Decreased Quality of Life in Individuals with Type 2 Diabetes Mellitus Is Associated with Emotional Distress
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Participants
2.3. Assessment Procedure
2.4. Statistical Analysis
3. Results
3.1. Demographic and Clinical Features
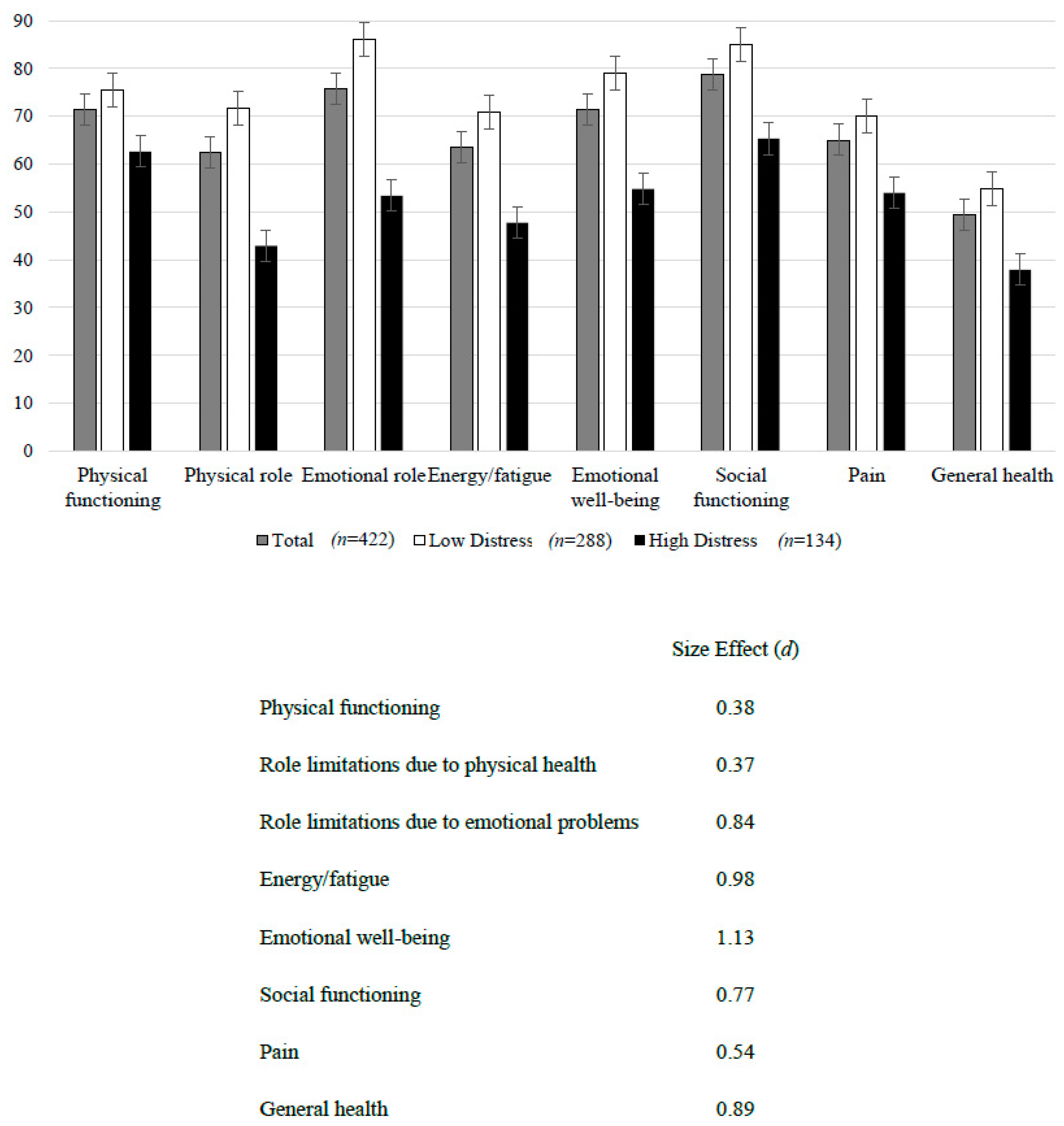
3.2. Health-Related Quality of Life
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cho, N.; Shaw, J.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.; Ohlrogge, A.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Prac. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Federation, I.D. IDF Diabetes Atlas; International Diabetes Federation: Brussels, Belgium, 2015. [Google Scholar]
- Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [CrossRef]
- DeFronzo, R.A.; Ferrannini, E.; Alberti, K.G.M.M.; Zimmet, P.; Alberti, G. International Textbook of Diabetes Mellitus, 2 Volume Set; John Wiley Sons: Hoboken, NJ, USA, 2015; Volume 1. [Google Scholar]
- Molina Iriarte, A.; Acevedo Giles, O.; Yáñez Sandoval, M.E.; Dávila Mendoza, R.; González Pedraza Avilés, A. Comparación de las prevalencias de duelo, depresión y calidad de vida asociados con la enfermedad entre pacientes con diabetes mellitus tipo 2 descontrolados y controlados. Rev. Espec. Médico-Quirúrgicas 2013, 18, 13–18. [Google Scholar]
- Juárez-Rojop, I.E.; Fortuny-Falconi, C.M.; González-Castro, T.B.; Tovilla-Zárate, C.A.; Villar-Soto, M.; Sanchez, E.R.; Hernández-Díaz, Y.; López-Narvaez, M.L.; Ble-Castillo, J.L.; Pérez-Hernández, N.; et al. Association between reduced quality of life and depression in patients with type 2 diabetes mellitus: A cohort study in a Mexican population. Neuropsychiatr. Dis. Treat. 2018, 14, 2511–2518. [Google Scholar] [CrossRef]
- Ogurtsova, K.; da Rocha Fernandes, J.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.; Cavan, D.; Shaw, J.; Makaroff, L. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Prac. 2017, 128, 40–50. [Google Scholar] [CrossRef]
- Rivera-Ledesma, A.; Lena, M.M.-L.; Sandoval-Ávila, R. Desajuste psicológico, calidad de vida y afrontamiento en pacientes diabéticos con insuficiencia renal crónica en diálisis peritoneal. Salud Ment. 2012, 35, 329–337. [Google Scholar]
- Granados, E.E.; Escalante, E. Estilos de personalidad y adherencia al tratamiento en pacientes con diabetes mellitus. Liberabit 2010, 16, 203–216. [Google Scholar]
- Lazcano Ortiz, M.; Salazar González, B.C. Estrés percibido y adaptación en pacientes con diabetes mellitus tipo 2. Aquichan 2007, 7, 77–84. [Google Scholar]
