Adverse Childhood Experiences and Early Pubertal Timing Among Girls: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion Criteria and Exclusion Criteria
2.3. Risk of Bias Assessment
2.4. Data Screening and Extraction
2.5. Effect Size Computation and Statistical Analyses
3. Results
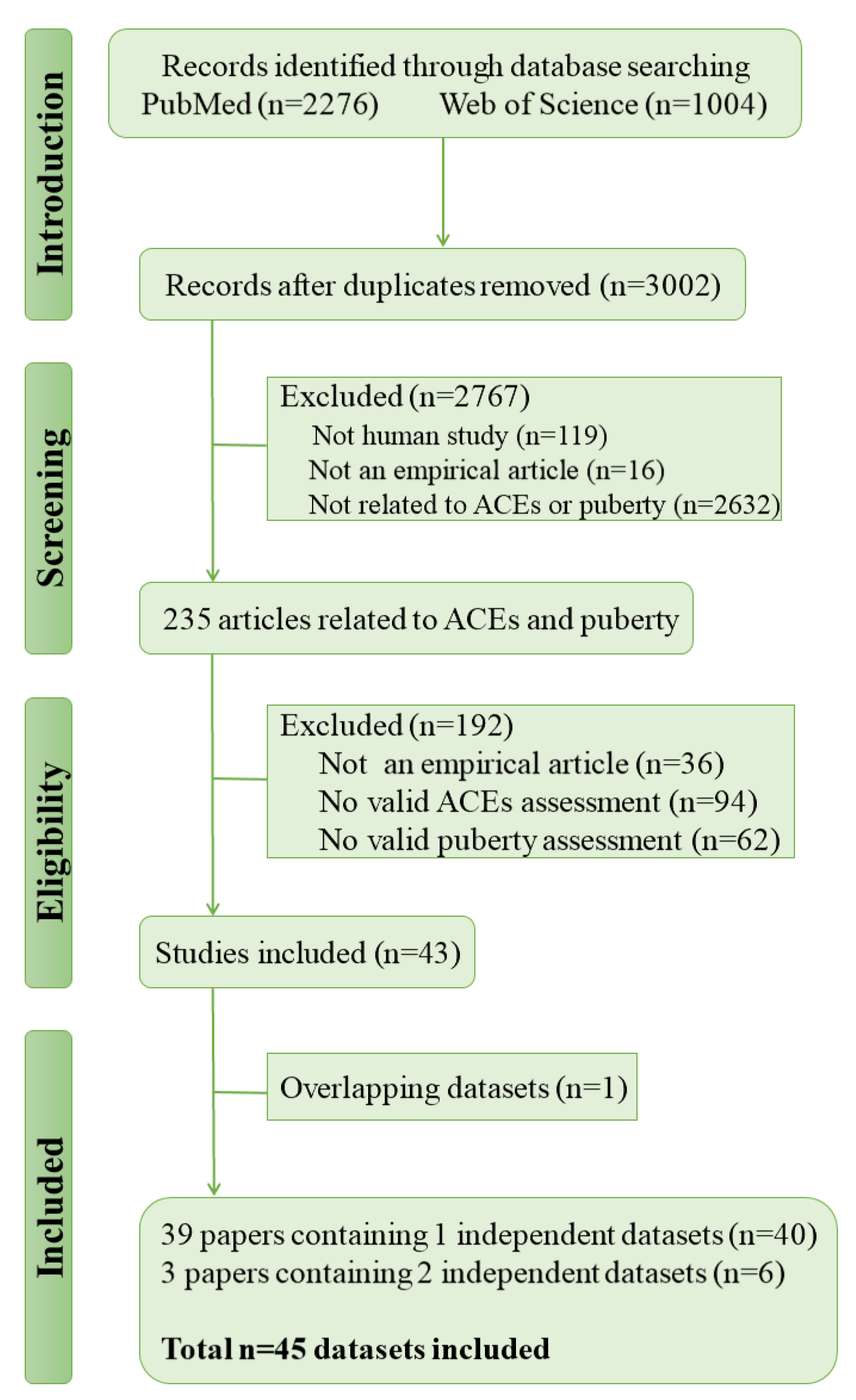
3.1. Search Results
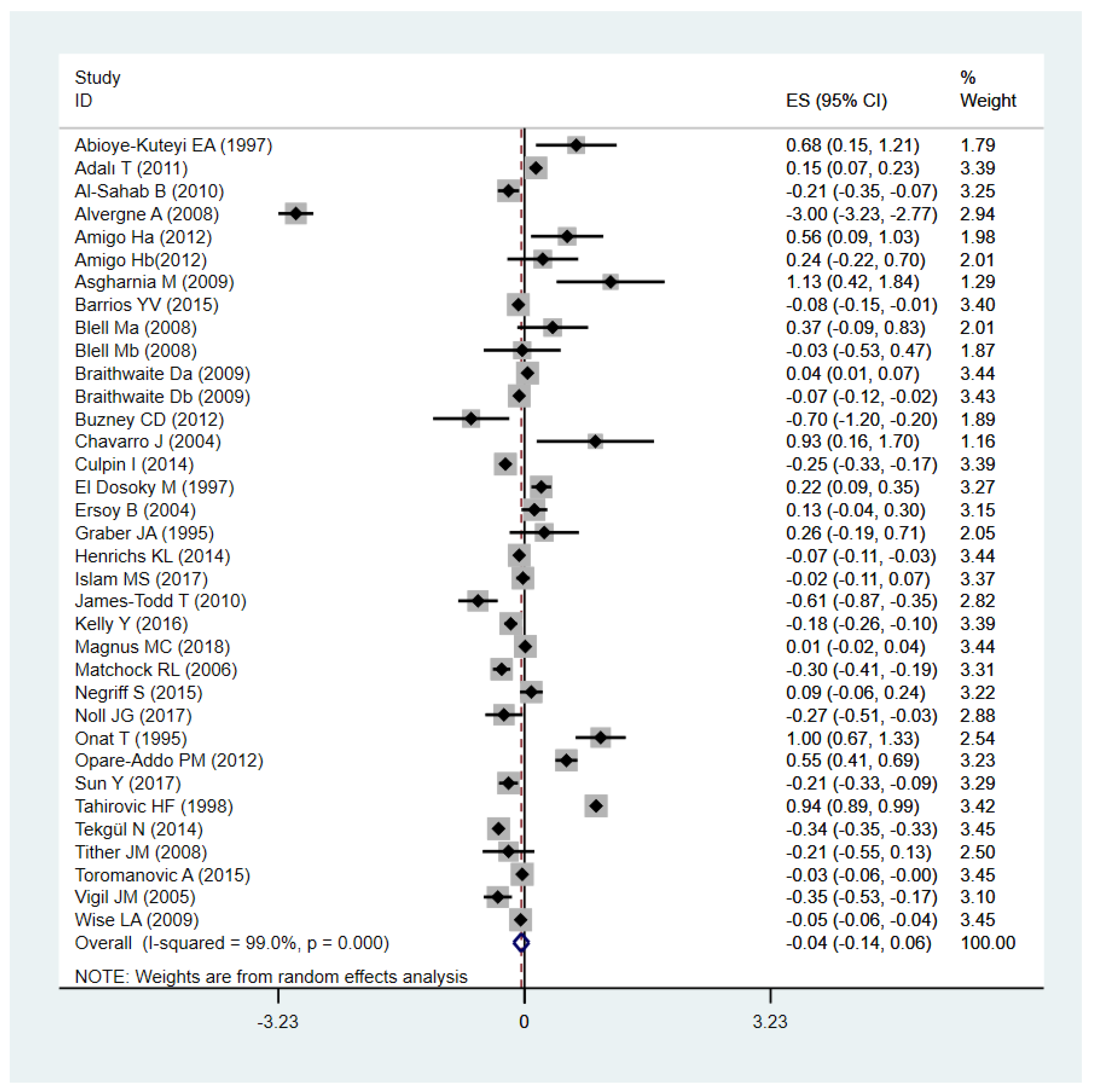
3.2. Association between Total ACEs and Early Pubertal Timing
3.3. Associations between Specific Types of ACEs and Early Pubertal Timing
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Perry, J.R.; Murray, A.; Day, F.R.; Ong, K.K. Molecular insights into the aetiology of female reproductive ageing. Nat. Rev. Endocrinol. 2015, 11, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, K.; Mouritsen, A.; Aksglaede, L.; Hagen, C.P.; Mogensen, S.S.; Juul, A. Recent secular trends in pubertal timing: Implications for evaluation and diagnosis of precocious puberty. Horm. Res. Paediatr. 2012, 77, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Hussain, M.A.; Islam, S.; Mahumud, R.A.; Biswas, T.; Islam, S.M.S. Age at menarche and its socioeconomic determinants among female students in an urban area in Bangladesh. Sex. Reprod. Healthc. 2017, 12, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Elks, C.E.; Ong, K.K.; Scott, R.A.; van der Schouw, Y.T.; Brand, J.S.; Wark, P.A.; Amiano, P.; Balkau, B.; Barricarte, A.; Boeing, H.; et al. Age at menarche and type 2 diabetes risk: The EPIC-InterAct study. Diabetes Care 2013, 36, 3526–3534. [Google Scholar] [CrossRef] [PubMed]
- Canoy, D.; Beral, V.; Balkwill, A.; Wright, F.L.; Kroll, M.E.; Reeves, G.K.; Green, J.; Cairns, B.J.; Million Women Study Collaborators. Age at menarche and risks of coronary heart and other vascular diseases in a large UK cohort. Circulation 2015, 131, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Charalampopoulos, D.; McLoughlin, A.; Elks, C.E.; Ong, K.K. Age at menarche and risks of all-cause and cardiovascular death: A systematic review and meta-analysis. Am. J. Epidemiol. 2014, 180, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Negriff, S.; Hillman, J.B.; Dorn, L.D. Does competence mediate the associations between puberty and internalizing or externalizing problems in adolescent girls? J. Adolesc. Health 2011, 49, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Himes, J.H. Examining the evidence for recent secular changes in the timing of puberty in US children in light of increases in the prevalence of obesity. Mol. Cell. Endocrinol. 2006, 254, 13–21. [Google Scholar] [CrossRef]
