Oxidative Stress Levels Induced by Mercury Exposure in Amazon Juvenile Populations in Brazil
Abstract
1. Introduction
2. Materials and Methods
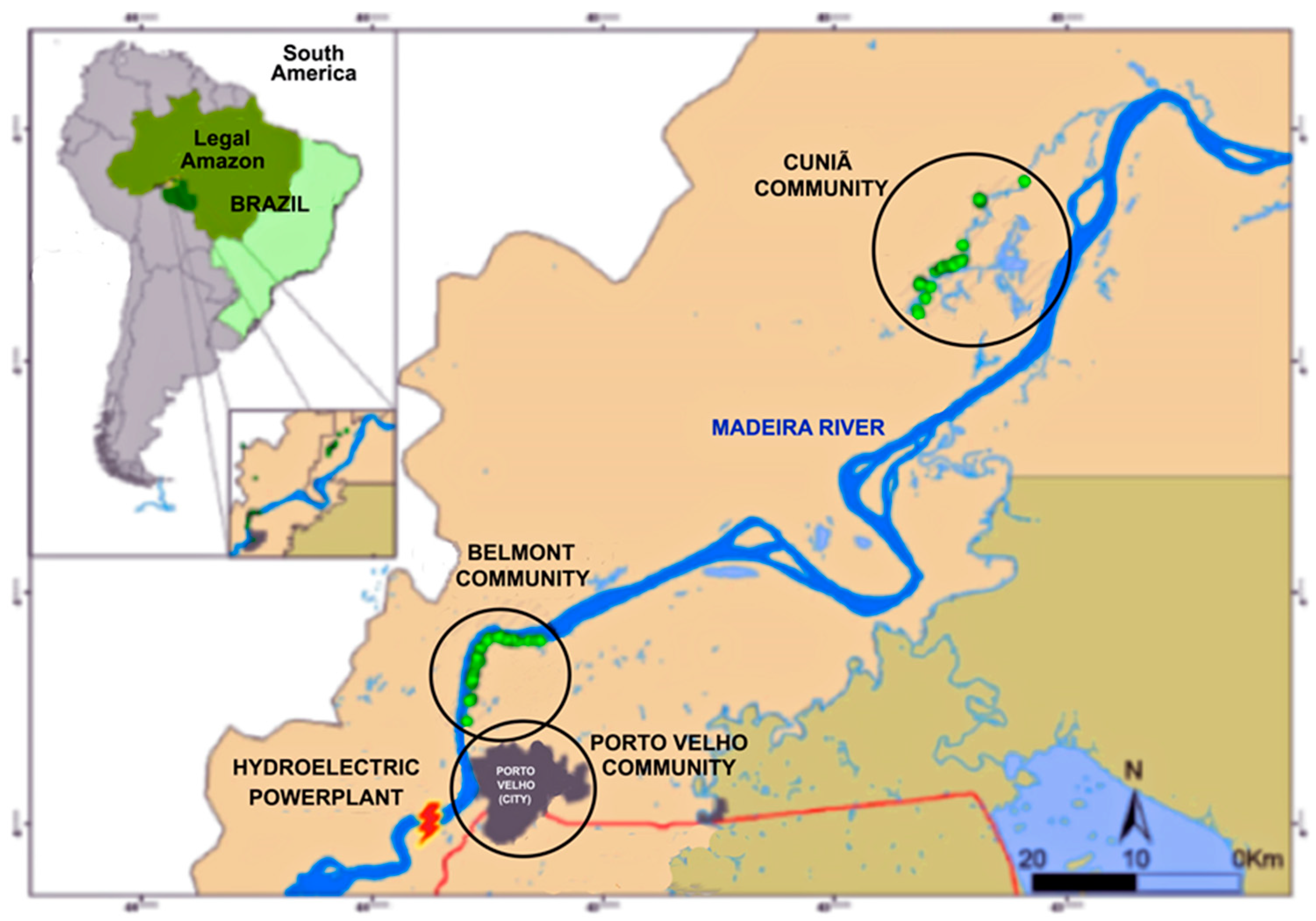
2.1. Study Population
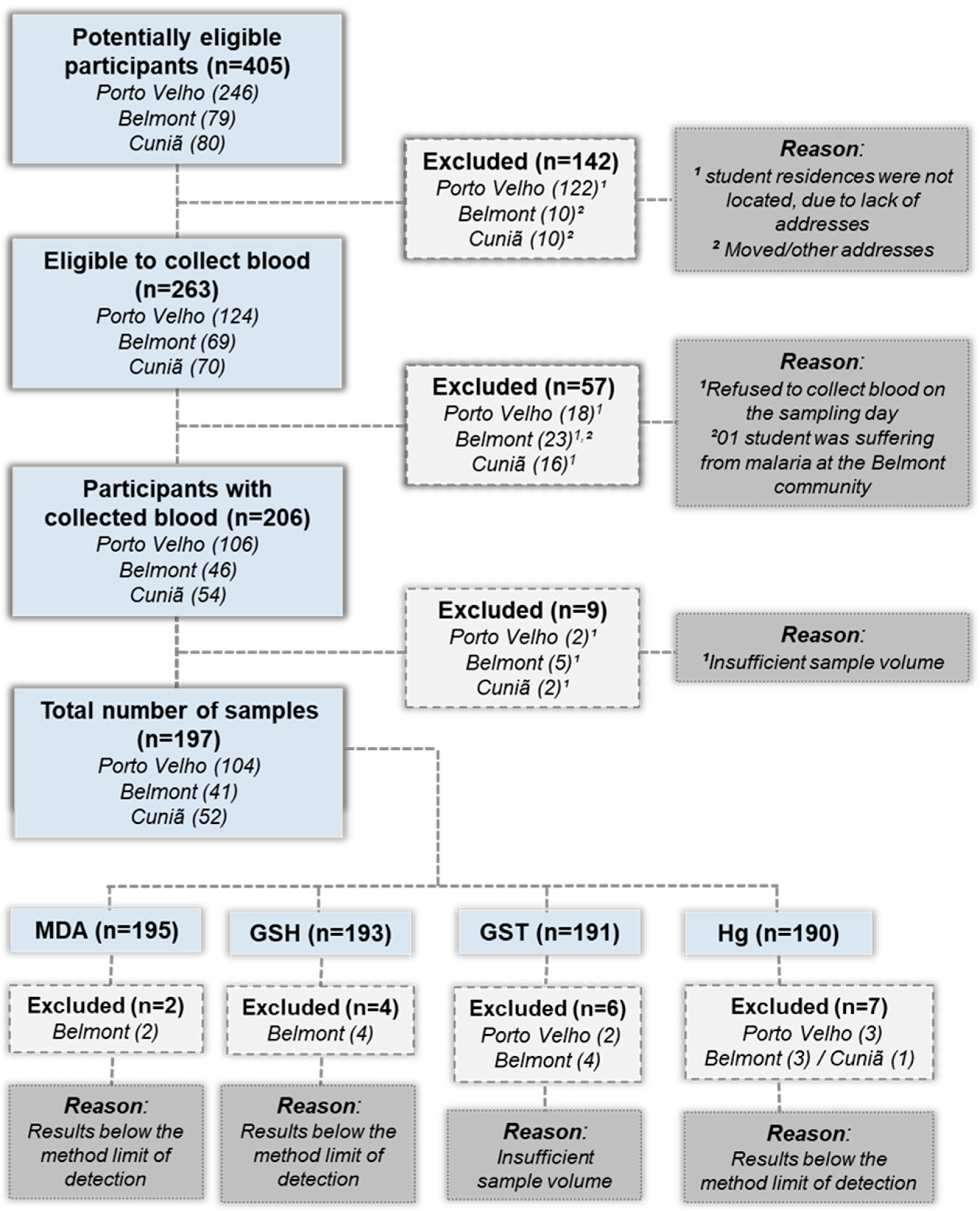
2.2. Study Design and Population
2.3. Collection and Fractionation of Blood Samples
2.4. Laboratory Analyses
2.4.1. Oxidative Stress Analyses
Determination of GST Activity
Determination of GSH Levels
Determination of MDA Levels
2.4.2. Determination of Hg Levels
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sies, H. Oxidative Stress: Introductory Remarks. In Oxidative Stress; Elsevier Academic Press INC: London, UK, 1985; pp. 1–8. [Google Scholar] [CrossRef]
- Vasconcelos, S.M.L.; Goulart, M.O.F.; Moura, J.B.D.F.; Manfredini, V.; Benfato, M.S.; Kubota, L.T. Espécies Reativas de Oxigênio e de Nitrogênio, Antioxidantes e Marcadores de Dano Oxidativo Em Sangue Humano: Principais Métodos Analíticos Para Sua Determinação. Quim. Nova 2007, 30, 1323–1338. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of Oxidative Damage in Human Disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Lushchak, V.I. Free Radicals, Reactive Oxygen Species, Oxidative Stress and Its Classification. Chem. Biol. Interact. 