- Ortiz, M.; Ortiz, E.; Gatica, A.; Gómez, D. Factores psicosociales asociados a la adherencia al tratamiento de la diabetes mellitus tipo 2. Ter. Psicol. 2011, 29, 5–11. [Google Scholar] [CrossRef]
- Schmidt, C.B.; van Loon, B.J.P.; Vergouwen, A.C.M.; Snoek, F.J.; Honig, A. Systematic review and meta-analysis of psychological interventions in people with diabetes and elevated diabetes-distress. Diabet. Med. 2018. [Google Scholar] [CrossRef]
- Babenko, A.Y.; Mosikian, A.A.; Lebedev, D.L.; Khrabrova, E.A.; Shlyakhto, E.V. Mental state, psychoemotional status, quality of life and treatment compliance in patients with Type 2 diabetes mellitus. J. Comp. Eff. Res. 2019, 8, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, P.; Dos Santos, M.; Zanetti, M.; Ferronato, A. Dificultades de los pacientes diabéticos para el control de la enfermedad: Sentimientos y comportamientos. Rev. Latino-Am. Enferm. 2007, 15, 1105–1112. [Google Scholar]
- Iriarte, A.M.; Acevedo Giles, O.; Sandoval, M.E.Y.; Dávila Mendoza, R.; González Pedraza Avilés, A. Comparison of prevalence of mourning, depression and quality of life related to disease between patients with uncontrolled and controlled diabetes mellitus type 2. Rev. Espec. Médico-Quirúrgicas 2013, 18, 13–18. [Google Scholar]
- Organization, W.H. Use of Glycated Haemoglobin (HbA1c) in Diagnosis of Diabetes Mellitus: Abbreviated Report of a WHO Consultation; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Durán-Arenas, L.; Gallegos-Carrillo, K.; Salinas-Escudero, G.; Martínez-Salgado, H. Towards a Mexican normative standard for measurement of the short format 36 health-related quality of life instrument. Salud Publica Mex. 2004, 46, 306–315. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zúniga, M.A.; Carrillo-Jiménez, G.T.; Fos, P.J.; Gandek, B.; Medina-Moreno, M.R. Evaluación del estado de salud con la Encuesta SF-36: Resultados preliminares en México. Salud Pública Méx. 1999, 41, 110–118. [Google Scholar] [CrossRef] [PubMed]
- McGuire, B.; Morrison, T.; Hermanns, N.; Skovlund, S.; Eldrup, E.; Gagliardino, J.; Kokoszka, A.; Matthews, D.; Pibernik-Okanović, M.; Rodríguez-Saldaña, J. Short-form measures of diabetes-related emotional distress: The Problem Areas in Diabetes Scale (PAID)-5 and PAID-1. Diabetologia 2010, 53, 66. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: Thames, UK, 2013. [Google Scholar]
- Sánchez-Cruz, J.F.; Hipólito-Lóenzo, A.; Mugártegui-Sánchez, S.G.; Yáñez-González, R.M. Estrés y Depresión asociados a la no adherencia al tratamiento en pacientes con Diabetes Mellitus tipo 2. Atención Fam. 2016, 23, 43–47. [Google Scholar] [CrossRef]