- Henrichs, K.L.; McCauley, H.L.; Miller, E.; Styne, D.M.; Saito, N.; Breslau, J. Early menarche and childhood adversities in a nationally representative sample. Int. J. Pediatr. Endocrinol. 2014, 2014, 14. [Google Scholar] [CrossRef]
- Suglia, S.F.; Koenen, K.C.; Boynton-Jarrett, R.; Chan, P.S.; Clark, C.J.; Danese, A.; Faith, M.S.; Goldstein, B.I.; Hayman, L.L.; Isasi, C.R.; et al. Childhood and Adolescent Adversity and Cardiometabolic Outcomes: A Scientific Statement From the American Heart Association. Circulation 2018, 137, e15–e28. [Google Scholar] [CrossRef]
- Kessler, R.C.; McLaughlin, K.A.; Green, J.G.; Gruber, M.J.; Sampson, N.A.; Zaslavsky, A.M.; Aguilar-Gaxiola, S.; Alhamzawi, A.O.; Alonso, J.; Angermeyer, M.; et al. Childhood adversities and adult psychopathology in the WHO World Mental Health Surveys. Br. J. Psychiatry 2010, 197, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Noll, J.G.; Trickett, P.K.; Long, J.D.; Negriff, S.; Susman, E.J.; Shalev, I.; Li, J.C.; Putnam, F.W. Childhood Sexual Abuse and Early Timing of Puberty. J. Adolesc. Health 2017, 60, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Obradović, J.; Hipwell, A. Psychopathology and social competence during the transition to adolescence: The role of family adversity and pubertal development. Dev. Psychopathol. 2010, 22, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Braithwaite, D.; Moore, D.H.; Lustig, R.H.; Epel, E.S.; Ong, K.K.; Rehkopf, D.H.; Wang, M.C.; Miller, S.M.; Hiatt, R.A. Socioeconomic status in relation to early menarche among black and white girls. Cancer Causes Control 2009, 20, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Negriff, S.; Blankson, A.N.; Trickett, P.K. Pubertal Timing and Tempo: Associations with Childhood Maltreatment. J. Res. Adolesc. 2015, 25, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Romans, S.E.; Martin, J.M.; Gendall, K.; Herbison, G.P. Age of menarche: The role of some psychosocial factors. Psychol. Med. 2003, 33, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Ellis, B.J. Timing of pubertal maturation in girls: An integrated life history approach. Psychol. Bull. 2004, 130, 920–958. [Google Scholar] [CrossRef]
- Belsky, J.; Steinberg, L.; Draper, P. Childhood experience, interpersonal development, and reproductive strategy: And evolutionary theory of socialization. Child Dev. 1991, 62, 647–670. [Google Scholar] [CrossRef]
- Ellis, B.J.; Essex, M.J. Family environments, adrenarche, and sexual maturation: A longitudinal test of a life history model. Child Dev. 2007, 78, 1799–1817. [Google Scholar] [CrossRef]
- Mendle, J.; Ryan, R.M.; McKone, K.M. Early Childhood Maltreatment and Pubertal Development: Replication in a Population-Based Sample. J. Res. Adolesc. 2016, 26, 595–602. [Google Scholar] [CrossRef]