2014, 224, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Rover Júnior, L.; Höehr, N.F.; Vellasco, A.P.; Kubota, L.T. Sistema Antioxidante Envolvendo o Ciclo Metabólico Da Glutationa Associado a Métodos Eletroanalíticos Na Avaliação Do Estresse Oxidativo. Quim. Nova 2001, 24, 112–119. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress: Oxidants and Antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Amorim, L.C.A. Os Biomarcadores e Sua Aplicação Na Avaliação Da Exposição Aos Agentes Químicos Ambientais. Rev. Bras. Epidemiol. 2003, 6, 158–170. [Google Scholar] [CrossRef]
- Limón-Pacheco, J.; Gonsebatt, M.E. The Role of Antioxidants and Antioxidant-Related Enzymes in Protective Responses to Environmentally Induced Oxidative Stress. Mutat. Genet. Toxicol. Environ. Mutagen. 2009, 674, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, C.D.; Watkins III, J.B. Fundamentos Em Toxicologia de Casarett e Doull, 2nd ed.; AMGH Editora Ltda: Porto Alegre, Brazil, 2012. [Google Scholar]
- Oga, S.; Camargo, M.M.A.; Batistuzzo, J.A.O. Fundamentos de Toxicologia, 3rd ed.; Atheneu Editora: São Paulo, Brazil, 2008. [Google Scholar]
- Pivetta, F.; Machado, J.M.H.; Araújo, U.C.; Moreira, M.D.F.R.; Apostoli, P. Monitoramento Biológico: Conceitos e Aplicações Em Saúde Pública. Cadernos de Saúde Pública 2001, 17, 545–554. [Google Scholar] [CrossRef]
- Farina, M.; Avila, D.S.; da Rocha, J.B.T.; Aschner, M. Metals, Oxidative Stress and Neurodegeneration: A Focus on Iron, Manganese and Mercury. Neurochem. Int. 2013, 62, 575–594. [Google Scholar] [CrossRef] [PubMed]
- Grotto, D.; Valentini, J.; Fillion, M.; Passos, C.J.S.; Garcia, S.C.; Mergler, D.; Barbosa, F. Mercury Exposure and Oxidative Stress in Communities of the Brazilian Amazon. Sci. Total Environ. 2010, 408, 806–811. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Biological Monitoring of Chemical Exposure in the Workplace: Guidelines—Vol. 1; WHO, Contribution to the International Programme on Chemical Safety (IPCS): Geneva, Switzerland, 1996. [Google Scholar]
- Agency for Toxic Substances and Disease Registry. (ATSDR)—Toxicological Profile: Mercury; Atlanta, GA, USA, 1999.