- Viveros, G.R.O.; Herrera, É.O. Capacidad predictiva de la adherencia al tratamiento en los modelos socio-cognitivos de creencias en salud. Psicol. Y Salud 2011, 21, 79–90. [Google Scholar]
- Pedraza Banderas, G.L.; Vega Valero, C.Z. Caracterización psicosocial de pacientes diabéticos mexicanos. Rev. Electrón. Psicol. Iztacala 2019, 21, 1371–1393. [Google Scholar]
- Le, T.K.; Curtis, B.; Kahle-Wrobleski, K.; Johnston, J.; Haldane, D.; Melfi, C. Treatment patterns and resource use among patients with comorbid diabetes mellitus and major depressive disorder. J. Med. Econ. 2011, 14, 440–447. [Google Scholar] [CrossRef]
- Bruno, B.A.; Choi, D.; Thorpe, K.E.; Yu, C.H. Relationship Among Diabetes Distress, Decisional Conflict, Quality of Life, and Patient Perception of Chronic Illness Care in a Cohort of Patients with Type 2 Diabetes and Other Comorbidities. Diabetes Care 2019, 42, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Anderson, B.J.; McKay, S.V. Barriers to glycemic control in youth with type 1 diabetes and type 2 diabetes. Pediatr. Diabetes 2011, 12, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Chan, O.; Inouye, K.; Akirav, E.; Park, E.; Riddell, M.C.; Vranic, M.; Matthews, S.G. Insulin alone increases hypothalamo-pituitary-adrenal activity, and diabetes lowers peak stress responses. Endocrinology 2005, 146, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Chong, A.C.; Vogt, M.C.; Hill, A.S.; Bruning, J.C.; Zeltser, L.M. Central insulin signaling modulates hypothalamus-pituitary-adrenal axis responsiveness. Mol. Metab. 2015, 4, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.F.; Chalmers, D.T.; Little, K.Y.; Watson, S.J.A.E. Bennett Research Award. Regulation of serotonin1A, glucocorticoid, and mineralocorticoid receptor in rat and human hippocampus: Implications for the neurobiology of depression. Biol. Psychiatry 1998, 43, 547–573. [Google Scholar] [CrossRef]
- Lyra, E.S.N.M.; Lam, M.P.; Soares, C.N.; Munoz, D.P.; Milev, R.; De Felice, F.G. Insulin Resistance as a Shared Pathogenic Mechanism Between Depression and Type 2 Diabetes. Front. Psychiatry 2019, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Starr, J.M.; Wardlaw, J.; Ferguson, K.; MacLullich, A.; Deary, I.J.; Marshall, I. Increased blood-brain barrier permeability in type II diabetes demonstrated by gadolinium magnetic resonance imaging. J. Neurol. Neurosurg. Psychiatry 2003, 74, 70–76. [Google Scholar] [CrossRef]
- Felger, J.C.; Haroon, E.; Woolwine, B.J.; Raison, C.L.; Miller, A.H. Interferon-alpha-induced inflammation is associated with reduced glucocorticoid negative feedback sensitivity and depression in patients with hepatitis C virus. Physiol. Behav. 2016, 166, 14–21. [Google Scholar] [CrossRef]