- Ruttle, P.L.; Shirtcliff, E.A.; Armstrong, J.M.; Klein, M.H.; Essex, M.J. Neuroendocrine coupling across adolescence and the longitudinal influence of early life stress. Dev. Psychobiol. 2015, 57, 688–704. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Deardorff, J.; Ekwaru, J.P.; Kushi, L.H.; Ellis, B.J.; Greenspan, L.C.; Mirabedi, A.; Landaverde, E.G.; Hiatt, R.A. Father absence, body mass index, and pubertal timing in girls: Differential effects by family income and ethnicity. J. Adolesc. Health 2011, 48, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Ullsperger, J.M.; Nikolas, M.A. A meta-analytic review of the association between pubertal timing and psychopathology in adolescence: Are there sex differences in risk? Psychol. Bull. 2017, 143, 903–938. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.K.; Mertz, D.; Loeb, M. Newcastle-Ottawa Scale: Comparing reviewers’ to authors’ assessments. BMC Med. Res. Methodol. 2014, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Agency for Healthcare Research and Quality. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. Available online: http://effectivehealthcare.ahrq.gov/index.cfm/search-for-guides-reviews-and-reports/?pageaction = displayproduct&productid = 318 (accessed on 13 July 2018).
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: New York, NY, USA, 1998. [Google Scholar]
- Effect Size Calculator. Available online: https://cebcp.org/practical-meta-analysis-effect-size-calculator/standardized-mean-difference-d/means-and-standard-deviations/ (accessed on 13 July 2018).
- Bornstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2009. [Google Scholar]
- Sánchez-Meca, J.; Marín-Martínez, F.; Chacón-Moscoso, S. Effect-size indices for dichotomized outcomes in meta-analysis. Psychol. Methods 2003, 8, 448–467. [Google Scholar] [CrossRef] [PubMed]
- Trotta, A.; Murray, R.M.; Fisher, H.L. The impact of childhood adversity on the persistence of psychotic symptoms: A systematic review and meta-analysis. Psychol. Med. 2015, 45, 2481–2498. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Abioye-Kuteyi, E.A.; Ojofeitimi, E.O.; Aina, O.I.; Kio, F.; Aluko, Y.; Mosuro, O. The influence of socioeconomic and nutritional status on menarche in Nigerian school girls. Nutr. Health 1997, 11, 185–195. [Google Scholar] [CrossRef]
- Adalı, T.; Koç, I. Menarcheal age in Turkey: Secular trend and socio-demographic correlates. Ann. Hum. Biol. 2011, 38, 345–353. [Google Scholar] [CrossRef]
- Al-Sahab, B.; Ardern, C.I.; Hamadeh, M.J.; Tamim, H. Age at menarche in Canada: Results from the National Longitudinal Survey of Children & Youth. BMC Public Health 2010, 10, 736. [Google Scholar] [CrossRef][Green Version]