- United Nations Environment Programme; International Labour Organization; World Health Organization (WHO). Methylmercury; International Program on Chemical Safety: Geneva, Switzerland, 1990. [Google Scholar]
- World Health Organization (WHO). Mercury Flyer—Exposure to Mercury; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- World Health Organization (WHO); United Nations Environment Programme (UNEP). Guidance for Identifying Populations at Risk from Mercury Exposure; WHO: Geneva, Switzerland, 2008; p. 167. [Google Scholar]
- Carneiro, M.F.H.; Grotto, D.; Barbosa, F. Inorganic and Methylmercury Levels in Plasma Are Differentially Associated with Age, Gender, and Oxidative Stress Markers in a Population Exposed to Mercury through Fish Consumption. J. Toxicol. Environ. Health Part A 2014, 77, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Méndez, J.A.; Arcega-Cabrera, F.E.; Fargher, L.F.; Moo-Puc, R.E. Mercury Levels Assessment and Its Relationship with Oxidative Stress Biomarkers in Children from Three Localities in Yucatan, Mexico. Sci. Total Environ. 2016, 543 Pt A, 187–196. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Garg, A. Chronic Effects of Toxic Environmental Exposures on Children’s Health. J. Toxicol. Clin. Toxicol. 2002, 40, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Mello-da-Silva, C.A.; Fruchtengarten, L. Riscos Químicos Ambientais à Saúde Da Criança. J. Pediatr. 2005, 81, S205–S211. [Google Scholar] [CrossRef]
- Hacon, S.H.S.; Barrocas, P.R.G.; Vasconcellos, A.C.S.D.; Barcellos, C.; Wasserman, J.C.; Campos, R.C. Um Panorama Dos Estudos Sobre Contaminação Por Mercúrio Na Amazônia Legal No Período de 1990 a 2005—Avanços e Lacunas. Geochim. Bras. 2009, 23, 29–48. [Google Scholar] [CrossRef]
- Boischio, A.A.P.; Barbosa, A. Exposição Ao Mercúrio Orgânico Em Populações Ribeirinhas Do Alto Madeira, Rondônia, 1991: Resultados Preliminares. Cadernos de Saúde Pública 1993, 9, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Al-Saleh, I.; Elkhatib, R. Effect of Mercury (Hg) Dental Amalgam Fillings on Renal and Oxidative Stress Biomarkers in Children. Sci. Total Environ. 2012, 431, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Dufault, R.; Schnoll, R.; Lukiw, W.J.; LeBlanc, B.; Cornett, C.; Patrick, L.; Wallinga, D.; Gilbert, S.G.; Crider, R. Mercury Exposure, Nutritional Deficiencies and Metabolic Disruptions May Affect Learning in Children. Behav. Brain Funct. 2009, 5, 44. [Google Scholar] [CrossRef]
- Guney, M.; Welfringer, B.; de Repentigny, C.; Zagury, G.J. Children’s Exposure to Mercury-Contaminated Soils: Exposure Assessment and Risk Characterization. Arch. Environ. Contam. Toxicol. 2013, 65, 345–355. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Guidance for Implementing the January 2001 Methylmercury Water Quality Criterion; EPA 823-R-10-001; USEPA: Washington, DC, USA, 2010.