- Clarke, J.R.; Lyra, E.S.N.M.; Figueiredo, C.P.; Frozza, R.L.; Ledo, J.H.; Beckman, D.; Katashima, C.K.; Razolli, D.; Carvalho, B.M.; Frazao, R.; et al. Alzheimer-associated Abeta oligomers impact the central nervous system to induce peripheral metabolic deregulation. EMBO Mol. Med. 2015, 7, 190–210. [Google Scholar] [CrossRef]
- Lourenco, M.V.; Clarke, J.R.; Frozza, R.L.; Bomfim, T.R.; Forny-Germano, L.; Batista, A.F.; Sathler, L.B.; Brito-Moreira, J.; Amaral, O.B.; Silva, C.A.; et al. TNF-alpha mediates PKR-dependent memory impairment and brain IRS-1 inhibition induced by Alzheimer’s beta-amyloid oligomers in mice and monkeys. Cell Metab. 2013, 18, 831–843. [Google Scholar] [CrossRef]
- Gupta, S.; Bi, R.; Kim, C.; Chiplunkar, S.; Yel, L.; Gollapudi, S. Role of NF-kappaB signaling pathway in increased tumor necrosis factor-alpha-induced apoptosis of lymphocytes in aged humans. Cell Death Differ. 2005, 12, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, X.; Yu, Y. The prevalence of impaired glucose regulation in psychiatric patients with sleep disorders and its relationship with altered hypothalamopituitary–adrenal and hypothalamopituitary–thyroid axis activity. Sleep Med. 2013, 14, 662–667. [Google Scholar] [CrossRef] [PubMed]
| Total Sample n = 422 | Low Distress n = 288 | High Distress n = 134 | Statistics | ||||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | ||
| Gender-Women | 294 | 69.7 | 195 | 67.7 | 99 | 73.9 | Fisher = 0.19 |
| Marital status-Married | 258 | 61.1 | 184 | 63.9 | 74 | 66.2 | Fisher = 0.10 |
| Socioeconomic status | Fisher = 0.20 | ||||||
| Medium | 187 | 44.3 | 134 | 46.5 | 53 | 39.6 | |
| Low | 231 | 54.7 | 151 | 52.4 | 80 | 59.7 | |
| Non-remunerated activity | 280 | 66.4 | 184 | 63.9 | 96 | 71.6 | Fisher = 0.12 |
| BMI—Overweight/obesity | 333 | 78.9 | 235 | 81.6 | 98 | 73.1 | Fisher = 0.06 |
| HbA1C ≥ 6.5 | 340 | 80.6 | 227 | 78.8 | 113 | 84.3 | Fisher = 0.18 |
| Medical comorbidity | |||||||
| -Hepatic | 54 | 12.8 | 28 | 9.7 | 26 | 19.4 | Fisher = 0.008 |
| -Renal | 103 | 24.4 | 65 | 22.6 | 38 | 28.4 | Fisher = 0.22 |
| -Neuropathy | 191 | 45.3 | 125 | 43.4 | 66 | 49.3 | Fisher = 0.29 |
| -Cardiovascular | 184 | 43.6 | 124 | 43.1 | 60 | 44.8 | Fisher = 0.74 |
| Current treatment | |||||||
| -Insulin | 296 | 70.1 | 194 | 67.4 | 102 | 76.1 | Fisher = 0.07 |
| -Hypoglycemic agent | 387 | 91.7 | 269 | 93.4 | 118 | 88.1 | Fisher = 0.08 |
| -Antihypertensive agent | 203 | 48.1 | 142 | 49.3 | 61 | 45.5 | Fisher = 0.53 |
| Substance use | |||||||
| -Alcohol—Yes | 67 | 15.9 | 43 | 14.9 | 24 | 17.9 | Fisher = 0.43 |
| -Marihuana—Yes | 2 | 0.5 | 1 | 0.3 | 1 | 0.7 | Fisher = 0.53 |