- Alvergne, A.; Faurie, C.; Raymond, M. Developmental plasticity of human reproductive development: Effects of early family environment in modern-day France. Physiol. Behav. 2008, 95, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Amigo, H.; Vásquez, S.; Bustos, P.; Ortiz, G.; Lara, M. Socioeconomic status and age at menarche in indigenous and non-indigenous Chilean adolescents. Cad. Saude Publica 2012, 28, 977–983. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Asgharnia, M.; Faraji, R.; Sharami, H.; Yadak, M.; Oudi, M. A study of menarcheal age in northern iran (rasht). Oman Med. J. 2009, 24, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Barrios, Y.V.; Sanchez, S.E.; Nicolaidis, C.; Garcia, P.J.; Gelaye, B.; Zhong, Q.; Williams, M.A. Childhood abuse and early menarche among Peruvian women. J. Adolesc. Health. 2015, 56, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Bleil, M.E.; Adler, N.E.; Appelhans, B.M.; Gregorich, S.E.; Sternfeld, B.; Cedars, M.I. Childhood adversity and pubertal timing: Understanding the origins of adulthood cardiovascular risk. Biol. Psychol. 2013, 93, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Blell, M.; Pollard, T.M.; Pearce, M.S. Predictors of age at menarche in the newcastle thousand families study. J. Biosoc. Sci. 2008, 40, 563–575. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Boynton-Jarrett, R.; Harville, E.W. A prospective study of childhood social hardships and age at menarche. Ann. Epidemiol. 2012, 22, 731–737. [Google Scholar] [CrossRef]
- Boynton-Jarrett, R.; Wright, R.J.; Putnam, F.W.; Hibert, E.L.; Michels, K.B.; Forman, M.R.; Rich-Edwards, J. Childhood abuse and age at menarche. J. Adolesc. Health 2013, 52, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Buzney, C.D.; DeCaro, J.A. Explanatory models of female pubertal timing: Discordances between cultural models of maturation and the recollection and interpretation of personal developmental experiences. Cult. Med. Psychiatry 2012, 36, 601–620. [Google Scholar] [CrossRef]
- Chavarro, J.; Villamor, E.; Narváez, J.; Hoyos, A. Socio-demographic predictors of age at menarche in a group of Colombian university women. Ann. Hum. Biol. 2004, 31, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Culpin, I.; Heron, J.; Araya, R.; Melotti, R.; Lewis, G.; Joinson, C. Father absence and timing of menarche in adolescent girls from a UK cohort: The mediating role of maternal depression and major financial problems. J. Adolesc. 2014, 37, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Junqueira Do Lago, M.; Faerstein, E.; De Souza Lopes, C.; Werneck, G.L. Family socio-economic background modified secular trends in age at menarche: Evidence from the Pró-Saúde Study (Rio de Janeiro, Brazil). Ann. Hum. Biol. 2003, 30, 347–352. [Google Scholar] [CrossRef] [PubMed]