- Vega, C.M.; Godoy, J.M.; Barrocas, P.R.G.; Gonçalves, R.A.; De Oliveira, B.F.A.; Jacobson, L.V.; Mourão, D.S.; Hacon, S.S. Selenium Levels in the Whole Blood of Children and Teenagers from Two Riparian Communities at the Madeira River Basin in the Western Brazilian Amazon. Biol. Trace Elem. Res. 2017, 175, 87–97. [Google Scholar] [CrossRef]
- Bastos, W.R.; Gomes, J.P.O.; Oliveira, R.C.; Almeida, R.; Nascimento, E.L.; Bernardi, J.V.E.; de Lacerda, L.D.; da Silveira, E.G.; Pfeiffer, W.C. Mercury in the Environment and Riverside Population in the Madeira River Basin, Amazon, Brazil. Sci. Total Environ. 2006, 368, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Bastos, W.R.; Dórea, J.G.; Bernardi, J.V.E.; Lauthartte, L.C.; Mussy, M.H.; Lacerda, L.D.; Malm, O. Mercury in Fish of the Madeira River (Temporal and Spatial Assessment), Brazilian Amazon. Environ. Res. 2015, 140, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Hacon, S.; Dórea, J.; Fonseca, M.; Oliveira, B.; Mourão, D.; Ruiz, C.; Gonçalves, R.; Mariani, C.; Bastos, W. The Influence of Changes in Lifestyle and Mercury Exposure in Riverine Populations of the Madeira River (Amazon Basin) near a Hydroelectric Project. Int. J. Environ. Res. Public Health 2014, 11, 2437–2455. [Google Scholar] [CrossRef] [PubMed]
- Maribel Vega Ruiz, C. Interações Mercúrio-Selênio: Uma Abordagem Integrada de Avaliação de Exposição Ao Mercúrio Em Populações Ribeirinhas No Município de Porto Velho, Rondônia; Pontifícia Universidade Católica do Rio de Janeiro (PUC-Rio/Brasil): Rio de Janeiro, Brazil, 2015. [Google Scholar] [CrossRef]
- Arguelles, S.; Garcia, S.; Maldonado, M.; Machado, A.; Ayala, A. Do the Serum Oxidative Stress Biomarkers Provide a Reasonable Index of the General Oxidative Stress Status? Biochim. Biophys. Acta Gen. Subj. 2004, 1674, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Veskoukis, A.S.; Nikolaidis, M.G.; Kyparos, A.; Kouretas, D. Blood Reflects Tissue Oxidative Stress Depending on Biomarker and Tissue Studied. Free Radic. Biol. Med. 2009, 47, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Veskoukis, A.S.; Paschalis, V.; Vrabas, I.S.; Dipla, K.; Zafeiridis, A.; Kyparos, A.; Nikolaidis, M.G. Blood Reflects Tissue Oxidative Stress: A Systematic Review. Biomarkers 2015, 20, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-Transferases. The First Enzymatic Step in Mercapturic Acid Formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [PubMed]
- Habdous, M.; Vincent-Viry, M.; Visvikis, S.; Siest, G. Rapid Spectrophotometric Method for Serum Glutathione S-Transferases Activity. Clin. Chim. Acta 2002, 326, 131–142. [Google Scholar] [CrossRef]
- Hu, M.L. Measurement of Protein Thiol Groups and Glutathione in Plasma. Methods Enzymol. 1994, 233, 380–385. [Google Scholar] [CrossRef]
- Palmer, C.D.; Lewis, M.E.; Geraghty, C.M.; Barbosa, F.; Parsons, P.J. Determination of Lead, Cadmium and Mercury in Blood for Assessment of Environmental Exposure: A Comparison between Inductively Coupled Plasma–Mass Spectrometry and Atomic Absorption Spectrometry. Spectrochim. Acta Part B At. Spectrosc. 2006, 61, 980–990. [Google Scholar] [CrossRef]
