy, J.J.; Hayes, M.H.B.; Kwapiński, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Hu, X.J.; Wang, J.S.; Liu, Y.G.; Li, X.; Zeng, G.M.; Bao, Z.L.; Zeng, X.X.; Chen, A.; Long, W.F. Adsorption of chromium (VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: Isotherms, kinetics and thermodynamics. J. Hazard. Mater. 2011, 185, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Tong, L.H.; Zhao, N.; Li, J.; Lv, Y.Z. Coupling interaction between porous biochar and nano zero valent iron/nano α-hydroxyl iron oxide improves the remediation efficiency of cadmium in aqueous solution. Chemosphere 2019, 219, 493–503. [Google Scholar] [CrossRef]
- Georgieva, V.; Zvezdova, D.; Vlaev, L. Non-isothermal kinetics of thermal degradation of chitosan. Chem. Cent. J. 2012, 6, 81. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Tang, M.; Chen, C.; Chen, M.; Luo, K.; Xu, J.; Zhou, D.; Wu, F. Efficient bacterial inactivation by transition metal catalyzed auto-oxidation of sulfite. Environ. Sci. Technol. 2017, 51, 12663–12671. [Google Scholar] [CrossRef]
- Jin, X.Y.; Zhuang, Z.C.; Yu, B.; Chen, Z.X.; Chen, Z.L. Functional chitosan-stabilized nanoscale zero-valent iron used to remove acid fuchsine with the assistance of ultrasound. Carbohydr. Polym. 2016, 136, 1085–1090. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, Y.G.; Liu, S.B.; Liu, H.Y.; Zeng, G.M.; Tan, X.F.; Yang, C.P.; Ding, Y.; Yan, Z.L.; Cai, X.X. Sorption performance and mechanisms of arsenic(V) removal by magnetic gelatin-modified biochar. Chem. Eng. J. 2017, 314, 223–231. [Google Scholar] [CrossRef]
- Mills, P.; Sullivan, J. A study of the core level electrons in iron and its three oxides by means of X-ray photoelectron spectroscopy. J. Phys. D Appl. Phys. 1983, 16, 723. [Google Scholar] [CrossRef]
- Warwick, M.E.A.; Kaunisto, K.; Carraro, G.; Gasparotto, A.; Maccato, C.; Barreca, D. A Study of Pt/α-Fe2O3 Nanocomposites by XPS. Surf. Sci. Spectra 2015, 22, 47–57. [Google Scholar] [CrossRef]
- Dewage, N.B.; Liyanage, A.S.; Pittman, C.U.; Mohan, D.; Mlsna, T. Fast nitrate and fluoride adsorption and magnetic separation from water on alpha-Fe2O3 and Fe3O4 dispersed on Douglas fir biochar. Bioresour. Technol. 2018, 263, 258–265. [Google Scholar] [CrossRef]
- Hawn, D.D.; de Koven, B.M. Deconvolution as a correction for photoelectron inelastic energy losses in the core level XPS spectra of iron oxides. Surf. Interface Anal. 1987, 10, 63–74. [Google Scholar] [CrossRef]
- Yan, Q.G.; Wan, C.; Liu, X.J.; Gao, J.S.; Yu, F.J.; Zhang, L.; Cai, Z.Y. Iron nanoparticles in situ encapsulated in biochar-based carbon as an effective catalyst for the conversion of biomass-derived syngas to liquid hydrocarbons. Green Chem. 2013, 15, 1631. [Google Scholar] [CrossRef]
- Dong, X.L.; Ma, L.N.Q.; Li, Y.C. Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J. Hazard. Mater. 2011, 190, 909–915. [Google Scholar] [CrossRef]
- Wang, F.; Liu, L.Y.; Liu, F.; Wang, L.G.; Ouyang, T.; Chang, C.T. Facile one-step synthesis of magnetically modified biochar with enhanced removal capacity for hexavalent chromium from aqueous solution. J. Taiwan Inst. Chem. Eng. 2017, 81, 414–418. [Google Scholar] [CrossRef]
- Park, J.H.; Ok, Y.S.; Kim, S.H.; Cho, J.S.; Heo, J.S.; Delaune, R.D.; Seo, D.C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, B.; Li, Y.; Creamer, A.E.; He, F. Adsorptive removal of arsenate from aqueous solutions by biochar supported zero-valent iron nanocomposite: Batch and continuous flow tests. J. Hazard. Mater. 2017, 322, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.J.; Zhang, T.H.; Ren, Q.; Kruse, A.; Cui, R.F. Polyethylene imine modified hydrochar adsorption for chromium (VI) and nickel (II) removal from aqueous solution. Bioresour. Technol. 2018, 247, 370–379. [Google Scholar] [CrossRef]
- Liu, Q.S.; Li, Y.J. Sorption and Reduction of Hexavalent Chromium from Aqueous Solutions by Surface Modified Biochars. Sep. Sci. Technol. 2015, 50, 2617–2624. [Google Scholar] [CrossRef]
- Luo, H.Y.; Lin, Q.T.; Zhang, X.F.; Huang, Z.F.; Liu, S.S.; Jiang, J.R.; Xiao, R.B.; Liao, X.Y. New insights into the formation and transformation of active species in nZVI/BC activated persulfate in alkaline solutions. Chem. Eng. J. 2019, 359, 1215–1223. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, G.G.; Liu, X.W.; Khan, Z.H.; Qiu, W.W.; Song, Z.G. Synthesis and adsorption of Fe-Mn-La-impregnated biochar composite as an adsorbent for As(III) removal from aqueous solutions. Environ. Pollut. 2019, 247, 128–135. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.-L.; Li, F.; Xie, X.J.; Li, Z.; Chen, L. Nanoscale Zero-Valent Iron and Chitosan Functionalized Eichhornia crassipes Biochar for Efficient Hexavalent Chromium Removal. Int. J. Environ. Res. Public Health 2019, 16, 3046. https://doi.org/10.3390/ijerph16173046
Chen X-L, Li F, Xie XJ, Li Z, Chen L. Nanoscale Zero-Valent Iron and Chitosan Functionalized Eichhornia crassipes Biochar for Efficient Hexavalent Chromium Removal. International Journal of Environmental Research and Public Health. 2019; 16(17):3046. https://doi.org/10.3390/ijerph16173046
Chicago/Turabian StyleChen, Xue-Li, Feng Li, Xiao Jie Xie, Zhi Li, and Long Chen. 2019. "Nanoscale Zero-Valent Iron and Chitosan Functionalized Eichhornia crassipes Biochar for Efficient Hexavalent Chromium Removal" International Journal of Environmental Research and Public Health 16, no. 17: 3046. https://doi.org/10.3390/ijerph16173046
APA StyleChen, X.-L., Li, F., Xie, X. J., Li, Z., & Chen, L. (2019). Nanoscale Zero-Valent Iron and Chitosan Functionalized Eichhornia crassipes Biochar for Efficient Hexavalent Chromium Removal. International Journal of Environmental Research and Public Health, 16(17), 3046. https://doi.org/10.3390/ijerph16173046