- El Dosoky, M.; Al Amoudi, F. Menarcheal age of school girls in the city of Jeddah, Saudia Arabia. J. Obstet. Gynaecol. 1997, 17, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Ersoy, B.; Balkan, C.; Gunay, T.; Onag, A.; Egemen, A. Effects of different socioeconomic conditions on menarche in Turkish female students. Early Hum. Dev. 2004, 76, 115–125. [Google Scholar] [CrossRef]
- Graber, J.A.; Brooks-Gunn, J.; Warren, M.P. The antecedents of menarcheal age: Heredity, family environment, and stressful life events. Child Dev. 1995, 66, 346–359. [Google Scholar] [CrossRef] [PubMed]
- James-Todd, T.; Tehranifar, P.; Rich-Edwards, J.; Titievsky, L.; Terry, M.B. The impact of socioeconomic status across early life on age at menarche among a racially diverse population of girls. Ann. Epidemiol. 2010, 20, 836–842. [Google Scholar] [CrossRef]
- Jorm, A.F.; Christensen, H.; Rodgers, B.; Jacomb, P.A.; Easteal, S. Association of adverse childhood experiences, age of menarche, and adult reproductive behavior: Does the androgen receptor gene play a role? Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2004, 125, 105–111. [Google Scholar] [CrossRef]
- Kelly, Y.; Zilanawala, A.; Sacker, A.; Hiatt, R.; Viner, R. Early puberty in 11-year-old girls: Millennium Cohort Study findings. Arch. Dis. Child. 2017, 102, 232–237. [Google Scholar] [CrossRef]
- Magnus, M.C.; Anderson, E.L.; Howe, L.D.; Joinson, C.J.; Penton-Voak, I.S.; Fraser, A. Childhood psychosocial adversity and female reproductive timing: A cohort study of the ALSPAC mothers. J. Epidemiol. Community Health 2018, 72, 34–40. [Google Scholar] [CrossRef]
- Matchock, R.L.; Susman, E.J. Family composition and menarcheal age: Anti-inbreeding strategies. Am. J. Hum. Biol. 2006, 18, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Mendle, J.; Turkheimer, E.; D’Onofrio, B.M.; Lynch, S.K.; Emery, R.E.; Slutske, W.S.; Martin, N.G. Family structure and age at menarche: A children-of-twins approach. Dev. Psychol. 2006, 42, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Moffitt, T.E.; Caspi, A.; Belsky, J.; Silva, P.A. Childhood experience and the onset of menarche: A test of a sociobiological model. Child Dev. 1992, 63, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Onat, T.; Ertem, B. Age at menarche: Relationships to socioeconomic status, growth rate in stature and weight, and skeletal and sexual maturation. Am. J. Hum. Biol. 1995, 7, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Opare-Addo, P.M.; Stowe, M.; Ankobea-Kokroe, F.; Zheng, T. Menarcheal and pubertal development and determining factors among schoolgirls in Kumasi, Ghana. J. Obstet. Gynaecol. 2012, 32, 159–165. [Google Scholar] [CrossRef]