- Barreiros, A.L.B.S.; David, J.M.; David, J.P. Estresse Oxidativo: Relação Entre Geração de Espécies Reativas e Defesa Do Organismo. Quim. Nova 2006, 29, 113–123. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N.J. Oxidants, Oxidative Stress and the Biology of Ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free Radicals, Antioxidants, and Human Disease: Curiosity, Cause, or Consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Kaynar, H.; Meral, M.; Turhan, H.; Keles, M.; Celik, G.; Akcay, F. Glutathione Peroxidase, Glutathione-S-Transferase, Catalase, Xanthine Oxidase, Cu–Zn Superoxide Dismutase Activities, Total Glutathione, Nitric Oxide, and Malondialdehyde Levels in Erythrocytes of Patients with Small Cell and Non-Small Cell Lung Cancer. Cancer Lett. 2005, 227, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, D.A.; Forman, H.J. Cellular Glutathione and Thiols Metabolism. Biochem. Pharmacol. 2002, 64, 1019–1026. [Google Scholar] [CrossRef]
- Ketterer, B.; Coles, B.; Meyer, D.J. The Role of Glutathione in Detoxication. Environ. Health Perspect. 1983, 49, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Huber, P.C.; Almeida, W.P.; Fátima, Â.D. Glutationa e Enzimas Relacionadas: Papel Biológico e Importância Em Processos Patológicos. Quim. Nova 2008, 31, 1170–1179. [Google Scholar] [CrossRef]
- Sheehan, D.; Meade, G.; Foley, V.M.; Dowd, C.A. Structure, Function and Evolution of Glutathione Transferases: Implications for Classification of Non-Mammalian Members of an Ancient Enzyme Superfamily. Biochem. J. 2001, 360 Pt 1, 1–16. [Google Scholar] [CrossRef]
- Gupta, A.; Bhatt, M.L.B.; Misra, M.K. Lipid Peroxidation and Antioxidant Status in Head and Neck Squamous Cell Carcinoma Patients. Oxid. Med. Cell. Longev. 2009, 2, 68–72. [Google Scholar] [CrossRef]
- Lykkesfeldt, J. Malondialdehyde as Biomarker of Oxidative Damage to Lipids Caused by Smoking. Clin. Chim. Acta 2007, 380, 50–58. [Google Scholar] [CrossRef]
- Niki, E. Lipid Peroxidation: Physiological Levels and Dual Biological Effects. Free Radic. Biol. Med. 2009, 47, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Yagi, K. Simple Assay for the Level of Total Lipid Peroxides in Serum or Plasma. In Free Radical and Antioxidant Protocols; Humana Press: NJ, USA, 1998; Volume 108, pp. 101–106. [Google Scholar] [CrossRef]
- Houston, M.C. Role of Mercury Toxicity in Hypertension, Cardiovascular Disease, and Stroke. J. Clin. Hypertens. 2011, 13, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.M.D.; Santos, R.C.C.D.; Lima, E.S. A Simple Automated Procedure for Thiol Measurement in Human Serum Samples. J. Bras. Patol. Med. Lab. 2006, 42, 345–350. [Google Scholar] [CrossRef]