| -Nicotine—Yes | 23 | 5.5 | 17 | 5.9 | 6 | 4.5 | Fisher = 0.65 |
| Psychiatric comorbidity | |||||||
| -Major depression—Yes | 46 | 10.9 | 25 | 8.7 | 21 | 15.7 | Fisher = 0.04 |
| -Manic episodes—Yes | 1 | 0.2 | 1 | 0.3 | - | Fisher = 1.00 | |
| -Alcohol use disorder—Yes | 2 | 0.5 | 1 | 0.3 | 1 | 0.7 | Fisher = 0.53 |
| -Psychosis—Yes | 1 | 0.2 | 1 | 0.3 | - | Fisher = 1.00 | |
| Mean | S.D. | Mean | S.D. | Mean | S.D. | ||
| Current age | 54.8 | 12.8 | 56.8 | 12.1 | 50.6 | 13.1 | t =1.2, p = 0.23 |
| Years of education | 6.9 | 4.4 | 7.1 | 4.5 | 6.6 | 4.4 | t = 0.2, p = 0.83 |
| Illness evolution (years) | 15.1 | 8.7 | 15.8 | 8.8 | 13.6 | 8.1 | t = 0.2, p = 0.82 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Pimienta, E.; González-Castro, T.B.; Fresan, A.; Juárez-Rojop, I.E.; Martínez-López, M.C.; Barjau-Madrigal, H.A.; Ramírez-González, I.R.; Martínez-Villaseñor, E.; Rodríguez-Sánchez, E.; Villar-Soto, M.; et al. Decreased Quality of Life in Individuals with Type 2 Diabetes Mellitus Is Associated with Emotional Distress. Int. J. Environ. Res. Public Health 2019, 16, 2652. https://doi.org/10.3390/ijerph16152652
Gómez-Pimienta E, González-Castro TB, Fresan A, Juárez-Rojop IE, Martínez-López MC, Barjau-Madrigal HA, Ramírez-González IR, Martínez-Villaseñor E, Rodríguez-Sánchez E, Villar-Soto M, et al. Decreased Quality of Life in Individuals with Type 2 Diabetes Mellitus Is Associated with Emotional Distress. International Journal of Environmental Research and Public Health. 2019; 16(15):2652. https://doi.org/10.3390/ijerph16152652
Chicago/Turabian StyleGómez-Pimienta, Elena, Thelma Beatriz González-Castro, Ana Fresan, Isela Esther Juárez-Rojop, Miriam Carolina Martínez-López, Hugo Adrián Barjau-Madrigal, Iris Rubí Ramírez-González, Esteban Martínez-Villaseñor, Esther Rodríguez-Sánchez, Mario Villar-Soto, and et al. 2019. "Decreased Quality of Life in Individuals with Type 2 Diabetes Mellitus Is Associated with Emotional Distress" International Journal of Environmental Research and Public Health 16, no. 15: 2652. https://doi.org/10.3390/ijerph16152652
APA StyleGómez-Pimienta, E., González-Castro, T. B., Fresan, A., Juárez-Rojop, I. E., Martínez-López, M. C., Barjau-Madrigal, H. A., Ramírez-González, I. R., Martínez-Villaseñor, E., Rodríguez-Sánchez, E., Villar-Soto, M., López-Narváez, M. L., Tovilla-Zárate, C. A., & Genis-Mendoza, A. D. (2019). Decreased Quality of Life in Individuals with Type 2 Diabetes Mellitus Is Associated with Emotional Distress. International Journal of Environmental Research and Public Health, 16(15), 2652. https://doi.org/10.3390/ijerph16152652