- Sun, Y.; Mensah, F.K.; Azzopardi, P.; Patton, G.C.; Wake, M. Childhood Social Disadvantage and Pubertal Timing: A National Birth Cohort from Australia. Pediatrics 2017, 139, e20164099. [Google Scholar] [CrossRef]
- Tahirović, H.F. Menarchal age and the stress of war: An example from Bosnia. Eur. J. Pediatr. 1998, 157, 978–980. [Google Scholar] [CrossRef]
- Tekgül, N.; Saltık, D.; Vatansever, K. Secular trend of menarche age in an immigrant urban city in Turkey: Izmir. Turk. J. Pediatr. 2014, 56, 138–143. [Google Scholar]
- Tither, J.M.; Ellis, B.J. Impact of fathers on daughters’ age at menarche: A genetically and environmentally controlled sibling study. Dev. Psychol. 2008, 44, 1409–1420. [Google Scholar] [CrossRef]
- Toromanović, A.; Tahirović, H. Effect of family disintegration on age at menarche. Acta Med. Acad. 2015, 44, 124–134. [Google Scholar] [CrossRef]
- Vigil, J.M.; Geary, D.C.; Byrd-Craven, J. A life history assessment of early childhood sexual abuse in women. Dev. Psychol. 2005, 41, 553–561. [Google Scholar] [CrossRef]
- Wise, L.A.; Palmer, J.R.; Rothman, E.F.; Rosenberg, L. Childhood abuse and early menarche: Findings from the black women’s health study. Am. J. Public Health 2009, 99 (Suppl 2), S460–S466. [Google Scholar] [CrossRef]
- Herman-Giddens, M.E.; Slora, E.J.; Wasserman, R.C.; Bourdony, C.J.; Bhapkar, M.V.; Koch, G.G.; Hasemeier, C.M. Secondary sexual characteristics and menses in young girls seen in office practice: A study from the Pediatric Research in Office Settings network. Pediatrics 1997, 99, 505–512. [Google Scholar] [CrossRef]
- Draper, P.; Harpending, H. Father absence and reproductive strategy: An evolutionary perspective. J. Anthropol. Res. 1982, 38, 255–273. [Google Scholar] [CrossRef]
- Ellis, B.J.; McFadyen-Ketchum, S.; Dodge, K.A.; Pettit, G.S.; Bates, J.E. Quality of early family relationships and individual differences in the timing of pubertal maturation in girls: A longitudinal test of an evolutionary model. J. Personal. Soc. Psychol. 1999, 77, 387–401. [Google Scholar] [CrossRef]
| Study | Study Design | N | Age (Year) | ACEs Type | ACEs Measure | Puberty Measure |
|---|---|---|---|---|---|---|
| Abioye-Kuteyi EA (1997) [33] | C | 60 | 14.2 | low SES | questionnaire | AAM |
| Adalı T(2011) [34] | C | 2789 | 15–49 | low SES | combination | AAM |
| Al-Sahab B (2010) [35] | C | 1403 | 14–17 | low SES | questionnaire | NO. |
| Alvergne A (2008) [36] | C | 708 | 20.9 | FB | questionnaire | AAM |
| Amigo H (a) (2012) [37] | C | 127 | 8–16 | low SES | interview | AAM |