- Farina, M.; Soares, F.A.; Feoli, A.; Roehring, C.; Brusque, A.M.; Rotta, L.; Perry, M.L.; Souza, D.O.; Rocha, J.B.T. In Vitro Effects of Selenite and Mercuric Chloride on Liver Thiobarbituric Acid-Reactive Substances and Non-Protein Thiols from Rats: Influences of Dietary Cholesterol and Polyunsaturated and Saturated Fatty Acids. Nutrition 2003, 19, 531–535. [Google Scholar] [CrossRef]
- Patrick, L. Mercury Toxicity and Antioxidants: Part 1: Role of Glutathione and Alpha-Lipoic Acid in the Treatment of Mercury Toxicity. Altern. Med. Rev. 2002, 7, 456–471. [Google Scholar] [PubMed]
- Stacchiotti, A.; Morandini, F.; Bettoni, F.; Schena, I.; Lavazza, A.; Grigolato, P.G.; Apostoli, P.; Rezzani, R.; Aleo, M.F. Stress Proteins and Oxidative Damage in a Renal Derived Cell Line Exposed to Inorganic Mercury and Lead. Toxicology 2009, 264, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Giustarini, D.; Tsikas, D.; Colombo, G.; Milzani, A.; Dalle-Donne, I.; Fanti, P.; Rossi, R. Pitfalls in the analysis of the physiological antioxidant glutathione (GSH) and its disulfide (GSSG) in biological samples: An elephant in the room. J. Chromatogr. B 2016, 1019, 21–28. [Google Scholar] [CrossRef]
- Lemire, M.; Mergler, D.; Fillion, M.; Passos, C.J.S.; Guimarães, J.R.D.; Davidson, R.; Lucotte, M. Elevated Blood Selenium Levels in the Brazilian Amazon. Sci. Total Environ. 2006, 366, 101–111. [Google Scholar] [CrossRef]
- Lemire, M.; Fillion, M.; Barbosa, F.; Guimarães, J.R.D.; Mergler, D. Elevated Levels of Selenium in the Typical Diet of Amazonian Riverside Populations. Sci. Total Environ. 2010, 408, 4076–4084. [Google Scholar] [CrossRef]
- Farina, M.; Aschner, M.; Rocha, J.B.T. Redox State in Mediating Methylmercury Neurotoxicity. In Methylmercury and Neurotoxicity; Springer: Boston, MA, USA, 2012; pp. 101–125. [Google Scholar] [CrossRef]
- Fonseca, M.D.F.; De Souza Hacon, S.; Grandjean, P.; Choi, A.L.; Bastos, W.R. Iron Status as a Covariate in Methylmercury-Associated Neurotoxicity Risk. Chemosphere 2014, 100, 89–96. [Google Scholar] [CrossRef]
- Barbieri, F.L.; Gardon, J. Hair Mercury Levels in Amazonian Populations: Spatial Distribution and Trends. Int. J. Health Geogr. 2009, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Xavier, O.L.P.; Asmus, C.I.R.F.; Tambellini, A.T.; Meyer, A.; Câmara, V.D.M. Contribuição Para o Estabelecimento de Níveis de Referência Para a Concentração de Mercúrio No Sangue de Crianças Na Cidade Do Rio de Janeiro. Cad. Saúde Colet. 2013, 21, 182–187. [Google Scholar] [CrossRef]
- Ziech, D.; Franco, R.; Georgakilas, A.G.; Georgakila, S.; Malamou-Mitsi, V.; Schoneveld, O.; Pappa, A.; Panayiotidis, M.I. The Role of Reactive Oxygen Species and Oxidative Stress in Environmental Carcinogenesis and Biomarker Development. Chem. Biol. Interact. 2010, 188, 334–339. [Google Scholar] [CrossRef] [PubMed]

| Communities | Belmont | Cuniã | Porto Velho | Total | p-Value |
|---|---|---|---|---|---|
| n = 41 | n = 52 | n = 104 | n = 197 | ||
| N (%) or Means (SD) | N (%) or Means (SD) | N (%) or Means (SD) | N (%) or Means (SD) | ||
| Gender | |||||
| Male | 19 (46.3%) | 22 (42.3%) | 37 (35.6%) | 78 (39.6%) | 0.492 |
| Female | 22 (53.7%) | 30 (57.7%) | 67 (64.4%) | 119 (60.4%) | |
| Age (yrs) | 11.3 (3.07) | 10.9 (2.51) | 11.1 (2.61) | 11.1 (2.68) | 0.807 |
| Age Group | |||||
| 5–11 yrs | 22 (53.7%) | 30 (57.7%) | 52 (50.0%) | 104 (53.0%) | 0.657 |
| 12–17 yrs | 19 (46.3%) | 22 (42.3%) | 52 (50.0%) | 93 (47.0%) | |
| BMI | 18.5 (3.73) | 17.5 (2.79) | 18.4 (3.74) | 18.2 (3.52) | 0.455 |
| Residence Time (yrs) | 6.36 (2.42) | 5.00 (1.56) | 5.54 (2.34) | 5.60 (2.25) | 0.255 |
| Fish Consumption | |||||
| 0–3 times/week | 33 (82.5%) | 12 (23.1%) | 100 (96.2%) | 145 (74.0%) | 0.000 1 |
| 3 or + times/week | 7 (17.5%) | 40 (76.9%) | 4 (3.8%) | 51 (26.0%) | |
| Oxidative Stress | |||||
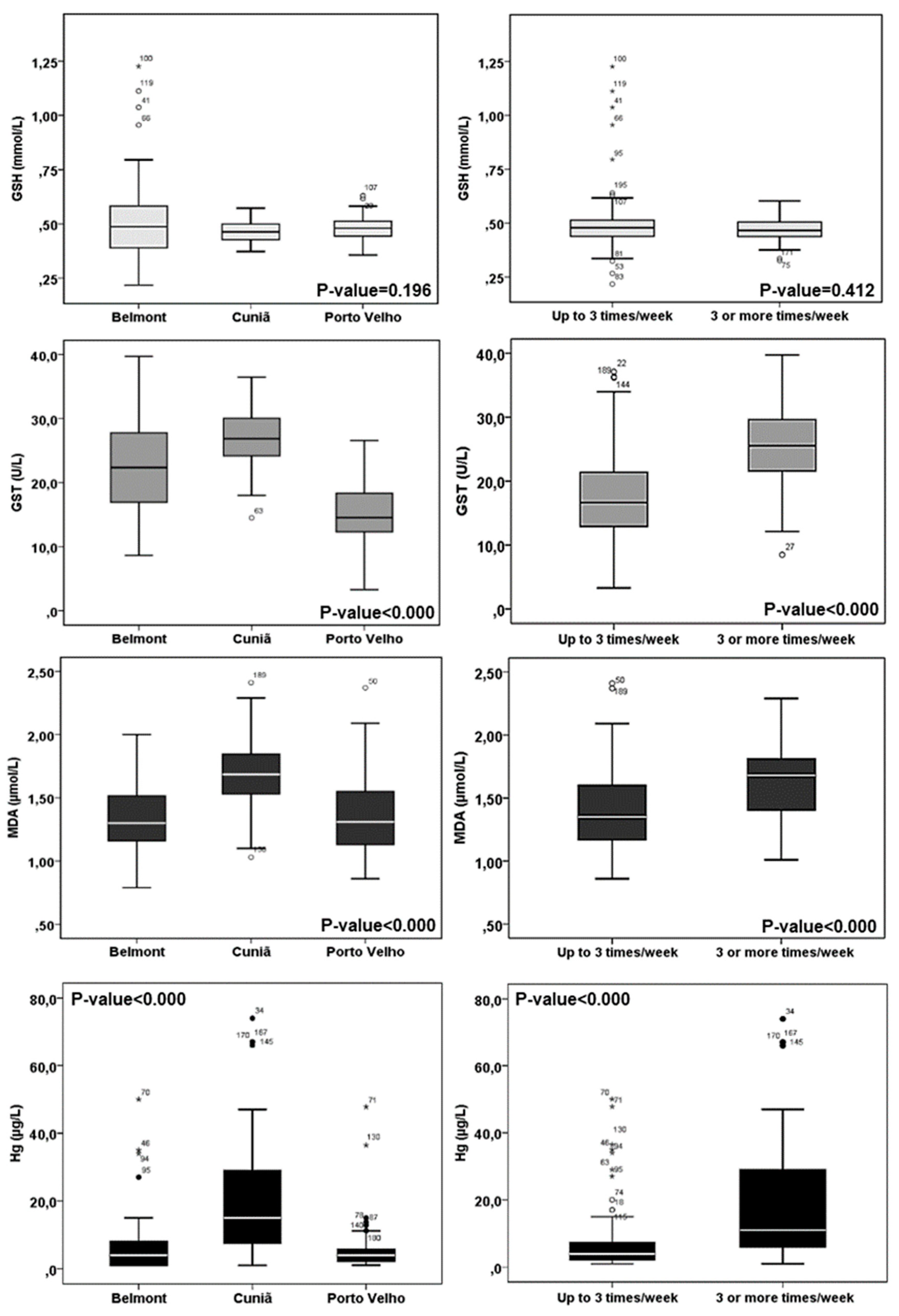
| GSH (mmol/L) | 0.53 (0.22) | 0.46 (0.04) | 0.47 (0.05) | 0.48 (0.11) | 0.196 |
| GST (U/L) | 22.4 (7.77) | 27.2 (4.93) | 15.2 (4.42) | 19.8 (7.50) | 0.000 2 |
| MDA (µg/L) | 1.34 (0.28) | 1.69 (0.27) | 1.37 (0.31) | 1.45 (0.32) | 0.000 3 |
| Hg (µg/L) | 7.84 (11.0) | 20.6 (18.0) | 5.22 (6.04) | 9.86 (13.1) | 0.000 2 |
| GSH | GST | MDA | Hg | Age | BMI | |
|---|---|---|---|---|---|---|
| GSH | 1 | |||||
| GST | 0.128 | 1 | ||||
| MDA | −0.048 | 0.277 ** | 1 | |||
| Hg | 0.142 | 0.388 ** | 0.229 ** | 1 | ||
| Age | 0.081 | −0.113 | −0.160 * | −0.072 | 1 | |
| BMI | −0.064 | −0.152 * | −0.051 | −0.035 | 0.489 ** | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carvalho, L.V.B.; Hacon, S.S.; Vega, C.M.; Vieira, J.A.; Larentis, A.L.; Mattos, R.C.O.C.; Valente, D.; Costa-Amaral, I.C.; Mourão, D.S.; Silva, G.P.; et al. Oxidative Stress Levels Induced by Mercury Exposure in Amazon Juvenile Populations in Brazil. Int. J. Environ. Res. Public Health 2019, 16, 2682. https://doi.org/10.3390/ijerph16152682
Carvalho LVB, Hacon SS, Vega CM, Vieira JA, Larentis AL, Mattos RCOC, Valente D, Costa-Amaral IC, Mourão DS, Silva GP, et al. Oxidative Stress Levels Induced by Mercury Exposure in Amazon Juvenile Populations in Brazil. International Journal of Environmental Research and Public Health. 2019; 16(15):2682. https://doi.org/10.3390/ijerph16152682
Chicago/Turabian StyleCarvalho, Leandro V.B., Sandra S. Hacon, Claudia M. Vega, Jucilene A. Vieira, Ariane L. Larentis, Rita C. O. C. Mattos, Daniel Valente, Isabele C. Costa-Amaral, Dennys S. Mourão, Gabriela P. Silva, and et al. 2019. "Oxidative Stress Levels Induced by Mercury Exposure in Amazon Juvenile Populations in Brazil" International Journal of Environmental Research and Public Health 16, no. 15: 2682. https://doi.org/10.3390/ijerph16152682
APA StyleCarvalho, L. V. B., Hacon, S. S., Vega, C. M., Vieira, J. A., Larentis, A. L., Mattos, R. C. O. C., Valente, D., Costa-Amaral, I. C., Mourão, D. S., Silva, G. P., & Oliveira, B. F. A. (2019). Oxidative Stress Levels Induced by Mercury Exposure in Amazon Juvenile Populations in Brazil. International Journal of Environmental Research and Public Health, 16(15), 2682. https://doi.org/10.3390/ijerph16152682