| Amigo H (b) (2012) [37] | C | 114 | 8–16 | low SES | interview | AAM |
| Asgharnia M (2009) [38] | C | 91 | 11–16 | low SES | combination | AAM |
| Barrios YV (2015) [39] | C | 1499 | 28 | PA, SA | questionnaire | NO. |
| Bleil ME (2013) [40] | C | 650 | 34.9 | FD | combination | AAM |
| Blell M (a) (2008) [41] | L | 94 | 49–51 | low SES | questionnaire | AAM |
| Blell M (b) (2008) [41] | L | 106 | 49–51 | low SES | questionnaire | AAM |
| Boynton-Jarrett R (2012) [42] | L | 4524 | 16 | PA, SA, neglect | combination | NO. |
| Boynton-Jarrett R (2013) [43] | L | 67,658 | 25–44 | abuse | questionnaire | NO. |
| Braithwaite D (a) (2009) [14] | L | 1091 | 18–19 | low SES | interview | NO. |
| Braithwaite D (b) (2009) [14] | L | 986 | 18–19 | low SES | interview | NO. |
| Buzney CD (2012) [44] | C | 70 | 19–25 | low SES | combination | AAM |
| Chavarro J (2004) [45] | C | 30 | 15−42 | low SES | questionnaire | AAM |
| Culpin I (2014) [46] | L | 3785 | 8–17 | FB | questionnaire | AAM |
| Deardorff J (2011) [23] | L | 444 | 8–10 | FB, low SES | interview | Tanner staging |
| Junqueira DLM (2003) [47] | C | 2053 | 9–19 | low SES | questionnaire | AAM |
| El Dosoky M (1997) [48] | C | 929 | 9–18 | low SES | questionnaire | AAM |
| Ersoy B (2004) [49] | C | 534 | 15.7 | low SES | combination | AAM |
| Graber JA (1995) [50] | L | 75 | 11.93 | FB, low SES | questionnaire | AAM |
| Henrichs KL (2014) [9] | C | 3288 | 45.7 | PA, SA, neglect, low SES, FB, FD | interview | NO. |
| Islam MS (2017) [3] | C | 680 | 14 | low SES | questionnaire | NO. |
| James-Todd T (2010) [51] | L | 237 | —a | low SES | questionnaire | AAM |
| Jorm AF (2004) [52] | C | 3702 | —a | PA, SA, neglect, FB | questionnaire | AAM |
| Kelly Y (2016) [53] | L | 5839 | 11.2 | low SES | interview | PDS |
| Magnus MC (2018) [54] | L | 8984 | 28.5 | Total adversity, SA, FD | questionnaire | AAM |
| Matchock RL (2006) [55] | C | 1896 | 20 | FB | questionnaire | AAM |
| Mendle J (2006) [56] | C | 1284 | 24.5 | FB, FD | interview | AAM |
| Mendle J (2016) [20] | L | 6273 | 28.7 | SA, PA, neglect | interview | AAM |
| Moffitt TE (1992) [57] | L | 416 | 15 | FB, low SES, FD | combination | AAM |
| Negriff S (2015) [15] | L | 213 | 8–13 | SA | questionnaire | PDS |
| Noll JG (2017) [12] | L | 173 | 6–16 | SA | interview | Tanner staging |
| Onat T (1995) [58] | L | 169 | 8.5–13.4 | low SES | questionnaire | AAM |
| Opare-Addo PM (2012) [59] | C | 720 | 7–17 | low SES | questionnaire | AAM |
| Romans SE (2003) [16] | C | 488 | 39.1 | low SES, PA, SA, FB | interview | NO. |
| Sun Y(2017) [60] | L | 1770 | 10–11 | low SES | questionnaires | PDS |
| Tahirovic HF (1998) [61] | C | 6077 | 8–17 | War | questionnaire | AAM |
| Tekgül N (2014) [62] | C | 61,293 | 15–49 | low SES | interview | AAM |
| Tither JM (2008) [63] | C | 136 | 16–44 | FB | questionnaire | AAM |
| Toromanović A (2015) [64] | C | 22,469 | 9–17.5 | FD | questionnaire | AAM |
| Vigil JM (2005) [65] | C | 616 | 26.9 | SA | questionnaire | AAM |
| Wise LA (2009) [66] | C | 35,330 | M = 38 | PA, SA | questionnaire | NO. |
| Types of Adversity | K | Cohen’s d (95% CI) | I2 (%), p Value |
|---|---|---|---|
| Sexual abuse | 12 | −0.14(−0.18, −0.11) | 72.4, <0.001 |
| Physical abuse | 8 | −0.03 (−0.07,0.01) | 91.5, <0.001 |
| Neglect | 4 | 0.02 (−0.1,0.14) | 72.8, 0.011 |
| Low SES | 25 | 0.07 (−0.03,0.18) | 97.4, <0.001 |
| Father absence | 12 | −0.40 (−0.63, −0.16) | 98.2, <0.001 |
| Family dysfunction | 11 | −0.08 (−0.11,−0.04) | 66.9, 0.001 |
| Moderator | Cohen’s d | 95% Confidence Interval | p-Value | I2 (%) | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| ACEs measure | |||||
| questionnaire | −0.06 | −0.22 | 0.10 | 0.493 | 99.1 |
| interview | −0.07 | −0.23 | 0.09 | 0.382 | 98.7 |
| combination | 0.03 | −0.14 | 0.20 | 0.728 | 86.3 |
| Puberty measure | |||||
| No. of early menarche | −0.050 | −0.090 | −0.011 | 0.012 | 81.3 |
| PDS | −0.109 | −0.268 | 0.050 | 0.180 | 82.1 |
| Tanner staging | −0.270 | −0.515 | −0.025 | 0.031 | — |
| age at menarche | 0.013 | −0.172 | 0.199 | 0.889 | 99.3 |
| Original variable type | |||||
| categorical | −0.073 | −0.114 | −0.031 | 0.001 | 82.1 |
| continuous | −0.011 | −0.254 | 0.233 | 0.931 | 99.5 |
| others | 0.098 | −0.137 | 0.333 | 0.415 | 65.3 |
| Study design | |||||
| cross-sectional | −0.050 | −0.194 | 0.094 | 0.492 | 99.3 |
| longitudinal | −0.051 | −0.141 | 0.038 | 0.262 | 91.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Zhang, D.; Sun, Y. Adverse Childhood Experiences and Early Pubertal Timing Among Girls: A Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 2887. https://doi.org/10.3390/ijerph16162887
Zhang L, Zhang D, Sun Y. Adverse Childhood Experiences and Early Pubertal Timing Among Girls: A Meta-Analysis. International Journal of Environmental Research and Public Health. 2019; 16(16):2887. https://doi.org/10.3390/ijerph16162887
Chicago/Turabian StyleZhang, Lei, Dandan Zhang, and Ying Sun. 2019. "Adverse Childhood Experiences and Early Pubertal Timing Among Girls: A Meta-Analysis" International Journal of Environmental Research and Public Health 16, no. 16: 2887. https://doi.org/10.3390/ijerph16162887
APA StyleZhang, L., Zhang, D., & Sun, Y. (2019). Adverse Childhood Experiences and Early Pubertal Timing Among Girls: A Meta-Analysis. International Journal of Environmental Research and Public Health, 16(16), 2887. https://doi.org/10.3390/ijerph16162887






